Research Article - (2025) Volume 9, Issue 3
Polycystic Ovary Syndrome: Clinical Assessment, Diagnosis and Challenges in its Management
Received Date: Jun 02, 2025 / Accepted Date: Jul 15, 2025 / Published Date: Jul 30, 2025
Copyright: ©2025 Lukman Yusuf, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Yusuf, L., Negash, S. (2025). Polycystic Ovary Syndrome: Clinical Assessment, Diagnosis and Challenges in its Management. J Gynecol Reprod Med, 9(3): 01-15.
Abstract
Polycystic ovarian syndrome (PCOS) is one of the most common endocrinopathological disorders affecting the hypothalamic, pituitary, ovarian and adrenal axis of women in their reproductive ages. It is recount able from fetal life in utero through menarche, puberty, the reproductive years, and menopause and beyond. It was first described in 1935. The prevalence rate is highly variable and ranges from 6 to 20%. It is classically articulated by oligo-amenorrhea, hyperandrogenism and polycystic ovarian morphology. Under the classification of the syndrome are registered several phenotypes and sub-phenotypes. The pathophysiology is not clearly understood and an array of several causal considerations is in existence. The presentations vary from person to person and it is not always easy to cure it or formulate a linear implementable universal scheduling; and therefore the management tends to be highly individualized.
PCOS is trending as a significant reproductive health related clinical condition in an Ethiopia setup accounting for up to 8.7% of our studied population. There exists a limited clinical data in the country and it deserves our attention within the context of its prevalence, investigations, available health care resources, exploring treatment availabilities and future opportunities impacting their outcomes. Hence, this article is intended to raise awareness among patients and clinicians, and above all empower service providers with up to date information on its presentations, clinical management and enlighten them with future prospects for research.
Keywords
PCOS, Oligo-Amenorrhea, Hyperandrogenism, Insulin Resistance, Metabolic Syndrome, Addis Ababa, Ethiopia
Abbreviations
AES: Androgen Excess (PCOS) Society
AFAB: Assigned Female at Birth
AMH: Anti-Müllerian hormone
ART: Assisted Reproductive Technology
BMI: Body Mass Index
COCP: Combined Oral Contraceptive Pills
CRP: C-reactive protein
CTS: Computed Tomography Scanning
DM: Diabetes Mellitus
FMT: Fecal Microbiota Transplant
FNPO: Follicle Number per Ovary
FSH: Follicle Stimulating Hormone
GnRH: Gonadotropin Releasing Hormone
HAIR-AN: Hyperandrogenemia, Insulin Resistance, Accanthosis Nigricans
hCRH: Human Corticotropin-Releasing Hormone HDL: High Density Lipoprotein
IUGR: Intra-uterine Growth Restriction
IVM: In Vitro Maturation
LDL: Low Density Lipoprotein
LH: Luteinizing Hormone
LOD: Laparoscopic Ovarian Drilling
MILD: Minimally Invasive Laparoscopic Drilling
MRI: Magnetic Resonance Imaging
NAFLD: Non-alcoholic Fatty Liver Disease
NGO: Non-Governmental Organization
NIH: National Institute of Health
PCOM: Polycystic Ovarian Morphology
PCOS: Polycystic Ovarian Syndrome
PRP: Platelet–Rich-Plasma
SGA: Small for Gestational Age
SLS: Stein-Leventhal Syndrome
USG: Ultrasonography
WHO: World Health Organization
Introduction
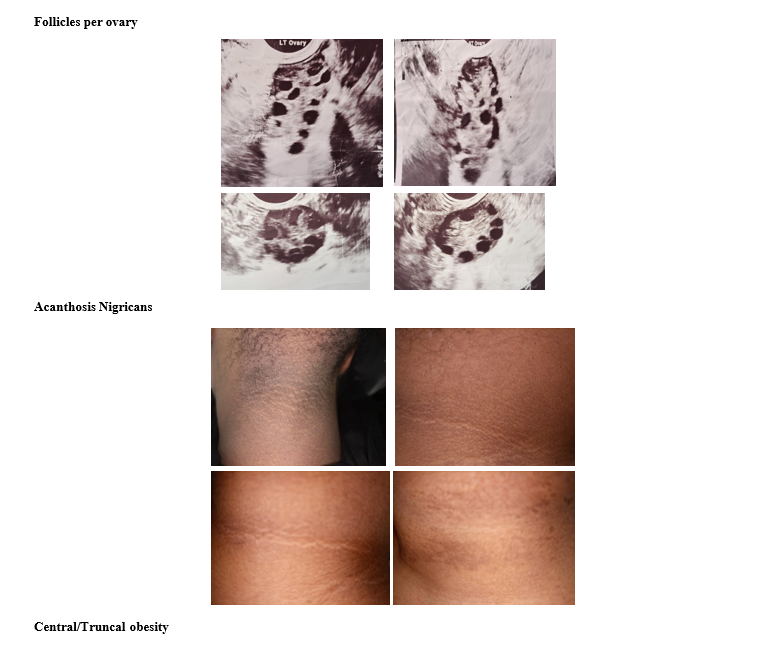
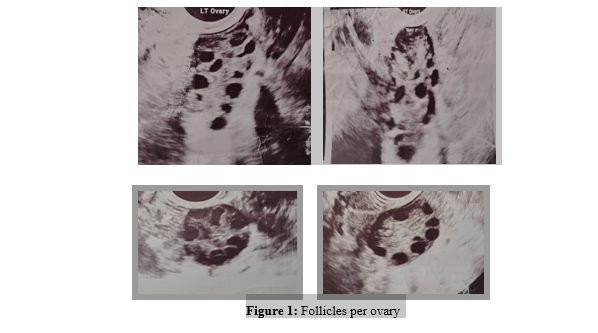
The size of a normal ovary on the average is 3.5cm long, 2.5cm wide and 1.5cm thick with 6-10 antral follicles and a volume of 3-6ml. On the other hand, the classic description of a polycystic ovarian morphology is profiled as 12 or more small follicles per ovary with necklace pattern or string of pearl sign that often do not mature and release an egg although the follicle number per ovary (FNPO) has been raised to >20 follicles with the introduction of advanced high resolution newer ultrasound technology. It is to be noted that each of these follicles may measure with a range of 2-9mm in diameter, a volume of 10ml or more in accordance with the enlarged ovarian sizes that are engaged in concurrent processes of luteinization of theca interna and reduction of granulosa cells(hyperthecosis).
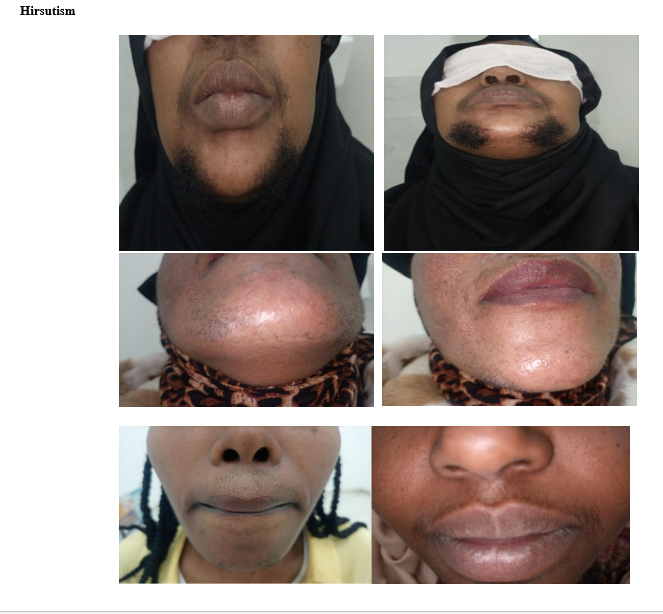
These descriptions of enlarged ovaries, multiple cysts or follicles need not be constantly present in all cases and hence not considered as an absolute phenomenon. Polycystic ovarian syndrome is mainly characterized by irregular menstrual bleeding (infrequent and prolonged), hyperandrogenemia (acne, hirsutism, oily facial skin, thinning of hair over the scalp, male type of baldness) and non-ovulating follicles often referred to as cysts. It generally affects 6-20% of women mostly in their second or third decade of life. PCOS heralds its commencement with the onset of menarche although its onset is understandably delayed especially when and if associated with obesity and in the adolescent [1-3].
PCOS is a common endocrine imbalance. The different set of combinations according to the commonly employed Rotterdam criteria (2003) includes ovarian dysfunction, hyperandrogenemia and polycystic ovaries whereby any two of three may hinge the diagnosis [4]. The clinical manifestations may encompass an abnormal, infrequent, prolonged and chronic an-ovulatory bleeding starting with onset of menarche. Subsequently the development of early adrenarche and pubarche, acne, oily facial skin and obesity as features of puberty overlapping with the entity is not an uncommon encounter. However, it is to be understood that thelarche is quite independent and assumed to be unaffected. The spectrum continues through out the woman’s life with obstetrical and gynecological serious complications and later in life with metabolic syndrome complexes [5].
The central theme of PCOS in women and people assigned female at birth (AFAB) is most commonly attributed to delayed fertility, subfertility or infertility even though most can still successfully carry out a pregnancy but yet prone to develop and complicate possibly encountering miscarriage, hypertensive disorders of pregnancy, gestational diabetes, macrosomia or even IUGR and SGA, preterm births or increased cesarean deliveries. Thus, the fertility challenges are enormous and this demands thorough understanding of the pathophysiology and its perspective management strategies [6].
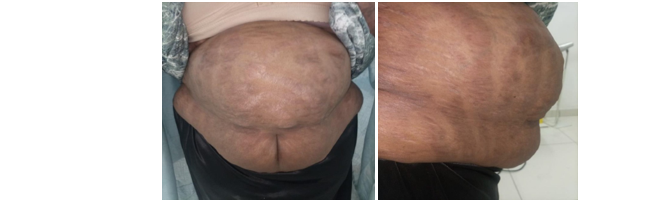
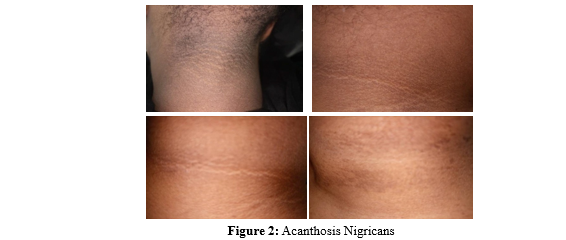
Of the multifold possible presentations, the dermatological manifestations that prevail in PCOS are demonstrated by darkening of the skin around the neck, axillae, breasts and groin which is referred to as acanthosis nigricans and sometimes skin tags in the axillae and neck. The presence of low grade inflammation with raised CRP (C - reactive protein) coexisting with leukocytosis or steatohepatitis may mask or complicate the presence of other infectious processes. The psychosocial hitches range from anxiety, depression, low self esteem, body image disruptions and to eating disorders or bulimia nervosa. The unopposed and continuous proliferation of the endometrial lining by hyper-estrogenic state which is not counter balanced by progesterone because of the absence or lack of the LH surge may eventually result in the development of endometrial cancer. Furthermore, under the umbrella of a metabolic syndrome exist a cluster of conditions comprising of high blood pressure, high blood sugar (Type II DM, pre-diabetes), obstructive sleep apnea, unhealthy lipid profiles contributing to severe non-alcoholic fatty liver disease (NAFLD) also named non-alcoholic steatohepatitis (NAHSH) all of which may markedly increase the risk of future cardiovascular diseases [7]. It is noteworthy to acknowledge the fact that age is the limit for the development of the metabolic syndrome and the cumulative insults incurred in relation to the continuum of the pathophysiology of PCOS over a lengthy period of time. The investigations are extensive and to reach at the diagnosis with the choice of the existing criteria is challenging and the rationale requires a deeper knowledge and substantiated approach.
The overall treatment strategy include the psychological support, medical therapy with combined oral contraceptive pills if a desire to achieve a pregnancy is not an issue; and when present a variety of options are offered to and include letrozole or clomiphene citrate, metformin or acarbose in case of insulin resistance and abnormal glycemic profile, myo-inositol and dopamine antagonists like bromocriptine or cabergoline in case of hyperprolactinemia; and further suggested advanced treatment modalities are to be reviewed. Additional hormonal treatments employed are glucocorticoids, gonadotropins (FSH/LH) and pure FSH/hCG. Strict follow up for ovarian hyperstimulation syndrome should always be considered and undertaken. Assisted Reproductive Technologies for thosewho can economically afford and opt for remains an alternative recommendation. The surgical options which are very much dependent on the sizes are laparoscopic drilling and wedge resection in order to circumvent the lengthy course of treatment and handiness of the other composite resources [1,8].
The above presentation is a brief clinical preview of the syndrome. The article intends to explore the very relevant and contemporary matters identifying areas for future undertaking and considers the management options in view of the abundant existing literature reviews.
Literature Review
PCOS was first described by two American gynecologists in 1935, and hence bearing their names and also referred to as Stein- Leventhal Syndrome (SLS). Furthermore, it is often called the “O” syndrome emphasizing and deciphered as over nourishment (obesity), ovarian confusion/dysfunction/disruption, ovarian cysts (polycystic ovary morphology) and over production of androgens and insulin. There are 16 different phenotypes and as well sub- phenotypes that impact the overall pictures of the syndrome [9, 10]. Of interest, a sub phenotype of PCOS designated as HAIR- AN syndrome is characterized by a triad of hyperandrogenism, insulin resistance and acanthosis nigricans. Under different classifications, there are four types of PCOS depicted as insulin- resistant, inflammatory, hidden-cause and pill-induced in addition to the four phenotypes (ABCD); and moreover those based on severity of the symptoms as asymptomatic, mild, classical and metabolic; and according to grouping in relation to response to hCRH. Under a new name, it is alluded to as a “functional metabolic-hyperandrogenic syndrome. In a nutshell, it is reasonable and sound rationale to have an elaborate knowledge in the existing concoction in the naming of this heterogeneous endocrinopathy and the designation of the variety of the diagnostic criteria although the consensual approach embraces the Rotterdam criteria (2003) [11-15].
PCOS is characterized by a wide range of signs and symptoms that commonly include infrequent abnormal uterine bleeding, hyperandrogenism, insulin resistant diabetes mellitus, obesity, dermatological, respiratory, endometrial cancer and as well other metabolic ailments. The factors blamed in the causation of PCOS are many and to name a few include in utero fetal androgen exposure, genetics and genetic inheritability, environment and pollutants, industrial compounds (bisphenyl A), gut microbiota and dysbiosis, smoking, insulin resistance and obesity and the latter with its causal and effect relationship. In the quest of definitive treatment of PCOS, molecular etiological diagnosis has made headways by targeting XIST, ETS2, ceRNA, mRNA and six other biomarkers especially by influencing ART outcomes [16-18].
PCOS remains a heterogeneous disorder and it is worth considering time factor and avoiding undue delay in establishing the diagnosis as 70% of such patients may not be totally diagnosed. Hence, it is of paramount importance that with thorough clinical assessment, appropriate and detailed hormonal and metabolic blood tests in conjunction with imaging trans-abdominal and trans-vaginal USG and as well pelvic MRI and CTS as deemed necessary in order to mitigate long term health effects and protracted delays in the final diagnosis and timely initiation of treatment [1].
The overall attempt to over simplify the conglomerate signs and symptoms of PCOS has never been so easy and conclusive and equally the management remains complex. To start with, the life style management approach promotes diet with reduced glycemic/ carbs intake, intermittent fasting and giving up smoking with regular physical exercise with the intention of aiming to reduce weight by at least 3-15% and decreasing adipose tissue and insulin resistance and minimizing peripheral aromatization, estrogens and further stimulation of FSH to avert the pathophysiological mechanisms in place. Myo-inositol and D-chiro-inositol increase insulin sensitivity, clinical pregnancy rates and improves quality of eggs and embryos in ART utilizing IVF and also under normal circumstances. Resveratrol, berberine and N-acetylcysteine (NAC) are emerging additional natural supplements and therapies with their diverse capability of modulating the disorders caused by PCOS [19,20].
Emotional wellbeing that may be expressed in a form of anxiety, depression, infertility, dismay with body image and related self esteem concerns ought to be stabilized. COCPs when pregnancy is not desired may regulate and reduce symptoms, hirsutism and subsequent development of endometrial cancer and can directly modify the feedback mechanism, shutdown ovarian functions and reduce outpouring of estrone. GnRH and SHBG can also be prescribed for those not intending to become pregnant [4,19].
The hyperandrogenism effect as stated above can be managed with COCPs but stronger pharmacological substances like spironolactone, flutamide, eflornithine, onandrolone, cyproterone acetate and finastride can be counted upon if the signs and symptoms of these happen to be of a major concern and focus of the patient but with dire caution weighing the pros and cons of such course of therapy. SGLT2 inhibitors, GLP-1 receptor agonists, kisspeptin agonists and spirolactine are of newer alternative medical and hormonal therapies [20]. Anti-diabetics (acarbose, metformin) have their place in countering the effect of insulin resistance with or without ovulation induction (clomiphene / letrozole) depending on the ultimate goal of trying to conceive [21].
Supplementary Vitamin D and calcium enhance ovulation, regularize menstrual cycles, improve androgenic effects and may help in weight loss. If administered in Vitamin D3 deficiency state, its supplementation alone has multifold therapeutic advantages by regulating and lowering blood testosterone levels, modifying insulin sensitivity, improving the menstrual periods and achieving eumenorrhoic states as well as promoting folliculogenesis, ovulation and concurrently imparting a counter effect on miscarriage and fostering healthy procreation and pregnancy outcomes. It is to be noted too that its anti-inflammatory property plays a significant role not only in ovulation but implantation processes, regulating genes and on metabolic disorders [22,23].
PCOS has been implicated in the causation of chronic low grade inflammatory processes affecting insulin sensitivity, ovarian confusion, and cardiovascular disease related risks. Aspirin has the property of reducing inflammation markers in PCOS. Thus, low dose aspirin has a place in the prevention of pre-eclampsia; improve blood flow and its blood thinning effect helps in facilitating fertility treatment and cardiovascular problems. The beneficial effects of anti-inflammatory properties of low dose aspirin in combination with clomiphene citrate, letrozole or tamoxifen is considered to be safe and effective [24-26]. The recent advanced fertility treatment protocols introduced gut microbiome–based probiotics with Fecal Microbiota Transplant (FMT), ovarian Platelet–Rich-Plasma (PRP) which is injected into the ovaries to restore the normal ovarian function and improve fertility and additionally an in vitro maturation (IVM) in a laboratory setting [27]. Surgical management alternatives depending on surface area ratio include Laparoscopic Ovarian Drilling (LOD) or Minimally Invasive Laparoscopic Drilling (MILD) and when and where it is not feasible; a wedge resection under mini laparotomy can be an offered choice [28].
The subject is diverse with multisystem involvement as a commonly encountered reproductive health issue. The diagnostic approaches are vast and the treatment options are ever changing and getting sophisticated. Cost, time and resourceful facilities and well trained personnel are required to promptly address the individual health needs. Therefore, it is timely to address this issue in our Ethiopian context and update our colleagues with up-to-date scientific information and enlighten and empower them with the broader overall management strategies
Methods
The study was conducted from January 1, 2024 to December 31, 2024 GC involving all patients attending the ObGyn unit of Yehuleshet Medical Systems. It is a privately owned institution with multidisciplinary units and providing high level clinical services.
The design was an institution-based descriptive cross-sectional study using a predesigned and selected relevant title-specific dependent and independent variables that could be reliant on availability, cost effectiveness, efficiency, standard and acceptability of the referred-to facilities. The study participants were all women in their reproductive age group of 18-45 years and basically fulfilling the Rotterdam criteria (2003). The data was collected in line with the predetermined essential set of variables.
The data processing and analysis was performed using Epi Info. The data quality assurance was ascertained in line with the selected set of variables. The results were presented using frequencies and percentages; tables and figures were used to present the findings. The findings of the study are to be used only for academic and public health matters.
The justification for adopting the Rotterdam criteria (2003) is based on the consensual approach that it is broad spectral including hyperandrogenic cases and thus impressively inclusive but compromising its sensitivity and specificity. In contrast, the Androgen Excess-PCOS Society (2006) is stringent and focuses on hyperandrogenemia both biochemically and clinically thus making it ideal to identify and highly specific for diagnosing the scenario but with lower sensitivity as it may miss milder cases. The NIH criteria (1990) also put emphasis on ovulatory dysfunction and hyperandrogenism with moderate sensitivity and specificity but lacking the inclusiveness to conduct such a study [29].
The inclusion criteria were all women presenting with irregular menses, signs and symptoms of hyperandrogenism and polycystic ovarian morphology in accordance with the Rotterdam Criteria (2003) and/or other additional suspected clinical presentations like metabolic syndrome and dermatological findings of topic specific significance as shown in Table I and Table 2 in keeping with the recognition of the other major criteria [4].
As PCOS is a diagnosis of exclusion, the clinical entities mimicking it to be excluded were in fact conditions that are actual differential diagnosis and/or clinical presentations short of copycatting the syndrome that would include premature ovarian failure, late onset Congenital Adrenal Hyperplasia, gonadal dysgenesis, Cushing syndrome, hypothyroidism, use or administration of androgenic steroids, familial or idiopathic hirsutism, virilizing ovarian tumors and metabolic syndromes.
|
Required Features |
Rotterdam (2003) |
AES (2006) |
NIH (1990) |
|
Two of the three but with no focus |
Two of the three but with focus |
All of the three but with focus |
|
|
Oligo-amenorrhea ( Ovarian dysfunction, oligo- anovulation ) |
Optional |
Required |
Required |
|
Hyperandrogenism ( Clinical / Biochemical ) |
Optional |
Required |
Required |
|
PCOS (US) |
Optional |
Not required |
Not required |
|
Exclusion of other disorders^ |
Required |
Required |
Required |
|
Dermatological* |
* |
* |
* |
Table 1: Diagnostic Criteria & supplementary clinical signs and symptoms
Congenital adrenal hyperplasia, gonadal dysgenesis, Cushing’s syndrome, hypothyroidism, use of or administration of androgenic steroids, familial or idiopathic hirsutism, metabolic syndromes and virilizing ovarian tumors.* Acanthosis nigricans, alopecia, male pattern of baldness, skin tags, acne, oily facial skin- Cross-cutting findings that could be clinically present; but not required in the specific criteria
This Table 1 relates to the three main criteria regarding history of menstrual irregularities (Oligo-amenorrhea), clinical signs and symptoms of hyperandrogenemia, ultrasound verified polycystic ovaries and dermatological effects of the syndrome and stressing the need to exclude other similar entities.
|
Rotterdam (2003) |
Phenotypes |
Frequency |
Percent |
Cum Percent |
|
|
HA + PCOM |
A |
16 |
9.6% |
9.6% |
|
|
OD + PCOM |
B |
150 |
90.4% |
100.0% |
|
|
Total 166 |
100.0% |
100.0% |
|
||
Table 2: Phenotype diagnosis under Rotterdam criteria (2003)
Table 2 summarizes the distribution of the clinical diagnosis of the study population according to the two phenotypes under Rotterdam criteria (2003).
HA: Hyperandrogenemia OD: Ovarian Dysfunction PCOM: Polycystic Ovarian Morphology
Results
A total of 2875 clients in their reproductive ages between 15 to 45 years visited the ObGy unit of the Yehuleshet Medical Center for obstetrical and gynecological alleged complaints and routine recalls for screening during the period extending from January 1, 2024 to December 31, 2024 GC. Of the 1903 patients who were worked up for history pertaining to fertility issues, abnormal uterine bleeding with an infrequent and out of range according to current understanding of the length of normal menstrual cycle varying from 24 days onwards and no more than 38 days, or other signs and symptoms related to the syndrome, 166 of them were identified and confirmed to have PCOS; and the calculated prevalence rate was put at 8.7% (166/1903). The mean age was 29.38+SD3.62 years with a range of 20-38 years.
The entrants were cleared off for any family history of similar illnesses and/or treatment for similar disorders. All possible differential diagnosis mimicking PCOS were excluded among the studied population. There was no admittance of smoking of cigarettes, hookah and other substances. They were all normo- glycemic, non-hypertensive and not on medication for impaired lipid profile. All of the subjects basically had both the trans- abdominal and trans-vaginal ultrasound examinations and the features of the reports for the establishment of the PCOS were as entered by the radiologists and concluded accordingly.
The studied population belonged to the two major religions namely of Christianity 114(68.7%) and Islam 52 (31.3%) and were predominantly from Ethiopia. The majority 94 (56.6%) were college graduates and governmental and non governmental employees while 43 (25.9%) reported as housewives and the rest self employed. The married group accounted for 156 (94.0%) and 10 (6%) reported to be single. Their fertility pattern revealed that 138 (83.1%), 23(13.9%) and 5(3.0%) were nullipara, primipara and those with two or more children, respectively. It was found out that only 32(19.3%) had history of either induced or spontaneous abortions in the past and none had ectopic pregnancy. Upon clinical presentation, the most frequented complaints related to a desire to have a child accounting (infertility) for 151 (91.0%), oligo- amenorrhoea in 14 (8.4%) patients and only one with hirsutism.
|
Variable |
Mean |
Median |
Mode |
Range |
|
Age (years) : |
29.38+SD3.62 |
30.00 |
28 |
20-38 |
|
Ethnicity: |
Frequency |
Percent |
Cum Percent |
Graph |
|
Amhara |
96 |
57.8% |
57.8% |
|
|
Somali |
40 |
24.1% |
81.9% |
|
|
Tigre |
11 |
6.6% |
88.5% |
|
|
Oromo |
9 |
5.4% |
93.9% |
|
|
Guraghe |
9 |
5.4% |
99.3% |
|
|
Others |
1 |
0.6% |
99.9% |
|
|
Total |
166 |
100.0% |
100.0% |
|
|
Occupation: |
|
|
|
|
|
Housewife |
43 |
25.9% |
25.9% |
|
|
Self employed |
28 |
16.9% |
42.8% |
|
|
Gov employee |
94 |
56.6% |
99.4% |
|
|
Unemployed |
11 |
0.6% |
0.6% |
|
|
Total |
166 |
100.0% |
100.0% |
|
Table3: Socio- Demographic Characteristics
The data analysis as shown in Table 3 revealed that most were young in their second and third decade of life, from within Ethiopia (Addis Ababa Administrative City, Federal RegionalStates including Somali region), married and belonging to various ethnic groupings. The government employees were mainly from Ethiopia and with fair income for the existing scale of payment.
|
Age at Menarche |
Frequency |
Percent |
Cum Percent |
Graph |
|
11 |
6 |
3.6% |
3.6% |
|
|
12 |
34 |
20.5% |
24.1% |
|
|
13 |
57 |
34.3% |
58.4% |
|
|
14 |
61 |
36.7% |
95.2% |
|
|
15 |
8 |
4.8% |
100.0% |
|
|
Total |
166 |
100.0% |
100.0% |
|
Table4 :Participate Age at Menarche in Years
Mean: 13.19 + .94 years Median: 13 years Range: 11-15 years
|
Fertility pattern |
Frequency |
Percent |
Cum Percent |
Graph |
|
Normal |
2 |
1.2% |
1.2% |
|
|
Hypogondotropic / hypogonadism (WHO I) |
1 |
0.6% |
1.8% |
|
|
Normogonadotropic/ anovulatory (WHO II) |
163 |
98.2% |
100.0% |
|
|
Hypergonadotropic/ hypogonadism (WHO III) -:-: |
- |
- |
- |
- |
|
Total |
166 |
100.0% |
100,0% |
|
Table5:WHO Based Classification of Anovulation
Excluded (Premature ovarian insufficiency/menopause) WHO I is practically hypothalamo-pituitary failure ( Kallman syndrome, crush diet, excessive exercise, stress) with low GnRH, FSH, LH and estrogen and the only case registered was suffering of infertility related stress and depression, while WHO III is hy- pergonadotropic hypogonadism suggestive of premature ovarian failure/menopause or gonadal dysgenesis. WHO II is practically eugonadotropic anovulatory disorder with normal or high LH and estrogen values and with responsive ovaries whereby a diagnosis of PCOS can be entertained.
|
Parameters |
Units |
Mean |
Median |
Mode |
Range |
|
LH |
(mIU/ml) |
24.90 +SD12.99 |
24.00 |
32.00 |
2-67 |
|
FSH |
(mIU/ml) |
8.39 + SD3.11 |
8.00 |
8.00 |
3-18 |
|
LH/FSH ratio |
2.97 : 1.0 |
||||
|
Estrogen |
(pg/ml) |
184.71 + SD64.09 |
181.50 |
128.00 |
48-435 |
|
Progesterone |
(ng/ml) |
1.81 +SD1.16 |
1.65 |
1.00 |
0.05-8.00 |
|
Prolactin |
(ng/dl) |
22.85 +SD23.74 |
20.00 |
23.00 |
2-300 |
|
Cholesterol |
(mg/dl) |
194.88 +SD45.66 |
202.00 |
245.00 |
11-245 |
|
Triglyceride |
(mg/dl) |
104.62 +SD28.51 |
107.50 |
78.00 |
55-176 |
|
HDL |
(mg/dl) |
43.97 +SD14.54 |
44.00 |
34.00 |
21-120 |
|
LDL |
(mg/dl) |
121.27 +SD27.58 |
120.00 |
120.00 |
30-120 |
|
LDL/HDL ratio 2.76:1.0 |
|||||
|
Testosterone |
(ng/dl) |
0.77+SD0.56 |
0.56 |
1.0 |
0.08-3.4 |
Table6:Biochemical Characteristics
The biochemical fertility panel, prolactin levels, lipid profile and testosterone that were determined are depicted in Table 6.
|
BMI (Kg/m2) |
Range (Kg/m2) |
Frequency |
Percent Cu |
m Percent |
Graph |
|
Normal |
18.5-24.9 |
26 |
15.7% |
15.7% |
|
|
Overweight |
25.0-29.9 |
63 |
38.0% |
53.6% |
|
|
Class I |
30.0-34.9 |
36 |
21.7% |
75.3% |
|
|
Class II |
35.0-39.9 |
35 |
21.1% |
96.4% |
|
|
Class III |
> 40 |
6 |
3.6% |
100.0% |
|
|
Total |
166 |
100.0% |
100.0% |
|
|
Table7 :Body Mass Index
Table 7 demonstrates the normal BMI (15.7%), overweight (38.0%) and obese patients of class I, II and III summed up accounting for 46.40%.
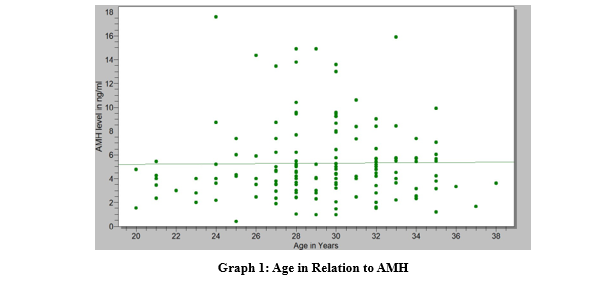
1 ng/ml = 7.18pmol/l
AMH values in ng/ml Age in years
Mean 5.41 SD + 3.28 Mean age 29.38 + SD 3.62
Median 4.5 Median 30
Range 0.40-17.6 Range 20 -38
The Graph shows the distribution of AMH across the ages of the study population and reveals clustering in the young/adolescents be- tween 24-35 years of age.
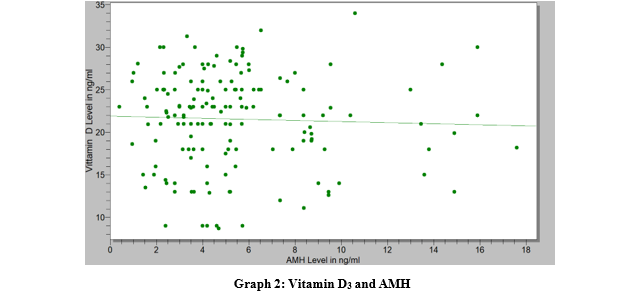
AMH values in ng/ml Vitamin D3 values in ng/ml
|
Mean |
5.41 SD + 3.28l |
Mean |
21.38 +SD 5.65 |
|
Median |
4.5 |
Median |
33.15 |
|
Range |
0.40-17.6 |
Range |
8.7-34 |
The Graph 2 demonstrates the relationship of vitamin D3 and AMH which gradually decreases with increase of the latter.
Discussion
The prevalence rate of PCOS is very much dependent on the diagnostic criteria employed and is subject to have regional and ethnic variations. These factors markedly have an effect on the prevalence rate, presenting symptoms and signs and treatment outcomes influencing comparability of the different studies and their applied generalizability. The rate in the current study is put at 8.7% which is quite comparable within the bracket of most other quotable publications of 6-20% [1-8]. Interestingly enough, it was found out and stated that Ethiopia was one of three countries (Ethiopia, Brazil, China) with the highest age-standardized rates of PCOS (2007-2017) among women of reproductive ages of 15-49 years with a report of 3.80% (3.08-4.52); and this was attributed to the fastest growing economy and population growth and exhibiting higher rates of obesity and exposure to chemical pesticides in the agricultural sector and a high prevalence of epilepsy as elaborated by the authors [30]. In another awareness study on prevalence and knowledge level on PCOS, 44 of 382 female Ethiopian adolescent students in Hyderabad, India with a mean age of 18.19 years were allegedly diagnosed with PCOS giving a rate of 11.5%; and 17.3% were rather documented as suspected in accordance with their study design [31]. Hence, PCOS is a complex clinical entity under so many names and diagnostic criteria, inherently associated with expensive and protracted investigations and often being an exclusion diagnosis and inherently with late initiation of treatment with no cure do shred doubt on the hypothesis, design, methodology, analytical power and comparability of the above studies.
The mean age at menarche for our study population was 13.19 years in contrast to the Americans which is 12.25 years with concomitantly lowered figures for blacks and Hispanics [32]. The difference is not impressive but the divergent trend is obvious as there are the interplay of differential multifactorial influences that include genetic, familial, ethnic, nutritional, health and activity, environmental, psychosocial development and socioeconomic factors. The features of PCOS tend to have common characteristics with the abnormal uterine bleeding consistent with the immature nature of the hypothalamo-pituitary-ovarian axis in the post menarche periods and also around puberty and adolescence with pronounced expression of hyperandrogenemia, irregular menses, obesity, hyperinsulinemia and even enlarged ovaries. It is obvious that non clinical undertakings on the topic, subsequent to the above factors, emphatically influence the outcome of the conclusion.
It is imperative that obesity is a salient feature of the syndrome and the findings and observations account for 40-80% of the list of disorders which is in confirmatory of our analysis of 46.4% without the inclusion of the overweight group of 38%. However, the distribution pattern does not warrant a comparative analysis because of the length of the study period and sample size. Nevertheless, it is not the symptom and/or sign complexes that determine or dictate the clinical scenario in general but the presenting symptom or sign as the main complaint which is very much consistent with the concern of the patients as causally connected to infertility and oligo-amenorrhea and not hirsutism, obesity or overweight in line with the fact that even lean or thin individuals can have PCOS and by the same token not all patients succumb to the criteria. The hormonal imbalance arising from the insulin resistance with the resultant hyperinsulinemia in patients with high BMI worsens the androgen excess and ovulation dysfunction. Infertility, hirsutism and risk of further metabolic syndrome allegedly increases with higher body mass indices but in our study especially regarding the latter, only 26(15.7%) had mild to moderate NAFLD/NAHSH. The perception of obesity in our population with resembling cultures relate to wealth, social status, beauty and bounty and is thus very much shaped by sociocultural and economic factors despite the associated catastrophic health risks and possible commonality with PCOS and underscoring its importance demands due attention and critical scrutiny [33-38]. The clients were relatively younger and the intent of employing sensitive, accessible, affordable and reproducible diagnostic means to detect and prevent the complicated course of the metabolic syndrome in general and mitigate their long-term sequelae requires a prospective, age related, long term and larger comprehensive population-based studies.
The altered LH: FSH ratio of 2.97 : 1.0 as in the current article instead of the 1:1 ratio in regular ovulatory cycles is suggestive of PCOS and clinically significant though it can not be considered diagnostic as the imbalance is demonstrated not in all patients. Its relevance lies on the fact that the elevated LH from pituitary gland results in hyperandrogenism which may physiologically arrests ovulation promoting anovulatory folliculogenesis and impaired metabolic syndrome with increased levels of insulin and insulin- like growth factor [39]. Serum testosterone level though expected to be higher than the findings, it can not be as absolute as DHEAS and 17 Hydroxyprogesterone which otherwise would have helped rule out adrenal causes of excess androgen production. The figures as presented ascertain that the estrogen and progesterone values are not seriously impaired and remained within normal limits.
The AMH level as determined in our study population as a reflection of the ovarian reserve is highly variable with a mean of 5.41 SD + 3.28, median of 4.5 and a range of 0.40-17.6 ng/ ml. Reasonably, high levels may be impacting and employed as a diagnostic tool and consistently correlate with the presentations as it is two to three times higher in PCOS than the normal women population. In general, poor to fair validity is documented based on the physiological changes that it peaks in adolescents of 20-25 years of age and in contrast varies across the menstrual cycle, and is lower in women with raised body mass index and gets suppressed by combined oral contraceptive pills and ovarian surgery. The different laboratories employ kits of different sources impacting clinical interpretations and approaches. They provide results in a single range, dichotomized in age groups, simple singular normal range specific for the result or use the classification of predictive of poor response in ART or ovulation induction, limited, optimal and hyperstimulation syndrome or PCOS [38]. An extreme caution should be exercised despite the fact that some studies have shown that high serum AMH levels correlate with PCOS and may even be employed in such a situation to replace ultrasound in the diagnosis of the syndrome [40].
Hyperprolactinemia, though may clinically interrelate with PCOS, is a distinct hormonal disorder that causes deranged menstrual function (oligo-amenorrhea), impaired fertility, galactorrhea and low estrogen levels characterized by decreased libido, vaginal dryness and dyspareunia. It is also known to cause visual disturbance, headache and osteopenia or osteoporosis. Therefore, its exclusion from the workup of the patient will ultimately impact the diagnosis and management.
The estrogen level can be normal or elevated as in our result but more so of E1 (estrone) which is increased in the circulation because of excess androgen converted to estrone in adipose tissue via aromatase and chronic or continuous E2 formation. Subsequently, the estradiol exposure without progesterone counterbalancing ascertains estrogen dominance with endometrial changes, behavioral/mood swings, breast tenderness and abdominal bloating. Hence, estrogen as one of the hormones raised in PCOS is of paramount importance in its late sequelae.
Relative hypovitaminosis of D3 with an increase in AMH level has been demonstrated in the presentation calling for an intervention thereby reinforcing the fact that there exists a positive correlation of Vitamin D3 and may help in regulating the high AMH levels in PCOS. Thus in managing PCOS in our patients, Vitamin D3 supplementation has been widely practiced as it posses multifold therapeutic advantages by regulating and lowering blood testosterone levels, stabilizing AMH, modifying insulin sensitivity, improving the menstrual periods and achieving eumenorrhoic states, promoting folliculogenesis, ovulation and concurrently imparting a counter effect on miscarriage and promoting healthy procreation and pregnancy outcomes. It is to be noted that its anti- inflammatory property in conjunction with aspirin also plays a significant role not only in ovulation but implantation processes and regulating genes [22].
The treatment modalities for those who were not married and had no intention of desiring to have a baby, COCPs remained our foremost course of therapy. Infertility remains the caveat and the management tends to be highly individualized; and in the light of the soaring costs of medical services we generally opted for life style modifications (carb reduction, exercise regimen with a target of 3-15% weight loss with well intended health tracking), orientating them with social media on available free videos and addressing their associated mental health worries. Supplements with myo-inositol and D-Chirro, folic acid, vitamin D3, letrozole and didroprogesterone from day 11 through day 25 of the menstrual cycle with timed intercourse coupled with ovulation kit tests and/ or signs and symptoms of ovulation (quality of vaginal discharge/ Spinnbarkeit, basal body temperature, mittelchmerz/mid cycle or ovulation pain). The treatment outcomes with the available modalities are variable and awaits time bound future analysis. If time seemed to be a factor, ART and/or ovarian tissue cryopreservation were advocated; and MLD, LOD and wedge resection remained dire interventionist alternatives. The introduction of advanced hormonal or stem cell therapies, ovarian platelet-rich plasma (PRP), In Vitro Maturation (IVM), targeted gene therapies and gut microbiome related options like probiotics and fecal microbiota transplant (FMT) if any are remote possibilities [41].
It is possible to manage excess androgens with their manifestations of acne, excess body hair, and male pattern of alopecia with thinning of hair over the scalp with COCPs, spironolactone, flutamide eflornithine, onandrolone, cyproterone acetate, finastride, The clinical problems were not the prime concern of our patients although contraceptive pills were administered to regularize the menses in the absence of desire to achieve a pregnancy, reduce the risk of endometrial hyperplasia and hirsutism. Anti diabetic drug, metformin was commonly prescribed to counter the insulin resistance with and without ovulation induction with letrozole and occasionally with clomiphene citrate in patients with a desire to achieve a pregnancy.
The psychological or mental health problems and subsequently the quality of life may be marred by anxiety, low self esteem, depression and resultant body image changes with overweight or obesity. Thus, it is obvious that such undertaking requires rigorous inter-professional, multi disciplinary complex long term approach and funded study design. The psychosocial support, rehabilitative measures and minimal emotional wellbeing may relief what otherwise has been expressed in a form of anxiety, depression, infertility and body image and related self esteem disorders were thoroughly discussed with the patients assuming it would give them a big relief [4]. The life style approach with dietary management reducing glycemic/carbs intake, decrease adipose tissue, insulin resistance, decrease peripheral aromatization resulting in excessive production of estrogens and stimulation of FSH, cessation of smoking of hookah or cigarettes and exercise to achieve weight reduction were routinely addressed and advised to personally reverse PCOS resorting to available free videos through the social media as most were quite educated. In a nutshell, a psychological and quality of life assessment is a distinct area worth exploring in order to design a coherent framework in the management of PCOS.
This presentation provides an insight into our experiences in our population and highlights the snapshot of current practices and future perspectives. The quirky phenomenon demands lengthy holistic approach with selective individualized framework and personalized management. It is highly recommended that a large scale national prospective and funded research should be conducted in the future.
Biases
a complex issue requiring lengthy and costly investigations; subjecting patients for financial burden with non-existent cost sharing mechanism and delayed diagnosis with late initiation of therapeutic interventions.
Recommendations
the study reinforces the need for future qualitative and quantitative research undertakings addressing the different challenges, contradictions and promoting best practices.
Acknowledgements
The corresponding author highly appreciates the staff at Yehuleshet Medical Center for the facilitation in the day to day data collection, digital services and their unreserved assistance in the day to day managing of the cards.
Ethical Declarations
Privacy, anonymity, and confidentiality was absolutely observed and maintained throughout the study period without involving any personal identifiers for or to any individuals, authorities or institutions. There was no need whatsoever for consent request and no institutional clearance was required. There was no coercion or inducement.
Authors Contribution
The authors exercised the norms and regulations in effect to writing, acknowledgement where indicated and finally collectively read, edited, and finalized the manuscript for submission for publication.
Competing Interest
The authors have no competing interests whatsoever.
Funding
The study is solely of self-funded and no internal or external source was sought. The brunt of the financial expenses in the course of the investigation was totally covered by the clients themselves.
References
- Escobar-Morreale, H. F. (2018). Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nature Reviews Endocrinology, 14(5), 270-284.
- Azziz, R., Carmina, E., Chen, Z., Dunaif, A., Laven, J. S., Legro, R. S., ... & Yildiz, B. O. (2016). Polycystic ovary syndrome. Nature reviews Disease primers, 2(1), 1-18.
- Mathur, A., & Tiwari, A. (2023). Prevalence of polycystic ovary syndromes (PCOS) in adolescent girls and young women: A questionnaire-based study. Indian Journal of Obstetrics and Gynecology Research, 10(3), 330-334.
- Teede, H. J., Misso, M. L., Costello, M. F., Dokras, A., Laven, J., Moran, L., ... & Norman, R. J. (2018). Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Human reproduction, 33(9), 1602-1618.
- Apter, D., Bützow, T., Laughlin, G. A., & Yen, S. S. (1994). Accelerated 24-hour luteinizing hormone pulsatile activity in adolescent girls with ovarian hyperandrogenism: relevance to the developmental phase of polycystic ovarian syndrome. The Journal of Clinical Endocrinology & Metabolism, 79(1), 119-
- Bahri Khomami, M., Shorakae, S., Hashemi, S., Harrison,C. L., Piltonen, T. T., Romualdi, D., ... & Mousa, A. (2024). Systematic review and meta-analysis of pregnancy outcomes in women with polycystic ovary syndrome. Nature communications, 15(1), 5591.
- Waghmare, S. V., & Shanoo, A. (2023). Polycystic Ovary Syndrome: A Literature Review With a Focus on Diagnosis, Pathophysiology, and Management. Cureus, 15(10).
- Legro, R. S., Arslanian, S. A., Ehrmann, D. A., Hoeger, K. M., Murad, M. H., Pasquali, R., & Welt, C. K. (2013). Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. The Journal of Clinical Endocrinology & Metabolism, 98(12), 4565-4592.
- Elasam, A. N., Ahmed, M. A., Ahmed, A. B., Sharif, M. E., Abusham, A., Hassan, B., & Adam, I. (2022). The prevalence and phenotypic manifestations of polycystic ovary syndrome (PCOS) among infertile Sudanese women: a cross-sectional study. BMC Women’s Health, 22(1), 165.
- Bellver, J., Rodríguez-Tabernero, L., Robles, A., Muñoz, E., Martínez, F., Landeras, J., ... & Group of interest in Reproductive Endocrinology (GIER) of the Spanish Fertility Society (SEF). (2018). Polycystic ovary syndrome throughout a woman’s life. Journal of assisted reproduction and genetics, 35, 25-39.
- Azziz, R., Carmina, E., Chen, Z., Dunaif, A., Laven, J. S., Legro, R. S., ... & Yildiz, B. O. (2016). Polycystic ovary syndrome. Nature reviews Disease primers, 2(1), 1-18.
- Lobo, R. A. (1995). A disorder without identity:” HCA,”” PCO,”” PCOD,”” PCOS,”” SLS”. what are we to call it?!. Fertility and Sterility, 63(6), 1158-1160.
- Carmina, E., & Lobo, R. A. (2022). Comparing lean and obese PCOS in different PCOS phenotypes: Evidence that the body weight is more important than the Rotterdam phenotype in influencing the metabolic status. Diagnostics, 12(10), 2313.
- Nandi, A., Chen, Z., Patel, R., & Poretsky, L. (2014). Polycystic ovary syndrome. Endocrinology and Metabolism Clinics, 43(1), 123-147.
- O’Brien, B., Dahiya, R., & Kimble, R. (2020). Hyperandrogenism, insulin resistance and acanthosis nigricans (HAIR-AN syndrome): an extreme subphenotype of polycystic ovary syndrome. BMJ Case Reports CP, 13(4), e231749.
- Berenji, E., Valipour Motlagh, A., Fathi, M., Esmaeili, M., Izadi, T., Rezvanian, P., ... & Nasr-Esfahani, M. H. (2024). Discovering therapeutic possibilities for polycystic ovary syndrome by targeting XIST and its associated ceRNA network through the analysis of transcriptome data. Scientific Reports, 14(1), 6180.
- Risal, S., Pei, Y., Lu, H., Manti, M., Fornes, R., Pui, H. P., ... & Stener-Victorin, E. (2019). Prenatal androgen exposure and transgenerational susceptibility to polycystic ovary syndrome. Nature Medicine, 25(12), 1894-1904.acanthosis nigricans (HAIR-AN) syndrome reflects adipose tissue dysfunction (“Adiposopathy” or “Sick Fat”) in Asian Indian girls. Dermatology, 237(5), 797-805.Agrawal, K., Mathur, R., Purwar, N., Mathur, S. K., & Mathur,D. K. (2021). Hyperandrogenism, insulin resistance, and
- Givens D.L, Woo C.H. (2016). Psychological and emotional aspects of polycystic ovary syndrome. Current Psychiatry Reports, 18(7), 1-8.
- Hummel P, McCall, J. (2019). Medical management of polycystic syndrome. American Family Physician, 99(3),165- 172.
- Hanjalic-Beck, A., Gabriel, B., Schaefer, W., Zahradnik, H. P., Schories, M., Tempfer, C., ... & Denschlag, D. (2010). Metformin versus acarbose therapy in patients with polycystic ovary syndrome (PCOS): a prospective randomised double- blind study. Gynecological Endocrinology, 26(9), 690-697.
- Krul-Poel, Y. H. M., Koenders, P. P., Steegers-Theunissen, R. P., Ten Boekel, E., Wee, M. T., Louwers, Y., ... & Simsek, S. (2018). Vitamin D and metabolic disturbances in polycystic ovary syndrome (PCOS): A cross-sectional study. PloS one, 13(12), e0204748.
- Wehr, E., Pilz, S., Schweighofer, N., Giuliani, A., Kopera, D., Pieber, T. R., & Obermayer-Pietsch, B. (2009). Association of hypovitaminosis D with metabolic disturbances in polycystic ovary syndrome. European Journal of Endocrinology, 161(4), 575-582.
- Tawfeek, M., Hassan, M., Ahmed, M., Mohamed, N., & Ahmed, N. (2021). Low dose Aspirin with clomid in pco. Minia Journal of Medical Research, 32(1), 100-106.
- Yu, Q., Wang, Z., Su, F., & Wang, M. (2021). Effectiveness and safety of aspirin combined with letrozole in the treatment of polycystic ovary syndrome: A systematic review and meta- analysis. Annals of Palliative Medicine, 10(4), 4632641- 4634641.
- Aref, N. K., Ahmed, W. A. S., Ahmed, M. R., & Sedik, W.F. (2019). A new look at low-dose aspirin: Co-administration with tamoxifen in ovulation induction in anovulatory PCOS women. Journal of gynecology obstetrics and human reproduction, 48(8), 673-675.
- Rasquin, L. I., Anastasopoulou, C., & Mayrin, J. V. (2017). Polycystic ovarian disease.
- Meier, R. K. (2018). Polycystic ovary syndrome. NursingClinics, 53(3), 407-420.
- ESHRE, T. R., & ASRM-Sponsored PCOS Consensus Workshop Group. (2004). Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertility and sterility, 81(1), 19- 25.
- Liu, J., Wu, Q., Hao, Y., Jiao, M., Wang, X., Jiang, S., & Han,L. (2021). Measuring the global disease burden of polycystic ovary syndrome in 194 countries: Global Burden of Disease Study 2017. Human Reproduction, 36(4), 1108-1119.
- Jahnavi, N., Harshitha, T., Adithi, J., Durdana, F., & Indracanti,M. (2024). Prevalence and Knowledge Levels of Polycystic Ovarian Syndrome among Female Adolescents in Educational Institutions in Hyderabad, India: A Cross-sectional Study. Ethiopian Journal of Health Sciences, 34(6).
- Biro, F. M., Pajak, A., Wolff, M. S., Pinney, S. M., Windham,G. C., Galvez, M. P., ... & Teitelbaum, S. L. (2018). Age of menarche in a longitudinal US cohort. Journal of pediatric and adolescent gynecology, 31(4), 339-345.
- Aden, E. H., Senbeta, A. M., Mekonnen, H. K., Hassan, S. M., & Mohammed, S. A. (2024). Magnitudes of overweight and obesity among adult and associated factors in jigjiga town, Somali region, Ethiopia. Human Nutrition & Metabolism, 37, 200282.
- Csace, I. (2016). Ethiopia demographic and health survey 2016. Addis Ababa, Ethiopia, and Rockville, Maryland, USA: CSA and ICF, 1-551.
- Beyene, E. T., Cha, S., & Jin, Y. (2024). Overweight and obesity trends and associated factors among reproductive women in Ethiopia. Global Health Action, 17(1), 2362728.
- Jemere, T., & Kefale, B. (2021). Metabolic syndrome and its associated factors in Ethiopia: A systematic review and meta- analysis. Journal of Diabetes & Metabolic Disorders, 20, 1021-1031.
- Fraissinet, A., Robin, G., Pigny, P., Lefebvre, T., Catteau-Jonard, S., & Dewailly, D. (2017). Use of the serum anti- Müllerian hormone assay as a surrogate for polycystic ovarian morphology: impact on diagnosis and phenotypic classification of polycystic ovary syndrome. Human Reproduction, 32(8), 1716-1722.
- Anagnostis, P., Tarlatzis, B. C., & Kauffman, R. P. (2018). Polycystic ovarian syndrome (PCOS): Long-term metabolic consequences. Metabolism, 86, 33-43.
- Liao, B., Qiao, J., & Pang, Y. (2021). Central regulation of PCOS: abnormal neuronal-reproductive-metabolic circuits in PCOS pathophysiology. Frontiers in endocrinology, 12, 667422.
- Albu, D., & Albu, A. (2019). The relationship between anti- Müllerian hormone serum level and body mass index in a large cohort of infertile patients. Endocrine, 63, 157-163.
- Singh, S., Pal, N., Shubham, S., Sarma, D. K., Verma, V., Marotta, F., & Kumar, M. (2023). Polycystic ovary syndrome: etiology, current management, and future therapeutics. Journal of clinical medicine, 12(4), 1454
Appendix