Research Article - (2022) Volume 3, Issue 2
Prevalence and Associated Risk Factors of Ascaris in Poultry in and Around Bahir Dar Zuria District; Northwest Ethiopia
2Departments of Veterinary Science, School of Animal Science and Veterinary Medicine,BahirDar Univers, Ethiopia
Received Date: Jan 31, 2022 / Accepted Date: Feb 07, 2022 / Published Date: Jun 14, 2022
Copyright: ©Copyright: ©2022 Birhan Agmas. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Birhan Agmas, Shewatatek Melaku. (2022). Prevalence and Associated Risk Factors of Ascaris in Poultry in and Around Bahir Dar Zuria District, Northwest Ethiopia. J Vet Heal Sci, 3(2), 173-177.
Abstract
A cross sectional study was conducted from April 2019 to June 2019 to investigate ascaris in chickens in and around Bahir Dar Zuria District, Northwest Ethiopia. A total of 351 chickens comprising of 168 exotic and 182 local were examined for ascaris infections. Out of these, 55 (15.67%) were found to be infected with ascaris. There was a statistically significant difference (p<0.05) in the prevalence between breeds of chickens in which higher infection rate was observed in local breed chickens (22.53%) than exotic breeds (8.88%). In addition, there was higher statistically significant difference (p<0.05) in the prevalence between body condition of chicken. Among this group poor body conditions chicken was higher (50%) than good body condition (4.56%). In addition, there was a statistically significant association (p<0.05) in prevalence between the different management systems where there was higher infection rate was recorded in extensive farming system (21.46%) compared to semi intensive (6.82%). There was also higher statistically significant difference among hygienic practice of chicken house in which the prevalence of the infection was 39.05% and 6.12% in poor and good hygiene. There was also high statistically significant difference among availability of dewormer, in which the prevalence of infection was 15.76% and 0% in non-available and available of dewormers. In conclusion the management system should be improved especially the housing and hygienic practice. Veterinarian think about deworming of parasites when supervise poultry farms to enhance productivity in addition to antibiotics. Since there are lack of access for dewormer for smallholder farmers veterinarian should be search alternate; easily accessible and affordable drug for poultry internal parasite.
Keywords
Ascaris, Chicken, Prevalence, Risk Factor
Introduction
Poultry are domesticated birds kept by human for the purpose of obtaining meat, eggs and sometimes feathers. They include birds like chicken, duck, goose and turkey. Poultry are kept in backyards or commercial production systems in most areas of the world. It is one of the most important sources of protein and farm manure (Jegede et al., 2015).The most commonly kept poultry are chickens (Gallus spp.), ducks (Carina spp.) Geese (Anser spp.) and turkeys (meleagris spp.). In Ethiopia among these domestic chickens (Gallus domesticus) are the most im¬portant and clearly domesticated. In this country, approximately 99% of birds is raised under the traditional back yard system [1].
Total chicken population in Ethiopia is estimated to be 56.5 million with native chicken representing 96.9%, hybrid chicken 0.54% and exotic breeds 2.56% [2]. Major cause of mortality in scavenging chickens kept under traditional system of manage¬ment in Ethiopia includes viral, protozoan and bacterial disease. However, the traditional poultry production system is character¬ized by low input, low output and periodic destruction of a large portion of the flock due to gastro-intestinal parasites.
Domestic birds are highly susceptible to infection with large number of internal parasites specially helminthes one. In heavily parasitized young birds, the common manifestation are stunted growth, emaciation, weakness and death in young, while in lay¬ing hens the egg production was lowered or entirely stopped [3]. Ascaridia galli was the great cause of losses due to reduction in weight of chicken [4].
Keeping poultry for commercial use has increased from time to time. But due to poor management practices, poor sanitation, less biosecurity measures, miss disinfection procedure, and poor awareness of disease decrease the production system [5]. There is starting of using antibiotics with commercial poultry farming in Ethiopia but they do not consider dew arming. Among inter¬nal parasitic poultry disease one is ascaris; that considered being the most important problem of local chicken as major causes of ill, loss of productivity and indicator of parasite infestation [6]. Prevalence of ascaris parasite in poultry farm became higher. For example 64% in Denmark, 67-88% in Germany, 84% in En¬gland and 97% in Netherlands [7-10].In Africa also increase the prevalence of infection for example 48.4% from Nigeria, 95.2% of from Zambia and 25.63% in Kenya [11-13]. Ascaris galli also reported from different parts of Ethiopia for example 35.6% from central Ethiopia [1].
But the prevalence of ascaris not estimated in our study area. In our preliminary survey, there was no deworming activity per¬formed in commercial and back yard poultry farming despite production loss due to parasite is high. Hence, surveying prev¬alence of ascaris and monitoring of the risk factors in poultry is of paramount importance in order to provide useful data regard¬ing to the parasite status in and around Bahir Dar Zuria district. This work result helps to plan the right management methods against poultry parasite risks. Detailed knowledge on the bur¬den and management practice is required to apply prevention management methods against poultry ascaris. Besides, this study can serve as baseline information for future work of interven¬tion measures on poultry ascaris. Therefore, the objective of this study was to assess the prevalence of ascaris and its associated risk factors in poultry in and around Bahir Dar Zuria Distric, Northwest Ethiopia
Material and Method
Description of Study Area
The study was conducted in Bahir Dar zuria Distrct North west Ethiopia. Bahir Dar is the capital city of Amhara regional state that is found 565 km distance from the capital city of Ethiopia (Addis Ababa). Bahir Dar zuria Distrct is geographically locat-ed at about 110, 37, N latitude and 370,28E longitude at an alti¬tude of 1912 m.a.s.l. The annual temperature is 290c and annual rainfall ranges from 1428-1521mm (CAES, 2015). The sampled peasant associations were tenta peasant association, Sebatamit peasant assciation, robit Peasant association and woreb Peasant association. Out of the total population 1086 people are partic¬ipate in poultry production (1025 male and 61 female). 70% of the land is covered by plain platues, various bush types, low weeds mainly evergreen landmasses planted with major agricul¬tural products on some semi humid highlands. Goats, equines, and poultry (Amhara Regional Administration Livestock Devel¬opment Agency records, 2017). The total population of poultry in Zenzelma are 4345, from this, 1320 are exotic and 3025 are local breeds.
Study Population and Study Design
The study population was all-exotic and local breed poultry found in and around Zenzeima. The study units were those poul-try that included in the study by chance and the investigator sam¬pled them.
Community based cross sectional quantitative study design was conducted in poultries to estimate the prevalence and risk factors of poultry ascaris in Bahir Dar zuria District from January 2019 to June 2019.
Sample Size Determination and Sampling Method
Sample Size Determination
Sample size for the study was calculated using the formula given by Thrusfield (2005) with precision level of 5%, confidence in- terval of 95% and considering of previous study of 35.6% from central Ethiopia (Ashenafi and Eshetu, 2004).
n=z2×P exp (1-_Pexp)
d2
Where; n=sample size, P exp=expected prevalence, d=absolute precision, z=1.96
n= (1.96)2*0.356(1-0.356)
(0.05)2
n = 351
Over all of 351 chickens were sampled.
Sampling Method
Systematic random sampling method was implemented for sam¬pling of chicken. Close-ended questioner was used to identify the managmental-associated risk factors. Fecal samples were collected per cloaca from selected study units. The information regards to age, sex, breed, body condition and different manage¬ment systems was recorded.
Sample Collection and Coprological Examination
One hundred twenty fecal samples comprising of 44 local and 76 exotic breeds of chickens were collected per cloaca. All sam¬ples were put in clean sample bottles containing 10% formalin as preservative and identified appropriately. The sample was processed in Bahir Dar university; Veterinary Parasitology Lab¬oratory. Laboratory examination was also involved faecal exam¬ination in household chickens. The samples were later processed in the veterinary Parasitology laboratory using the salt floatation technique (Kassa, 2005). Identification of ascaris galli eggs was done using a standard microscope under ×10 objective magnifi¬cation (fig.
It is the largest nematode in birds, with females measuring 72 to 112 mm long (Figure 2.1.). The body is semitransparent, creamy-white, and cylindrical. The anterior end is characterized by a prominent mouth, which is surrounded by three large, tri-lobed lips. The edges of the lips bear teeth-like denticles. Where¬as males are relatively shorter and smaller (measuring 50 to 76 mm long), with a distinct pointed and curved tail (Ashour, 1994). The body is entirely covered with a thick proteinaceous struc¬ture called cuticle. The cuticle is striated transversely through the length of the body and cuticularalae are poorly developed. Two conspicuous papillae are situated on the dorsal lip and one on each of the sub lips (Lalchhandama et al., 2009).

Figure 2.1a: Adult A.galli (Tarbiat et al., 2015).
Ascaridia eggs are ovoid in shape, about 50x80 micrometers, and have thick, albuminous shells (Tarbiet et al., 2015).
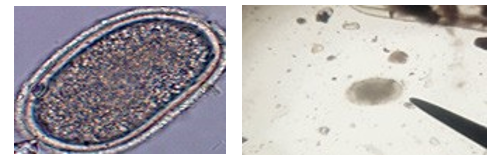
Figure 2.1b: Eggs of Ascaris galli
Data Analysis and Management
The information obtained from laboratory test and observation was entered on the spreadsheet of Microsoft excel work sheet 2007and STATA version 12 was used to coded and analyzed the sampled data. .Chi-square test was used to asses weather there is a statistical significant difference in ascaris galli infection be¬tween host and management factors. A statistically significant association between variables was considered to exist if the cal¬culated p-value is less than 0.05 with 95% confidence level.
Result and Discussion
Out of 351 examined chicken 55 of them were found to be pos¬itive. The overall prevalence ascaris in poultry in our study area was 15.67%.
Table 3.1: Prevalence of Ascaris In Poultry In Relation To Host Factors In Bahir Dar Zuria District, 2019.
|
Variables |
Categories |
No-examined |
No-positive |
X2 |
P-value |
|
Breed |
Local |
182 |
41(22.53%) |
4.3822 |
0.036 |
|
Exotic |
169 |
15(8.88%) |
|||
|
Sex |
Male |
52 |
14(26.92%) |
2.2671 |
0.132 |
|
Female |
299 |
41(13.71%) |
|||
|
Age |
Grower |
166 |
32(19.28%) |
0.9781 |
0.323 |
|
Adult |
184 |
23(12.50%) |
|||
|
Body condition |
Poor |
88 |
44(50%) |
35.0391 |
0.000 |
|
Good |
263 |
12(4.56%) |
Table 3.2: Prevalence Of Ascaris In Poultry In Relation To Different Management Factors In Bahir Dar Zuria District In 2019.
|
Management variables |
Categories |
No-examined |
No-ascaris-+Ve (%) |
X2 |
P-value |
|
Farming system |
Extensive |
219 |
47(21.46%) |
4.5399 |
0.033 |
|
Semi- intensive |
132 |
9(6.82%) |
|
|
|
|
Feeding |
Scavenging |
239 |
44(18.41%) |
1.1753 |
0.278 |
|
Additional |
112 |
12(10.71%) |
|
|
|
|
Hygiene |
Poor |
105 |
41(39.05%) |
20.5137 |
0.000 |
|
Good |
245 |
15(6.12%) |
|
|
|
|
Availability of deworming in owner |
Available |
2 |
0 |
16.23 |
0.001 |
|
Unavailable |
349 |
55(15.76%) |
|
|
This finding was in lined with the previous report of with a prev-alence of 15.1% from Bahir Dar. However, our finding prev-alence of ascaris was much lower than the previous reported works in central Ethiopia by with prevalence of 55.26% and from Haramaya 38% by [17, 18]. This might be due to difference in management system, deworming practice and agro-ecological conditions of the study area [1].
In the present study the prevalence of infection in local breed (22.58%) was significantly higher (P<0.05) than the exotic breed (8.62%). This result agreed with the previous studies conduct¬ed in Addis Ababa by [19]. He reports a high prevalence rate of ascaris (80%) in local chicken. This is not uncommon be¬cause of their free-range mode of management practice which allows them free access to virtually all types of environment and hence, predisposing them to various forms of infections [20]. In addition, domestic chickens feed widely which makes them more predisposed to infection. The duration for the local breed to reach table size is much longer compared to the exotic breeds, which fed usually on artificial diets [21].
In our study, there is no association in prevalence between asca¬ris galli and the risk factors (Age and Sex) of the ascaris positive chicken (p>0.05). This result agrees with the previous studies conducted in [14, 15] and Morocco by [22]. There was no natu¬ral affinity of ascaris to either sex and age of the host of chick¬ens. But this study contrary with another work of Sonaiya (1990) and Matur and his colleagues [20] those reported females and grower chickens were more infected with ascaris parasite than males and adult in both local and exotic breeds [23].
In our study, management system have higher statistically sig-nificance especially housing system, hygienic practice and free range strategies (p<0.05) with ascaris in poultry [24, 25]. Be-cause more management system in this area was extensive, the chickens seek there feed from the soil and contaminated with in-fective stage of parasite and living organisms, which serve as in-termediate host this, leads to higher chance of infection with as¬caris. In addition, there is higher statistically significant (p<0.05) in related to body condition with prevalence 50% and 4.44% in poor and good body condition chickens [26-30].
Conclusion & Recommendations
Generally, ascaris infection is one of the non-negligible prob¬lem and prevalent in this study area especially in local breed, poor body condition and poor managed chickens. In addition to other factors, ascaris is one of the causes of lowering chick¬en production in this area. Based on the above conclusions the following recommendations were forwarded: The management system should be improved especially the housing and hygienic practice [31-43]. In addition regular deworming and treatment of sick chicken should be done. Since there are lack of access for dewormer for smallholder farmers veterinarian should be search alternate; easily accessible and affordable drug for poultry inter¬nal parasite by considering ascaris like other disease.
Acknowledgements
We would like to thank to Bahir Dar University; School of Ani-mal Science and Veterinary Medicine; Veterinary Clinic workers and Veterinary Parasitological Laboratory for their contribution by providing the materials and laboratory possessing. We would also like to thank BVSc. veterinary science graduate of 2019 who helped us data collection process. Lastly, gratitude goes to poultry farm owners for their cooperation and allowing conduct¬ing this study on their farms during the study period.
References
- Ashenafi, H., & Eshetu, Y. (2004). Study on gastrointestinal helminths of local chickens in central Ethiopia. Revue de médecine vétérinaire, 155(10), 504-507.
- Jegede O., Asadu I., OparaM. Obeta S. (2014). Central Statistical Authority. Agricultural Sample Survey. Addis Ababa, Ethiopia. Statistical Bulletin 2: 331
- Mulugeta, A., Chanie, M., & Bogale, B. (2012). Major constraints of village poultry production in Demba Gofa District of Southern Region, Ethiopia.
- Nnadi, P. A., & George, S. O. (2010). A cross-sectional survey on parasites of chickens in selected villages in the subhumid zones of South-Eastern Nigeria. Journal of parasitology research, 2010.
- Bsrat, A., Tesfay, T., & Tekle, Y. (2014). Clinical, gross and histopathological study on common local chicken diseases in Enderta District, South East Tigray. European Journal of Biological Sciences, 6(4), 95-103.
- Yami, A. (1995). Poultry production in Ethiopia. World'sPoultry Science Journal, 51(2), 197-201.
- Eshetu, Y., Mulualem, E., Ibrahim, H., Berhanu, A., & Aberra, K. (2001). Study of gastro-intestinal helminths of scavenging chickens in four rural districts of Amhara region, Ethiopia. Revue scientifique et technique (International Office of Epizootics), 20(3), 791-796.
- Permin, A., Bisgaard, M., Frandsen, F., Pearman, M., Kold, J., & Nansen, P. (1999). Prevalence of gastrointestinal helminths in different poultry production systems. British poul-try science, 40(4), 439-443.
- Kaufmann, F., DaÅ?, G., Sohnrey, B., & Gauly, M. (2011). Helminth infections in laying hens kept in organic free-range systems in Germany. Livestock Science, 141(2-3), 182-187.
- Sherwin, C. M., Nasr, M. A. F., Gale, E., Petek, M. E. T. Ä°.N., Stafford, K., Turp, M., & Coles, G. C. (2013). Prevalence of nematode infection and faecal egg counts in free-range laying hens: relations to housing and husbandry. British poultry science, 54(1), 12-23.
- Thapa, S., Hinrichsen, L. K., Brenninkmeyer, C., Gunnars-son, S., Heerkens, J. L., Verwer, C.,...& Mejer, H. (2015). Prevalence and magnitude of helminth infections in organic laying hens (Gallus gallus domesticus) across Europe. Veterinary Parasitology, 214(1-2), 118-12.
- Nnadi, P. A., & George, S. O. (2010). A cross-sectional survey on parasites of chickens in selected villages in the subhumid zones of South-Eastern Nigeria. Journal of parasitology research, 2010.
- Phiri, I. K., Phiri, A. M., Ziela, M., Chota, A., Masuku, M., & Monrad, J. (2007). Prevalence and distribution of gastrointestinal helminths and their effects on weight gain in free-range chickens in Central Zambia. Tropical Animal Health and Production, 39(4), 309-315.
- Kaingu, F. B., Kibor, A. C., Shivairo, R., Kutima, H., Oke-no, T. O., Waihenya, R., & Kahi, A. K. (2010). Prevalence of gastro-intestinal helminthes and coccidia in indigenous chicken from different agro-climatic zones in Kenya. African Journal of Agricultural Research, 5(6), 458-462.
- Bayu, K. (2005). Standard veterinary laboratory diagnostic manual. Vol. III. MOA Addis Ababa.
- Lalchhandama, K., Roy, B., & Dutta, B. K. (2009). Anthel-mintic activity of Acacia oxyphylla stem bark against Ascaridia galli. Pharmaceutical Biology, 47(7), 578-583.
- Tarbiat, B., Jansson, D. S., & Höglund, J. (2015). Environmental tolerance of free-living stages of the poultry round-worm Ascaridia galli. Veterinary parasitology, 209(1-2), 101-107.
- Beyene, K., Bogale, B., & Chanie, M. (2014). Study on effects and occurrence of nematodes in local and exotic chickens in and around Bahir Dar, Northwest Ethiopia. American-Eurasian Journal of Science Research, 9(3), 62-66.
- Tesfaheywet Z., Amare E. and Hailu Z. (2012). Helminthosis of Chickens in Selected Small Scale Commercial Poultry Farms in and around Haramaya Woreda, Southeastern Ethiopia. Veterinary Advance.2: 462-468
- Gedion, Y. (1991). Preliminary survey of ectoparasites and gastrointestinal tract helminthes of local chickens in and around Dire Dawa. DVM Thesis. Faculty of Veterinary Medicine. Addis Ababa University. Ethiopia.
- Frantovo, D. (2000). Some parasitic nematodes (Nematoda) of birds (Aves) in the Czech Republic. Acta Societatis Zoological Bohemicae, 33, 45-65.
- Oniye, S. J., Audu, P. A., Adebote, D. A., Kwaghe, B. B., Ajanusi, O. J., & Nfor, M. B. (2001). Survey of helminth parasites of laughing dove (Streptopelia senegalensis) in Zaria Nigeria. African Journal of Natural Sciences, 4, 65-66.
- Hassouni, T., & Belghyti, D. (2006). Distribution of gastrointestinal helminths in chicken farms in the Gharb region— Morocco. Parasitology research, 99(2), 181-183.
- Sonaiya, E. B. (1990, October). The context and prospects for development of smallholder rural poultry production in Africa. In CTA-Seminar proceedings on smallholder rural poultry production (pp. 9-13).
- Matur, B. M., Dawam, N. N., & Malann, Y. D. (2010). Gastrointestinal helminth parasites of local and exotic chickens slaughtered in Gwagwalada, Abuja (FCT), Nigeria. New York Science Journal, 3(5), 96-99.
- Mulugeta, A., Chanie, M., & Bogale, B. (2012). Major constraints of village poultry production in Demba Gofa District of Southern Region, Ethiopia.
- Jegede O., Asadu I., OparaM., Obeta S. (2014). Central Statistical Authority (CSA. 2014). Agricultural Sample Survey. Addis Ababa, Ethiopia. Statistical Bulletin 2: 331.
- Jacobs, D., Fox, M., Gibbons, L., & Hermosilla, C. (2015). Principles of veterinary parasitology. John Wiley & Sons.
- Luna-Olivares, L. A., Ferdushy, T., Kyvsgaard, N. C., Ne-jsum, P., Thamsborg, S. M., Roepstorff, A., & Iburg, T. M. (2012). Localization of Ascaridia galli larvae in the jejunum of chickens 3 days post infection. Veterinary parasitology, 185(2-4), 186-193.
- Chadfield, M., Permin, A., Nansen, P., & Bisgaard, M. (2001). Investigation of the parasitic nematode Ascaridia galli (Shrank 1788) as a potential vector for Salmonella enterica dissemination in poultry. Parasitology Research, 87(4), 317-325.
- Martı´n-Pacho J., Montoya M., Aranguena T., Toro C., Mor-chon R., Atxutegi C. & Simon F. (2005). A coprological and serological survey for the prevalence of Ascaridia spp. in laying hens. Journal of Veterinary Medicine. 52: 238-242.
- Maurer V.,Amsler Z.,Perler E.and Heckendorn F. (2009). Poultry litter as source of gastrointestinal helminth infections .veterinary parasitiolgy.161 (3-4):255-260.
- North western Amhara livestock recoreds.2017
- Permin A., Pearman M., Wansen P., BisgaardM. & Frands-enF.1997. On investigation in different media for embryo-nation of Ascaridia galli eggs. Helminthologia. 34: 75-79.
- Permin A. and J. Hansen. 1999. The epidemiology,diagnosis and control of parasites in poultry. eds, FAO. Rome. 15-47.
- Sherwin, C. M., Nasr, M. A. F., Gale, E., Petek, M. E. T. Ä°.N., Stafford, K., Turp, M., & Coles, G. C. (2013). Prevalence of nematode infection and faecal egg counts in free-range laying hens: relations to housing and husbandry. British poultry science, 54(1), 12-23.
- Soulsby, E. J. L. (1982). Helminths. Arthropods and Proto-zoa of domesticated animals, 291.
- Taylor, M. A., Coop, R. L., & Wall, R. L. (2007). Parasites of poultry and gamebirds. Veterinary parasitology, 3, 459-557.
- Tesfaheywet Z., Amare E. and Hailu Z. 2012. Helminthosis of Chickens in Selected Small Scale Commercial Poultry Farms in and around Haramaya Woreda, Southeastern Ethiopia. Veterinary Advance.2: 462-468.
- Thrusfield M. (2005). Veterinary Epidemiology. 3rd ed. Singapore black well science: 233-236.
- Wongrak, K., DaÅ?, G., Moors, E., Sohnrey, B., & Gauly, M. (2014). Establishment of gastro-intestinal helminth infections in free-range chickens: a longitudinal on farm study. Berl Münch Tierarztl Wochenschr, 127, 305-13.
- Yamaguti, S. (1961). Systema Helminthum...: The Nematodes of Vertebrates. Interscience publishers.
- Eshetu, Y., Mulualem, E., Ibrahim, H., Berhanu, A., & Aberra, K. (2001). Study of gastro-intestinal helminths of scavenging chickens in four rural districts of Amhara region, Ethiopia. Revue scientifique et technique (International Office of Epizootics), 20(3), 791-796.


