Research Article - (2025) Volume 4, Issue 3
Prevalence of Dengue Fever and Malaria Patient, Among Febrile Patients in Kassala State in Sudan 2023
2Microbiology /Medical Laboratory Science, Khartoum University, Sudan
Received Date: Aug 19, 2025 / Accepted Date: Sep 08, 2025 / Published Date: Sep 18, 2025
Copyright: ©2025 Dr. Zinab Abdellatef Othman Blal, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Blal, Z. A. O., Badia, N. I. M., Musa, A. M. A., Alnor, J. A. A. D., Ahmed, H. M. I., et al. (2025). Al-Madain College for Medical Sciences and Technology Medical laboratory sciences Prevalence of Dengue Fever and Malaria Patient , among febrile patients in Kassala State in Sudan 2023. Archives Clin Med Microbiol, 4(3), 01-09.
Abstract
Background: Prevalence of dengue virus and malaria parasite or co-infection dengue virus and malaria parasite among febrile patients in Kassala State in Sudan. Malaria and dengue are common mosquito-borne diseases around the world that cause high mortality and morbidity. The number of cases of both diseases is currently rising in Sudan and is associated with climate and environmental changes. This study was done to determine the prevalence of dengue virus and malaria parasite in febrile patients in Kassala State in Sudan.
Methods: This is a cross-sectional analytical study was carried out on the target population (Khartoum state) (2023). The study included a total of (100) Blood specimens were collected from 100 consenting subjects with febrile complaints attending four police hospital in Kassala State in Sudan. Serological determination of dengue virus Igm was carried out using the enzyme linked Immunochromatographic assay (ICT)detection of dengue virus antibodies or surface antigen, sample was collected, IBM statistic SPSS (V 25) was used for statistical analysis.
Results: If it is inferred that there is a relationship between malaria and dengue fever, we have a morale level of 0.05 and it is inferred that there is a strong relationship between malaria and dengue fever. If you have dengue fever, you have malaria, so malaria has greatly affected dengue fever.
Conclusion: The study concluded that prevalence of malaria and dengue co-infections was estimated high, severity of clinical symptoms of patients with malaria and dengue co-infections was observed, and young ages were more affected by the co-infections and that malaria has greatly affected dengue fever. finding highlights, the necessity of clear understanding of the epidemiology of malaria and dengue co-infections is essential to inform decision-makers to institute an appropriate control strategy for both diseases.
Keywords
AI-Driven Platforms, Freelance Economy, Creative Industries, Business Model Innovation, Trust and UX Design, Digital Labour Markets
Chapter One
Introduction
Malaria has been well documented as an ancient disease in Egyptian and Chinese writing beginning in 2700 bc. By 200 bc, malaria was identified in Rome, spread throughout Europe during the twelfth century, and arrived in England by the fourteenth century.Malaria is endemic in more than 90 countries with a population of 2400 million people, representing 40% of the world’s population.
At least 90% of deaths caused by malaria occur in Africa. Plasmodium falciparum is the major species associated with deadly infections throughout the world. Unfortunately, prevention remains a complex problem, and no drug is universally effective for all Plasmodium species. In addition, of the five species that infect humans, P. vivax and P. falciparum cause 95% of infections. P. vivax may be responsible for 80% of the infections, because this species has the widest distribution in the tropics, subtropics, and temperate zones. P. falciparum is generally confined to the tropics, P. malaria is sporadically distributed, and P. ovale is confined mainly to central West Africa and some South Pacific islands. The fifth human malaria, Plasmodium knowlesi, a malaria parasite of long-tailed macaque monkeys, has been confirmed in human cases from Malaysian Borneo, Thailand, Myanmar, and the Philippines. The vector for malaria is the female anopheline mosquito. When the vector takes a blood meal, sporozoites contained in the salivary glands of the mosquito are discharged into the puncture wound (Figure 49-1). Within an hour, these infective sporozoites are carried via the blood to the liver, where they penetrate hepatocytes and begin to grow, initiating the preerythrocytic or primary exoerythrocytic cycle. The sporozoites become round or oval and begin dividing repeatedly. Schizogony results in large numbers of exoerythrocytic merozoites. Once these merozoites leave the liver, they invade the red blood cells (RBCs), initiating the erythrocytic cycle. A dormant schizogony may occur in P. vivax and P. ovale organisms, which remain quiescent in the liver. These resting stages have been termed hypnozoites and lead to a true relapse, often within 1 year or up to more than 5 years later. Delayed schizogony does not occur in P. falciparum, P. malaria, or P. knowlesi. [1] .
Both malaria and dengue are known to be rapidly spreading mosquito-borne diseases and are of high importance in terms of morbidity and mortality, posing a worldwide public health problem due to the global expansion of their vectors [2]. Malaria is a protozoan parasitic infection caused by apicomplexan Plasmodium spp. which is usually transmitted by Anopheles spp. mosquitoes. Five parasite species cause malaria in humans; nevertheless, two of these species– P. falciparum and P. vivax–pose the greatest threat. Plasmodium falciparum is the deadliest malaria parasite and the most prevalent on the African continent [3]. Dengue infection is caused by any of the four distinct serotypes (DEN-1, 2, 3, and 4) of single-stranded RNA flavivirus, and is mainly transmitted by Aedes aegypti mosquitoes [4]. Dengue is considered the most important arbovirus in terms of morbidity and mortality. The incidence of this disease has increased dramatically, especially the incidence of the more severe dengue hemorrhagic fever (DHF) [5]. Malaria and dengue are endemic in similar tropical regions, and therefore, may result in the possibility of co-infection. Urban demographic expansion, deforestation, and agricultural settlements in peri-urban areas are known causes of the probability increasing of concurrent infections of these two diseases [6]. Malaria is endemic in Sudan, whereas dengue has established itself in an epidemic transmission cycle [7]. The major outbreaks of dengue in Sudan were caused by serotypes 1, 2, and 3 that circulated in different parts of Sudan [8,9]. Over 80%, 58%, and 33% of the cases tested for dengue in West, Central, and East Darfur, respectively, also tested positive for malaria, according to an analysis of the Sudanese records keeping track of health facilities. This interesting observation points out the high level of co-infections with malaria amongst Sudanese patients with severe symptoms who tested positive for dengue fever and has implications for vector control approach and disease surveillance activities [10]. Kassala witnessed increased cases of dengue during the last three years according to the local clinical services records [11]. Malaria has become a major health problem in the last few years with incidence rate of 140 cases per 10,000 populations in Kassala state [12]. Clinical manifestations include symptoms that are non-specific and cannot be distinguished from other febrile illnesses. In addition, the misdiagnosis of both malaria and dengue infections may also lead to ambiguity in terms of disease burden in this area. Conducting a laboratory survey for malaria and dengue co-infections in Kassala city is necessary to assess the burden of concurrent infections and will reveal the potential possibility of existing co-infections. Therefore, this study was conducted to assess the burden and the risk factors associated with malaria and dengue co-infections in Kassala, eastern Sudan. [13].
Justification
An observation points out high level of co-infections with malaria amongst Sudanese patients has implications for vector control approach and disease surveillance activities [10]. Studying the current prevalence of dengue fever among malaria patients will be useful in future therapeutic studies. There a few previous study conducted about this study in Sudan.
Objectives
General Objective
To estimate the prevalence of dengue fever among malaria patients at Kassala state hospitals, 2023
Specific Objectives
• To respond to potential co-infection early to defense mechanism
• To improve the method of detection.
• To detect epidemics early and to respond to potential epidemics effectively
• To strengthen monitoring and evaluation to ensure optimal programme implementation, management and performances
Chapter Two
Literature Review
Introduction
Dengue may be the most widespread arboviral illness worldwide. Most patients with dengue infection have only mild disease or classic dengue fever, with influenza-like symptoms, severe headache, and aching joints and muscles. However, in a small percentage of patients—maybe half a million people every year— potentially lethal forms of dengue called dengue hemorrhagic fever and dengue shock syndrome develop. It has been known for many years that antibodies directed against either of the two surface proteins of the dengue virus (the precursor membrane protein and the envelope protein) can neutralize infectivity and confer protection, although envelope protein is known to be the more important protective antigen. Paradoxically, antibodies against either protein can also enhance infectivity, depending on their specificity, avidity, and concentration. Dejnirattisai et al. 1 recently reported that after a secondary dengue infection, a large proportion of the human antibody repertoire is directed against the precursor membrane protein on immature virus particles. This observation might be pertinent to the development of inactivated dengue vaccines.
Two to three billion people in tropical and subtropical climates are at risk for dengue infection, and an estimated 50 million infections occur each year, mostly in urban areas. The four dengue serotypes are transmitted by mosquitoes, and the ongoing spread of serotypes and of the vectors (mosquitoes) has now reached more than 100 countries, including the United States, variable, particularly with age. Young children might simply have fever and a rash, whereas classic dengue fever is more likely to develop in older children and adults. If dengue fever progresses to dengue hemorrhagic fever at the time of effervescence, it generally manifests as a capillary leak syndrome accompanied by thrombosis [14].
Dengue virus disease is a major public health problem in developing and tropical countries, and has been continuously spreading to new geographical areas. Among travelers returning from low and middle-income countries dengue is the second most diagnosed cause of fever after malaria.
Dengue Virus
Dengue virus (DENV) is a mosquito-borne virus that primarily spreads through the bites of infected Aedes mosquitoes, particularly Aedes aegypti and Aedes albopictus. The virus belongs to the Flaviviridae family and has four distinct serotypes: DENV-1, DENV-2, DENV-3, and DENV-4.
Transmission
1. Mosquito-Borne Transmission:
- Primary Vectors: Dengue virus (DENV) is primarily transmitted by Aedes aegypti and Aedes albopictus mosquitoes.
- Transmission Cycle:
- Infection of Mosquito: A mosquito becomes infected when it bites a person who has dengue virus in their blood.
- Virus Replication in Mosquito: The virus replicates inside the mosquito, typically taking 8-12 days.
- Transmission to Humans: The infected mosquito can then transmit the virus to other humans through subsequent bites.
- Geographic Distribution: Dengue is endemic in more than 100 countries, primarily in tropical and subtropical regions, affecting urban and semi-urban areas where these mosquitoes thrive [15, 16].
2. Vertical Transmission (Maternal)
- From Mother to Fetus: Pregnant women infected with DENV can transmit the virus to their fetus, especially during the later stages of pregnancy. This can result in preterm birth, low birth weight, and other complications [17].
3. Blood Transfusion and Organ Transplants
- Blood Transfusion: Dengue can be transmitted through bloodtransfusions from infected donors.
- Organ Transplants: Rare cases of transmission through organ transplants have been reported [18].
4. Needle Stick Injuries
- Healthcare Workers: Healthcare workers are at risk of transmission through accidental needle stick injuries, particularly in regions with high dengue prevalence [19].
Pathogenicity of Dengue Virus
1. Initial Infection and Viral Replication
- Entry and Spread: Dengue virus (DENV) is introduced into the human bloodstream through the bite of an infected Aedes mosquito. The virus initially targets dendritic cells and macrophages in the skin, then spreads to lymph nodes and other organs, leading to viremia.
- Immune Response: The body's immune response involves the activation of T-cells, production of antibodies, and release of cytokines. This response helps control the infection but can also contribute to disease severity.
2. Clinical Manifestations
- Dengue Fever: Characterized by high fever, severe headache, retro-orbital pain, joint and muscle pain (often referred to as "breakbone fever"), rash, and mild bleeding (e.g., nose or gum bleeding). Symptoms usually appear 4-10 days after infection and last 2-7 days.
- Severe Dengue (Dengue Hemorrhagic Fever/Dengue Shock Syndrome): In some cases, the disease progresses to severe dengue, which is marked by plasma leakage, fluid accumulation, respiratory distress, severe bleeding, and organ impairment. This can lead to shock (dengue shock syndrome) and can be fatal without proper medical care.
3. Pathogenesis of Severe Dengue
- Antibody-Dependent Enhancement (ADE): A well-documented phenomenon where a person previously infected with one DENV serotype experiences a more severe infection upon subsequent infection with a different serotype. Non-neutralizing antibodies from the first infection facilitate viral entry into host cells, leading to increased viral load and severe disease.
- Cytokine Storm: Severe dengue is associated with an excessive immune response, resulting in the release of large amounts of cytokines. This cytokine storm increases vascular permeability, leading to plasma leakage, fluid accumulation, and shock.
Malaria
• Malaria Life Cycle
The life cycle of malaria involves two hosts: the human and the Anopheles mosquito. The cycle can be divided into several stages:
1. Sporozoite Stage:
- Transmission: Infected Anopheles mosquitoes inject sporozoites into the human bloodstream during a bite.
- Liver Stage: Sporozoites travel to the liver and infect hepatocytes (liver cells), where they mature into schizonts [20].
2. Schizont Stage:
- Liver: Schizonts rupture, releasing merozoites into the bloodstream [21].
. Erythrocytic Cycle:
- Red Blood Cell Invasion: Merozoites invade red blood cells (RBCs) and develop into ring-stage trophozoites.
- Trophozoite Stage: Trophozoites mature into schizonts within RBCs.
- Rupture and Release: Schizonts rupture RBCs, releasing more merozoites to infect additional RBCs, causing cyclic fever episodes [22].
4. Sexual Stage (Gametocytes):
- Some merozoites differentiate into male and female gametocytes, which circulate in the bloodstream.
5. Mosquito Stage:
- Gametocytes: When another mosquito bites the infected human, it ingests gametocytes [23].
- Fertilization: In the mosquito's gut, gametocytes fertilize to form zygotes, which develop into ookinetes.
- Oocyst Formation: Ookinetes penetrate the mosquito gut wall and develop into oocysts.
- Sporozoites: Oocysts mature and release sporozoites, which migrate to the mosquito's salivary glands, ready to infect another human host [24].
Pathogenicity of Malaria
Malaria's pathogenic effects are primarily due to the destruction of red blood cells and the body's immune response to the infection: Red Blood Cell Destruction: *Plasmodium* parasites invade red blood cells (RBCs), leading to their destruction. This destruction is responsible for the characteristic symptoms of malaria, such as fever, chills, and anemia.
Cyclic Fever: The life cycle of *Plasmodium* involves periodic bursts of parasites from RBCs, leading to synchronized fever spikes. This cycle is linked to the release of toxins and the body's immune response.
Immune System Evasion: *Plasmodium* has evolved mechanisms to evade the host's immune system. For instance, it alters the surface proteins of infected RBCs to avoid detection and destruction. Cerebral Malaria: In severe cases, *Plasmodium falciparum* can cause cerebral malaria, where parasites accumulate in the brain's blood vessels, leading to neurological symptoms and potential long-term complications.
Immune Response: The body's immune response to malaria can contribute to pathology. For example, cytokine release can lead to inflammation and contribute to symptoms and disease severity. Anemia: The destruction of RBCs leads to anemia, causing fatigue, weakness, and pallor [25-27].
Dengue virus- malaria co-infection Dengue virus and malaria co-infection is a complex scenario with significant clinical implications due to the overlapping symptoms and potential for severe disease outcomes. Here’s a focused overview of dengue-malaria co-infection:
Clinical Overview
1. Symptom Overlap: Both dengue and malaria can present with high fever, headache, and body aches, making diagnosis challenging. Dengue may also cause rash and hemorrhagic symptoms, while malaria can lead to chills, sweating, and splenomegaly.
2. Complications: Co-infection may increase the risk of severe manifestations:
- Dengue: Malaria can aggravate the risk of bleeding, shock, and other severe forms of dengue.
- Malaria: Dengue might complicate malaria by increasing the risk of severe anemia or organ failure.
3. Diagnosis: Differentiating between the two infections requires:
- Malaria: Blood smears, rapid diagnostic tests, or PCR.
- Dengue: Dengue NS1 antigen test, dengue IgM/IgG antibodies, or PCR.
4. Management: Treatment requires addressing both infections simultaneously:
- Malaria: Treated with antimalarial medications such as artemisinin-based combination therapies (ACTs).
- Dengue: Managed with supportive care, hydration, and monitoring for severe complications. No specific antiviral treatment is available for dengue [28,29].
Previous Studies
Objective: To detect dengue virus IgM (DENV-IgM) antibodies in febrile hospitalized patients in Khartoum State (Sudan). Materials and methods: This was a descriptive, cross-sectional study investigating 90 febrile patients attending some Khartoum State hospitals. The study was conducted during the period from October to December 2019. The enzyme linked immuno-sorbent assay (ELISA) method was used for detection of IgM antibodies of dengue virus in all serum specimens collected.
Results: From the 90 febrile patients investigated, 53 (58.9%) were males and 37 (41.1%) were females. 8 febrile patients (8.9%) were found positive for dengue virus IgM antibodies. The age incidence of all patients ranged between 1-75 years. Conclusion: In this study a low seroprevalence rate (8.9%) of dengue fever virus IgM antibodies was detected. There was a significant correlation between malaria and dengue fever infection [30].
Chapter Three
Methodology
A cross-sectional analytical descriptive observational study was carried out on the target population (Kassala state).
Study Area
Kassala police hospital in Kassala State in Sudan
Study Population
This study will cover 100 patients at Khartoum locality who fulfilled criteria:
Inclusion Criteria
-Males and females who are age group range (20 - 70).
Exlusion Criteria
-People who are more than 70 years less than 20 years.
Sample Size
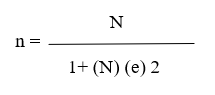
N = total number of population
E = error estimation (usually 0.05)
n = sample size
Due to time factor, any cooperating person in the area of Khartoum, all available data were considered, a sample of 100 was taken.
Sample Frame
A sample of (100) person who are a residence in Kassala police hospital in 2023
Sample Method
Finger prick, whole blood, serum or plasma can be used. In the method 1 drop of patient’s sample is added to the circular well of the cassette using the micro pipette provided. After allowing the sample to absorb completely , 2 drops of buffer reagent are added to the square well. The test results are read after 15 min. A pink line in the IgM and control areas is indicative of primary dengue. A pink line in the IgG and control areas or in the IgG, IgM and control areas is indicative of secondary dengue. A pink line only mean that no IgM, IgG antibody have been detected and the and the reagent have performed satisfactory.
Sample Technique
• Immunochromatographic for Malaria (ICT)
Rapid test is direct binding test to detection of malaria antigen in serum.
Principle
Refers to migration of liquid across the surface of a nitrocellulose membrane.
- Malaria antigen from a lyzed blood sample is reacted with mauve particles.
- the antigen antibody colloidal gold complex migrates along the nitrocellulose membrane where it become bound (captured)by a line of specific monoclonal antibody, produce a pink line in the test result area. This line can be seen after a washing buffer has removed the background haemoglobin.
Procedure
In the method 1 drop of patient’s sample is added to the circular well of the cassette using the micro pipette provided. After allowing the sample to absorb completely, 2 drops of buffer reagent are added to the square well. The test results are read after 15 min.
A pink line in the IgM and control areas is indicative of primary dengue.
A pink line in the IgG and control areas or in the IgG, IgM and control areas is indicative of secondary dengue. A pink line only means that no IgM, IgG antibody have been detected and the and the reagent have performed satisfactory
Interpretation
A pink line in the test without line in control.
Quality Control
ICT control has been done on kits, we make control positive and negative at stac national laboratory (the reference laboratory).
Data CollectionTools
Data was collected anonymously through a self-administered questionnaire, prepared in English, and a blood sample is collected.
Data Analysis
Data entry and analysis will be done using statistical package of social science program SPSS version 25.0 (IBM SPSS Inc. Chicago, IL). Descriptive statistics (i.e., frequencies, percentages, mean, p. value and correlation) will be performed.
Ethical Consideration
Approval was taken from research committee, Al-Madain College for Medical Sciences and Technology.
Participants’ consent will be taken while collecting the data for the study. Participants will be informed about the objective and purpose of the study in the informed consent. And only participant voluntarily willing to take part will be included. No financial benefits will be offered to participants.
Chapter Four
Data Presentation, Analysis Results
From 100 people 93 present with positive dengue fever 93.9% , 6 patients with negative dengue fever 6.1% ,17 patients present with positive malaria 17.2% and 82 patients present with negative malaria 82.8% Population genders: Male represented 54 patients 54.5% Female represented 45 patients 45.5% Total co infection with dengue fever and malaria 15.2% positive and 84.8% negative
Methods
Frequencies and percentages have used to know the statistics with mean and STD. Crosstabulation and correlation was done.
Malaria Among Participants
|
Answer |
Frequency |
Percent |
|
Positive |
17 |
17.2% |
|
Negative |
82 |
82.8% |
|
Total |
99 |
100.0% |
Table No (4.1): Shows the Population’s Malaria Results
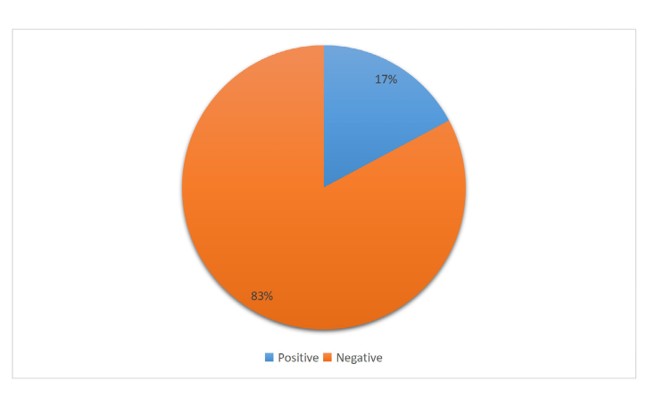
Figure No (4.1): Malaria Results
Dengue Fever Infection Among Participants
|
Answer |
Frequency |
Percent |
|
Positive |
93 |
93.9% |
|
Negative |
6 |
6.1% |
|
Total |
99 |
100.0% |
Table No (4.2): Shows The Population’s Dengue Fever Results
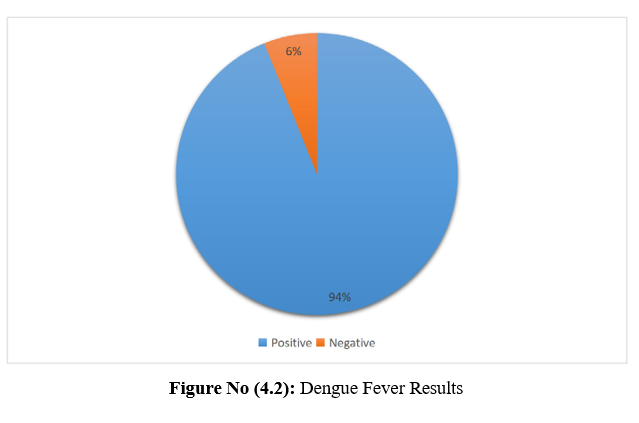
Figure No (4.2): Dengue Fever Results
Cross Tabulation of Malaria and Dengue Fever
|
% within Dengue |
Dengue fever |
Total |
Chi-Square |
P. value |
Sig |
||
|
Positive |
Negative |
|
|
|
|
||
|
Malaria |
Positive |
12.8% |
60.0% |
15.2% |
8.239a |
0.004 |
Sig |
|
Negative |
87.2% |
40.0% |
84.8% |
||||
|
Total |
100.0% |
100.0% |
100.0% |
|
|
|
|
|
*P. value considered significant when < 0.05 |
|||||||
Table No (4.3): Malaria * Dengue Crosstabulation
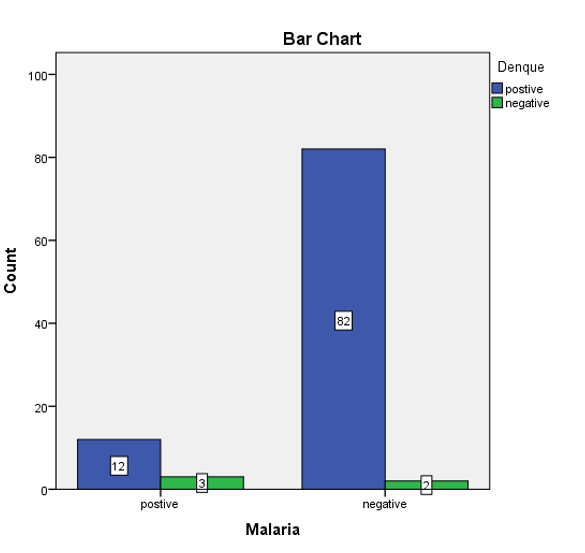
Figure No (4.3): Malaria
Chapter Five
Discussion, Conclusion and Recommendations
Discussion
Malaria and dengue co-infections are considered of great public health importance, especially in the tropics. This condition is usually underreported and could have fatal outcomes if left undiagnosed. However, malaria and dengue co-infections cause febrile illness with similar symptoms that are not easy to differentiate clinically. Therefore, understanding the distribution of malaria and dengue co-infections is vital for improving accurate diagnosis and proper therapeutic interventions. Information provided by this study can be used as guidance for clinicians, policymakers, and public health workers for choosing appropriate diagnostics and treatment in Sudan and similar endemic areas.
Therefore, our study was designed to estimate the estimate the prevalence of dengue fever among malaria patients at Kassala state hospitals among participants a high percent of males (54.5%) age mean (43.6 ± 12.8), Most of them were positive results for dengue fever (93.9%) revealing that less than 20.0% were positive for malaria, in compare with other studies those results were somehow similar the study done in Sudan by Al-Sheikh and Al Fadhil A. Omer (2020) which showed a low seroprevalence rate (8.9%) of dengue fever virus IgM antibodies was detected. There was a significant correlation between malaria and dengue fever infection [30].
If it is inferred that there is a relationship between malaria and dengue fever, we have a morale level of 0.05 and it is inferred that there is a strong relationship between malaria and dengue fever. If you have dengue fever, you have malaria, so malaria has greatly affected dengue fever.
Conclusion
The study concluded that prevalence of malaria and dengue co- infections was estimated high, severity of clinical symptoms ofpatients with malaria and dengue co-infections was observed, and young ages were more affected by the co-infections and that malaria has greatly affected dengue fever.
Recommendations
This finding highlights the necessity of clear understanding of the epidemiology of malaria and dengue co-infections is essential to inform decision-makers to institute an appropriate control strategy for both diseases. More research on malaria and dengue co- infections is needed in other endemic areas of Sudan. In addition, more vector studies are required for a better understanding of their role in transmission of malaria and dengue fever.
Dedication
First of all, we would like to thank Allah, without his guidance we would never have been able to accomplish this. Our great teacher and messenger, Mohammed (May Allah bless and grant him), who taught us the purpose of life. And to our parents who have never failed to give us financial and moral support, for giving all our needs during the time we developed our system and for teaching us that even the largest task can be accomplished if it is done one step as a time. We dedicate this Project to all the people who have worked hard to help us complete this project.
Acknowledgement
Innumerable thanks and gratitude are due to Allah the Almighty Who has given us guidance and inspiration to complete this study and present it to the University Our sincere appreciation, thanks and respect are due to Dr. Zinab Abdellatef Othman Blal, for her valuable and inspiring guidance and supervision.
Thank you to the interviewees, who so generously took time out of their schedules to participate in our research and make this project possible.
Finally, thanks are due to all those who kindly supported, encour- aged and assisted us in any way during the different processes of writing this study.
References
- Baird, J. K. (2009). Resistance to therapies for infection by Plasmodium vivax. Clinical microbiology reviews, 22(3), 508-534.
- Wiwanitkit, V. (2011). Concurrent malaria and dengue infection: a brief summary and comment. Asian Pacific journal of tropical biomedicine, 1(4), 326-327.
- World Health Organization. (2022). Fact sheet about malaria.Geneva, Switzerland. Dec.
- Prevention, C. C. (2020). for DC and. CDC-Malaria-About Malaria-Biology. CDC-Centers Dis Control Prev.
- Monath, T. P., & Vasconcelos, P. F. (2015). Yellow fever. Journal of clinical virology, 64, 160-173.
- Magalhães, B. M., Siqueira, A. M., Alexandre, M. A., Souza,M. S., Gimaque, J. B., Bastos, M. S., ... & Mourão, M. P. (2014). P. vivax malaria and dengue fever co-infection: a cross-sectional study in the Brazilian Amazon. PLoS neglected tropical diseases, 8(10), e3239.
- Abdalla, S. I., Malik, E. M., & Ali, K. M. (2007). The burden of malaria in Sudan: incidence, mortality and disability– adjusted life–years. Malaria journal, 6(1), 97.
- Ahmed, A., Ali, Y., Elmagboul, B., Mohamed, O., Elduma, A., Bashab, H., ... & Higazi, T. (2019). Dengue fever in the Darfur area, Western Sudan. Emerging infectious diseases, 25(11), 2126.
- Hamid, Z., Hamid, T., Alsedig, K., Abdallah, T., Elaagip, A., Ahmed, A., ... & Hamid, M. A. (2019). Molecular investigation of dengue virus serotype 2 circulation in Kassala state, Sudan. Japanese Journal of Infectious Diseases, 72(1), 58-61.
- WHO. (2015). Malaria and VHF outbreak in Darfur, Sudan. Situation Report No 04, 04 November 2015, Federal Ministry of Health, Sudan. World Health Organization, Geneva, Switzerland.
- Bagahizel, A., Elkhatim, S. (2023). Assessment of the readiness and response toward the dengue fever outbreak in Sudan: a qualitative exploration. Population Medicine. 5(Supplement):A976.
- WHO. (2011). Emergency preparedness and humanitarian action (EHA). Sudan, Sudan Health Highlights World Health Organization, Geneva, Switzerland.
- Sudan Demographics 2020 (Population, Age, Sex, Trends)— Worldometer. (2020). (accessed Feb. 13, 2023).
- Schmidt, A. C. (2010). Response to dengue fever–the good,the bad, and the ugly. N Engl J Med, 363(5), 484-487.
- World Health Organization [WHO Dengue Factsheet]
- Centers for Disease Control and Prevention (CDC)
- Guzman, M. G., & Harris, E. (2015). Dengue. The Lancet, 385(9966), 453-465.
- Bhatt, S., Gething, P. W., Brady, O. J., Messina, J. P., Farlow,A. W., Moyes, C. L., ... & Hay, S. I. (2013). The global distribution and burden of dengue. Nature, 496(7446), 504- 507.
- Halstead, S. B. (2007). Dengue. The lancet, 370(9599), 1644-1652.
- Gardner, M. J., Hall, N., Fung, E., White, O., Berriman, M., Hyman, R. W., ... & Barrell, B. (2002). Genome sequence of the human malaria parasite Plasmodium falciparum. Nature, 419(6906), 498-511.
- Chen, Q., Schlichtherle, M., & Wahlgren, M. (2000). Molecular aspects of severe malaria. Clinical microbiology reviews, 13(3), 439-450.
- Cowman, A. F., Berry, D., & Baum, J. (2012). The cellular and molecular basis for malaria parasite invasion of the human red blood cell. Journal of cell Biology, 198(6), 961-971.
- Rts, S. C. T. P. (2015). Efficacy and safety of RTS, S/AS01 malaria vaccine with or without a booster dose in infants and children in Africa: final results of a phase 3, individually randomised, controlled trial. Lancet, 386(9988), 31-45.
- Sinden, R. E., et al. (2012). Developing transmission- blocking strategies for malaria control. PLOS Pathogens, 8(12), e1002982.
- Hemingway, J., Ranson, H., Magill, A., Kolaczinski, J.,Fornadel, C., Gimnig, J., ... & Hamon, N. (2016). Averting a malaria disaster: will insecticide resistance derail malaria control?. The Lancet, 387(10029), 1785-1788.
- Moody, A. (2002). Rapid diagnostic tests for malariaparasites. Clinical microbiology reviews, 15(1), 66-78.
- Johnston, S. P., Pieniazek, N. J., Xayavong, M. V., Slemenda, S. B., Wilkins, P. P., & Da Silva, A. J. (2006). PCR as a confirmatory technique for laboratory diagnosis of malaria. Journal of clinical microbiology, 44(3), 1087-1089.
- Manigauha, A., & Koirala, S. (2022). "Malaria and dengueco-infection: A review." Journal of Vector Borne Diseases, 59(1), 1-9. doi:10.4103/0972-9062.324911.
- Manigauha, A., & Koirala, S. (2022). "Malaria and dengue co-infection: A review." *Journal of Vector Borne Diseases*, 59(1), 1-9. doi:10.4103/0972-9062.324911.
- Sheikh, A. I., Omer, A. I. F. A. (2020). Detection of Dengue Virus IgM Antibodies in Febrile Hospitalized Patients in Khartoum State (Sudan) S Manal, African Journal of Medical Sciences 5 (3).
Appendix I

Al-Madain College for Medical Sciences and Technology Medical laboratory sciences
Questionnaire
1. Gender:
Male ( ) Female ( )
2. Age in years: ……………….
3. Malaria infection?
Positive ( ) Negative ( )
4. Dengue fever infection?
Positive ( ) Negative ( )



