Research Article - (2024) Volume 2, Issue 1
Post-Chemotherapy Cardiotoxicity in Women With Breast Cancer
2Colonia Centro, Facultad de Medicina, Benemerita Universidad Autonoma de Puebla, 4 Sur #104, Puebla , Mexico
3Hospital Regional de Alta Especialidad del Bajio, Servicio de Oncologia Radioterapia, Blvd. Milenio #130, Colonia San Carlos La Roncha, Leon 37660, Mexico
4Hospital Regional de Alta Especialidad del Bajio, Servicio de Cardiologia, Blvd, Milenio #130, Colonia San Carlos La Roncha, Leon 37660, Mexico
Received Date: Dec 22, 2023 / Accepted Date: Jan 10, 2024 / Published Date: Jan 24, 2024
Copyright: ©Â©2024 Maria Maldonado-Vega, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Maldonado-Vega, M., Cejudo-Arteaga, S., Ramirez-Reyes, M. A., Farias-Serratos, F., Teniente-Valente, R. (2024). Post-Chemotherapy Cardiotoxicity in Women With Breast Cancer. Int Internal Med J, 2(1), 01-13.
Abstract
Chemotherapy (CT) with anthracyclines can generate cardiotoxicity. The immunological system is part of the response to treatment with CT. Women with breast cancer were evaluated during their CT treatment, assessing their response to cardiotoxicity according to the treatment regimens and the classification of the cancer. Single CT and combined CT groups were made up (anthracyclines, Trastuzumab, and taxanes). Cardiotoxicity was measured as the Left Ventricle Expulsion Function (LVEF). The hematological response was measured start and at the end of treatment as the Lymphocyte/Macrophage Index (L/M) and the Lymphocyte/Platelet Index (L/P) The study included 451 women with breast cancer, among whom 230 underwent follow-up with LVEF on at least three occasions. Eight study groups were conformed based on their CT treatment. The comparison with LVEF was obtained between the baseline and the last LVEF. Combined CT diminished the LVEF, contrary to the CT of a single compound, which in addition showed a recovery from LVEF. The combined CT caused functional loss in terms of LVEF in 27% of the studied population. The L/M increased, and the L/P diminished in patients with the combined therapy. This suggests the participation of inflammation processes and vascular damage. Human epidermal growth factor receptor 2-positive (HER2+) and triple- negative patients have less survival, above all when the LVEF baseline was <55%. Anthracycline CT combined with other CT reduced the LVEF in women with breast cancer due to cardiotoxicity associated LVEF dysfunction. The use of only taxanes not exerted cardiotoxicity effect.
Keywords
LVEF, Cardiotoxicity, Breast Cancer, Anthracyclines, Trastuzumab, L/M Index, L/P index, Mexican Women.
Introduction
Chemotherapy (CT) in breast cancer has increased the survival of women with this diagnosis [1]. Anthracyclines, hormone therapy, and radiotherapy (RT) limit the growth of tumor and healthy cells. The secondary effects of the damage depend on individual sensitivity, the treatment regimen, and the dosing. Human Epidermal Growth Factor Receptor Type 2-positive (HER2+) breast cancer presents in 15% of Mexican women and treatment of the latter is based on hormone therapy with Tratuzumab [2-4]. Cardiological control is necessary in patients, above all when the CT is combined with anthracyclines, requiring the assessment of the ventricular function and of the function of harmful proteins within the care protocols. Cardiotoxicities represent the diminution of the LVEF because of the dysfunction of the myocytes in their contractility and cardiac insufficiency due to cell death [5-7]. The anthracyclines give rise to damage to the cardiomyocytes and to their mitochondria because of the increase of oxidative stress and the rise of free iron, which provokes molecular oxygen and apoptosis. These conditions can be reduced by the volume-correction, interruption of the oncological treatment, or cardioprotector medication [8-11].
Diminution of the LVEF by CT in women with breast cancer is added to other factors such as the following: age; obesity; smoking; hypertension; the cancer type (HER2+), and hypercholesterolemia [6,12]. Additionally, CT and radiotherapy (RT) affect the vascular endothelial cells, in addition to the aging of the heart and the presence of inflammatory processes generated from the deposit of visceral adipose tissue. Cardiotoxicity is dependent on the compound used; Trastuzumab interferes with the signaling of the neuregulin-1/HER, which is a cardioprotector; damage and susceptibility in cardiomyocytes is greater after exposure to the anthracyclines, with the diminution of the LVEF of between 4.4 and 18-6% and the increase of the cardiotoxicity-index ranges from 0.8-4-1% [11,13,14]. Cardiotoxicity related to Trastuzumab is reversible after the treatment interruption of 6-7 months, where partial or complete recovery of the LVEF is up to 80% spontaneously or with medical treatment; this condition does not occur with the anthracyclines, while patients with LVEF recovery tolerate and continue with Trastuzumab or with 5-Fluorouracil [14-17].
In RT for breast cancer, there exists the risk of cardiac insufficiency due to the inflammatory and profibrotic environment, leading to endothelial dysfunction, atherosclerosis, and myocardial fibrosis, as well as coronary arteriopathy or myocardiopathy. The left ventricular apex and the ventricular segments irrigated by the left anterior descending coronary artery are vulnerable to radiation due to their proximity to the thoracic wall. In breast cancer, the contrast of the RT of the left side vs. that of the right side entertains the risk of 29% and 22% of coronary disease and cardiac apoptosis, respectively [13,18,19].
The genetic basis of cancer is related with13/72 genes. with variants that involve cardiotoxicity with the diminution of the LVEF due to CT with Doxorubicin and Trastuzumab [20]. HER2+ type breast cancer is a homodimer that, on the use of Trastuzumab, a substitution takes place of interleukin for valine (HER 655 Ile/Val SNP) in the transmembrane domain, modifying the configuration to the heterozygote and an active state. On the other hand, the substitution of the proline for the alanine (SNP HER2 1170 Pro/ Ala) of the carboxyl domain leads to a structure of a secondary amide, allowing for the union of H and greater stability. These changes in the cardiomyocytes are highly dependent on HER2+ signaling for growth, yield, and survival. Thus, the blocking of HER2+ by Trastuzumab exerts damage and functional alteration [14].
Cardiotoxicity with a reduction of ≥10% in the baseline of the LVEF has a lower limit of 55% with symptoms of damage [5]. Other studies sustain that cardiac insufficiency occurs due to muscular weakness and a relaxation of a longer time in the contraction, causing difficulty in breathing, little tolerance for exercise, and much fatigue. The low LVEF implies ventricular thickening, which gives rise to a reduction in the ejection and low blood perfusion toward the tissues [13]. To evaluate this information in patients with CT treatment permits the physician to obtain clinical information for treatment adjustment and clinical follow-up. The work analyzed the effect of cardiotoxicity by single and combined CT and in Mexican women with breast cancer; the LVEF was analyzed, in addition to the immunological response of the lymphocytes, monocytes, and platelets.
Methods
This was a retrospective, observational, descriptive, and longitudinal study that was registered with the Ethics Committee of the Hospital Regional de Alta Especialidad del Bajío CEI-064- 2021. We declare that all methods were carried out in accordance with relevant guidelines and regulations and due to the retrospective nature of the study the hospital's research ethics committee waived the need of obtaining informed consent.
Population
The study included 451 cases of women diagnosed with breast cancer who were diagnosed and treated during the period from 2009-2019 and receiving surveillance monitoring in 2021. Of these women, the LVEF was studied in 230 (51%). The economic status of the study population corresponded to women living in rural areas with a low-to-medium economic level. The information was obtained from the electronic registry and hospitalization records deriving from the institutional archive. The follow-up of the cases was carried out at 5 years with the registry of recurrences.
From the clinical studies, the values of the leukocytes, monocytes, platelets, CA-15-3, and Ki-67 were obtained at diagnosis and at the end of the oncological treatments. The data were organized based on chemotherapy treatment into eight groups with treatments with a single compound with and multiplex-compound neoadjuvant therapy: Without chemotherapy; anthracyclines; Trastuzumab; taxanes, and the combination of these.
The group without CT corresponded to patients who, due to advanced disease, were only monitored palliatively for pain control; therefore, we may not have all of the follow-up information.
Percentage with Obesity
The study population was found to be 73% of women with overweight and obesity and who were considered a risk factor for treatment and chronic diseases [3].
Percentage of Left Ventricular Ejection Fraction (LVEF)
The LVEF was performed as a cardiac gamma gram, using a radioactive-material (thallium) MultiMate nuclide (MUGA) scanner, which was subsequently analyzed by imaging in the heart chambers and analysis by Wall Motion Score Index (WMSI).
The ejection fraction or LVEF is equal to the end-diastolic volume minus the end-systolic volume, divided by the end-diastolic volume and multiplied by 100. The normal LVEF ranges from 53-73%. An LVEF below 53% for women is considered low. An LVEF of less than 45% is considered a potential indicator of heart problems.
Percentage data were obtained by taking as a basis the initial LVEF test prior to any treatment and calculating the percentage value in the last LVEF test conducted at the end of treatment. The data were considered with reduction, maintenance and/or recovery with respect to the baseline.
The six patients who had an LVEF of less than 50% prior to CT treatment were eliminated from the post-CT analysis.
From among the study groups, a representative image was taken between the study groups at baseline and at the end of the LVEF analysis to contrast the changes and/or the maintenance of the LVEF.
The images annex the baseline comparison group and after treatment, according to the treatment group.
Cardiotoxicity Definition by CT
Cardiotoxicity was considered as a reduction of ≥10% with respect to the baseline LVEF with a lower limit without symptoms of damage [5,18].
Cardiac Risk Factors
The chronic diseases of the patients were taken into consideration: Diabetes mellitus, arterial hypertension, smoking or exposure to wood smoke, and the ingestion of alcohol are considered as risk factors for manifesting cardiotoxicity in addition to the treatment.
Statistics Analysis
The software program used was SPSS version 25 for Windows (IBM, Armonk, NY, USA). The information was organized into a database to obtain means, minimums and maximums, standard deviations of the quantitative variables, and frequencies in the qualitative variables. The quantitative variables were analyzed by multiple comparison (ANOVA) and the Tukey test; the significant difference in the comparisons was p <0.05. Student t two-tailed tests and Chi-square tests were applied to qualitative variables to assess significance. Linear correlation tests between variables were applied, as was the Chi-square test for risk factors.
Results
The data were obtained for assigning the patients into eight groups according to the CT applied. Groups 2-4 received one CT treatment, and groups 5-8 received more than two CT. By group, the percentage distribution of patients in these groups was the following:1) Without CT (2%); 2) Anthracyclines (3%); 3) Trastuzumab (4%); 4) Taxanes (3%); 5) Anthracyclines+Trastuzumab (2%); 6) Trastuzumab+Taxanes (7%); 7) Anthracyclines+Taxanes (36%), and 8) Anthracyclines+Trastuzumab+Taxanes (22%). The majority of patients were treated with CT of more than two compounds (groups 7 and 8) (Table 1). A total of 230 patients had an LVEF follow-up. Average age was 57-60 years, which corresponded to the CT groups with a single compound (anthracyclines and Trastuzumab), while for combined CT, the average age was 50-54 years. The highest Ki-67 presented in the group without treatment, in the patients without treatment, and in patients with combined therapy. The comorbilities were Diabetes mellitus, arterial hypertension, smoking, and obesity. In these groups, these illnesses are manifested at a time of more than 5 years; thus, they would not be related as CT effects. However, they are factors that could modify the response to CT, in addition to exerting effects on the endothelial tissue and on the myocardium in patients with breast cancer. The Body mass index (BMI) of overweight and obesity was observed in all the study groups.
|
Treatments chemotherapy |
Age at diagnosis (years) |
Menopause level |
Ki-67 (%) |
DM (years) 20% |
AHT (years) 28% |
Smoker (years) 22% |
IBM |
|
1)Without CT |
64 |
3 |
80 * |
15 |
15 |
2 |
32 |
|
2)Anthracyclines |
57 |
3 |
24 |
5 |
3 |
25 * |
34 |
|
3)Trastuzumab |
60 |
3 |
24 |
10 |
14 |
40 * |
29 |
|
4)Taxanes |
50 |
2 |
35 |
9 |
9 |
25 * |
32 |
|
5)Anthracyclines + Trastuzumab |
50 * |
2 |
40 |
13 |
15 |
35 * |
26 |
|
6)Trastuzumab + Taxanes |
54 |
2 |
49 * |
14 |
5 |
32 * |
28 |
|
7)Anthracyclines + Taxanes |
50 * |
2 |
45 * |
5 |
7 |
15 * |
29 |
|
8)Anthracyclines + Trastuzumab + Taxanes |
50 * |
2 |
48 * |
9 |
9 |
22 * |
29 |
DM = Diabetes mellitus; AHT = Arterial HyperTension. IBM= Index Body Mass; Chi2 p <0.05.
Table 1: Characteristics of Patients With Breast Cancer and Cardiovascular Risk Factors at Diagnosis
The average accumulated dose of CT shown in Table 2 corresponds to the consideration based on body surface/person. The dose-per- patient took into consideration the grade, the clinical state, the hormone receptors of the cancer, and age. The CT doses were found within the limits registered in protocols of breast cancer. The baseline of LVEF was reported as normal; after the CT of a single compound (groups 2, 3, and 4) did not exhibit changes, or even demonstrated recovery (positive). In terms of contrast, the combined CT (groups 5, 6, 7, and 8) presented a statistically significant LVEF, affecting least number of the studied population. The negative reduction of the LVEF is depicted in the groups of anthracyclines in combination with Trastuzumab. All the groups treated with single or combined CT revealed a statistically significant (p<0.04) reduction in platelets counts, with the combined treatment showing the greatest reduction. The average differences in the LFEV at the end of CT are presented in Figure 1; in the CT of a single compound, the FVEF was positive, with statistically significant changes between the groups.
Additionally, this condition is in contrast with the combined CT with negative effect and reductions between 3% and 6% in LVEF with significant differences between these groups. The differences observed by CT per group concerning the LVEF are depicted in Table 3. The single-compound CT, although exhibiting negative data regarding the LVEF, represents a one-fourth part of the patients with combined CT, with a reduction of around 10% in the LVEF in many cases.
|
Treatments chemotherapy |
Cumulative Dose (mg) |
LVEF Baseline (%) |
LVEF After (%) |
Modification LVEF (%) |
Platelets Baseline (cell x 10-3 uL) |
Platelets After (cell x 10-3 uL) |
|
1)Without CT |
0 |
61 |
65 |
7.84 |
254 |
230 * |
|
2)Anthracyclines |
4,359 |
64 |
65 |
3.31 |
289 |
210 * |
|
3)Trastuzumab |
6,683 |
68 |
68 |
1.70 |
258 |
217 * |
|
4)Taxanes |
1,106 |
64 |
69 |
8.63 |
326 |
287 * |
|
5)Anthracyclines + Trastuzumab |
11,983 |
65 |
61 |
-4.50 * |
322 |
207 * |
|
6)Trastuzumab + Taxanes |
8,584 |
64 |
61 |
-4.00 * |
289 |
240 * |
|
7)Anthracyclines + Taxanes |
7,133 |
66 |
64 |
-2.30 * |
290 |
232 * |
|
8)Anthracyclines +Trastuzumab + Taxanes |
13,347 |
66 |
64 |
-2.82 * |
281 |
233 * |
Mean baseline = without treatment, after = the end of chemotherapy treatments. Student t test (*) p <0.05two-tailed tests; Lymphocytes/platelets (L/P)
Table 2: Effect of Single and Combined Chemotherapy Treatment on LVEF Modification and Platelet Status
Figure 1: Effect of Treatment with Anthracyclines, Trastuzumab, and Taxanes in Women with Breast Cancer and the Effect of Cardiotoxicity Assessed by the LVEF Response. In the ANOVA Test, there were Significant Differences Between Single Treatments (p = 0.0001) and Differences in Combination Treatments (p = 0.002).
|
Treatments chemotherapy |
Single |
FEC |
FAC |
AC |
EC |
CMF |
TCH |
CBP |
|
2)Anthracyclines |
|
|
-5.6 |
-6.1 |
|
|
|
|
|
3)Trastuzumab |
-9.7 |
|
|
|
|
|
|
|
|
4)Taxanes |
-1.4 |
|
|
|
|
|
|
|
|
5)Anthracyclines + Trastuzumab |
|
-19.3 |
-20.0 |
|
-10.0 |
-8.4 |
|
|
|
6)Trastuzumab + Taxanes |
-11.3 |
|
|
|
|
|
-25.2 |
|
|
7)Anthracyclines + Taxanes |
|
-16.5 |
-8.9 |
-8.6 |
-28.2 |
-13.2 |
|
-5.8 |
|
8)Anthracyclines + Trastuzumab + Taxanes |
|
-11.4 |
-11.6 |
-16.9 |
-13.8 |
|
-9.8 |
-12.9 |
FEC = 5-Fluoroucil+ Epirubicin, Cyclophosphamide; FAC = 5-Fluoroucil+Adriamycin+ Cyclophosphamide; AC = Adriamycin+ Cyclophosphamide; EC = Epirubicin+Cyclophosphamide; CMF = Cyclophosphamide+Methotrexate+5-Fluoroucil; TCH = Trastuzumab+ Cyclophosphamide; CBP = Carboplatin. Mean value of LVEF in cases with negative effect. the single treatments subsequently recovered the LVEF.
Table 3: Negative Effect of the Anthracycline Type and their Combinations Used in Patients with Breast Cancer on the LVEF at the End of Treatment.
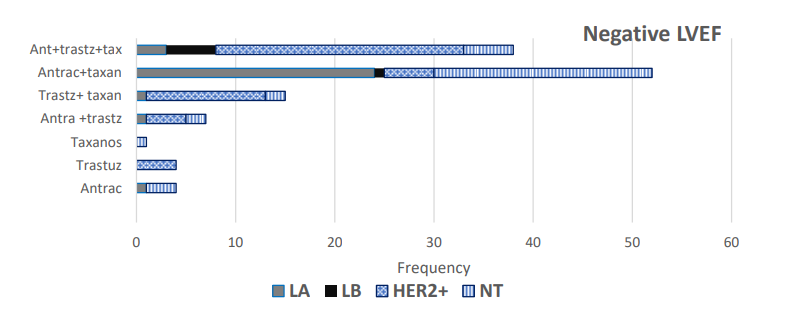
Figure 2: Distribution of Patients by Breast-Cancer Type in Single-Compound CT. Only Patients with a Reduction Response in the LVEF. LA = luminal A, LB = luminal B, HER2+ = Human Epidermal Growth Factor Receptor 2-positive. NT = Triple-Negative.
Figure 2 presents the distribution of patients based on the histological type of breast cancer and the CT. The majority groups were the following: LA; HER2+, and triple- negative. CT combined with anthracyclines+Trastuzumab+taxanes and anthracyclines+taxanes explains the reduction of the LVEF by means of their having many patients treated with this cancer.
Regarding the antigen CA 15-3, the baseline value of many patients was reduced after CT, except in group 7, where all the cases of triple negative and HER2+ were involved (Figure 2) (Table 4). It was observed that the combined CT involves an increase in the Lymphocyte/Macrophage Index (L/M) at the end of treatment, while the Lymphocyte/Platelet Index (L/P) increased at CT, independently of its being the single or combined treatment. The data suggest the inflammatory response, above all in the combined CT, response not observed in the independent counts of lymphocytes, monocytes and platelets.
|
Treatments (mg /m2) |
CA 15-3 Baseline (U/mL) |
CA 15-3 After (U/mL) |
L/M Baseline Index |
L/M After Index |
L/P Baseline Index |
L/P After Index |
|
1)Without CT |
10.4 |
12.6 * |
2.30 |
0.89 |
26 |
31* |
|
2)Anthracyclines |
22.0 |
14.1* |
3.33 |
2.13* |
28 |
41* |
|
3)Trastuzumab |
14.3 |
13.1 * |
3.00 |
2.83 |
26 |
29* |
|
4)Taxanes |
14.0 |
17.4* |
2.23 |
2.51 |
27 |
26 |
|
5)Anthracyclines +Trastuzumab |
29.4 |
7.1 * |
2.60 |
2.76 |
23 |
29* |
|
6)Trastuzumab + Taxanes |
32.2 |
7.1* |
2.67 |
3.37* |
25 |
28* |
|
7)Anthracyclines + Taxanes |
19.0 |
50.2 * |
2.65 |
3.02 * |
25 |
27* |
|
8)Anthracyclines+Trastuzumab + Taxanes |
21.3 |
15.0 * |
2.56 |
3.17 * |
24 |
28* |
CA-15-3 (cancer antigen). Multivariate ANOVA analysis and Tukey test p <0.05 (*) = statistical significance with respect to the individual treatment. L/M = Lymphocytes/Macrophages; L/P = Lymphocytes/Platelets.
Table 4: Effect of Chemotherapeutic Treatment with Anthracyclines, Hormone Therapy, and Taxanes Singly and Combined in Women with Breast Cancer on Cardiotoxicity Response, Tumor Marker, and the Lymphocyte/Macrophage Index (L/M).
In the face of the cardiological affectation caused by CT with greater sensitivity in the HER2 type. In Table 5, the distribution is presented of HER2+ patients with respect to the baseline of the LVEF. These patients had an average age of 52 years, with the extreme ages of 23 years and 85 years. It was observed that the CT with the greatest effect on the LVEF occurs in the group with a baseline of >66 years, where the average LVEF was 63 years, with a range of 33-79 years. Nonetheless, lowest survival presents in the groups with an LVEF baseline of >55-65 years and of >66 years. Contrary to the response demonstrated previously in the groups by CT, these patients exhibited a diminution of platelets and an increase in Interval Normalized Ratio (INR) coagulation time (Figures 3.a and 3.b), platelet response is dependent on the type of cancer and the combined CT, we suppose that the 64% treated patients with RT in addition exerts an influence on the platelet count, even when low toxicity is supposed for Modulated- Emission Radiotherapy (MERT) with low dispersion to other organs, finding a survival of above 65% for this HER2+ group.
|
|
LVEF BASELINE (%) |
||
|
LVEF Baseline |
< 55 (n = 11) |
>55.1 – 65 (n = 75) |
> 66 (n = 87) |
|
Age (years) |
52 [40 – 68] |
51 [30 - 78] |
52 [23 - 85] |
|
LVEF after |
66 [54 – 71] |
65 [60 – 79] |
63 [36 – 79] * |
|
Recovers LVEF (%) |
7 / 11 (63.6) |
30 / 75 (40) |
6 / 87 (7) |
|
INR baseline (second) |
1.040 |
0.975 |
1.001 |
|
INR after (second) |
1.142 |
1.434 |
1.082 |
|
Anthracyclines (mg/m2) |
4,765 [2,409 – 6,920] |
5,275 [360 – 16,560] |
5,259 [480 -10,250] |
|
Trastuzumab (mg/m2) |
7,446 [5,092 -10,200] |
7,511 [1,440 -11,621] |
6,897 [1,800 -22,572] |
|
Taxanes (mg/m2) |
738 [182 -1,560] |
1,211 [60 - 32,140] |
758 [110 – 2,680] |
|
Radiotherapy cases (%) |
7/11 (64) |
48/75 (64) |
59/87 (68) |
|
Left breast |
6 |
37 |
43 |
|
Right breast |
3 |
35 |
40 |
|
Two-way breast |
2 |
3 |
4 |
|
Survivors (%) |
9/11 (82) |
49/75 (65) |
57/87 (65) |
Table 5: Characteristics of HER2+ Breast-Cancer Patients on the Effects of Cardiotoxicity Following Drug Treatment and Radiotherapy
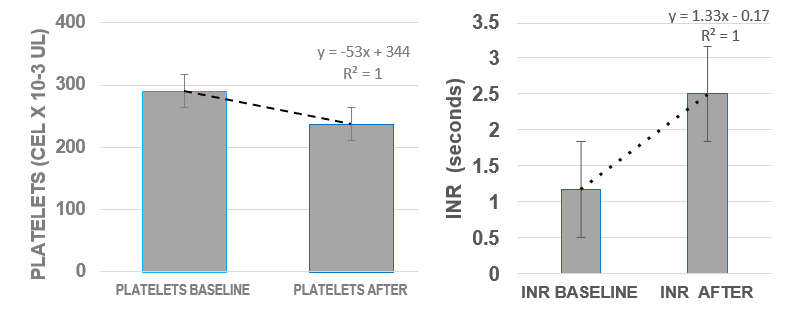
Figure 3.a: Effect of CT in HER2 Patients Diagnosed with Breast Cancer on Platelets at Baseline and After CT (anthracyclines+ Trastuzumab+taxanes).
Figure 3.b: Effect of CT in HER2 Patients Diagnosed with Breast Cancer on INR at Baseline and After CT (anthracyclines+Trastuzumab+ taxanes)
In Figures 4.a and 4.b. the combined CT (anthracyclines+Trastuzumab+taxanes) increase in platelets count and monocytes suggests damage at the vascular-wall level, which could initiate the platelet attraction repair response process plus other cells. In addition, the CT could generate an oxidative environment with cellular damage. Under these conditions, an increase is observed in the L/M and in the L/P, suggesting inflammatory processes and vascular damage in the patients during the treatment.
Figure 4.a: Effect of CT in Patients Diagnosed with Breast Cancer on the Lymphocyte/Macrophage Index (L/M) at Baseline and after CT (anthracyclines+ Trastuzumab+taxanes).
Figure 4.b. Effect of CT in Patients Diagnosed with Breast Cancer on the Lymphocyte/Platelet Index (L/P) at Baseline and After CT (anthracyclines+Trastuzumab+ taxanes).
The multifactorial analysis (Table 6) revealed that, among surviving and deceased patients, the statistical significance on comparing CT with a single compound with the combined CT, L/M are evident with single treatments in surviving women with single treatments, but in the combined therapies, significant differences were observed in the surviving women as well as in those who were deceased, while the L/P was only significant in some of the groups with combined therapy. It has been shown that the combined CT has a greater effect on the bodily economy of the patients, demonstrating that the hematological response and the cardiological response are intimately related.
|
Treatments |
|
PLATELETS |
CA 15-3 |
L/M INDEX |
L/P INDEX |
|
1)Without CT |
Live |
- |
- |
0.015* |
- |
|
Dead |
- |
- |
- |
- |
|
|
2)Anthracyclines |
Live |
- |
- |
0.020* |
- |
|
Dead |
- |
- |
- |
- |
|
|
3)Trastuzumab |
Live |
- |
- |
0.020* |
- |
|
Dead |
- |
- |
- |
- |
|
|
4)Taxanes |
Live |
- |
- |
0.006* |
- |
|
Dead |
- |
- |
- |
- |
|
|
5)Anthracyclines + Trastuzumab |
Live |
- |
0.004* |
0.024* |
0.02* |
|
Dead |
- |
- |
- |
- |
|
|
6)Trastuzumab + Taxanes |
Live |
- |
- |
0.017* |
- |
|
Dead |
- |
- |
0.001* |
0.04* |
|
|
7)Anthracyclines + Taxanes |
Live |
5.76 E-07 |
|
0.0018* |
|
|
Dead |
7.58 E-04 |
0.003* |
0.003* |
0.024* |
|
|
8) Anthracyclines + Trastuzumab + Taxanes |
Live |
3.55 E-06 |
0.010* |
0.001* |
0.003* |
|
Dead |
5.19 E-04 |
0.0001* |
0.021* |
- |
(-) data not statistically significant. (*) p <0.05 ANOVA analysis comparison between groups.
Table 6: Statistical Comparison Between Breast-Cancer Treatment in Survivors vs. Deceased Patients on the Effect On Platelets, the CA15-3 Antigen, the L/MI, and the L/PI
In Figure 5. The baseline image has a small area and represents normal function in contrast to the second image at the end of treatment which has a larger area and signifies myocardial damage and reduced left ventricular ejection, consistent with the effects that have been shown by type of treatment.
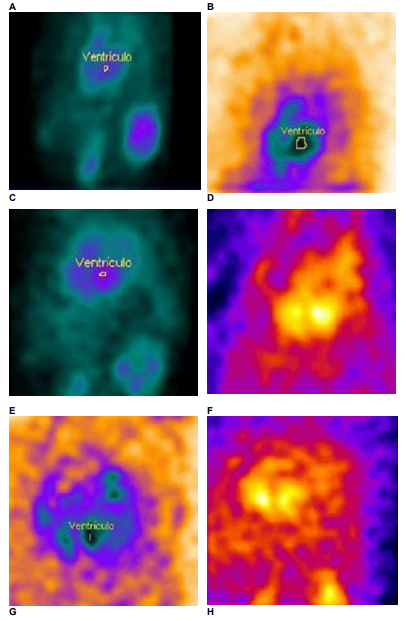
Figure 5: MUGA scan in patients with breast cancer, baseline LVEF and after chemotherapy. ventrículo= ventricle A) LVEF baseline(65.1%) and B) LVEF after (64.8%) anthracyclines + trastuzumab; C) LVEF baseline (79.5%) and D) LVEF after (40.9%) trastuzumab+taxanes; E) LVEF baseline (62.4%) and F) LVEF after (59.2) anthracyclines + taxanes; G) LVEF baseline (64.7%) and H) LVEF after (52%) anthracyclines + trastuzumab + taxanes. The baseline image has a small area and represents normal function in contrast to the second image at the end of treatment which has a larger area and signifies myocardial damage and reduced left ventricular ejection.
Discussion
In Mexican women with breast cancer, CT generates 27% of cardiotoxicity, nearly three times higher than that reported in similar studies with 10% [21]. The data indicate that single- component CT or its combination depends on the effect of inhibition and/or cellular death; the effects on the LVEF have been observed as reversible and irreversible, above all in combined therapies. We suppose that the comorbility of diseases and behaviors such as hypertension and smoking exert an influence on the LVEF. Also, the women of the present study presented a BMI greater than 26% in 73% of this population, with overweight and obesity very close to cardiotoxicity and the presence of diabetes, arterial hypertension, and smoking habits of 20%, 28%, and 22%, respectively, conditions that affect, among other organs, the endothelial and cardiac tissue.
CT-associated cardiotoxicity has been demonstrated during treatment; the cases affected are managed for recovery in the LVEF index; however, there is a residual effect in a small group of patients with 3% and 6% LVEF, less than those that established by the cardiological guides (a reduction of >10% from baseline) for its functional-damage classification [2,18]. Not with standing this, among the damages due to the disease and to the chronic illnesses, the data appear to imply systemic and endovascular damage, which contribute to an oxidative environment that is maintained during the treatment with CT. Therefore, the conditions of risk and cardiological dysfunction will be present, as others have reported [22,23]. The oncological–clinical results of the CT of patients with breast cancer considers the predisposition to damage in the cardiac function, above all in the presence of overweight and obesity; it has been demonstrated that the adipose tissue participates in the production of estrogens; thus, it is assured that these are factors of resistance to the hormone treatment (Anastrozole, Letrozole, and Exemestrane) [12,24].
While CT considers specific criteria of the patients, such as age, cancer type, pathological grade, and stage of disease advance, among these patients the election of chemotherapeutics and the intensity of application will limit the tumor response; however, it is important to provide follow- up for other organs, such as renal and hepatic function in the processing and elimination of metabolites [25]. It is important to consider the aldosterone–angiotensin–renin axis that contributes to the control of the arterial pressure and vascular integrity, a function that will be affected in patients who were from the beginning identified as diabetic and hypertensive; this would explain the changes between the baseline LVEF and the diminution of the LVEF after treatment in the studied patients, with damage to the vascular endothelium and consequently, to the cardiological function [22,23,26-28].
The cardiotoxicity analyzed as the diminution of the LVEF was statistically significant (p<0.001) in all the combined-therapy groups, where the greatest reduction occurred in the use of anthracyclines+ Trastuzumab (-4.50%) and Trastuzumab+taxanes (-4.00%). The use of taxanes in the combined LVEF therapy appears not to affect the LVEF, which seems to exert a cardiovascular protection effect, as has been suggested in other studies [10,18]. The single therapies do not affect the LVEF; this suggests a recovery in the cardiac-tissue cells, as has been demonstrated in therapies with Trastuzumab, on with drawing these chemicals, or on clinical control in the administration of the treatment [14,29,30].
It has been demonstrated that CT affects the breast-cancer cells on inhibiting the tyrosine kinase and the Vascular Endothelial Growth Factor (VEGF), but this also implies undesirable effects. It was identified that the activated kinase protein AMP and the platelet- derived growth factor are elements related with cardiomyocyte function; this explains the vascular damage associated with pharmaceuticals, which increases the concentration of reactive species and the limitation of nitrogen oxide (NO) in vasodilatation and that is influenced by the CT, contributing an increase of the systemic post-burden that will drive the cardiological function in patients with breast cancer [12,23,31]. The endovascular damage observed indirectly with changes involving a decrease platelet count in the combined therapies can be implicated in the recovery of the LVEF.
On the other hand, to the calculation of index, the high L/M and L/P in the combined CT treatments are expected in high-risk cancer, because the hematological system will exhibit a pressure response; in addition, the lymphocytes, macrophages, and platelets, under this condition, add up to overactivity in patients with overweight and obesity, who show a response of the lymphocytes and macrophages participating in a chronic low-grade inflammation. In this situation, the combined treatments increased with the pressure in the hematological response in addition to the damage in the LVEF.
In terms of cardiotoxicity in Mexican women with breast cancer, the LVEF demonstrated recovery in a treatment of a single compound, while the use of anthracyclines and their combination always showed a reduction of the LVEF. It has been reported that the anthracyclines provoke irreversible cellular damage during the first months, with damage to and the death of the cardiomyocytes. In addition, there occurs the accumulation of reactive oxygen species (ROS) that harm the cellular membrane. Although the oxidative environment is not limited to the heart, due to that the drugs demonstrate a systemic distribution, the anthracyclines reach the hematological cells, with a response in the L/M and L/P increase, explained as the multiorgan inflammatory state [1,8,13,20,32].
The toxicity and the hematological malignancy of the anthracyclines is limited to women older than 65 years of age, as stipulated in the international protocols; therefore, they are utilized in younger patients with breast cancer in whom a better immunological response is expected, with a lesser effect on their physical condition for them to be able to endure the treatment. The patients who received combined therapy for the control of the cancer in this study showed that nearly 49% had a reduction of the LVEF without recovery, in this manner confirming the damage to cardiomyocytes due to the oxidative environment and cellular death.
The patients identified as HER2+ and triple-negative treated with the combined regimens demonstrated the greatest number of cases with LVEF diminution. The patients treated with a single compound, such as Trastuzumab and taxanes, were differentiated for showing recovery in the LVEF. In the multiple comparison tests with statistical significance, age at diagnosis was the most important factor (p = 0.014) in the age extremes of 23 years and more than 70 years; the final L/M increased in all the groups in which anthracyclines were employed (p = 0.000, 0.006, and 0.0159) with an impact on the reduction of the CA153 antigen. The percentage of patients with HER2+ who did not survive their baseline LVEF was under 55%, even on their being treated with Trastuzumab, the treatment that was expected to provide the recovery of their LVEF, this signifying that the baseline function exerts an influence on the cardiotoxicity deriving from the CT treatment in breast cancer.
CT combined with anthracyclines affects the cardiomyocytes through oxidative stress and cellular death, which are inflammatory processes that affect the LVEF, implying the thickening of the LVEF area with MUGA images, reflecting a cardiac dysfunction (Figure 5).
At present, the control of the cancer indicates management with iRNA (interference RNA), in terms of their being inserted into the proteins that form complexes of control in the synthesis of the proteins and enzymes that silence genes linked with the development of cancer. The groups advanced in the theme ensure and trust in that there will be an impact on a diminution in the secondary effects caused by the currently employed drugs [33,34].
Conclusion
The patients administered with breast cancer in CT composed of multiple oncological drugs experience a deterioration in the left ventricular expulsion function (LVEF) in 27% of patients, and in the hematological response, with high L/M and L/P index as the indirect response of the inflammatory processes and endovascular damage, in Mexican women with breast cancer.
The use of anthracyclines exerts greater damage in the LVEF compared with the combination with taxanes and Tratuzumab. In contrast with single-compound CT, such as the taxanes, these have a lesser cardiotoxicity effect in the LFEV.
The use of Trastuzumab in HER2+ patients does not reveal a difference of cardiotoxicity with respect to other histological classifications.
Ethical Declarations
The authors declare that the information in the documentation respected the confidential data of the participants, and that there was no commercial and financial relationship that could be interpreted as potential conflict of interest. All data generated or analysed during this study are included in this published article CAMAMA L-M repository. The datasets generated and/or analysed during the current study are not publicly available due the authorised protocol limited the dissemination of study participant data, but are available from the corresponding author on reasonable request.
Contributions of the Authors
(SC-A data capture, data management); (MAR-R Reviewer of data and patient follow-up and the treatment surveillance of the radiotherapy treatment); (FF-S reviewer and translation of the English language); (RT-V Reviewer of the LVEF data, cardiotoxicity assessment), and (MM-V Document conceptualization, data capture, statistical capture, review).
Finding
This work was evaluated by the Investigation and Ethical and Investigation Committees of the Hospital Regional de Alta Especialidad del Bajío, León, Guanajuato, Mexico. The study utilized the respective databases; therefore, there was no implication of funds in the investigation. The follow-up of the patients and of the goods employed in their treatment correspond to the health-care regimens of the population, which is that is the responsibility of the Federal Secretary of Health, which includes the Hospitals High Specialties distributed.
Conflict of Interest
The authors declare that we have no conflict of interest in the information, data, or opinions expressed in this document. throughout the Mexican Republic.
Data Availability
Data available for review in the repository are integrated.
References
- Lee Chuy, K., & Yu, A. F. (2019). Cardiotoxicity of contemporary breast cancer treatments. Current treatment options in oncology, 20, 1-16.
- Nicolazzi, M. A., Carnicelli, A., Fuorlo, M., Scaldaferri, A., Masetti, R., Landolfi, R., & Favuzzi, A. M. R. (2018). Anthracycline and trastuzumab-induced cardiotoxicity in breast cancer. European Review for Medical & Pharmacological Sciences, 22(7).
- Cejudo-Arteaga, S., Guerrero-Ramos, M. Á., Kuri-Exome, R., Martínez-Cordero, E., Farias-Serratos, F., & Maldonado- Vega, M. (2022). Epidemiology of Breast Cancer in Mexican Women with Obesity as a Risk Factor. International Journal of Molecular Sciences, 23(18), 10742.
- Cameron, D., Piccart-Gebhart, M. J., Gelber, R. D., Procter, M.,Goldhirsch, A., de Azambuja, E., ... & Jackisch, C. (2017). 11years' follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive early breast cancer: final analysis of the HERceptin Adjuvant (HERA) trial. The Lancet, 389(10075), 1195-1205.
- Houbois, C. P., Nolan, M., Somerset, E., Shalmon, T., Esmaeilzadeh, M., Lamacie, M. M., ... & Thavendiranathan,P. (2021). Serial cardiovascular magnetic resonance strain measurements to identify cardiotoxicity in breast cancer: comparison with echocardiography. Cardiovascular Imaging, 14(5), 962.
- Jerusalén C, Lancellotti P, and Bae KS. HER2+ breast cancer treatment and cardiotoxicity: follow-up and management. Breast Cancer Res. Treat. 177(2), 237-250 (2019).
- Narayan, V., & Ky, B. (2018). Common cardiovascular complications of cancer therapy: epidemiology, risk prediction, and prevention. Annual review of medicine, 69, 97-111.
- O’Brien, M. E., Wigler, N., Inbar, M. C. B. C. S. G., Rosso, R., Grischke, E., Santoro, A., ... & CAELYX Breast Cancer Study Group. (2004). Reduced cardiotoxicity and comparable efficacy in a phase IIItrial of pegylated liposomal doxorubicin HCl (CAELYX™/Doxil®) versus conventional doxorubicin forfirst-line treatment of metastatic breast cancer. Annals of oncology, 15(3), 440-449.
- Padegimas, A., Clasen, S., & Ky, B. (2020). Cardioprotective strategies to prevent breast cancer therapy-induced cardiotoxicity. Trends in cardiovascular medicine, 30(1), 22- 28.
- Shah, A. N., & Gradishar, W. J. (2018). Adjuvant anthracyclines in breast cancer: what is their role?. The oncologist, 23(10), 1153-1161.
- Lee Chuy, K., & Yu, A. F. (2019). Cardiotoxicity of contemporary breast cancer treatments. Current treatment options in oncology, 20, 1-16.
- Khosrow-Khavar, F., Filion, K. B., Al-Qurashi, S., Torabi, N., Bouganim, N., Suissa, S., & Azoulay, L. (2017). Cardiotoxicity of aromatase inhibitors and tamoxifen in postmenopausal women with breast cancer: a systematic review and meta- analysis of randomized controlled trials. Annals of Oncology, 28(3), 487-496.
- Norton, N., Crook, J. E., Wang, L., Olson, J. E., Kachergus, J. M., Serie, D. J., ... & Fairweather, D. (2020). Association of genetic variants at TRPC6 with chemotherapy-related heart failure. Frontiers in Cardiovascular Medicine, 7, 142.
- Lunardi, M., Al-Habbaa, A., Abdelshafy, M., Davey, M. G., Elkoumy, A., Ganly, S., ... & Soliman, O. (2022). Genetic and RNA-related molecular markers of trastuzumab- chemotherapy-associated cardiotoxicity in HER2 positive breast cancer: a systematic review. BMC cancer, 22(1), 1-14.
- Udagawa, C., Nakamura, H., Ohnishi, H., Tamura, K., Shimoi, T., Yoshida, M., ... & Zembutsu, H. (2018). Whole exome sequencing to identify genetic markers for trastuzumabâ? induced cardiotoxicity. Cancer science, 109(2), 446-452.
- Giunta G, De Abreu M, Peralta S, Garagoli F, et al. Consenso deCardiología-Oncológica. Sociedad Argentina de Cardiología.Área de Consensos y Normas. 87(5), 10-96 (2019)
- Navarrete, S. (2018). Cáncer y cardiotoxicidad en la mujer. Revista Colombiana de Cardiología, 25, 144-153.
- Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, Deswal A, Drazner MH, Dunlay SM, Evers L R, Fang JC, Fedson SE, Fonarow GC, Hayek SS, Hernández AF, Kittleson MM, Lee CS, Link, MS, and Yancy W. AHA/ ACC/HFSA. Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 145(18), e895-e1032 (2022)
- Meattini, I., Poortmans, P. M., Aznar, M. C., Becherini, C., Bonzano, E., Cardinale, D., ... & Livi, L. (2021). Association of breast cancer irradiation with cardiac toxic effects: a narrative review. JAMA oncology, 7(6), 924-932.
- Serie, D. J., Crook, J. E., Necela, B. M., Axenfeld, B. C.,Dockter, T. J., Colon-Otero, G., ... & Norton, N. (2017). Breast cancer clinical trial of chemotherapy and Trastuzumab: potential tool to identify cardiac modifying variants of dilated cardiomyopathy. Journal of Cardiovascular Development and Disease, 4(2), 6.
- Murguía, S., Pagnussat, F., Simeone, A., Lujambio, M., Delorenzi, M., Acquistapace, F., ... & Ferrando Castagnetto,F. (2023). Perfil de liberação de biomarcadores miocárdicos em pacientes com doenças hemato-oncológicas que recebem transplante de células-tronco hematopoéticas. Revista Uruguaya de Cardiología, 38(1).
- Wu, C., Lu, H., Cassis, L.A., & Daugherty,A. (2011). Molecular and pathophysiological features of angiotensinogen: a mini review. North American journal of medicine & science, 4(4), 183.
- Cai, H., & Harrison, D. G. (2000). Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circulation research, 87(10), 840-844.
- Madkour, L. H. (2022). RNA Delivery Function for Anticancer Therapeutics. CRC Press.
- Reuvekamp, E. J., Bulten, B. F., Nieuwenhuis, A. A., Meekes, M. R. A., de Haan, A. F. J., Tol, J., ... & de Geus- Oei, L. F. (2016). Does diastolic dysfunction precede systolic dysfunction in trastuzumab-induced cardiotoxicity? Assessment with multigated radionuclide angiography (MUGA). Journal of nuclear cardiology, 23, 824-832.
- Foëx, P., & Sear, J. W. (2004). Hypertension: pathophysiology and treatment. Continuing education in anaesthesia, critical care & pain, 4(3), 71-75.
- SALUD, SEDENA, SEMAR. Guía práctica clínica. Consumo de tabaco y humo ajeno. Evidencias y recomendaciones. Consejo de Salubridad General. 1-66 (2012).
- Slamon, D. J., Leyland-Jones, B., Shak, S., Fuchs, H., Paton, V., Bajamonde, A., ... & Norton, L. (2001). Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. New England journal of medicine, 344(11), 783-792.
- Fallah-Rad, N., Walker, J. R., Wassef, A., Lytwyn, M., Bohonis, S., Fang, T., ... & Jassal, D. S. (2011). The utility of cardiac biomarkers, tissue velocity and strain imaging, and cardiac magnetic resonance imaging in predicting early left ventricular dysfunction in patients with human epidermal growth factor receptor II–positive breast cancer treated with adjuvant trastuzumab therapy. Journal of the American College of Cardiology, 57(22), 2263-2270.
- Ewer MS, Vooletich MT, Durand J, et al. Reversibilidad de la cardiotoxicidad relacionada con Trastuzumab: nuevos conocimientos basados en el curso clínico y la respuesta al tratamiento médico. J. Clin. Oncol. 23, 7820-7826 (2005).
- Varghese, S. S., Johnston, W. J., Eekhoudt, C. R., Keats, M. R., Jassal, D. S., & Grandy, S. A. (2021). Exercise to reduce anthracycline-mediated cardiovascular complications in breast cancer survivors. Current Oncology, 28(5), 4139-4156.
- Ramos-Peñafiel, C. O., Santos-González, B., Flores-López,E. N., Galván-Flores, F., Hernández-Vázquez, L., Santoyo- Sánchez, A., ... & Martínez-Tovar, A. (2020). Utilidad de los índices neutrófilo/linfocito, monocito/linfocito y linfocito/ plaqueta para el pronóstico de complicaciones asociadas a COVID-19. Gaceta médica de México, 156(5), 413-419.
- Meattini, I., Poortmans, P. M., Aznar, M. C., Becherini, C., Bonzano, E., Cardinale, D., ... & Livi, L. (2021). Association of breast cancer irradiation with cardiac toxic effects: a narrative review. JAMA oncology, 7(6), 924-932.
- Habeeb, E., Aldosari, S., Saghir, S. A., Cheema, M., Momenah, T., Husain, K., ... & Ansari, R. A. (2022). Role of environmental toxicants in the development of hypertensive and cardiovascular diseases. Toxicology Reports, 9, 521-533.




