Research Article - (2025) Volume 3, Issue 4
Prevalence of Disorder of Villous Maturation and the Pattern of CD15 Immunohistochemical Expression in Placentas with Late Intrauterine Fetal Death
2Professor and Head of the Department, Department of Pathology, Sir Salimullah Medical College, Dhaka, Bangladesh
3Associate Professor, Department of Pathology, Sir Salimullah Medical College, Dhaka, Bangladesh
4Indoor Medical Oficer, Department of Thoracic Surgery, Dhaka Medical College Hospital, Dhaka, Bangladesh
5Lecturer, Department of Pathology, Manikgonj Medical College, Manikgonj, Bangladesh
6Department of Pathology, Uttara Adhunik Medical College, Dhaka, Bangladesh
Received Date: Jun 19, 2025 / Accepted Date: Jul 15, 2025 / Published Date: Jul 23, 2025
Copyright: ©Â©2025 Saba, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Saba, S., Begum, S., Ferdous, J. N., Billah, M., Tahira, U. Q., et. al. (2025). Prevalence of Disorder of Villous Maturation and the Pattern of CD15 Immunohistochemical Expression in Placentas with Late Intrauterine Fetal Death. Int Internal Med J, 3(4), 01-09.
Abstract
Background: Disorder of villous maturation is one of the main reasons for antenatal hypoxia and fetal deaths. Meticulous microscopic and CD15 immunohistochemical studies of such cases may provide valuable information regarding the cause of intrauterine fetal death (IUFDs), can help to avoid diagnostic dilemmas and can offer potential treatment options for its prevention in future pregnancies.
Objective: To determine the types of disorders associated with villous maturation in the placenta in late intrauterine fetal death patients, identify delayed villous maturation via histopathological and CD15 immunohistochemical studies and determine its association with recurrent pregnancy loss.
Method: A cross -sectional study was carried out in the Department of Pathology, Sir Salimullah Medical College Mitford Hospital, Dhaka, from March 2021 to January 2023. A total of 80 patients with late IUFDs were included in this study. The placentas of the dead newborns were histopathologically analyzed, and CD15 immunohistochemistry was performed.
Result: Among the 80 patients, 40 (50.0%) were multiparas, 36 (45.0%) were primis, and only 4 (5.0%) were grand multipara patients. Twenty (25.0%) patients had a history of one IUFD, and 4 (5.0%) patients had a history of multiple IUDFs. Twenty-eight (35%) patients had disorders of villous maturation, among whom 19 (23.8%) had delayed villous maturation (DVM), 4 (5.0%) had malformed villi, 3 (3.8%) had distal villous hypoplasia, 1 (1.3%) had deficiency of intermediate villi and 1 (1.3%) had accelerated villous maturation. CD15 immunostaining was performed in 53 patients, including villitis of unknown origin, disorders of villous maturation and villous edema. Among them, 34 (64.15%) had negative CD15 expression, whereas 19 (35.8%) had positive CD15 expression. All 19 patients with delayed villous maturation had positive CD15 expression (100%), which confirmed their histological diagnosis and ensured the complete exclusion of other false positive cases. Among them, 8 (42.1%) had moderate CD15 expression and 11 (57.89%) had severe CD15 expression. Among the 13 multipara patients with DVM, 12 (92.3%) had a history of previous IUFD. Among the remaining 31 multipara patients with no DVM, 11 (35.5%) had a previous history of IUFD. A significant difference in a history of previous intrauterine fetal death was observed between the DVM present and absent groups (p=0.001).
Conclusion: This study revealed that disorders of villous maturation are among the main causes of late IUFD and that DVM shows a distinctive pattern of CD15 immunohistochemical expression. Moreover, this study shed light on the association between DVM and recurrent pregnancy loss. The identification of DVM can guide physicians in decision making regarding the management of the patients and prevention of further pregnancy losses.
Keywords
Placenta, Late IUFD, DVM, CD15, IHC
Introduction
According to the International Classification for Diseases 11th revision (ICD-11), fetal death prior to complete expulsion or extraction from the mother of a product of conception at or after 28 weeks or weighing greater than 1000 g or after 28 weeks is called late intrauterine fetal death (IUFD). According to the World Health Organization, in 2022, the stillbirth rate in Bangladesh was 25 per 1000 total births, which is still high. The human placenta is a discoid, choriodecidua organ that acts as a portal connecting the fetus with the uterine wall of the mother via the umbilical cord. Despite its undeniable role in human fetal development, the study of the placenta has lagged behind that of fetuses1 Intrauterine fetal death may be caused by maternal, fetal or placental factors. The placental factors include umbilical cord and placental disc abnormalities such as retroplacental hemorrhage, circumvallation, marginal cord, true knot, calcification, inflammatory reactions, circulatory compromise (maternal or fetal), abnormalities in villous maturation, hemorrhage, necrosis, villous edema, perivillous fibrin deposition and other conditions [1-5]. Among all the placental factors, disorders of villous maturation constitute 44.0%, of which delayed villous maturation comprises 22% [6].
The placenta undergoes profound developmental changes throughout the conception period, progressing from mesenchymal to immature intermediate to stem villi to mature intermediate to mature terminal villi, which are collectively referred to as villous maturation. This process is characterized by a steadily increasing number of villous capillaries and the formation of vasculosyncytial membranes within terminal villi via the merging of syncytiotrophoblasts with the villous capillary endothelium. It shortens the oxygen diffusion distance between the fetal and maternal blood. This is the main mechanism by which the placenta meets the growing fetal demand in the last 2 months of pregnancy [6]. Thus, defective villous tree maturation can cause decreased vasculosyncytial membrane formation, resulting in placental dysfunction and fetal hypoxia [6].
Disorders of villous maturation are ramification disorders of the villous tree, whereby the observed morphology chronologically differs from what should be normal for the gestational age [7]. Its etiology is unknown, with diabetes, ABO incompatibility and viral disease being the most important risk factors.7 Persistent villous immaturity, with a predominance of immature villi and a deficiency of terminal villi, increases the risk of stillbirths approximately 70- fold and the risk of recurrence 10-fold [8]. The recurrence rate of DVM in subsequent pregnancies is approximately 5% [6].
Turowski and Vogel, (2018) suggested a classification of maturation disorders according to the morphology of the placenta, which is described below- [9]
Accelerated Villous Maturation:
- It is diagnosed by identifying a diffuse pattern of term- appearing villi with increased syncytial knots (>33%) and intervillous fibrin, usually alternating with areas of villous paucity prior to term.
- Accelerated maturation can be observed in FGR, preeclampsia, and preterm labor,; in twin or triplet placentas and in the hyperviscosity of fetal blood.
Delayed Villous Maturation:
- Characteristic morphology shows villi of increased villous diameter, with a cellular stroma and increased extracellular matrix. Capillaries are centrally placed, and vasculosyncytial membranes are reduced, which can be found a few weeks before or close to term, rarely before 34 weeks of gestation.
- For DVM, more than 30% of the villi within the basal 2/3rd of the placental parenchyma is immature. The immature intermediate/terminal villous ratio is increased.
- It is associated with maternal metabolic disorders and obesity, intrauterine hypoxia, fetal chronic diseases, disorders of the central nervous system in children, congenital anomalies, FGR, and fetal death [9,10].
- The risk of recurrence in subsequent pregnancies is greater.
-
Arrest of Villous Maturation:
-
Deficient ramification of immature intermediate villi with large diameters and lumpy shapes. The stroma is primitive mesenchymal, embryonic, loosely reticular with Hofbauer cells and few capillaries with decreased VSM. The chorionic epithelium is flat and single layered with few knots and cytotrophoblast cells .
-
Seen in early miscarriage and late pregnancy, maternal obesity with and without diabetes, syndromes and chromosome aberration (trisomy).
-
Recurrence risk is assumed to depend on the severity of the disease.
- Distal Villous Hypoplasia:
-
- The villi are thin and relatively elongated with increased intervillous space and syncytial knots. It is more commonly associated with MVM in early pregnancy (<32 weeks of gestation). Features in the lower two-thirds and involving at least 30% of 1 full-thickness parenchymal slide are diagnostic.
- Early-onset FGR and preeclampsia may lead to adverse fetal outcomes [11]
- Deficiency of Intermediate Villi:
-
- This disease is characterized by the absence of intermediate villi, with only stem villi and slender terminal villi. The stroma is partly fibrous and contains vasculosyncytial membranes with regressive syncytial knots in both villous types.
- It is clinically associated with preeclmapsia, HELLP syndrome, chronic hypertension, collagenous syndromes, prematurity, placental hypoplasia, IUFD and FGR.
-
In most cases of asymptomatic villous immaturity, the only method of identification is histopathological observation of the placenta. However, delayed villous maturation and villitis can mimic the appearance of villous edema and can be microscopically associated with severe fetal erythroblastosis and/or nonspecific chronic villitis of unknown etiology [1,7,11]. Therefore, by performing CD15 immunostaining, DVM can be confirmed to be differentiated from other similar histopathological entities, which is highly important, as patient management differs across different diagnoses.
-
CD15 is a stage-specific embryonic antigen (SSEA-1) known to be expressed in the immature endothelium. CD15 expression mirrors cytoplasmic maturity, and its expression is increased in the G1 phase of the cell cycle. The normal placenta contains CD15+ endothelium with physiological villous immaturity in the first and second trimesters. Gradual loss of CD15 positivity at term indicates trophoblastic differentiation, spiral artery remodeling and placental maturity. Therefore, CD15 positivity in the villous endothelium at term is an important indicator of delayed villous maturation. As endothelial cells perform many essential functions, endothelial dysfunction leads to vascular disease and can cause distress to the fetus, thus causing fetal death [6,7].
Therefore, CD15 immunostaining can differentiate DVM from other cases, which may help physicians treat patients accordingly and take necessary measures to prevent further recurrence [7].
Method
This was a cross-sectional, descriptive and observational study. A total of 80 mothers aged 18 years or above with a diagnosis of unexplained late IUFD at a gestational age of 28 weeks or above (maternal and fetal screening performed till 28 weeks and yielded normal results) were included in this study. The maternal exclusion criteria included: diabetes, hypertension, eclampsia, preeclampsia, covid, dengue and other co morbidities. The fetal exclusion criteria included: fetal with congenital anomaly and macerated fetus. The study was conducted at the Department of Pathology, Sir Salimullah Medical College, Dhaka, Bangladesh, from March 2021 to January 2023.
The placentas were collected from the Department of Obstetrics and Gynecology, Sir Salimullah Medical College Mitford Hospital, Dhaka, immediately after delivery and fixed with 10% neutral buffered formalin for 24 hours. The specimens were subsequently processed and examined at the Department of Pathology, SSMC, for histopathological examination.
Placentas were weighed after the cord and membrane were removed. After meticulous gross examination, 6 blocks were submitted: 1 block to include a roll of the extraplacental membranes from the rupture edge to the placental margin, 2 cross sections of the umbilical cord (one from the fetal end and another 5 cm from the placental insertion end), and 3 blocks each containing a full thickness or upper 3rd or lower 3rd section of placenta parenchyma. The slides were stained with H&E.
Microscopic examination of the placentas was performed, and slides from 53 cases (disorders of villous maturation, edema and villitis) were subjected to CD15 immunostaining. Immunostaining was evaluated via light microscopy. The percentage of positive staining was determined in 5 highpower fields on each slide, and the average percentage of CD15-positive cells was calculated.
Primary antibody: FLEX monoclonal mouse anti-human antibody to CD15 (clone Carb-3). The secondary antibody used was EnVision FLEX/Horseraddish Peroxidase SM802.
Scoring System (Table I):
Positive: Circumferential CD15-positive expression in the macrovasculatures (chorionic plate and stem vessels), microvasculatures (terminal villi) or both in >30% of the villi of the placental parenchyma was considered pathological placental villous immaturity.
|
CD15 grading of placental immaturity |
Assessment of endothelial fetoplacental immaturity by CD15 expression |
Similar CD15 status of EC in normal pregnancy |
Assessment of structural immaturity of the placenta |
||
|
Macrovascular ECs |
Microvascular ECs |
Immature villi |
Mature villi |
||
|
Grade I(severe immaturity) |
Immaturity (CD15+) |
Immaturity (CD15+) |
I trimester |
Increased >50% |
Deficiency |
|
Grade II(moderate immaturity) |
|
II trimester |
Increased 30%-50% |
Decreased |
|
|
Grade III(maturity) |
Maturity (CD15-) |
Maturity (CD15-) |
III trimester |
Normal |
Normal |
Table 1: CD15 Immunophenotyping for Postpartum Assessment of the Grade of Pathological Placental Immaturity [7].
Negative: Positive expression in <30% of the villi of the placental parenchyma was considered normal maturity (Grade 3).7
Positive and negative controls: Tissue from the Vermiform Appendix, diffuse epithelial membrane staining was used as a positive control for CD15. The negative control was normal term placental tissue.
Results
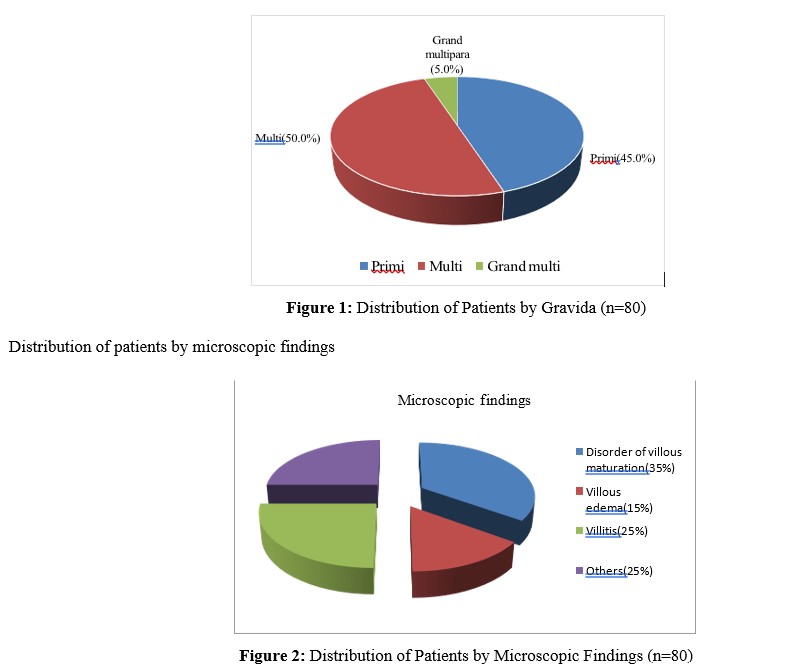
Distribution of patients by gravida
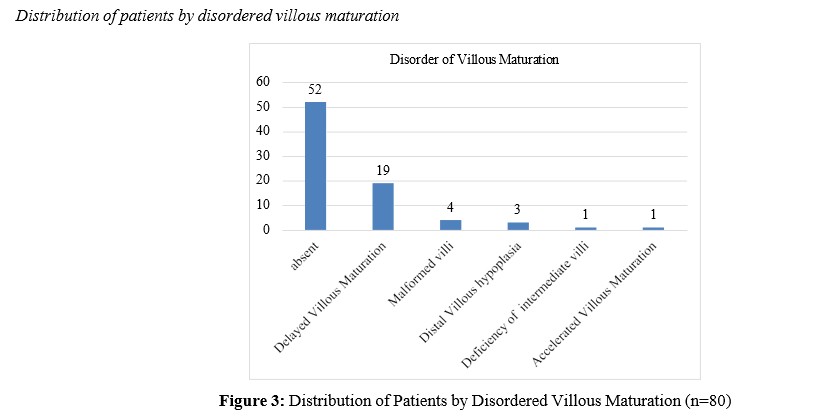
Distribution of patients by CD15 immunohistochemical expression
|
CD 15 expression |
Frequency (n) |
Percentage (%) |
|
Negative |
34 |
64.15 |
|
Positive |
19 |
35.8 |
Distribution of CD15 expression in patients with delayed villous maturation
Table 2: Distribution of Patients by CD15 Expression (n=53)
|
CD 15 expression |
Frequency (n) |
Percentage (%) |
|
Negative |
0 |
0.0 |
|
Positive |
19 |
100.0 |
|
Moderate |
8 |
42.1 |
|
Severe |
11 |
57.89 |
Table 3: Distribution of CD15 Expression in Patients with Delayed Villous Maturation (n=19)
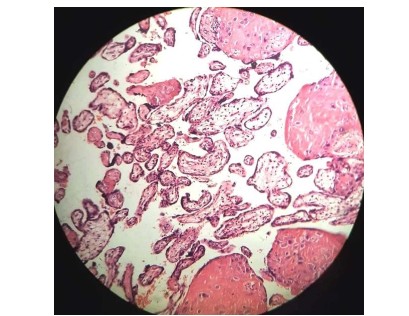
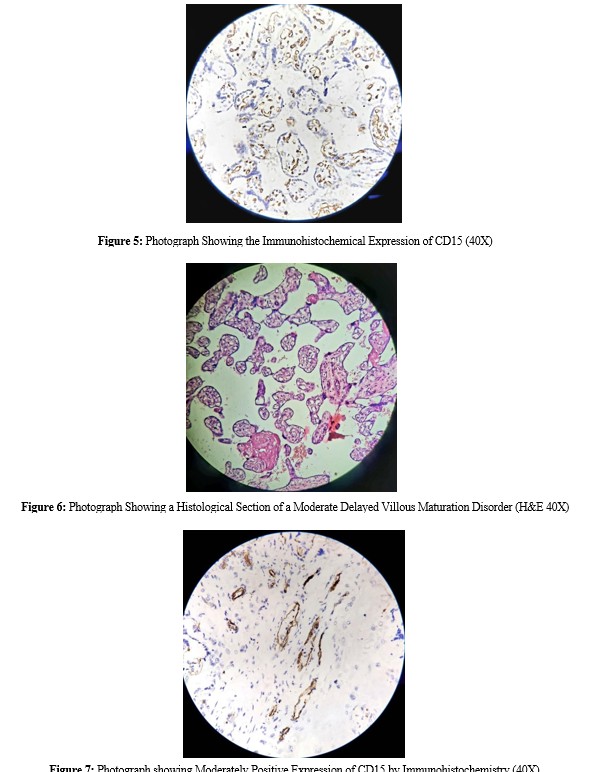
Figure 4: Photograph Showing a Histological Section of a Severe Case of Delayed Villous Maturation (H&E 40X)
Distribution of patients by previous history of intrauterine fetal death
|
History of intrauterine fetal death |
Frequency (n) |
Percentage (%) |
|
Absent |
56 |
70.0 |
|
Single |
20 |
25.0 |
|
Multiple |
4 |
5.0 |
Association between a history of previous IUFD and delayed villous maturation
Table 4: Distribution of Patients by Previous History of Intrauterine Fetal Death (IUFD) (n=80)
|
History of previous IUFD |
Delayed villous maturation |
p value |
|
|
|
Present |
Absent |
|
|
Absent |
1 (7.7%) |
20 (64.5%) |
0.001 |
|
Present |
12 (92.3%) |
11 (35.5%) |
|
Fisher’s exact test (n=44)
Table 5: Association between a History of Previous IUFD and Delayed Villous Maturation
Discussion
In our previous study, we aimed to categorize fetal deaths by different gross and histopathological findings to analyze the underlying mechanisms and to determine how many unexplained fetal deaths remain unexplained after examination of the placenta [1]. One of the major causes was the disorder of villous maturation. In our current study, we aimed to further categorize different types of villous maturation disorders, CD15 immunopositivity in the endothelium of delayed villous maturation, their grading and their association with recurrent pregnancy loss.
In the present study, a total of 80 cases of late IUFD were examined histopathologically. Among them, 50% were multiparous, 45% were primiparous, and 5% were grand multiparous patients (Figure 1). The majority of the patients (70.0%) did not have any previous history of intrauterine death (IUFD), while 20 (25.0%) patients had a history of one IUFD, and 4 (5.0%) patients had a history of multiple IUFDs (table 4).
In the present study, 28 (35%) patients presented with disorders of villous maturation. The patterns revealed that 19 (23.8%) patients had delayed villous maturation, 4 (5.0%) had malformed villi, 3 (3.8%) had distal villous hypoplasia, 1 (1.3%) had deficiency of intermediate villi, and 1 (1.3%) had accelerated villous maturation (figure 2,3). In 2009, Korteweg and his colleagues12 reported delayed villous maturation in 4.3% and accelerated villous maturation in 12% of stillbirth cases. Gibbins et al., (2016) reported delayed villous maturation in 9.9% and accelerated villous maturation in 7.4% of fetal death cases [13]. Ananthan et al., (2019) reported that delayed villous maturation (DVM), 38.82%, was significantly present in stillbirths and that the incidence of distal villous hypoplasia (DVH) was 44 (51.76), which supports the findings of the present study [5]. Disorders of villous maturation [delayed villous maturation, 22% (31/143); accelerated villous maturation, 20% (28/143); and maturation arrest, 4% (5/143)] were significantly more common in cases of fetal death according to the study of Jaiman et al., (2020) [6]. In the study conducted by Kulkarni et al., (2021), delayed villous maturation was found in 5.9% of fetal deaths, accelerated villous maturation in 17.4%, and distal villous hypoplasia in 41.3% of fetal deaths [3]. The above findings are almost comparable with those of the present study.
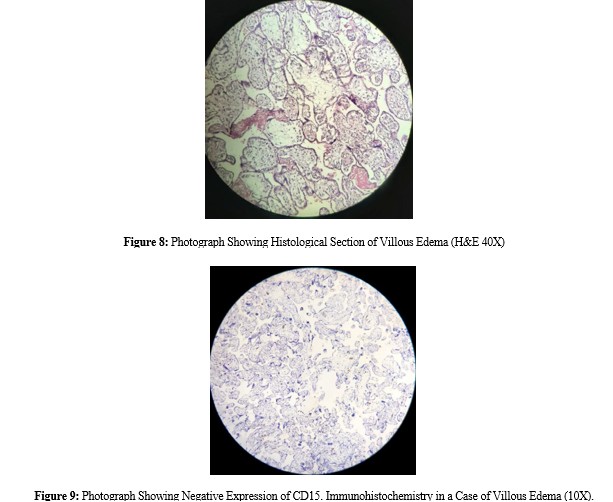
CD15 immunohistochemistry was performed on samples from 53 patients, including villitis of unknown origin, disorders of villous maturation and villous edema (villous edema, figure 8). Thirty- four (64.15%) patients had negative CD15 expression (figure 9), whereas 19 (35.8%) had positive CD15 expression (figure 5,7, table II). All 19 patients who expressed CD15 immunopositivity were histologically compatible with delayed villous maturation (DVM) (figure 4,6), which strongly supports the theory of Seidmann et al., (2014), who reported that only DVM shows CD15 immunopositivity. Among them, 8 (42.1%) had moderate CD15 expression, and 11 (57.89%) had severe CD15 expression. Eighty- five placentas were diagnosed with delayed villous maturation microscopically [7]. Seidmann et al., (2014)7 reported that 64 out of 111 normal and pathological term placentas presented moderate immaturity (30–50%) with moderately positive CD15 immunostaining, and 21 of them presented severe immaturity (>50%) with strongly positive CD15 immunostaining.
Among the 44 multipara patients, 13 patients had DVM, and 12 (92.3%) had a history of previous IUFD. The remaining 31 patients had no DVM, and among them, 11 (35.5%) had a history of previous IUFD. A significant difference in the number of patients with a history of previous IUFD was observed between the groups (p=0.001) (Table III). Stallmach et al., (2001) also reported that the risk of recurrent stillbirth was 10-fold greater than that at baseline and occurred mostly after 35 weeks of gestation [8].
Conclusion
Delayed villous maturity can cause significant fetal hypoxia and hence unexplained fetal death in apparently healthy mother and fetus. However, it can be prevented in subsequent pregnancies by taking necessary measures. In this context, the diagnosis of DVM, both histopathologically and immunohistochemically, is highly valuable. Our current study revealed that patients with delayed villous maturation disorder express positive CD15 expression in the immature villous endothelium and show a statistically significant association with recurrent pregnancy loss. We propose that immature CD15+ endothelium in the term placenta is an important diagnostic marker of DVM.
Human Ethics Declarations: This research work has been approved by Institutional Ethics Committee of Sir Salimullah Medical college Mitford Hospital, Dhaka (Ref: SSMC/2021/321).
Consent to Participate Declaration: Informed written consents were taken from all the patients or patient’s relatives before collecting their placentas for histopathological and immunohistochemical studies.
Acknowledgments: We thank Biostatistician Dr. Farzana Azam Tuli for assisting in the data analysis.
Author Contributions: Saba S prepared the research protocol and was responsible for data collection, arranging the data analysis and preparing the manuscript. Begum S, Ferdous J N and Billah M were responsible for conceptualizing the study and assisted in protocol development, manuscript preparation and proof reading. Tahira UQ and Ahmed S helped in grossing.
Funding: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflicts of Interest: None.
References
- Ujwala, C. H., Guruvare, S., Bhat, S. S., Rai, L., & Rao, S. (2013). Evaluation of placenta in foetal demise and foetal growth restriction. Journal of Clinical and Diagnostic Research: JCDR, 7(11), 2530.
- Kulkarni, A. D., Palaniappan, N., & Evans, M. J. (2017). Placental Pathology and Stillbirth: A review of the literature and guidelines for the less experienced. Journal of fetal medicine, 4(04), 177-185.
- Malusi, Z., Schubert, P. T., Theron, G. B., & Wright, C. A. (2019). The value of histopathology of the placenta in a tertiary referral hospital in South Africa.
- Ananthan, A., Nanavati, R., Sathe, P., & Balasubramanian, H. (2019). Placental findings in singleton stillbirths: a case-control study. Journal of tropical pediatrics, 65(1), 21-28.
- Jaiman, S., Romero, R., Pacora, P., Jung, E. J., Kacerovsky, M., Bhatti, G., ... & Hsu, C. D. (2020). Placental delayed villous maturation is associated with evidence of chronic fetal hypoxia. Journal of perinatal medicine, 48(5), 516-518.
- Seidmann, L., Suhan, T., Unger, R., Gerein, V., & Kirkpatrick, C. J. (2014). Transient CD15-positive endothelial phenotype in the human placenta correlates with physiological and pathological fetoplacental immaturity. European Journal of Obstetrics & Gynecology and Reproductive Biology, 180, 172-179.
- Stallmach, T., & Hebisch, G. (2004). Placental pathology: its impact on explaining prenatal and perinatal death. Virchows Archiv, 445, 9-16.
- Turowski, G., & Vogel, M. (2018). Reâ?view and view on maturation disorders in the placenta. Apmis, 126(7), 602-612.
- Khong, T. Y., Mooney, E. E., Ariel, I., Balmus, N. C., Boyd, T. K., Brundler, M. A., ... & Gordijn, S. J. (2016). Sampling and definitions of placental lesions: Amsterdam placental workshop group consensus statement. Archives of pathology & laboratory medicine, 140(7), 698-713.
- Rebecca, N. (2011). Manual of pathology of the human placenta. Springer-verlag New York Incorporated.
- Korteweg, F. J., Erwich, J. J. H., Holm, J. P., Ravisé, J. M., van der Meer, J., Veeger, N. J., & Timmer, A. (2009). Diverse placental pathologies as the main causes of fetal death. Obstetrics & Gynecology, 114(4), 809-817.
- Gibbins, K. J., Silver, R. M., Pinar, H., Reddy, U. M., Parker, C. B., Thorsten, V., ... & Goldenberg, R. L. (2016). Stillbirth, hypertensive disorders of pregnancy, and placental pathology. Placenta, 43, 61-68.




