Research Article - (2025) Volume 9, Issue 2
Prevalence of Antibiotic Prescription in the End-Of-Life Care of Hospitalised Geriatric Patients Presented Fever or Hypothermia Running Title: Antibiotic Prescription in the End-Of-Life Care
2Université de Limoges, Laboratoire Vie-Santé, UR 24 134, Institut OmegaHealth, Faculté de Médecine, 87025 Limoges, France
3CHU de Limoges, Service d’Accompagnement et de Soins Palliatifs, F-87042, Limoges, 2 Avenue Martin-Luther King, France
4Service des maladies infectieuses et tropicales, CHU de Limoges, 87042 Limoges cedex, INSERM, U1094, Institut d’Epidémiologie et de Neurologie Tropicale, GEIST, Univ. Limoges, CHU Limoges, Limoges, France
Received Date: May 16, 2025 / Accepted Date: Jun 25, 2025 / Published Date: Jul 08, 2025
Copyright: ©2025 Professor Achille TCHALLA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Festou, B., Quesnel, P. A., Ndiaye, A., Faucher, J. F., Tchalla, A. (2025). Prevalence of Antibiotic Prescription in the End-Of-Life Care of Hospitalised Geriatric Patients Presented Fever or Hypothermia. Archives of Infect Diseases & Therapy, 9(2), 01-08.
Abstract
Currently, population aging is a public health issue. End-of-life situations of older patients in hospital are increasingly frequent and raise therapeutic and ethical questions. Patients at the end of their lives, particularly older patients, are more often exposed to infections. The place of antibiotic therapy is central in terminal palliative care. Nevertheless, the use of antibiotics should always be assessed, especially because of the development of bacterial resistance. Therefore, this study assessed the prevalence of antibiotic use in older people at the end of life. This descriptive retrospective study included all patients aged 75 years and older, hospitalised in 2018 at the Limoges University Hospital Centre and at the hospital in Saint-Yrieix la Perche who died in hospital. The primary outcome was the use of an antibiotic in the last 10 days of life. Of the 338 patients included, 237 (70.1%) were treated with an antibiotic and 233 (68.9%) experienced hyperthermia > 38.3°C or hypothermia < 35°C. The most frequent infections were pulmonary (n = 141, 43.3%) and urinary tract (n = 67, 20.1%). Of the patients, 23.3% did not have an established infection diagnosis. Samples were taken from 29% of the patients. Escherichia coli and Staphylococcus aureus were the bacteria most often isolated. Of the patients, 54.4% (n = 129) died while taking antibiotics. For 25.7% (n = 61) of the patients, a doctor decided to stop treatment early. Many older patients at the end of their lives are concerned about antibiotic therapy. In the absence of scientific proof of the effectiveness of antibiotics to relieve patients at the end of life, these treatments should be studied to develop guidelines about their use in terminal palliative care.
Keywords
Antibiotics, End-of-life, Geriatric patients, Prevalence
Abbreviations
3CGs: third-Generation Cephalosporins;
CBES: Cytobacteriological Examination of the sputum;
CBEU: Cytobacteriological Examination of the Urine;
ERENA: Espace de Réflexion Ethique Nouvelle-Aquitaine;
GIM: Geriatric Internal Medicine;
GR: Geriatric Rehabililtation;
GM: General Medicine;
PCU: Palliative Care Unit;
PEG: Post-Emergency Geriatric;
SD: Standard Deviation;
UHC: University Hospital Center
Introduction
All the projections of ageing show an increase in life expectancy in the general population, along with an increase in the proportion of people over 75 [1,2]. Ageing is a major public health issue because of the care necessary for frail people, but also an economic issue, especially with regard to the cost of dependency [3-5]. Older patients are exposed to more bacterial infections because of immunosenescence and the many geriatric syndromes or clinical situations encountered in older patients (bladder catheters, prostheses, and other medical devices) [6-8].
In recent years, the consumption of antibiotics has increased in France; worldwide it increased by 65% between 2000 and 2015 in a study conducted in 76 countries [9,10]. Numerous studies have shown increases in bacterial resistance. The sensitivity of the bacteria most often encountered in geriatric settings to the most frequently used antibiotics is tending to decrease, for example Escherichia coli and quinolones or third-generation cephalosporins (3GCs) [11].
Antibiotics are frequently used in 27% to 97.5% of patients in palliative care, depending on the study [12], especially at the end of life. Patients in terminal palliative care are exposed to infections, most often pulmonary or urinary [13-19]. Treatments using 3GCs, amoxicillin + clavulanic acid, or quinolones are threatened by bacterial resistance [11,17,20,21].
Numerous studies have proven the curative effectiveness of antibiotic therapy. To our knowledge, however, its effectiveness on patient comfort at the end of life has not been demonstrated. Indeed, the side effects may be preponderant [15,16,22-24]. Very few studies have examined the use of antibiotic therapy in end- stage palliative care, and even fewer in a geriatric setting, with most of them having been conducted in the palliative phase of neoplasia.
Therefore, our study assessed the prevalence and conditions of the use of antibiotics at the end of life in hospitalised older patients
Material and Method
Design and Settings
This retrospective multicentre observational study included patients from the Dupuytren University Hospital Centre in Limoges and the Jacques Boutard Hospital in Saint-Yrieix la Perche.
Study Objectives
The main study objective was to assess the prevalence of antibiotic use in the last 10 days of life of hospitalised older patients. Secondary objectives were to describe the conditions of antibiotics use at the end of life (context, infection site, and method of treatment) and to analyse doctors’ decisions to continue the treatment.
Patients
The medical records of all patients hospitalised between January 1 and December 31, 2018, who died during their hospitalisation were studied. Patients were hospitalised in the Post-Emergency Geriatric (PEG), Geriatric Internal Medicine (GIM), Geriatric Rehabilitation (GR), or General Medicine (GM) departments or the Palliative Care Unit (PCU).
Inclusion criteria were an age of 75 years or older, hospitalisation longer than 72h, a body temperature > 38.3°C or < 35°C or a suspected or confirmed bacterial infection in the last 10 days of life. Patients who died outside of hospital or presented with a non- bacterial infection were excluded.
Outcomes
The main outcome measure was the prescription of antibiotics for cure and not for prophylaxis during the last 10 days of life. Secondary endpoints were the presence of fever, samples collected, infection site, bacteria isolated, antibiotic used, route of administration, duration of antibiotic therapy, early stoppage of the antibiotic therapy, number of days between stopping antibiotic therapy and death, end of antibiotic therapy before death, or death before the end of antibiotic therapy.
Data Collection
The hospital centre and the department in which the patient died, length of hospitalisation, and patient age, sex, and history were extracted from the patients' computerised medical records.
Statistical Analysis
The prevalence of end-of-life antibiotic use in the geriatric population, proportion of sites infected, premature stoppage of antibiotics, and average duration of antibiotic therapy were calculated. Qualitative variables are described as numbers and percentages. Quantitative variables are described as the median, mean and standard deviation. All data were analysed using Microsoft Excel.
Ethics
This study has been approved the ethics committee of Limoges University Hospital Center.
Results
Demographic Data
Of the 637 patients over 75 who died between January 1, 2018, and January 31, 2019, 21 were admitted to hospital in 2017 and 38 in 2018. Among the remaining 578, 138 were hospitalised for less than 72 hours, 100 did not have hyperthermia, hypothermia or infection, and two had non-bacterial infections. In all, 338 (58.5%) patients were included (Fig 1). Table 1 summarises the characteristics of the included patients.
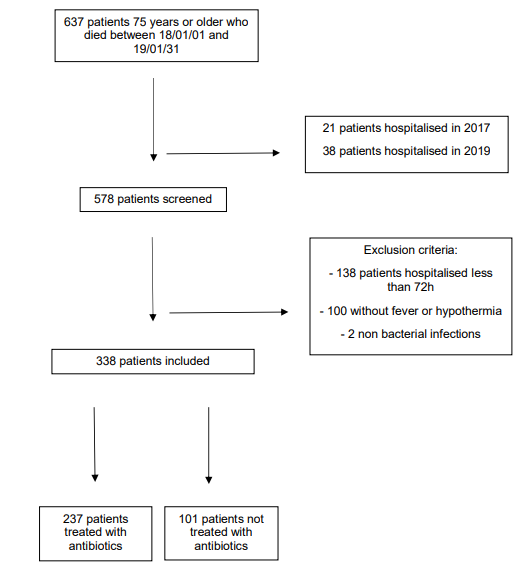
Figure 1: The APALIM Study Flow Chart
|
Variables |
Total1
n = 338 (%) |
Patients treated1
n = 237 (%) |
Patients not treated1
n = 101 (%) |
p value² |
|
Males |
177 (52.4) |
131 (55) |
46 (46) |
0.12 |
|
Mean age (SD) years |
87 (6.2) |
88 (6.2) |
86 (6) |
0.017 |
|
Site of hospitalisation |
|
|
|
0.011 |
|
Patients from UHC |
226 (66.9) |
137 (58) |
58 (58) |
|
|
Patients from Saint-Yrieix la Perche |
112 (33.1) |
100 (42) |
43 (43) |
|
|
Department |
|
|
|
< 0.001 |
|
Hospitalisation in acute unit (PEG + GIM + GM) |
163 (48) |
113 (48) |
49 (49) |
|
|
Hospitalisation in GR |
111 (32.8) |
88 (37) |
24 (24) |
|
|
Hospitalisation in PCU |
64 (18.9) |
36 (15) |
28 (28) |
|
|
Duration of hospitalisation |
|
|
|
0.8 |
|
Duration of hospitalisation, mean (SD), in days |
14 (11) |
14 (9.6) |
15 (13,2) |
|
|
Duration of hospitalisation, median (SD), in days |
12 (11) |
12 (9.6) |
11.5 (13.2) |
|
|
Neurocognitive impairment |
136 (40.2) |
98 (41) |
39 (39) |
0.7 |
|
Falls |
129 (38.2) |
97 (41) |
35 (35) |
0.3 |
|
Hypertension |
221 (65.4) |
161 (68) |
60 (60) |
0.2 |
|
Ischemic heart disease |
69 (20.4) |
55 (23) |
17 (17) |
0.2 |
|
Atrial fibrillation |
125 (37) |
87 (37) |
37 (37) |
> 0.9 |
|
Heart failure |
125 (37) |
114 (48) |
27 (27) |
< 0.001 |
|
Stroke |
101 (29.9) |
71 (30) |
35 (35) |
0.4 |
|
Diabetes |
78 (23.1) |
64 (27) |
16 (16) |
0.03 |
|
Obliterative arteriopathy of the lower limbs |
36 (10.7) |
30 (13) |
8 (8) |
0.2 |
|
Active cancer |
75 (22.2) |
43 (18) |
38 (38) |
< 0.001 |
|
Active haemopathy |
36 (10.7) |
24 (10) |
13 (13) |
0.4 |
|
Chronic obstructive pulmonary disease |
45 (13.3) |
37 (16) |
8 (8) |
0.061 |
|
Anxio-depressive syndrome |
130 (38.5) |
88 (37) |
42 (42) |
0.4 |
|
Chronic renal failure |
56 (16.8) |
48 (20) |
8 (8) |
0.006 |
|
Pace-Maker |
37 (20) |
30 (13) |
7 (7) |
0.13 |
|
Orthopaedic material |
79 (23.4) |
56 (24) |
23 (23) |
> 0.9 |
|
Mean Charlson Comorbidity Index |
7.2 |
7.2 |
7.2 |
> 0.9 |
|
SD, Standard Deviation; UHC, University Hospital Centre; PEG, Post-Emergency Geriatric department; GIM, Geriatric Internal Medicine department; GR, Geriatric Rehabilitation department; GM, General Medicine department; PCU, |
||||

Table 1: Patient Characteristics According to the Antibiotic Therapy Group.
Patients hospitalised in acute care units were treated significantly more often than patients in the PCU. Patients with diabetes, heart failure, chronic renal failure, or active cancer were also treated significantly more often. 3.2. Primary Outcome: Use of Antibiotics Of the 338 patients included, 237 received antibiotic therapy in the last 10 days of their lives. This was 70.1% of the total population studied. Patients hospitalised in palliative care were treated less often with antibiotics: 56.3% versus 69.9% in acute geriatric departments and 79.3% in geriatric rehabilitation. Patients with fewer comorbidities were treated more often (Fig 2).
Figure 2: Prevalence of Antibiotic Prescriptions According to Comorbidities
Secondary Outcomes
Overall, 233 patients (68.9%) had a temperature above 38.3°C or hypothermia below 35°C. Of these, 141 (61%) were treated with antibiotics. In comparison, 105 patients had no fever and 96 were treated (91%). Of the 338 patients included, the fever was considered to be of neoplastic origin in 12 (3.6%). For the 326 others, the most frequent infection was pulmonary infection (141 cases, 43.3%) followed by urinary tract infections (67 cases, 20.1%). In 76 patients (23.3%), no infection was diagnosed, but 19.7% were treated with an antibiotic (Table 2).
|
Site of infection |
n = 326 (%) |
|
Pulmonary tract |
141 (43.3) |
|
Urinary tract |
67 (20.1) |
|
Digestive tract |
22 (6.7) |
|
Cutaneous |
20 (6.1) |
|
Bones |
8 (2.5) |
|
Endocarditis |
5 (1.5) |
|
Oral |
5 (1.5) |
|
Unknown |
76 (23.3) |
|
Samples |
n = 287 (%) |
|
Number of patients with samples |
202 |
|
Number of positive samples |
112 (39) |
|
Blood cultures |
140 (41.4) |
|
CBEU |
116 (34.3) |
|
Stool cultures |
16 (4.7) |
|
Puncture or swab |
11 (3.3) |
|
CBES |
5(1.5) |
|
Bacteria isolated |
n = 112 (%) |
|
Escherichia coli |
38 (33.9) |
|
Staphylococcus aureus |
22 (19.6) |
|
Klebsiella pneumonia |
8 (7.1) |
|
Pseudomonas aeruginosa |
7 (6.3) |
|
Enterococcus faecalis |
7 (6.3) |
|
Clostridium dificile |
5 (4.5) |
|
Proteus mirabilis |
4 (3.6) |
|
Others |
21 (18.2) |
|
Antibiotics prescribed |
n = 237 (%) |
|
3GC |
117 (49.4) |
|
Amoxicillin + clavulanate |
92 (38.9) |
|
Metronidazole |
48 (20.3) |
|
Quinolones |
43 (18.1) |
|
Amoxicillin |
13 (5.5) |
|
Piperacillin + Tazobactam |
7 (3) |
|
Aminoglycoside |
7 (3) |
|
Vancomycin |
7 (3) |
|
Trimethoprim + Sulfamethoxazole |
6 (2.5) |
|
Rifampicin |
6 (2.5) |
|
Penicillin M |
4 (1.2) |
|
Carbapenem |
2 (0.8) |
|
Others |
18 (7.6) |
|
CBEU, cytobacteriological examination of the urine; CBES, cytobacteriological examination of the sputum; 3GC: third-generation cephalosporin |
|
Table 2: Type and Management of Infections
Blood was cultured in 140 patients (41.4%); cytobacteriological examination of the urine (CBEU) was performed in 116 (34.3%); 16 (4.7%) stool cultures were performed; 11 patients (3.3%) had a puncture or swab; and 5 (1.5%) had cytobacteriological examinations of the sputum (CBES).
A total of 112 different bacteria were isolated from 98 patients (29%). The most prevalent bacterium was E. coli (38 cases; 33.9% of all bacteria isolated), followed by Staphylococcus aureus (22 strains isolated, 19.6%) (Table 2).
The most widely used antibiotics were 3GCs (117 patients, 49.4% of patients treated), followed by amoxicillin + clavulanate (92 patients, 38.9%), metronidazole (48 patients, 20.3%), and quinolones (43 patients, 18.1%). Other antibiotics were rarely used (Table 2).
To administer these antibiotics, the intravenous route was preferred (190 patients, 80.2%), followed by oral (69 patients, 29.1%) and subcutaneous (40 patients, 16.9%) routes.
The average duration of antibiotic treatment was 7.4±5.9 (median 6) days.
The antibiotic therapy was completed in 46 (19.4%) patients several days before they died, while 129 (54.4%) patients died while taking antibiotics, of whom 39 (30.2%) had been treated for less than 72 hours. For 61(25.7%) patients, a doctor decided to stop the antibiotic treatment early, on average 3.9±2.1 days before death (Table 3).'
|
Variables |
|
|
Duration of antibiotic treatment, mean (SD) days |
7.4 (5.9) |
|
Completed antibiotic therapy before dying, n (%) |
46 (19.4) |
|
Patients died while taking antibiotics, n (%) |
129 (54.4) |
|
Patients died after taking antibiotics for less than 72 h, n (%) |
39 (30.2) |
|
Early suspension of treatment before death, n (%) |
61 (25.7) |
Table 3: Clinical Outcomes Related to Antibiotics.
Discussion
In the last 10 days of their lives, 338 patients had a suspected infection or hyper/hypothermia. This represents 58.5% of the patients hospitalised in 2018 who died in hospital. A large majority of our patients were treated with antibiotics. Patients hospitalised in acute care units were treated more often than patients hospitalised in the PCU, probably because the initial goal was to cure the infection before a decision to limit care was made.
Concerning comorbidities, patients with diabetes or heart failure were treated more often. We hypothesised that physicians preferred to treat because decompensation could lead to discomfort, such as pulmonary oedema. Patients with cancer were also treated more often, possibly because they are at risk of more severe infections and more discomfort. Perhaps they were also readily treated in cases of suspected infections because of their frailty.
Patients with a Charlson Comorbidity Index ≤ 8 were treated more often. We postulate that physicians gave antibiotics to fewer frail patients because they had more serious conditions at the occurrence of infection.
Unless older patients may not have fever even in case of infection, we decided to include only patients with hyperthermia or hypothermia. Effectively, some patients could be overtreated by antibiotics based only on biological criterias like C-Reactive Protein (CRP) or leucocytosis which are unspecifics of infection. This choice has been made to limit the risk of inclusion bias.
Regarding infected sites, our results are consistent with previous studies [17,19,25]. Pulmonary and urinary tract infections were encountered most frequently. Of the patients, 23.3% had inaccurate infection diagnoses, due to the absence of clear clinical symptoms and unclear information in the medical record.
Samples were collected from less than half the patients, which may be explained by an undetected fever or maintaining the patients’ comfort at the end of life with fewer injections. Urine samples are difficult to collect in patients already probed or incontinent.
Bacteria were isolated in a minority of patients, due to the small numbers of samples collected. Escherichia coli, especially in urinary tract infections, and Staphylococcus aureus were isolated the most frequently. These results are comparable with previous studies [16,19,25].
The antibiotics used preferentially were third-generationcephalosporins (± metronidazole), amoxicillin + clavulanate, and quinolones. This is consistent with the most common types of infection: pulmonary and urinary tract infections.
Many patients died while taking antibiotics (54.4%). A majority had been treated for more than 72 hours. Thus, in almost two thirds of cases, the doctor decided to maintain the antibiotic therapy despite the lack of improvement in the patient with treatment. For 30.2% of the patients, the treatment was not re-evaluated because the patient died within 72 hours of initiating the treatment. Doctors decided to stop treatment early in 25.7% of the patients treated. These results show that the trend is to maintain antibiotic treatment at the end of life, although practices remain heterogeneous.
Lastly, patients are frequently concerned about antibiotic therapy in their last days of life, but only 19.4% of them completed their treatment before dying. Our results are consistent with previous studies in other countries and confirm the need to conduct other studies to make recommendations about this subject.
Our study has many strengths. It is one of the few geriatric studies dealing with antibiotic use and end-of-life infection. To our knowledge, it is also the only French study on the subject. Much relevant data were collected because the study was multicentric. Systematic reading of the medical records enabled us to collect interesting data on the use of antibiotic therapy at the end of life in the geriatric population.
However, the retrospective nature of the study is a weakness. There is information bias because data may be missing from the medical records. There may also be confusion bias with overestimation of the number of urinary tract infections. The CBEU frequently identifies asymptomatic bacteriuria, and antibiotics may be initiated without obvious symptoms explaining the fever. Likewise, treatment for pneumonia may be initiated in a patient with bronchial congestion without other clinical signs. These observations may reflect the over-prescription of antibiotics because the congestion may be linked to cardiac decompensation or to salivary stagnation. In addition, infections may be diagnosed wrongly based only on hyperthermia in patients with cancer or active haemopathy.
For some, the question of antibiotic treatment at the end of life is an integral part of palliative care, although there is no evidence of their effectiveness. Studies should establish the usefulness of antibiotics with regard to infected patients’ comfort during their last days of life.
Conclusion
The use of antibiotics at the end of life in older patients is very common in hospitals. In view of the lack of scientific evidence about the effectiveness of antibiotic therapy on patient comfort at the end of life, prospective studies must be conducted to propose recommendations for the management of infections in this context
Authors Contribution
BF collected all data’s and drafted the initial manuscript. PAQ, AN, JFF and AT made substantive intellectual contributions to the conception and design of the study as well as interpretation of data. BF and AT were involved in critically reviewing and revising the manuscript. All authors read and approved the final manuscript and agreed to be accountable for all aspects of the work.
References
- Population projections to 2070.
- Robert-Bobée, I. (2006). Projections de population pour la France métropolitaine à l’horizon 2050. INSEE première, 1089(4).
- Analyse de l’activité hospitalière. (2017).
- Heam, J. C., Mikou, M., & Ferreti, C. (2018). Les dépenses de santé en 2018: Résultats des comptes de la santé.
- Roussel, R. (2017). Personnes âgées dépendantes: les dépenses de prise en charge pourraient doubler en part de PIB d’ici à 2060. Études et résultats, 1032.
- Selton-Suty, C., Célard, M., Le Moing, V., Doco-Lecompte, T., Chirouze, C., Iung, B., ... & Hoen, B. (2012). Preeminence of Staphylococcus aureus in infective endocarditis: a 1-year population-based survey. Clinical infectious diseases, 54(9), 1230-1239.
- Rapport d’activité. (2018). Epidémiologie. (2017). CNRP.
- Lucado, J., Gould, C., & Elixhauser, A. (2012). Clostridiumdifficile infections (CDI) in hospital stays, 2009.
- La consommation d’antibio en france en 2016.pdf.
- Klein, E. Y., Van Boeckel, T. P., Martinez, E. M., Pant, S.,Gandra, S., Levin, S. A., ... & Laxminarayan, R. (2018). Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proceedings of the National Academy of Sciences, 115(15), E3463-E3470.
- Rapport d’activité de l’ONERBA, 2017.pdf.
- Marra, A. R., Puig-Asensio, M., Balkenende, E., Livorsi, D. J., Goto, M., & Perencevich, E. N. (2021). Antibiotic use during end-of-life care: a systematic literature review and meta- analysis. Infection Control & Hospital Epidemiology, 42(5), 523-529.
- Albrecht, J. S., McGregor, J. C., Fromme, E. K., Bearden, D. T., & Furuno, J. P. (2013). A nationwide analysis of antibiotic use in hospice care in the final week of life. Journal of painand symptom management, 46(4), 483-490.
- Lam, P. T., Chan, K. S., Tse, C. Y., & Leung, M. W. (2005).Retrospective analysis of antibiotic use and survival in advanced cancer patients with infections. Journal of pain andsymptom management, 30(6), 536-543.
- Oh, D. Y., Kim, J. H., Kim, D. W., Im, S. A., Kim, T. Y., Heo,D. S., ... & Kim, N. K. (2006). Antibiotic use during the last days of life in cancer patients. European journal of cancer care, 15(1), 74-79.
- Rosenberg, J. H., Albrecht, J. S., Fromme, E. K., Noble,B. N., McGregor, J. C., Comer, A. C., & Furuno, J. P. (2013). Antimicrobial use for symptom management in patients receiving hospice and palliative care: a systematic review. Journal of palliative medicine, 16(12), 1568-1574.
- Nakagawa, S., Toya, Y., Okamoto, Y., Tsuneto, S., Goya, S., Tanimukai, H., ... & Uejima, E. (2010). Can anti-infective drugs improve the infection-related symptoms of patients with cancer during the terminal stages of their lives?. Journal of palliative medicine, 13(5), 535-540.
- Ahronheim, J. C., Morrison, R. S., Baskin, S. A., Morris, J., & Meier, D. E. (1996). Treatment of the dying in the acute care hospital: advanced dementia and metastatic cancer. Archives of Internal Medicine, 156(18), 2094-2100.
- Nagy-Agren, S., & Haley, H. B. (2002). Management of infections in palliative care patients with advanced cancer. Journal of pain and symptom management, 24(1), 64- 70.
- D’Agata, E., & Mitchell, S. L. (2008). Patterns of antimicrobial use among nursing home residents with advanced dementia. Archives of Internal Medicine, 168(4), 357-362.
- Thompson, A. J., Silveira, M. J., Vitale, C. A., & Malani, P. N. (2012). Antimicrobial use at the end of life among hospitalized patients with advanced cancer. American Journal of Hospice and Palliative Medicine®, 29(8), 599-603.
- Reinbolt, R. E., Shenk, A. M., White, P. H., & Navari, R. M. (2005). Symptomatic treatment of infections in patients with advanced cancer receiving hospice care. Journal of pain and symptom management, 30(2), 175-182.
- Clayton, J., Fardell, B., Hutton-Potts, J., Webb, D., & Chye, R. (2003). Parenteral antibiotics in a palliative care unit: prospective analysis of current practice. Palliative medicine, 17(1), 44-48.
- Stiel, S., Krumm, N., Pestinger, M., Lindena, G., Nauck, F., Ostgathe, C., ... & Elsner, F. (2012). Antibiotics in palliative medicine—results from a prospective epidemiological investigation from the HOPE survey. Supportive care in cancer, 20, 325-333.
- Vitetta, L., Kenner, D., & Sali, A. (2000). Bacterial infections in terminally ill hospice patients. Journal of pain and symptom management, 20(5), 326-334.
- Dyer, J., Vaux, L., Broom, A., & Broom, J. (2019). Antimicrobial use in patients at the end of life in an Australian hospital. Infection, Disease & Health, 24(2), 92-97.



