Research Article - (2025) Volume 4, Issue 1
Potential Geographic Distributions of Ceiba Pentandra Under Current and Future Climate Conditions in Benin, West Africa
2SOS Biodiversity. 04 BP 1005 Cotonou, Benin
Received Date: Nov 10, 2024 / Accepted Date: Dec 10, 2024 / Published Date: Jan 27, 2025
Copyright: ©Â©2025 Sunday Berlioz Kakpo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kakpo, S. B., Ganglo, J. C. (2025). Potential Geographic Distributions of Ceiba Pentandra Under Current and Future Climate Conditions in Benin, West Africa. World J Forest Res, 4(1), 01-11.
Abstract
The 21st century is marked by growing losses of biodiversity particularly in tropical regions. These losses result not only from direct anthropogenic effects but also from climate change; research must therefore consider impacts of climate change on future potential distributions of species. Ceiba pentandra (L) Gaert, a high-value multiple-use and keystone species of tropical environments, deserves particular attention. This study aims to model the ecological niche of C. pentandra, assess the impacts of climate change on its potential distribution by 2055 and 2085, and assess the effectiveness of protected areas for conservation both at present and in the future. Bioclimatic variables and soil data layers were used for modeling with maximum entropy approaches and resulting distributional predictions were overlaid on the existing protected areas network. Results showed that the distribution of C. pentandra is governed by a combination of effects of climate (temperature and precipitation) and substrates (soils); its distributional potential in West Africa trended weakly northward in future decades. The relationship of the species distribution to protected areas of Benin suggests the poor potential of present protected areas to conserve populations of species. Protected area networks must be strengthened to improve the protection of forests in suitable areas; protected areas located in medium-suitable areas could be enriched for C. pentandra because many such areas are anticipated to become more suitable in the future for the species.
Keywords
Species Distribution Modeling, Climate Change, Ceiba Pentandra, West Africa
Introduction
Global biodiversity is concentrated in tropical countries, and their complement of species is often unique [1,2]. Climate change and human activities have resulted in biodiversity losses at 100-1000 times the normal rate of extinction of plant and animal species [3-5]. Knowledge of current and future distributions of species is crucial to designing feasible biodiversity conservation policies [6,7]. Conservation assessment and planning require information on the spatial distribution of biodiversity, often across broad regions [8,9]. Ecological niche models are used widely to predict potential geographic distributions of species [10-14]. These models and their resulting predictions are used to anticipate the impacts of climate change on species and ecosystems [15,16]. These models estimate relationships between environmental conditions and occurrences of species, which can in turn be mapped to predict potential geographical distributions of species [17,18]. It is important to assess the effects of climate change because the interactive effects of anthropogenic and climatic disturbances could lead to changes in habitat, ecological requirements of species, and demographic decline [19].
In West Africa, distributions of many species have been studied. For instance, Fandohan et al. assessed the vulnerability of protected areas to the invasive plant Chromolaena odorata under current and future conditions [20]. Dossou et al., analyzed climate change impacts on populations of Lannea microcarpa in Benin [21]. Hounkpèvi et al., assessed suitability for cultivation and in situ conservation of the black plum (Vitex doniana Sweet) in Benin [22]. Idohou et al. evaluated spatio-temporal potential range dynamics for eight economically important wild palms under present and future climate conditions across West Africa [23]. Kakpo et al. studied the spatial distribution and impacts of climate change on the endangered Milicia excelsa in Benin [24]. Akaffou et al. studied the current and future distribution of Chromolaena odorata (L.) and Hopea odorata Roxb in the Banco National Park (Ivory Coast) [25].
Ceiba pentandra (L) Gaert, or the Kapok tree (Bombacaceae), is a species of dense tropical forests that deserves special attention, because of its multiple uses in human nutrition, medicine, and the timber industry [26-32]. In this paper, we aim to model the ecological niche of C. pentandra, to assess the impacts of climate change on the potential distribution of the species for the 2055 and 2085 time horizons, and to assess the effectiveness of protected areas for conservation of this species under present and future climates.
Materials and Methods
Study Area
The study was carried out in Benin Republic, located between 6°10’ and 12°50’ N and 1° to 3°40’ E in West Africa (Figures 1 and 2). West Africa (4°-20°N; 20°W- 40°E) is open to the Atlantic Ocean. It extends from Nigeria to the coasts of Senegal. This part of Africa is characterized by a dry tropical climate. There are four types: a desert climate in the north (annual rainfall < 200 mm); Sahelian in the center (annual rainfall between 200 mm and 600 mm); Sudanian (annual rainfall between 600 mm and 1200 mm); and Guinean (annual rainfall > 1200mm).
<img src="https://www.opastpublishers.com/scholarly-images/8683-685e3a4288283-potential-geographic-distributions-of-ceiba-pentandra-under-.jpg" style="width:500px;height:250px;">
Benin's climatic profile shows two contrasting climatic zones (Guinean vs. Sudanian) and a transitional zone (Sudano-Guinean). The Guinean zone (between 6°25’ and 7°30’ N) is characterized by a subequatorial climate with four seasons (two rainy and two dry). The rainfall of about 1200 mm per year is bimodal mostly from March to July and September to November. The temperature varies between 25 and 29 °C, and the relative humidity varies between 69 % and 97 %. The Sudanian zone (9°45’ - 12°25’ N) has a tropical dry climate with two equal-length seasons (rainy and dry). The mean annual rainfall in this zone is often less than 1000 mm and occurs mainly from May to September. The relative humidity varies from 18% to 99% and the temperature from 24 to 31°C. The Sudano-Guinean (from 7°30’ to 9°45’ N) is a transitional zone with two rainy seasons merging into an unimodal regime. The annual rainfall fluctuates between 900 and 1110 mm, the temperature is between 25 and 29°C, and the relative humidity is from 31 % to 98 [33,22].
<img src="https://www.opastpublishers.com/scholarly-images/8683-685e3a90b34ca-potential-geographic-distributions-of-ceiba-pentandra-under-.jpg" style="width:500px;height:350px;">
Input Data
In all, 1246 records were obtained from the Global Biodiversity Information Facility and our fieldwork in Benin [34]. In Benin, most of the occurrence records are concentrated in the South of Benin (Figure 2). Data cleaning is important in ecological niche modeling because it determines the quality of the resulting models. Occurrences with problematic geographic coordinates (i.e., the value of latitude not between -90 and +90, or longitude not between -180 and +180) were removed; we have checked if coordinate problems were not errors of reversal. When duplicate occurrences were available from sites, only one was retained. Finally, occurrences with geographic coordinates that did not correspond to the countries mentioned in the data record were also removed. In the end, 255 occurrence points were used for the species across its range in the West African region (Figure 1)
Models were run using data on climate, land cover, and soils. Current (1950–2000) climate summaries were obtained from WorldClim version 1.4 (available at www.worldclim.org/bioclim) and future (2055 and 2085 years) climate summaries were drawn from AFRICLIM version 3.0 (available at www.york.ac.uk/ environment/research/kite/resources/; Platts et al., 2015) at a resolution of 2.5’ which corresponds approximately to 4.6 km spatial resolution in West Africa [20,23]. Through the CORDEX initiative, outputs from regional climate models (RCMs) have become available for Africa [35,36]. AFRCLIM provides high- resolution ensemble climate data for Africa including, estimates for 17 bioclimatic variables (Bio1-Bio7 and Bio10-Bio17) derived from average monthly maximum and minimum temperature and precipitation data [37]. We added detailed land cover (available at http://gcmd.nasa.gov) and soil layers (available at http://glcf. umd.edu/index.shtml) to analyses. The inclusion of soil data in models that estimate the habitat suitability of a plant species has been recommended when the resolution of the study is < 2000 km2 [38,39].
For projections of future climatic conditions, predictions from two models were used: AfriClim-Ensemble and the Model for Interdisciplinary Research on Climate Change (MIROC5). Africlim-Ensemble is a set of summary statistics (mean, minmean, maxmean) over 18 pairwise combinations of 5 Regional Climate Models (RCMs) driven by 10 General Circulation Models (GCMs). MIROC5 is among the most used models currently available for simulating global climate response to increasing greenhouse gas concentration. The projections were run under two of the four scenarios developed by the Intergovernmental Panel on Climate Change (IPCC) in its Fifth Assessment Report (AR5): representative concentration pathway (RCP) 4.5 and RCP 8.5, for the 2055 and 2085 time horizons. By the mid-21st century, RCP
4.5 projects temperatures to rise above industrial level by at least 1.48°C in West Africa, with atmospheric CO2 reaching 500 ppm [40]. Under the more extreme RCP 8.5 projections, temperatures are predicted to rise by 2.8°C and atmospheric CO2 to reach >550 ppm [40]. These climate projections were statistically downscaled to match the bioclim variables using the delta method [41].
The Protected Area Network (PAN) map of Benin was obtained from the World Database on Protected Areas (available at www. protectedplanet.net) and used to assess the in-situ conservation of the species in the country under current and future climates [42].
Modeling the Distribution of the Species
The maximum entropy species distribution model algorithm (MaxEnt, version 3.3.3k) was used for modeling the ecological niche of C. pentandra in Benin. This tool, requiring presence data only, is one of the best-performing algorithms among those available, is very widely adapted, and is relatively robust for small sample sizes [43]. It is a machine-learning method that estimates ecological niches based on known occurrences of species across a study area by calculating the probability distribution of maximum entropy, subject to the constraint that the expected value of each feature under this estimated distribution should match its empirical average [44]. Although several conceptual ambiguities and uncertainties exist about bioclimatic envelope modeling, MaxEnt remains an important modeling tool in assessing the potential impacts of climate change on species distributions [45,46].
Before modeling, occurrence data were cleaned up by removing duplicate records in grid cells to reduce sampling bias resulting from oversampling of some sites [47]. Maxent models were developed using: 25,000 background points, 10 bootstrap replicate analyses, a maximum of 500 iterations, a convergence threshold of 0.00001, and a default prevalence of 0.7. A jackknife test was performed on the selected bioclimatic variables to identify variables that contribute most to model predictions. Variables selected were then subjected to correlation tests, to select least-correlated suitability predictor variables (r <0.80) [48,49]. Models were simplified to the highest threshold that included 95% of the calibration data, an approach that prioritizes correct prediction of presences over correct prediction of absences and takes into account problems regarding occurrence data quality [18].
To capture the fullest range of variation in each bioclimatic factor, we performed the modeling process using occurrence and climatic data for the whole of West Africa. The outputs of MaxEnt were then clipped on Benin, to mark out the study area. To assess the potential of protected areas to conserve C. pentandra, population the protected area network of Benin was overlaid on model outputs, and relevant areas were estimated in ArcGIS 10.2.2.
Model Evaluation
Two criteria were used to evaluate the performance i.e. goodness- of-fit and predictive power of the model: the area under the receiver operating characteristic curve and the true skill statistic (TSS) [47,50,51]. The AUC is the probability that a randomly chosen presence point of the species will be ranked as more suitable than a randomly chosen absence point [47]. A model is considered to have a good fit when its AUC is close to one (AUC ≥ 0.75) [47]. The TSS is the capacity of the model to accurately detect true presences (sensitivity) and true absences (specificity). A model with TSS ≤ 0 indicates a random prediction, while a model with a TSS close to 1 (TSS > 0.5) has a good predictive power [50]. To assess the model performance the occurrence data were divided randomly into maxent that are 70 % for calibration and 30 % for evaluation.
Results
Variable Importance and Model Performance
Our analyses of variable contributions and correlations among variables identified Soil Type and four bioclimatic variables as contributing importantly to model quality (Table 1): annual precipitation, mean diurnal range, temperature seasonality, and temperature annual range. These variables all had significant effects on model gain. Soil was the most informative predictor because its inclusion in models considerably affected the gain.
|
Variables |
Definition |
Percent contribution (%) |
Permutation importance |
|
Soil |
Type of soil |
55 |
40 |
|
bio4 |
Temperature Seasonality (standard deviation *100) |
21.2 |
16.2 |
|
bio7 |
Temperature Annual Range (Max Temperature of Warmest Month - Min Temperature of Coldest Month) |
11 |
36 |
|
bio12 |
Annual Precipitation (mm) |
7,3 |
2.2 |
|
bio2 |
Mean Diurnal Range of temperature (10 x °C) |
5,5 |
5.6 |
Table 1: Contribution of Predictor Variables to the Model
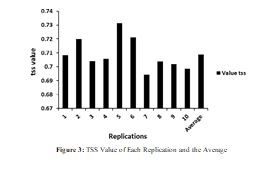
The model had a very goodness-of-fit (cross-validated average AUC = 0.977) and a very good predictive power (TSS = 0.71) (Figure 3). Then, the models indicated an excellent performance. The threshold for the habitat suitability discrimination was 0.34.
Areas with occurrence probability above this threshold were then considered as suitable for the species, the remaining being considered as unsuitable areas.
Habitat Suitability for West Africa and Benin
Across West Africa, our models showed C. pentandra as distributed along the coast (Figure 4), including portions of Nigeria, Benin, Togo, Ghana, Côte d’Ivoire, Liberia, Sierra Leone, Guinea, Guinea-Bissau, Senegal, and Gambia. Father northern West Africa away from the coasts, was not suitable for the species. Overall, around 18.3% of West Africa was identified as suitable versus 81.7% not suitable (Table 2).
In West Africa, an increase in suitable area was noticed in the species’ future distribution (Figure 4) for RCP 4.5 and RCP 8.5, in the form of a range extension toward the north into the Sahel. Indeed, suitable areas were projected to increase by 7.1–10.3% and 6.9-10.8% under AFRICLIM-Ensemble projections for 2055 and 2085, respectively (Figure 4). Under MIROC5 projections, the suitable area extended by 14.7-19.3% and 13.8-14.1% for 2055 and 2085, respectively (Figure 4). More in detail in Benin, the same tendencies were observed for the RCP 4.5 and RCP 8.5. The highly suitable area extended toward the northern and mainly the Sudanian-Guinean climatic zone (Figure 5). In the country, it was noticed an increase of 1.9–10.9% and 1.2-3.2% under AFRICLIM- Ensemble projections for 2055 and 2085, respectively (Figure 5). Under MIROC5 projections the suitable area extended more by 11.7-24.4% and 15.6-29.9% for 2055 and 2085, respectively (Figure 5).
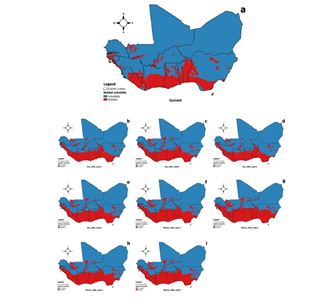
Figure 4: Potential distribution of Ceiba pentandra in West Africa under current and future climatic conditions. Panel (a) current climatic conditions, (b, c, d, and e) 2055 and 2085 horizon with RCPs 4.5 and 8.5 for AFRICLIM-Ensemble, respectively, and (f, g, h, and i) 2055 and 2085 horizon with RCPs 4.5 and 8.5 for MIROC5, respectively.
In Benin, the species showed a high suitability in the Guinean climatic zone. The Sudanian-Guinean zone is not so suitable for the species. But toward the East and the Northeastern of this climatic zone, low suitable areas were noticed for the species. In the Sudanian zone, the suitability area of species was low, but toward the north and northeast of this climatic zone, the ecological conditions were unsuitable for the species. Regarding the suitability of species in Benin, approximately 10.21% of the area of Benin was highly suitable, 34.20% medium, 38.95% low, and 16.64% unsuitable (Table 3). It is noteworthy that only 0.9% of protected areas (lama, Agrimey, and Djigbé reserve) contained the highly suitable area of the species (Figure 5).
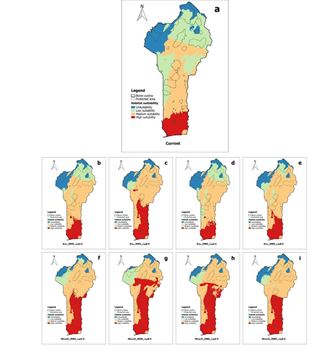
Figure 5: Potential distribution of Ceiba pentandra in Benin under current and future climatic conditions. Panel (a) current climatic conditions, (b, c, d, and e) 2055 and 2085 horizon with RCPs 4.5 and 8.5 for AFRICLIM-Ensemble, respectively, and (f, g, h and i) are 2055 and 2085 horizon with RCPs 4.5 and 8.5 for MIROC5, respectively.
|
Suitability |
Current |
AFRICLIM-Ensemble |
MIROC5 |
|||||||||||||||
|
2055 |
2085 |
2055 |
2085 |
|||||||||||||||
|
Area |
% |
4.5 |
8.5 |
4.5 |
8.5 |
4.5 |
8.5 |
4.5 |
8.5 |
|||||||||
|
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
|||
|
Unsuitable |
411.63 |
81.70 |
375.93 |
|
359.71 |
|
377.04 |
|
357.00 |
|
337.34 |
|
314.47 |
|
341.93 |
|
340.84 |
|
|
Suitable |
92.21 |
18.30 |
127.91 |
7.09 |
144.13 |
10.30 |
126.80 |
6.87 |
146.83 |
10.84 |
166.49 |
14.74 |
189.37 |
19.28 |
161.90 |
13.83 |
163.00 |
14.05 |
|
Sub-total |
503.84 |
|
503.84 |
|
503.84 |
|
503.84 |
|
503.84 |
|
503.84 |
|
503.84 |
|
503.84 |
|
503.84 |
|
Tr: Trends (%) ; Area (Km2/104)
Table 2: Potential Habitat Suitability for Ceiba Pentandra in West Africa Under Current and Future Climatic Conditions
|
Suitability |
Current |
AFRICLIM-Ensemble |
MIROC5 |
|||||||||||||||
|
2055 |
2085 |
2055 |
2085 |
|||||||||||||||
|
Area |
% |
4.5 |
|
8.5 |
4.5 |
8.5 |
4.5 |
8.5 |
4.5 |
8.5 |
||||||||
|
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
Area |
Tr † |
|||
|
High |
1.17 |
16.64 |
1.38 |
1.88 |
2.42 |
10.92 |
1.54 |
3.21 |
1.53 |
3.15 |
2.51 |
11.72 |
3.96 |
24.37 |
4.59 |
29.89 |
2.96 |
15.59 |
|
Medium |
3.92 |
38.95 |
6.29 |
|
6.55 |
|
5.85 |
|
6.94 |
|
6.37 |
|
5.59 |
|
4.95 |
|
6.59 |
|
|
Low |
4.46 |
34.20 |
1.95 |
|
0.80 |
|
2.18 |
|
1.19 |
|
1.22 |
|
1.05 |
|
1.10 |
|
1.34 |
|
|
Unsuitable |
1.91 |
10.21 |
1.84 |
|
1.68 |
|
1.89 |
|
1.79 |
|
1.35 |
|
0.86 |
|
0.81 |
|
0.57 |
|
|
Sub-total |
11.46 |
|
11.46 |
|
11.46 |
|
11.46 |
|
11.46 |
|
11.46 |
|
11.46 |
|
11.46 |
|
11.46 |
|
Tr: Trends (%) ; Area (Km2/104)
Table 3: Potential Habitat Suitability for Ceiba Pentandra in Benin Under Current and Future Climatic Conditions
Discussion
Ecological niche modeling is considered a powerful tool with which to estimate and predict the potential distributions of species [52]. This approach uses information on environmental requirements derived from known occurrences to predict potential distributions of species under current or possible future conditions, which can guide sustainable management plans [44,45,53,54]. Moreover, projections of our models do not integrate any demographic parameter (fruitfulness, mortality, growth) and dynamics of populations (dispersion, migration, competition) [47]. The migration and stray capacitance of species are the factors determining the potential impact of climatic change on their habitats [47]. When the constraints of dispersion are not taken into account, the temperature increase could expand the distribution area of certain species [15,55,56].
The results of this study reveal that the model performed well in predicting suitable conditions for Ceiba pentandra. So, the models were judged significantly better than random.
On the whole, our results from this modeling showed that C. pentandra distributions are influenced by combinations of effects of climate variables and soil characteristics. The influence of climatic factors is consistent because both water availability and temperature are the main factors that control the geographic distributions of species in tropical zones. From a mechanistic point of view, it is desirable to predict the distribution of biotic entities based on direct gradients (such as temperature and precipitation) that are believed to be the causal, driving forces for their distribution
and abundance [10]. Although indirect gradients (as the soil) are variables that have no direct physiological relevance for a species performance they are most easily measured in the field and are often used because of their good correlation with observed species patterns [57]. Indirect gradients can be applied within a limited geographical extent without significant errors [10].
C. pentandra in West Africa is anticipated to extend somewhat farther north in future decades. This increase in habitat suitability can be explained by changes projected for the bioclimatic parameters, particularly temperature, but this expansion may be limited by water availability. Under both the Africlim-Ensemble and the MIROC5 projections, the increase in suitable area was noticed in all coastal countries of West Africa, and the expansion was greater under RCP 8.5 than under RCP 4.5, particularly under the MIROC5 model. These spatial differences in future range projections could be attributed to local differences in the magnitude of climate change as projected by each climate model and representative concentration pathway [20,58].
In Benin, our results showed that at present, C. pentandra is more subservient in the Guinean climatic zone. But in the future, it is noticed an expansion of the species towards the north of the country. Part of the Sudanian-Guinean climatic zone became very favorable to species. Although the modification of the climate was favorable to the expansion of species, it remains limited all the same. The principal factors limiting the distribution observed were the biotic factors. The biotic factors, like competition, affect the distribution of species at local scales [38].
Assessment of the relationship of C. pentandra species distribution to protected areas of Benin showed the poor potential of protected areas to conserve populations of species in the current climate. Only 0.9% of protected areas (Lama, Agrimey, and Djigbé reserve) contained highly suitable areas for the species. The situation improved in future projections in particular for climatic models MIROC5. Indeed, in sub-Saharan Africa, the future climate threats are not fully making out even though sub-Saharan Africa has been cited as one of the most vulnerable areas to climate change [59]. The threats increase in protected areas exponentially because of human population expansion, illicit exploitation of resources, and fragmentation of the habitat [60,61]. These pressures and threats destroy the species' habitats and can cause the extinction of species.
Conclusion
Our results offer scientific bases for a planning and decision- support tool for the conservation of the high-value species Ceiba pentandra. Our results showed an increase in suitable areas for the species in all coastal countries of West Africa. Even though the habitat suitability of C. pentandra is projected to increase, its reproduction under future climate might be affected either positively or negatively. The species may have undergone several physiological adaptations in response to past climates, but under the current rapid climate change, the expansion of the species in new areas will likely require important energy-dependant adjustments in morphological, physiological, or behavioral traits of the species and this could have negative impacts on its reproduction. Worth mentioning pollinators of species, for example, bats might be affected by climate change. An urgent conservation measure is to strengthen the protection of protected areas and unclassified forests in highly suitable areas. Protected areas in areas only moderately suitable require enrichment in C. pentandra because many such areas are anticipated to become highly suitable in the future. Studies are required on the genetic diversity of the species to build a consistent base for sustainable management of the species under climate change.
References
1. Gaikwad, J., & Chavan, V. (2006). Open access and biodiversity conservation: challenges and potentials for the developing world. Data Science Journal, 5, 1-17.
2. Ganglo, J. C., & Kakpo, S. B. (2016). Completeness of digital accessible knowledge of plants of Benin and priorities for future inventory and data discovery. Biodiversity Informatics, 11(1), 23-39.
3. Jenkins, M. (2003). Prospects for biodiversity. science, 302(5648), 1175-1177.
4. Loreau, M., Oteng-Yeboah, A., Arroyo, M. T., Babin, D., Barbault, R., Donoghue, M., ... & Watson, R. T. (2006). Diversity without representation. Nature, 442(7100), 245-
246.
5. Djoghlaf, A. (2007). Climate change is real, the time to act is now. In: Gincana 3: Biological Diversity and Climate Change. Convention on Biological Diversity (CBD), 1-32.
6. Hortal, J., & Lobo, J. M. (2001). A preliminary methodological approach to model the spatial distribution of biodiversity attributes. In Spatio-temporal modelling of environmental processes. Proceedings of the 1st Spanish workshop on spatio- temporal modelling of environmental processes. Publicacions de la Universitat Jaume I. Castelló de la Plana (pp. 211-229).
7. Burgman, M. A., Lindenmayer, D. B., & Elith, J. (2005). Managing landscapes for conservation under uncertainty. Ecology, 86(8), 2007-2017.
8. Margules, C. R., & Pressey, R. L. (2000). Systematic conservation planning. Nature, 405(6783), 243-253.
9. Ferrier, S., Manion, G., Elith, J., & Richardson, K. (2007). Using generalized dissimilarity modelling to analyse and predict patterns of beta diversity in regional biodiversity assessment. Diversity and distributions, 13(3), 252-264.
10. Guisan, A., & Zimmermann, N. E. (2000). Predictive habitat distribution models in ecology. Ecological modelling, 135(2- 3), 147-186.
11. Hosseinian Yousefkhani, S. S., Rastegar-Pouyani, E., & Aliabadian, M. (2016). Ecological niche differentiation and taxonomic distinction between Eremias strauchi strauchi and Eremias strauchi kopetdaghica (Squamata: Lacertidae) on the Iranian Plateau based on ecological niche modeling. Italian Journal of Zoology, 83(3), 408-416.
12. Ochieng, A. O., Nanyingi, M., Kipruto, E., Ondiba, I. M., Amimo, F. A., Oludhe, C., ... & Estambale, B. B. (2016). Ecological niche modelling of Rift Valley fever virus vectors in Baringo, Kenya. Infection ecology & epidemiology, 6(1), 32322.
13. Kalboussi, M., & Achour, H. (2018). Modelling the spatial distribution of snake species in northwestern Tunisia using maximum entropy (Maxent) and Geographic Information System (GIS). Journal of Forestry Research, 29, 233-245.
14. Moua, Y., Roux, E., Seyler, F., & Briolant, S. (2020). Correcting the effect of sampling bias in species distribution modeling–A new method in the case of a low number of presence data. Ecological Informatics, 57, 101086.
15. Araújo, M. B., Thuiller, W., & Pearson, R. G. (2006). Climate warming and the decline of amphibians and reptiles in Europe. Journal of biogeography, 33(10), 1712-1728.
16. Ganglo, J. C., Djotan, G. K., Gbètoho, J. A., Kakpo, S. B., Aoudji, A. K., Koura, K., & Tessi, R. Y. (2017). Ecological niche modeling and strategies for the conservation of Dialium guineense Willd.(Black velvet) in West Africa. International Journal of Biodiversity and Conservation, 9(12), 373-388.
17. Jiménez-Valverde, A., & Lobo, J. M. (2007). Threshold criteria for conversion of probability of species presence to either–or presence–absence. Acta oecologica, 31(3), 361-369.
18. Peterson, A. T., Soberón, J., Pearson, R. G., Anderson, R. P., Martínez-Meyer, E., Nakamura, M., & Araújo, M. B. (2011). Ecological niches and geographic distributions (MPB-49). In Ecological niches and geographic distributions (MPB-49). Princeton University Press.
19. Atauchi, P. J., Aucca-Chutas, C., Ferro, G., & Prieto-Torres,
D. A. (2020). Present and future potential distribution of the endangered Anairetes alpinus (Passeriformes: Tyrannidae) under global climate change scenarios. Journal of Ornithology, 161(3), 723-738.
20. Fandohan, A. B., Oduor, A. M., Sodé, A. I., Wu, L., Cuni- Sanchez, A., Assédé, E., & Gouwakinnou, G. N. (2015). Modeling vulnerability of protected areas to invasion by Chromolaena odorata under current and future climates. Ecosystem Health and Sustainability, 1(6), 1-12.
21. DOSSOU, E. M., LOUGBEGNON, T. O., HOUESSOU, L. G., & CODJIA, J. T. C. (2016). Analyse de l’impact du changement climatique sur l’aire de distribution actuelle et future de Lannea microcarpa Engl. et K. Krause au Bénin, Afrique de l’Ouest.
22. Hounkpèvi, A., Tosso, F., Gbèmavo, D. S. J. C., Kouassi, E. K., Koné, D., & Glèlè Kakaï, R. (2016). Climate and potential habitat suitability for cultivation and in situ conservation of the black plum (Vitex doniana Sweet) in Benin, West Africa. International Journal of Agronomy and Agricultural Research, 8(4), 67-80.
23. Idohou, R., Assogbadjo, A. E., Kakai, R. G., & Peterson, A. T. (2017). Spatio-temporal dynamic of suitable areas for species conservation in West Africa: eight economically important wild palms under present and future climates. Agroforestry Systems, 91, 527-540.
24. Kakpo, S. B., Aoudji, A. K. N., Gnanguènon-Guéssè, D., Gbètoho, A. J., Koura, K., Djotan, G. K., & Ganglo, J. C. (2021). Spatial distribution and impacts of climate change on Milicia excelsa in Benin, West Africa. Journal of Forestry Research, 32, 143-150.
25. Akaffou, S. V. E., Abrou, N. E. J., & Tiébré, M. S. (2020). Current and future distribution of Chromolaena odorata (L.) RM. King & H. Roxb (Compositae) and Hopea odorata Roxb(Dipterocarpaceae) in the Banco national park. IOSR Journal of Pharmacy and Biological Sciences (IOSR-JPBS), 15(2), 06-14.
26. Orwa, C., Mutua, A., Kindt, R., Jamnadass, R., & Anthony, S. (2009). Arbutus unedo L. Agroforestree Database: a tree reference and selection guide, version, 4.
27. Ibitoye, A. A. (2010). Chemical composition and nutritive significance of Ceiba pentandra leaves. Applied Tropical Agriculture 15(1 & 2), 36-38.
28. Enechi, O. C., Peter, C. D., Ugwu, O. P. C., Udeh, S. M. C., & Omeh, Y. S. (2013). Evaluation of the nutritional potential of Ceiba pentandra leaves. Mintage Journal of Pharmaceutical & Medical Sciences, 2(3), 25-27.
29. Ezigbo, V. O., Odinma, S. C., Duruaku, I. J., & Onyema, C. T. (2013). Preliminary phytochemical screening and antibacterial activity of stem bark extracts of. Ceiba Pentandra. J. Appl. Chem, 6, 42-44.
30. Raimi, M. M., Oyekanmi, A. M., & Farombi, A. G. (2014). Proximate and phytochemical composition of leaves of Ceiba pentandra, Manihot esculentus and Abelmoschus esculentus in Southwestern Nigeria. Sci Res J, 2, 30-34.
31. Anosike, C. A., C. ugwu, J., Ojeli, P. C., & Abugu, S. C. (2014). Anti-ulcerogenic effects and anti-oxidative properties of Ceiba pentandra leaves on alloxan-induced diabetic rats. European Journal of Medicinal Plants 4(4), 458-472.
32. Duvall, C. S. (2009). Ceiba pentandra (L.) Gaertn. Prota, 16, 14.
33. Fandohan, B., Assogbadjo, A. E., Glèlè Kakaï, R. L., & Sinsin, B. (2011). Effectiveness of a protected areas network in the conservation of Tamarindus indica (Leguminosea– Caesalpinioideae) in Benin. African Journal of Ecology, 49(1), 40-50.
34. GBIF. (2016). Occurrence data download. Denmark: Global Biodiversity Information Facility.
35. Jones, C., Giorgi, F., & Asrar, G. (2011). The coordinated regional downscaling experiment (CORDEX). An international downscaling link to CMIP5. CLIVAR exchanges, 56.
36. Platts, P. J., Omeny, P., & Marchant, R. (2015). AFRICLIM: high-resolution climate projections for ecological applications in Africa. African Journal of ecology, 103-108.
37. Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G., & Jarvis, A. (2005). Very high resolution interpolated climate surfaces for global land areas. International Journal of Climatology: A Journal of the Royal Meteorological Society, 25(15), 1965-1978.
38. Pearson, R. G., & Dawson, T. P. (2003). Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful?. Global ecology and biogeography, 12(5), 361-371.
39. Taylor, S., & Kumar, L. (2013). Potential distribution of an invasive species under climate change scenarios using CLIMEX and soil drainage: a case study of Lantana camara
L. in Queensland, Australia. Journal of environmental management, 114, 414-422.
40. IPCC. (2013). Climate Change 2013: The Physical Science Basis. IPCC Working Group I Contribution to the IPCC Fifth Assessment Report. Cambridge University Press, New York, USA.
41. Ramírez Villegas, J., & Jarvis, A. (2010). Downscaling global circulation model outputs: the delta method decision and policy analysis Working Paper No. 1.
42. UNEP-WCMC. (2016). World database on protected areas user manual 1.4.
43. Pearson, R. G., Raxworthy, C. J., Nakamura, M., & Townsend Peterson, A. (2007). Predicting species distributions from small numbers of occurrence records: a test case using cryptic geckos in Madagascar. Journal of biogeography, 34(1), 102- 117.
44. Phillips, S. J., Anderson, R. P., & Schapire, R. E. (2006). Maximum entropy modeling of species geographic distributions. Ecological modelling, 190(3-4), 231-259.
45. Schwartz, M. W. (2012). Using niche models with climate projections to inform conservation management decisions. Biological Conservation, 155, 149-156.
46. Elith, J., Phillips, S. J., Hastie, T., Dudík, M., Chee, Y. E., & Yates, C. J. (2011). A statistical explanation of MaxEnt for ecologists. Diversity and distributions, 17(1), 43-57.
47. Elith*, J., H. Graham*, C., P. Anderson, R., Dudík, M., Ferrier, S., Guisan, A., ... & E. Zimmermann, N. (2006). Novel methods improve prediction of species’ distributions from occurrence data. Ecography, 29(2), 129-151.
48. Zuur, A. F., Ieno, E. N., & Elphick, C. S. (2010). A protocol for data exploration to avoid common statistical problems. Methods in ecology and evolution, 1(1), 3-14.
49. Elith, J., Kearney, M., & Phillips, S. (2010). The art of modelling range-shifting species. Methods in Ecology and Evolution, 1(4), 330-342.
50. Fielding, A. H., & Bell, J. F. (1997). A review of methods for the assessment of prediction errors in conservation presence/ absence models. Environmental conservation, 24(1), 38-49.
51. Allouche, O., Tsoar, A., & Kadmon, R. (2006). Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). Journal of applied ecology, 43(6), 1223-1232.
52. Syfert, M. M., Smith, M. J., & Coomes, D. A. (2013). The effects of sampling bias and model complexity on the predictive performance of MaxEnt species distribution models. PloS one, 8(2), e55158.
53. Ihlow, F., Courant, J., Secondi, J., Herrel, A., Rebelo, R., Measey, G. J., ... & Rödder, D. (2016). Impacts of climate change on the global invasion potential of the African clawed frog Xenopus laevis. PLoS One, 11(6), e0154869.
54. Sommer, J. H., Kreft, H., Kier, G., Jetz, W., Mutke, J., & Barthlott, W. (2010). Projected impacts of climate change on regional capacities for global plant species richness. Proceedings of the Royal Society B: Biological Sciences, 277(1692), 2271-2280.
55. Sharma, S., & Jackson, D. A. (2008). Predicting smallmouth bass (Micropterus dolomieu) occurrence across North America under climate change: a comparison of statistical approaches. Canadian Journal of Fisheries and Aquatic Sciences, 65(3), 471-481.
56. Buse, J., & Griebeler, E. M. (2011). Incorporating classified dispersal assumptions in predictive distribution models–A case study with grasshoppers and bush-crickets. Ecological Modelling, 222(13), 2130-2141.
57. Guisan, A., Weiss, S. B., & Weiss, A. D. (1999). GLM versus CCA spatial modeling of plant species distribution. Plant ecology, 143, 107-122.
58. Salzmann, U., & Hoelzmann, P. (2005). The Dahomey Gap: an abrupt climatically induced rain forest fragmentation in West Africa during the late Holocene. The Holocene, 15(2), 190-199.
59. Niang, I., Osman-Elasha, B., Githeko, A., Yanda, P. Z., Medany, M., Vogel, A., ... & Nyong, A. (2008). Africa climate change 2007: Impacts, adaptation and vulnerability: contribution of working group II to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge.
60. Clerici, N., Bodini, A., Eva, H., Grégoire, J. M., Dulieu, D., & Paolini, C. (2007). Increased isolation of two Biosphere Reserves and surrounding protected areas (WAP ecological complex, West Africa). Journal for Nature Conservation, 15(1), 26-40.
61. Houehanou, T. D., Assogbadjo, A. E., Kakaï, R. G., Kyndt, T., Houinato, M., & Sinsin, B. (2013). How far a protected area contributes to conserve habitat species composition and population structure of endangered African tree species (Benin, West Africa). Ecological Complexity, 13, 60-68.




