Research Article - (2022) Volume 3, Issue 3
Pollutant Emissions in Livestock Buildings: Influence of Indoor Environment, Rearing Systems, And Manure Management
2Universidad de Navarra, Department of Construction, Building Services and Structures, Campus Universitario, 31009, Pamplona, Spain
3Universidad de Navarra, Department of Environmental Biology, Campus Universitario, 31009, Pamplona, Spain
Received Date: Jul 05, 2022 / Accepted Date: Jul 14, 2022 / Published Date: Jul 28, 2022
Copyright: ©Copyright: ┬®2022 Leonardo de Brito Andrade, et al. This is an openaccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Leonardo de Brito Andrade, C
Abstract
The issue of air pollutants from livestock buildings is prevalent in the literature. Because they and their emissions impact both animal production and livestock building users as well as the outdoor environment. This paper aims to compile and review data available in the scientific literature on the types of pollutants for a better understanding of their generation form, their distribution according to the kind of animal, and the main factors affecting their generation and concentration, i.e., the rearing system, the indoor microclimate, and the manure management. The elevated generation of pollutants in animal buildings is tied to the dense occupancy in this industrial activity. The indoor air quality is defined according to the type of livestock in animal housing, considering its welfare needs, and the types and concentrations of pollutants generated as a function of the family of animal and the management used in production. The main gases generated are CH4 , CO2 , H2 S, NH3 , N2 O, in addition to particulate matter and airborne microorganisms such as fungi and bacteria that very negatively affect the health of animals and users of the animal buildings. Furthermore, knowledge about the main contaminants generated, the form of generation, their origin, their concentrations, and their distribution throughout the shed is essential to achieve a permanent and adequate indoor air quality and, with that, a high-quality product that will lead to high production yield without neglecting animal welfare.
Keywords
Pollutant Emissions; Livestock Buildings; Animal Buildings; Indoor Air Quality; GHG Emissions
Introduction
FAO data show that from 1961 to 2018, the world population increased by 147% while the total meat production (all types) increased by 380% [1]. Another fact is that livestock production represents 50% of the total agricultural product and supports many developing countries [2, 3].
The animal production figures are a direct result of human consumption. As of 2019, chickens, pigs, goats and sheep, and cattle and buffaloes were reared for meat production amounting to 27.5 x 109 (billion) live animals (of which chicken were about 23 billion), while almost 234 million cows were used for milk production. In contrast, in the egg production sector, there were 7.5 billion laying hen. Fig. 1 illustrates the development of animal production and the corresponding growth of greenhouse gas emissions in the livestock industry.
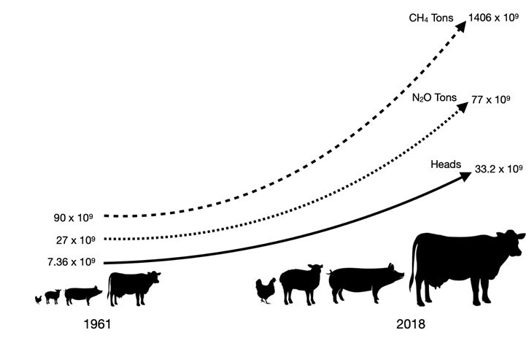
Figure 1: Evolution of Production and Animal GHG Impact In The Last 47 years (FAO 2020)
A significant intensification in livestock farming production has occurred because of the increase in both livestock buildings and indoor animal crowding in search for higher productivity. Other factors that enable the production growth can also be cited, such as using feed of higher nutritional value, improvement of pharmaceuticals, routine vaccination, and improvement of the infrastructure and feed efficiencies [4].
Collectively known as Animal Feeding Operations (AFO) and occurring within facilities where animals are concentrated or confined, these factors contributed to increasing animal production [5]. Currently, such facilities represent the most extensive, worldwide method for industrial-scale livestock production [6]. The impact of livestock production on surroundings is also relevant, causing effects on air, water, soil, biodiversity, and climate change, resulting in increased local and global environmental concerns. Therefore, reducing the pollutant emissions from management in animal buildings, emphasizing Indoor Air Quality (IAQ) improvement for the AFO, can now be considered the main research topic [7].
Massive efforts have been invested in the basic research to achieve new conceptions on air pollution. Several research projects have focused on identifying, quantifying, characterizing, and modeling air pollutant emissions in animal buildings through improving sampling and monitoring devices and developing mitigation methods. Results have yielded practical knowledge about what determines IAQ in different types of livestock production .
Some introductory examples of research that lead to this knowledge can be considered [8]. Monitored the environment of three different laying-hen housing systems: conventional cage, enriched colony, and typical aviary, and concluded that the IAQ was similar in conventional cage and enriched colony, both with ammonia and particulate matter concentrations bellow the typical aviary. On the other hand, found that while cage-free housing better agrees with natural behaviors of hens (foraging, dustbathing, wing-flapping, etc.), IAQ was lower than in the conventional system [9]. tested two types of laying-hen houses (high-rise and manure-belt) and verified the influence of the house design resulting in worse IAQ in a high-rise as compared to manure-belt, where a strong correlation was observed between IAQ and climate parameters (temperature and airflow rate) and animal conditions, influencing the results [10].
Different building materials also exposed to potential influence in IAQ. Found that selecting floor material became critical for IAQ. They compared two commonly-used systems in pig houses: fully slatted floor and deep fermented litter and concluded that the IAQ was worse in the case of the slatted floor [11].
This article aims to compile and discuss information regarding the influence of the indoor microclimate, the rearing system, and the management of animal manure on the Indoor Air Quality in livestock buildings, focusing on atmospheric pollutant emissions from intensive animal production (Table 1).
Table 1: Key Aspects of Review
|
Key aspects |
Details |
|
Environment conditions |
Influence of temperature, relative humidity, and airflow. Diurnal patterns. Influence of seasons. |
|
Rearing system |
Rearing system applied. Bedding and building materials. |
|
Manure management |
Characteristics (chemical, physical, size/magnitude). Management/movement/storage inside the building. |
Methodology for Searching and Selecting Scientific Papers
We searched articles in the Scopus® database using the following keywords: “pollutant emissions”, “animal buildings”, “indoor air quality”, “livestock animals”, and “GHG emissions”, without restrictions for year of publication or type of article. Therefore, publications such as books, periodicals, conference reports, technical reports, regulations, and technical guidelines were initially included in the result sets. Articles in the first result (Round 1) were then examined and their cited literature was recursively checked (up to six additional rounds) for additional relevant articles, which were then directly retrieved and examined (Fig. 2). A total of 295 technical or scientific documents were listed, although after detailed individual examination not all ended up yielding data relevant to this review. Most documents turned out to be regular scientific research papers, including a sizable number of review and discussion papers, but very relevant data emerged also from other types of documents such as inventories, databases, and technical reports
Figure 2: Rounds of Literature Review
Figure 3: Breakdown of the Selected Articles According To the Type of Document, the Pollutants Considered/Researched, and the Animals Studied, and the Factor of Influence Considered
Fig. 3 shows the non-exclusive breakdown of retrieved documents according to type of paper, pollutant, livestock, and three main influence factors– indoor environment, rearing system, and manure management.
Most of the documents analyzed (more than 45%) were related to poultry and pigs. Unsurprisingly, the most cited pollutant was ammonia (in more than 20% of publications). Although it is not the most dangerous pollutant in the animal industry, it is perhaps the biggest problem in terms of the concentration of pollutants produced in indoor livestock, mainly because of the large poultry production.
Approximately 1/5 of the documents analyzed discussed the theme of air pollutants in indoor animal production in a general or generic way, without specifying the type of animal. Considering the relevance that poultry and pig farming have on indoor animal production, we could expect that a large fraction of such generic papers would also apply to pigs and poultry.
Among the three factors analyzed in this article that influence the production and emission of pollutants in an animal building, the breeding system was the most cited and considered in the publications studied. However, the influence of the building’s indoor microclimate was above the influence of manure management in number of publications.
Pollutants in Livestock Buildings
Human activities release greenhouse gas (GHG) into the atmosphere, although GHG can also occur naturally in the (IPCC 2006). In 1997, many countries approved the Kyoto Protocol intending on reducing anthropogenic GHG emissions, and in 1999 the Gothenburg Protocol agreed to reduce emissions of ammonia (NH3), sulfur, nitric oxides, and volatile organic compounds (VOC), reinforcing the previous agreement. Pursuant to this, European Union countries are required to reduce GHG emissions by adopting the Gothenburg and Kyoto protocols, in the Directive 2001/81/EC (European Commission Publication 2001), and the Directive 2010/75/EU, known as Industrial Emission Directive (IED) (European Commission Publication 2010).
Animal production is a significant source of emissions. Currently, gaseous emissions from livestock production are considered an important issue because of their impact on health and the environment; and they have been taken in by public agencies and government agendas. Livestock processes play an important role in climate change and may cause negative impacts on the ecosystems, including air pollution [12-14].
Atmospheric water vapor (H2O), carbon dioxide (CO2), methane (CH4), and nitrous oxide (N2O), are well-known GHG related to and the thermal equilibrium of the biosphere, and their emission trends have been tied to climate change. Water vapor has a very variable content on the atmosphere and follows the water and climate cycles, and CO2 is tightly related to both natural phenomena (i.e., respiration, decomposition, plant intake and ocean absorption and immobilization) and anthropic intervention (fuel burning, vegetation cover change). On the other hand, CH4 and N2O are of particular interest as their CO2 eq values are respectively 21x and 310x by mol. Moreover, N2O emissions contribute to the depletion of ozone, via stratospheric conversion of N2O to nitric oxide (NO). CH4 and N2O emissions from various livestock sectors have been estimated in many countries following the Intergovernmental Panel on Climate Change (IPCC) guidelines [15-19].
The magnitude of GHG emissions from livestock production depends basically on the type of animal, rearing method/system, manure management, and indoor/outdoor climate conditions [20-27]. (Fig 4).
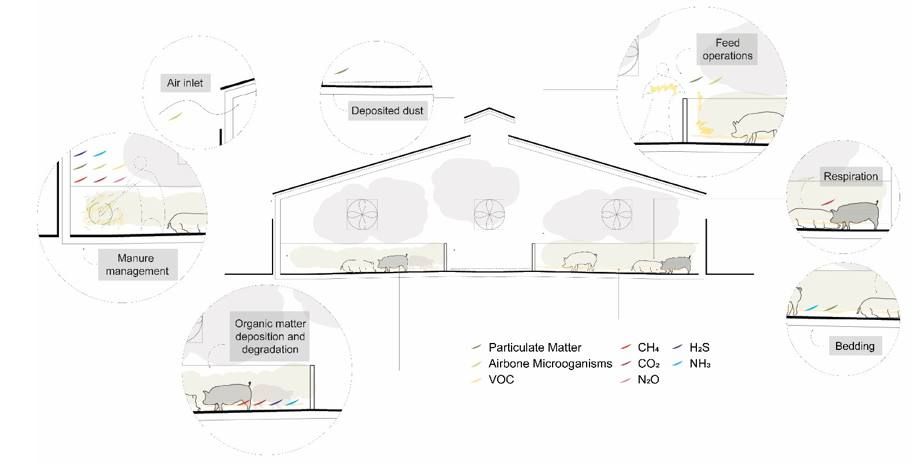
Figure 4: Scheme of Pollutant Production in Animal Building
Several papers have identified the factors that affect emissions in animal buildings, mainly related to different climate environments and management methods (rearing), and usually focusing on odor and environmental impacts that reach the outside by ventilation [28-31]. Some authors proposed enteric fermentation and manure management as two key source categories for overall livestock-related GHG emissions [32-34].
Enteric fermentation and manure management are the main sources of GHG emissions from animal production. In 2018, 46% of CO2 emissions, 78% of CH4 emissions and 6% of N2O emissions in agriculture from enteric fermentation + manure management [1]. In Spain, livestock contributed over 35% of all CH4 emissions in 2017, of which 75% came from cattle (62% from meat cattle alone) [35].
Many papers reported emission rates. The main pollutants described in animal buildings are airborne microorganisms, CH4, CO2, H2S, NH3, N2O, PM and VOC. Some authors classified animal pollutants into four groups: gases, odors, particulate matter, and volatile organic compounds [36-38].
NH3 is one of the most recognized harmful element that is produced from animal wastes worldwide, 65% of anthropogenic emissions of NH3 originate in the livestock sector. Poultry production emissions are higher than any other animal production, mainly because of a dense animal occupation, and constitute the major environmental problem for poultry farming. In contrast, while poultry buildings are a significant source of CO2, CH4, NH3, and N2O emissions generating from the bedding, animal excreta, or uric acid decomposing into urea, followed by NH3 and CO2 volatilization promoted by urease enzyme, CH4 and N2O emissions from poultry facilities usually are lower than that of cattle or pig production swine production buildings usually show high concentrations of NH3, CO2, and PM, that have been found to affect negatively the health of both animals and humans [39-55].
Thus, the type of contaminants (and, indeed, also their effects in both animals’ and worker’s health) will differ according to the type of both animal production and management in the animal buildings, although their global effect must also take into account how much each type of production and management represents in the sector. For example, pork meat is more consumed than poultry across the world, and therefore their contribution to the global emission form NH3 becomes even more significant [56].
While a reduction of meat consumption would most certainly lead to a corresponding reduction in emissions, current expectations are that consumption will instead grow alongside world population and expansion of indoor animal production, with a projected increase in global meat consumption by 70% by 2050, mainly concentrating in developing countries with more intensive animal production [56]. To counter this trend it is necessary to develop strategies to reduce pollutants in the livestock production building, where air quality is worse due to higher emissions per square meter [45].
We will discuss below the main air pollutants found in livestock buildings, considering the effects on animal health and wellbeing, production efficiency, and the subsequent environmental impacts.
Airborne Microorganisms Fungal Spores and Bacteria
Airborne microorganisms (mainly bacteria, fungi, actinomycetes, viruses, pollen, and some archaea) are omnipresent, lifted from the soil, water/seawater, vegetation, and other places [57, 58].
One of the most common airborne microorganisms is fungal spores, which can be hundreds of times more frequent than other particles like pollen grains. On average, one person inhales an air content of nearly 10m3/day mostly containing fungal spores ranging from 0.65 to 3.3μm in size [59-64].
High rates of airborne microorganisms occur in animal building and their impact is not restricted to the buildings themselves, as they can spread through natural airflow, affecting the IAQ and increasing the regional health risk. Indoor animal housings influence the transmission of airborne microorganisms significantly and may contribute to contamination of industries of food processing, according to the type of activity. For example, in laying-hen houses, where bacterial species are presented at high concentrations leading to food safety problems because bioparticles may be deposited to the eggshell, contaminating the table eggs [65-78].
Fusarium species are frequently found in animal feed [67]. Detected airborne spores of Fusarium species during grain handling from 32 farms in Finland. While concentrations were low, they found Fusaria in 77% of grain and feed samples. A large variety of trichothecenes (a group of mycotoxins) from Fusaria have been identified from different types of cereals for animal feed in different geographical regions [79].
Fecal contamination is another well-known bacterial problem that some authors noticed. In chicken-slaughtering facilities, the presence of Escherichia coli in chicken carcasses, Staphylococcus aureus in slaughtering environments, Pseudomonas aeruginosa in food processing environments are pathogens related to hygiene habits of employees [80-83]. Alerted to a high rate of airborne microorganisms measured, highlighting the importance of maintaining a low microbial level before the processing stage.
Both fungal spores and bacteria usually attach to solid particulates, although they can also be found as individual bacterial particles [84]. In animal buildings, airborne microorganisms usually occur in feed zones, animal bedding, and manure (bedding + excreta), where manure is the most important.
A significant concern about these aerial contaminants is, in many cases, their relevant effect on the health of animals and farmers, as they can cause diseases such as allergic reactions or asthma. Hence, poor IAQ and building emissions of airborne microorganisms are key indicators of workspace health for farmers, animal welfare, farm efficiency and productivity, food safety, and environmental impact, and a reduction of bioparticle levels to ensure healthy and safe conditions at the animal production workplace is as desirable as, and likely leading to, safer exhaust air from animal buildings [43, 52 and 83].
Table 2 condenses the main aspects of airborne microorganisms.
Table 2: Airborne Microorganisms in Livestock Buildings
|
Origin |
|
|
Characteristics |
|
|
Facilitators |
|
|
Effects |
|
Methane (CH4)
In 2018, agriculture produced more than 142 million tons of methane through burning, cultivation activities, manure management (7%) and most importantly, enteric fermentation by bacteria in the digestive tracts of animals (71%) [1]. this scenario affords a deep concern because CH4 is a GHG emission, and the livestock production does not stop growing.
Enteric fermentation is a natural process inherent to the largely anaerobic nature of digestion (especially in ruminants), and emissions will depend on the population size and trophic habit of livestock. On the other hand, manure management prompts a temporal succession of microbial processes, where substrates are converted into volatile fatty acids, CO2, and hydrogen (H2), increasing the temperature of the manure, and converting these products into methane [84-89].
CH4 production from manure may be estimated based on volatile solids in the excreted or organic matter. It is affected by low oxygen content, high temperature, high moisture in manure, a high level of degradable organic matter, a low redox potential, a neutral pH, and a C/N ratio between 15 and 30.
Dairy farms and cattle production, in general, is taken as the most important source of CH4 emissions in indoor livestock, as the involved ruminants produce more methane per unit feed than other types such as pigs or poultry. Methane depends on each animal physiological stage, and so emissions have a large range of variation. It can be influenced by age and herd of the animals, and diet quality and feed intake [90-92].
The type of housing in dairy farms (free stall barns with solid floors, barns with slatted floors and a collection pit below, bedded pack barns, an open lots) and the kind of stored manure may also affect the release of CH4 emissions [93-98]. Manure characteristics can significantly vary as they depend on the production factors, such as the type of building, rearing, feed, and facilities (straw supply, slurry separation, etc.).
Table 3 summarizes the main aspects of methane in farming
Table 3: Methane in Livestock Buildings
|
Origin |
|
|
Characteristics |
moisture content, a low redox potential, a neutral pH, and a C/N ratio of between 15 and 30.
|
|
Facilitators |
|
|
Effects |
|
Carbon Dioxide (CO2)
CO2 in animal production is a considerable problem when, in livestock confinement buildings, the production is overly dense, i.e., too many animals sharing, and breathing in, a confined space, however large. CO2 can also be originated from manure breakdown, although for both cases, breathing and manure, there are consequences on animal health and welfare when high concentrations of gas are reached.
Although CO2 is renewable and non-toxic at normal atmospheric concentrations, the release of considerable amounts contributes to global warming. CO2 concentration is normally used to estimate ventilation rate in animal houses and, consequently, the contaminated airflow is exhaled from the indoor to outdoor, leading to environmental impact [99-101].
The animal production types most related to high population densities are poultry and swine. In broiler houses, thousands of animals are reared together, and high CO2 concentrations from breathing are reached, with negative effects due to both the direct effect of the gas and the decrease in the oxygen concentration [102].
According to when the instantaneous CO2 concentration reaches 2, 4%, effects can be noticed in broilers. Still higher concentrations may lead to more severe health problems such as gasp and convulsions. However, lower concentrations held during longer exposure times could also affect the poultry health; for example, when broilers are exposed up to 6,000 ppm of CO2 for two weeks their body-weight is depressed and late mortality increases [103, 104].
To minimize these problems, regulations were established, and concentration limits were assigned. For example, the European Council Directive 2007/43/EC (European Commission Publication 2007) prescribes a maximum density of 33kg of broilers per m2 for non-monitored productions, and 42kg of broilers per m2 when a program of both CO2 monitoring and environmental control to keep CO2 concentration below 3,000 ppm are enforced. These requirements are used for both animal welfare and quality-meat production in security-food [43].
On the other hand, in swine production with a high-density of animals, in addition to exhalation by pigs CO2 comes from manure breakdown [89]. In manure, according to CO2 may have originated from three sources: 1) the rapid hydrolysis of urea into NH3 and CO2 catalyzed by the enzyme urease; 2) the anaerobic fermentation of organic matter into intermediate volatile fatty acids, CH4 and CO2; and, 3) the aerobic degradation of organic matter [105, 106].
Table 4 summarizes the characteristics of CO2 in animal production.
Table 4: Carbon Dioxide in Livestock Buildings
|
Origin |
|
|
Characteristics |
|
|
Facilitators |
|
|
Effects |
|
Hydrogen Sulfide (H2S)
As a key component of the sulfur cycle, H2S is a colorless, potentially harmful gas (although in very low concentration has low effect) produced in nature through the anaerobic breakdown of sulfate by bacteria. Nevertheless, hydrogen sulfide can be produced from human activities through various industrial practices and by the degradation of sulfur-containing protein in mammals [107].
In livestock production, H2S usually derives from manure breakdown (anaerobic decomposition) through two distinct ways: 1) mineralization of organic sulfur compounds; and 2) reduction of oxidized inorganic sulfur compounds [107].
Generally, low H2S concentrations are easily perceived, and long or extend gas exposure are taken as toxic and acutely dangerous to humans and animals: injury with chronic exposure at 10ppm and serious injury or death at > 500 ppm [108, 109]. Even at a low gas concentration of less than 0.3 ppm, H2S can be noticed by the human nose as an odorous gas with its unpleasant “rotten egg” smell. However, at higher concentrations (> 100 ppm) it may dangerously dampen the sense of smell (NRC 2009), negating the warning potential of odor.
Furthermore, H2S is corrosive, explosive (at 4.3–45% by volume in air), and flammable (260°C ignition temperature). These features have led authors to consider H2S one of the most dangerous gases in animal buildings and manure storage.
In livestock production, pig rearing is known as the animal production which has severe problems with H2S. And, in ruminants, the generation of large quantities of hydrogen sulfide depresses ruminal motility and cause severe distress to the nervous and respiratory systems [111].
Details of H2S in animal production are shown in Table 5.
Table 5: Hydrogen Sulfide in Livestock Buildings
|
Origin |
|
|
Characteristics |
|
|
Facilitators |
|
|
Effects |
|
Ammonia (NH3)
The microbial decomposition of the organic part of the manure is the main source of ammonia in animal houses. NH3 is generated from animal excreta (urine and feces) present on the floors of the buildings, generally beddings and pits. Research has shown that ammonia release depends on when in the day urea is deposited, although the enzymatic degradation of urea may occur over time [112, 113].
When mixed into the atmosphere, the ammonia lifetime tends to be short (five days or less), and it is generally located near its generation site. However, when NH3 is associated with other substances, mainly PM, it can form ammonium aerosols, such as ammonium sulfate, -nitrate, and -chloride. In aerosol form, NH3 can be transported far from the source by airflow and can increase its lifetime up to 15 days [114].
Besides, NH3 is also considered a significant environmental impact agent because it may contribute to the acidification of soil and nitrogen deposition in ecosystems when emissions from the indoor livestock production reach the outdoor atmospheric environment [115]. Additionally, NH3 emissions can generate nitrous oxide (N2O), a GHG, and secondary particles [116].
NH3 emissions from agricultural activities amount to more than 94% of the total anthropogenic emissions, and 75% come from manure management in livestock production. From all livestock activities, cattle, swine, and poultry generate 53%, 25%, and 15% of NH3 emissions, respectively [117-119].
Ammonia is the main contaminant in poultry buildings. Its high capacity to latch on to other particles and substances because of its sharply hydrophilic base can make it pervasive, decreasing health, welfare, and performance of the animals, for instance impacting feed intake and weight gain [120-123].
Indoor swine production also suffers from the consequences of poor IAQ by ammonia contamination, through respiratory diseases in piglets and farmworkers, and seriously impacts ecosystems as well [124-126]. Because of the extent of ammonia-related issues in swine production, the European Integrated Pollution Prevention and Control convention mandates mitigation actions for NH3 emission following Best Available Techniques (BAT) principles in pig fattening buildings with more than 2000 animals [54].
Details on the origin, characteristics, facilitators, and effects of ammonia in indoor animal production are presented in Table 6.
Table 6: Ammonia in Livestock Buildings
|
Origin |
|
|
Characteristics |
|
|
Facilitators |
|
|
Effects |
respiratory diseases and bacterial contamination of the lungs.
|
Nitrous Oxide (N2O)
Although N2O origins are still in need of much research, it is suggested that, worldwide, more than 65% of N2O emissions come from agricultural activities and almost 50% are produced from animal manure: manure management, manure applied to soils, and manure left on pasture [1]. There is thus a significant drive to mitigate this GHG emission through improved manure management, avoiding, for example, emissions from leaching, runoff, and volatile nitrogen from wastes deposited in pasture or lagoons [127, 128].
N2O from livestock depends on the chemical and organic composition of the manure (oxygen, nitrogen, carbon, and liquid content), its storage and management, and on the bacteria responsible for decomposition process [128,129].
Before N2O can be emitted from manure, ammonification of urea (either direct in urine from ruminants or indirect, through the conversion of uric acid to urea, in excreta from birds) must happen first [129]. The ammonification process is well understood and described for urine and uric acid, excreted by cattle/pigs and poultry, respectively. The ammonium produced is then oxidized first to nitrite and then to nitrate by nitrifying bacteria under the conditions of a sufficient supply of oxygen (nitrification). Denitrification can then occur in nitrified slurry (in which ammonium has been oxidized to nitrate) and in soils (nitrate from chemical fertilizers) when denitrifying bacteria reduce nitrates back to gaseous forms that escape to the atmosphere [130].
Besides becoming a GHG emission, the production of N2O from manure reduces the nitrogen content of animal waste. This fact decreases the value of animal manure as an organic fertilizer in crops, especially to organic food production farms [131].
GHG emissions from stored animal manure and different managements of manure have been widely researched by many authors [132-136]. However, in contrast to NH3, fewer data on the emissions of N2O from animal houses are available [137-140]. The published data suggest, however, that N2O concentrations in livestock production are generally low. In a study in a fattening pig house, reported quite low N2O concentrations (0.32–0.5 ppm) independent of the airflow characteristic and periods of rearing, and concluded that N2O emissions were negligible. The authors explained that not both aerobic and anaerobic conditions could be observed to nitrification and denitrification processes, respectively, in the waste slurry [141].
In poultry houses, N2O emissions are very low and lower than other broiler pollutants, like NH3, and lower than in other livestock categories, e.g., dairy cattle and swine. Low N2O concentrations have also been observed in laying-hen houses. Moreover, reported low nitrate content in hen manure, resulting in low denitrification [142].
On the other hand, even though it may represent a small percentage of all emissions as compared to other gasses, N2O is a strong GHG, having a global warming potential almost three hundred times higher than that of CO2 and a long residence time and its emission from animal manure occurs in all animal buildings globally (Table 7). Therefore, finding ways to mitigate its production through manure management becomes an important task [33].
Table 7: Nitrous Oxide in Livestock Buildings
|
Origin |
|
|
Characteristics |
|
|
Facilitators |
|
|
Effects |
|
Particulate Matter (PM)
PM is composed of fine airborne solid and/or liquid particles containing oxygen, carbon, silicon, phosphorus, nitrogen, and other substances. Normally PM is classified according to their size and the most common categories are 10, 2.5, or 1μm aerodynamic diameter which are usually known as PM10, PM2.5, and PM1 respectively Table 8 [143, 144].
The electrostatic attraction on PM causes particle agglomeration and may significantly alter both size category and attached content, such as hazardous matter like bacteria and/or viruses added on PM [145]. Therefore, PM might become a hazard. The health effects of PM have been exhaustively studied, and no completely safe level of PM has been found.
PM reduce IAQ in livestock production, compromising the health and welfare of animals and farmers, and by spreading to the neighboring areas it becomes a pollutant causing environmental impact [146, 147].
Stated that PM load data, i.e. levels, spatial distribution, and time-and frequency-related changes, are important to estimate the health impact on animals and farmers and to define PM mitigation actions such as indoor airflow assurance [148, 149].
Many authors reported that poultry and swine production generate more PM in livestock buildings when compared to e.g. dairy barns. Horse stalls is a particular case of livestock building that attracted research because PM considerably affects the health of horses when they are inhaled [150-159].
Table 8: Particulate Matter in Livestock Buildings
|
Origin |
|
|
Characteristics |
|
|
Facilitators |
|
|
Effects |
|
Volatile Organic Compounds (VOC)
A complex variety of volatile organic compounds (VOC) accompany other pollutant emissions such as gases, bioaerosols, particles and odors in animal husbandry [160, 161]. This commonly occurs in livestock farms where the pollutant profile varies according to the farm’s sections: indoor environment, manure storage, accumulated wastewater and the air above the surfaces of these waters, compost, and lagoons [162-164].
VOC are carbon-containing molecules that, under normal conditions of temperature and pressure, vaporize and enter the local atmosphere according to their specific vapor pressure points. They are seen as a large group of organic chemical products, formed by molecules of different functional groups that present different physical-chemical behaviors, but having in common a certain volatility. The most common examples are volatile fatty acids, alcohols, aldehydes, amines, aromatic hydrocarbons, carbonates, esters, ethers, ketones, sulfides, disulfides, mercaptans and heterocyclic nitrogen compounds [165-168].
VOC can attach to the surface of solid particles, i.e., PM, and be thus transported to, and spread through, the atmosphere [169]. However, VOCs can also be found at various places on a livestock farm that are completely decoupled from PM. For example, in silage products more than 50 different types of VOCs have been detected through emission monitoring. VOC contribute to tropospheric ozone production, which causes adverse health effects [170-173]. One of the biggest problems associated with the presence of VOC in a rural industry is their generally unpleasant odor, which causes discomfort of workers and neighbors (Table 9). Odor is defined by ISO 5492:2008 (ISO 2008) as an organoleptic attribute perceived by the olfactory organ (including nerves) when it smells certain volatile, pleasant or unpleasant substances [174]. The odor can be considered to occur due to the interaction of different volatile chemical species, such as sulfur compounds (for example, sulfides, and mercaptans), nitrogen compounds (for example, ammonia, amines) and volatile organic compounds (for example, esters, acids, aldehydes, ketones, alcohols).
Malodorous conditions are usually associated with harmful air pollutants and unhealthy air conditions [175-177]. Unpleasant odors can cause negative emotional reactions in people, resulting in an important decreasing of the quality of life in the areas surrounding the livestock farms, and discomfort due to the generation of odors in animal production is one of the main sources of complaints from people who are close to animal farms. As a result, over the years there has been a greater emphasis on controlling the impact of air pollutants exhaled from the livestock buildings and spread out in neighboring areas [178-184].
For evaluating odor concentration, olfactometry sensory measurements have been used as a standard method, and has been used to quantify odor concentrations from the animal slurry applied on the field [185]. The level of odor depends basically on the organic and inorganic odorous compounds, and therefore these could be measured as proxies for odor sensitivity. However, a recent study investigated the odor emissions from cow and pig slurries used on the soil used dynamic olfactometry, without specifically quantifying VOC [186].
Research have indicated that VOC can be considered biomarkers of decomposition associated to mortality, suggesting their use in animal production practices [187-191]. Several studies have explored the possibility of diagnosing pathologies in animals by identifying the VOCs produced by pathogens, pathogen-host interactions and biochemical pathways [192]. For example, VOC analysis has been explored as a method to diagnose bovine respiratory diseases, brucellosis, and bovine tuberculosis [193-195]. In fact, VOC emitted from different areas of the living body can be considered as individual 'fingerprints', and pathological processes (such as infection and endogenous metabolic disorders) can influence those ‘fingerprints’ either by producing new VOC or changing their normal proportions. One of the main advantages of these techniques is that they are non-invasive diagnostic tools that do not require any manipulation of the animals. For these reasons, exploring volatile organic compounds is an area of research of increasing interest in veterinary medicine [196, 197].
Table 9: VOC in Livestock Buildings
|
Origin |
|
|
Characteristics |
|
|
Facilitators |
|
|
Effects |
|
Factors Affecting the Production of Polluting Emissions
Many variables can influence the generation and emission of gases, particulate matter, and microbial agents in livestock buildings. Temperature and relative air humidity, type of floor, presence of certain materials, movement and handling of indoor manure, the season of the year and period of the day are just a few examples [42].
Important decisions, such as the rearing system used in animal buildings, volume of production and stock, age of the herd, feed programs, indoor climate program, or manure management, are often dependent on the size and complexity of any given farm. In turn, all these choices affect IAQ and the production and spreading of pollutants. Different animal housing systems may thus result in different pollutant emissions for the same livestock as much as the different livestock do.
Argued that promoting positive responses in zootechnical performance of animals through welfare practices, like better building design based on animal wellbeing, implementation of bioclimatic strategies to control the indoor climate, or improvement in sanitary status, may lead to a decrease in emission levels. Improvements in animal welfare may indeed carry investment and operating costs, but high taxation on emissions generated can drive decisions in favor of mitigation techniques [89].
Now we will introduce a literature review for each group of variables and their relationship with the generated pollutants.
Influence of Indoor Environment
An indoor environment refers to a closed place, usually a shed, where animals in production are confined and are subject to a certain environment, determined by natural, artificial means, or both. However, the microclimate of these indoor environments, mainly temperature (T), relative humidity (RH), and ventilation, is frequently challenged by local climate parameters varying with the seasons and the airflow patterns.
Indoor T and RH changes depend primarily on the geographical location of the building. The greater the seasonal climatic amplitude, the greater variation of the indoor microclimate’s T and RH. And the higher the T and RH, the lower the indoor animal activity and movement of animals, resulting in deterioration of animal welfare and, consequently, reduced productivity, matched by a corresponding reduction of pollutant emissions, as verified by in dairy cow buildings where increased air temperature resulted in reduced production and less release of CH4 by cows.
On the other hand, warm seasons leading to high indoor temperatures result in an increase of the chemical production of air pollutants due to the temperature effect on the chemical activity (catalyzing effect of chemical reactions). However, high temperatures demand more efficient airflow through the shed to remove heated air and allow fresher air in. Thus, the emissions will likely distribute more evenly throughout the building but will also be exhaled from livestock buildings in larger quantities, leading to important environmental impacts in surrounding areas [198, 199].
Rural sheds are usually equipped with air cooling systems to avoid losses in productivity from poor animal welfare when temperature rises. These systems are installed when natural ventilation alone is not able to reduce thermal impacts on production. On the other hand, when the T of the region is low and the buildings require maintenance of the internal heat, i.e., a warmed environment, the flow of fresh and cold air along the indoor building tends to be drastically reduced.
Thus, pollutant emission dynamics are strongly altered by the indoor microclimate both in production / concentration and in the spreading through the indoor area. Confirmed the seasonality in aerial pollutant emissions in pig houses, particularly to NH3 and H2S concentrations, emphasizing the influence of the airflow through the building, which tends to be lower during the cold seasons than in the warm seasons. Seasonal variations in the GHG emissions by animals can thus happen, and regular measurements to ensure representative emissions data throughout the year become important.
In general, all pollutant emissions in all types of animal production differ throughout the year. In laying-hen houses, differences in NH3 and CO2 concentrations in different seasons were verified by, with higher concentrations in summer than in winter. However, there is a tendency for indoor N2O levels to increase in cold seasons in laying-hen cage houses because of less ventilation. In contrast, reported that N2O atmospheric emissions were higher in the mild and warm seasons because the ventilation led to an improved release of this aerial pollutant.
In regions where the seasons have no significant variation in the outdoor temperature, indoor emissions tend to change minimally during the year, because the indoor climate remains constant throughout the year if the airflow pattern can be maintained. PM and indoor microclimate change have been extensively studied and correlated. Particulate matter have a strong dependence on RH and airflow [200-204]. Observed a positive correlation between PM and RH, though not between PM and T.
PM emissions can be taken as the major problem in horse stables during the cold seasons. But when an optimized ventilation system is applied, the respiratory diseases roughly decrease, indicating the importance of maintaining IAQ strategies independent of seasons and periods of the day in horse stalls [205].
Temperature and relative air humidity are two important factors for developing microorganisms, especially fungi, promoting the acceleration of the decomposition of organic matter, such as feed and manure. Explained that a high RH could promote a catalytic effect, i.e., acceleration in microorganism reproduction, such as bacteria, fungi, and some parasites, resulting in a fast proliferation of these biological organisms and, thus, animal diseases [99].
Another effect of the high RH is increasing the speed of decomposition of organic compounds (excreta, feed, and manure), providing ideal conditions for microorganism growth and raising the airborne pollutant concentration [206]. In indoor environments, higher T and RH are associated with the higher generation, release, and dispersal of fungal spore as observed by in cattle sheds [207]. Confirmed the seasonality in aerial pollutant emissions in pig houses, particularly NH3 and H2S, emphasizing the influence of the airflow through the building, which tends to be lower during the cold seasons than during the warm seasons. Ensuring outdoor air exchange in livestock confined production is always essential to reduce temperature and relative air humidity, and to renew internal air evacuating contaminated air. This airflow maintains IAQ and therefore, promotes animal welfare and productivity.
Adequate openings to natural ventilation are thus desirable in animal buildings, but when these are not possible or enough, mechanical ventilation should be considered to allow sufficient airflow through the shed according to IAQ requirements in animal production.
In cold climates, animals may not require as much fresh air to reduce temperature and achieve welfare, but nonetheless a renewal airflow is necessary to remove pollutants. On the other hand, in warm climates and hot and humid climates, a specific ventilation program must be designed to avoid poor IAQ associated to high T and RH. This higher ventilation rate can also assist in diluting pollutant concentration along the sheds, such as e.g. bioparticles as observed by and in laying-hen houses.
The sheds can be naturally ventilated through large side openings and roof and ridge openings, although sheds with large openings in cold climates may have difficulty maintaining appropriate thermal conditions with too much cold air entering the building [208]. This bioclimatic strategy drives fresh air by wind pressure or convection, allowing warmer air to escape through the roof openings while suctioning cold air through the lower openings. Both wind effect and convective (buoyancy) force lead to an economic reduction of electrical energy using mechanical ventilation [209, 210].
However, alerted that in buildings with large openings, it is almost impossible to clean the exhaust air, resulting in environmental pollution by exhalation to the atmosphere of NH3, CH4 and other airborne pollutants [208].
Mechanical systems demand substantial investments and running costs in the purchase and maintenance of equipment and consume electrical energy, increasing the cost of production besides producing an important noise level when they are switched on [211]. However, mechanically-controlled ventilation also facilitates removal of indoor pollutants before exhaust, as air can be driven through point cleaning systems before release [212,112].
Estimating pollutant emission rates in naturally-ventilated buildings can be much more difficult than in mechanically-ventilated buildings, given the complex connections between the outdoor wind and the indoor environment.
In open buildings, air exchange rates depend on both indoor parameters, such as temperature gradient and airflow, and outdoor parameters, like wind speed and surrounding topography [212]. These authors observed wide spatial and temporal variations in the concentration of CO2, NH3 and CH4, inside a naturally-ventilated barn. Modelled the concentration and spreading of NH3 above manure in livestock houses as a function of ventilation rate, air inlet conditions and temperature [112].
A significant influence of temperature and airflow (especially airflow momentum and intensity turbulence) on the rates of NH3 release from manure has been observed [213-215]. However, inside the building, airflow turbulence and natural wind variation reduce the accuracy of velocity data, and induce uncertainty about the airflow gradients and turbulence above surfaces that can potentially release pollutants, such as manure, bedding, and slurry [112].
As explained earlier, in a cold climate or weather environment livestock buildings reduce air exchange in order to maintain thermal comfort but must ensure enough internal airflow (called minimum ventilation rate) to control IAQ.
When minimum ventilation is used as hygiene air in animal houses, the focus should be placed on the worst pollutant emission, as it should afford the best relative IAQ improvement, rather than on some ready measurement. For example, in horse stables the minimum ventilation is usually established only to keep RH or CO2 levels within a threshold. However, this can result in dangerous levels of NH3 and PM, reducing the air quality and leading to poor health and welfare of the horses, as their respiratory tract particularly sensitive to high PM concentration [216-220]. Low ventilation rate in combination with type of bedding material and hygiene practices also influence the NH3 and PM concentrations in horse stables [221, 222].
Linked pollutant concentrations with airflow speed to establish a possible correlation between them. The authors observed that there was an indirect correlation between PM and NH3 with airflow speed.
Measured pollutant emissions in a riding school stable before and after installing a ventilation system and reported the positive influence of an adequate ventilation program to maintain the levels of CO2, NH3, airborne microorganisms and PM below the respective hazardous concentrations for horses [223].
Ventilation systems can effectively remove odor nuisance in livestock buildings, but an inadequate airflow, on the other hand, can influence the odor propagation through the indoor areas due to the low intensity of air renewal [224]. Explained that pollutant emissions should effectively guide airflow patterns to animal buildings, i.e., the higher the concentration of pollutants in the air, the greater the airflow for a specific volume. However, described that when minimum ventilation is used to maintain air quality in cold climates, refreshing indoor air, the building's ventilation design must be carefully studied to obtain an effective cleaning of the indoor air, avoiding the maintenance of air pollutants in the closed environment [225].
Some modifications in animal buildings have been tested to increase the efficiency of the ventilation system to mitigate pollutant emissions. Partial Pit Ventilation (PPV) is a concept largely used in swine houses, and the results have been published as a successful solution for reducing emissions and increasing efficiency of the ventilation system in reducing the concentration of emissions [226]. Showed that PPV can benefit energy savings by making up for 10–30% of the maximum ventilation rate and, therefore, allowing for a corresponding reduction of electricity dedicated to mechanical ventilation. Studied NH3 emissions when a PPV system was used in a fattening pig house and observed a reduction of 42.6% in ammonia concentration as respects to a conventional ventilation system. Similar results were found by.
In dairy cattle buildings, and also found increased efficiency in the reduction of emissions through experiments and numerical simulations, with a pollutant removal rate by pits in excess of 80%, depending on the airflow condition above the floor. On the other hand, found that the use of PPV leads to cumulate 64–83% of ammonia emissions in cattle buildings.
Influence of Rearing Systems
Pollutants in animal sheds are a function of the type of animal production – broilers, swine, cattle, eggs, etc. –, the rearing period or phase, for example, maternity or termination stage, and other variables. They affect the health and wellbeing of the animals, productivity, and sustainability [227].
The rearing system can influence the generation of aerial pollutants because it affects several parameters, such as animal activities, airflow, feed management, and, principally, manure management. Materials used in bedding also strongly influence emissions.
Hygiene in the shed, especially in relation to manure, and controlled storage of the feed are determining factors to avoid contamination of the environment, mainly by microorganisms and also fungi, that can grow on aged animal food, moldy hay, manure (bedding + excreta) and that are usually associated with individual bacterial particles or attach to PM [228, 229].
An unhygienic workplace could thus result in a high quantity of fungal spores being released in the air, causing infections or triggering respiratory disease both in farmers and animals [230]. Some authors have suggested that this problem can be alleviated by minimizing airborne microorganism levels in livestock buildings through severe mitigation actions to achieve healthy working conditions and quality in the animal growing environment [231, 232].
The bedding can also be considered a favorable place for the proliferation of microorganisms and production of PM. The movement of animals on it, which depends significantly on the type of rearing used, is a particular factor. For instance, in the cage-free hen house method, the high PM and airborne bacteria concentrations derived from the movements of the animals on the bedding and differed substantially from conventional cage houses.
Disinfecting the litter could potentially reduce the contamination by litter bacteria such as Gram− bacteria and protect crops and pasture zones from fecal bacteria when the bedding is removed and used as a fertilizer support [233, 234].
Studied cattle shed sections and observed a higher concentration of Nigrospora in the feed storage, especially the stacks of straw, which might serve as a local source of fungal spores. They found the toxic fungus A. flavus in stored straws and fodders for cows and called for an efficient IAQ program to prevent health hazards. Similar calls have been made by other authors, although some recognize that there is still insufficient comparative data about NH3, GHG, and PM emissions from shed sections. Understanding the mechanisms of aerial pollutant emissions throughout sections will require stronger statistical correlations of different variables, connecting cause and effect.
In livestock indoor production, diurnal patterns associated to the rearing systems have been found in the dynamics of pollutant emissions. Likewise, the seasons can also introduce patterns especially when seasonality is strong, e.g., distinctly hot and cold seasons throughout the year. Both types of patterns combine to regulate the activity behavior of animals (feed intake, movement on the bed, natural behaviors in general), which in turn influence contaminant mobilization patterns. For example, lower activity in pig houses reduces PM concentrations and emission rates at night because of the changes according to both temporal and spatial behavior of the animals. Observed that in poultry houses, PM emissions were higher during daytime and summer, and that they were higher in open aviaries than in cage systems. Similarly, observed that increased hen activity on the floor litter during the afternoon generated higher pollutant emission and bacterial particle levels than in the morning [235].
However, the period of the day did not seem to influence NH3 concentration in an enriched cage laying hen facility, while differences in CO2 emissions were low in a study by.
Feeding intervals within a given type of management or even a feeder area when animals are free to access feed significantly interfere with the production of particulates and move part of the feed to the floor or bedding in these areas, even becoming the main factor for higher PM emission rates in swine [236].
In dairy production, feeding times of the cows also condition the development of the pollutant concentration rate because of increased animal activity. A correlation between gas emissions and feed programs in dairy buildings exists.
Different production systems may lead to variable emission rates. In a study where different laying-hen houses were considered, observed that in aviary house systems, total bacteria concentrations and emission rates were much higher than both conventional cage house and enriched colony house systems.
In equine buildings, the inhalation exposure to PM was higher in stabled horses versus no-stabled horses, principally when the activities of the horses were walking and eating [237, 238].
Trying to mitigate high concentrations of PM in horse stables, investigated an alternative polymeric material, woody PET, for stall bedding. However, the use of woody PET resulted in increased PM concentration over natural straw, which is still considered the best bedding material for stables even when horse activity is high.
Several nutritional factors affect the rate of enteric CH4 production in ruminants, such as feed concentrate composition, the maturity of harvested forages, and the inclusion of maize silage at the expense of grass silage. However, although detailed information was available about nutritional conditions on dairy cattle farms, alerted that a large number of differences among farms other than nutrition makes it difficult to ascertain the effect of feeding systems on CH4 release [239, 240].
Influence of Manure Management
Total anthropogenic CH4 emission from enteric fermentation and manure management in farming activities is almost 80%, and the methane released in the biosphere from agriculture is about 40% (FAO 2020). Manure management is the most discussed factor regarding emissions and the most important source of NH3 N2O.
When Mnaure remains stored inside the building untill the end of the production cycle emissions are higher than when manure is frequently removed from the building, which dramatically reduces emissions. Emissions from manure when it is taken out to external areas may also decrease because these outside areas have typically lower temperatures than indoor areas. Temperature, wind speed and airflow directions, pH and volume of the manure, among other factors, influence the emission dynamics of manure stored in livestock buildings [241-243].
N2O is not likely to be produced from manure stored indoors in pits beneath the slatted floors, though this is the most common excreta storage system in pig and cattle farming. N2O emissions are expected in housing systems that are based on solid manure. In these systems, animal excreta are either already in the form of solid manure (poultry) or are being collected in, e.g., straw or wood shavings (pig and cattle). However, manure deposited on the floor and/or pit promotes the release of NH3, though ammonia production and concentration will depend on the characteristics of the manure, the rearing, the microclimate in the building, and the airflow above the stored manure surface.
In recent decades, bedded systems have been used in swine buildings, achieving better welfare, odor nuisance, and GHG emissions than in the most common system based on a slatted floor where the animal excreta fell on a pit used to store slurry, although other authors could not observe a significant reduction in the indoor aerial pollutant concentrations when partly-slatted floors were used in lieu of fully slatted floors [244].
Whereas several factors influence emissions of N2O and complex interactions must occur among different sources, several authors found that certain materials, for example, mixed with animal waste, may lead to N2O reduction and thus can be considered efficient actions of emission mitigation. For instance, the addition of porous materials in an animal slurry to absorb the liquid phase can slow down the chemical reactions and, consequently, achieve aerial pollutant production. Verified that N2O almost reached zero-emission when maize silage and wood chips were added to pig slurry. However, other mixing materials such as straw may not be as efficient, as they can modify the physical characteristics of the animal waste, enhancing the alternate aerobic and anaerobic conditions that may promote N2O emissions. Nonetheless, the use of straw in dairy cows bedding decreased total GHG emissions when the manure was stored indoor, even though there were significantly pollutant emission differences among the seasons of the year (warm, mild, and cold seasons) because of the temperature-dependent variation of the microorganism activity. When compared to wooden chips, chopped straw used to cover manure slurry from dairy cattle decreased aeration, resulting in an regarding emissions and the most important source of NH3 N2O. and increase of GHG emissions.
In free-stall barns, manure is normally removed every few hours When manure remains stored inside the building until the end of to once a day by scraping or flushing. With this rapid removal of the manure, CH4 does not have time to build and indoor emissions are low. In contrast, with a slatted floor manure accumulates in a pit under the floor from a few weeks up to several months, and in a bedded pack barn, manure accumulates on the floor for a few months as more bedding material is being added over the winter to absorb moisture. Under the aerobic and anaerobic conditions found within the pit or manure pack, N2O and CH4 emissions become much greater, respectively. Manure also accumulates on an open lot, but the manure is spread in a thinner layer where the more aerobic conditions induce less GHG emission. On the other hand, the effect of high compaction of the bedding may also become less favorable for the generation of contaminants, especially those aerobic processes that require oxygen.
Displacement of manure is often related to the release of GHG. If there is not enough ventilation in the shed, manure movements within the shed will thus elevate indoor contaminant levels. Manure buildup promotes emissions and requires additional manure handling, compounding the problem. In any case, GHG will eventually find their way to the atmosphere through building exhalation. On the other hand, frequent removal of manure from the building may reduce indoor emissions and enhance IAQ.
Discussion
There is a significant, demand-driven increase in the production of animal by-products worldwide. This is only possible through a considerable improvement and intensification of livestock production in controlled and closed buildings. However, emissions of pollutants such as airborne microorganisms, CH4, CO2, H2S, NH3, N2O, VOC and PM are intrinsic to the processes, and therefore IAQ is strongly affected and requires continuous and specialized attention including mechanical ventilation and constant monitoring for pollutant abatement.
Low IAQ affect both livestock and workers, and can also cause serious environmental impact problems on the neighborhood when the gases are exhaled from animal buildings. Achieving desirable IAQ following the environment guidelines of each country requires identification of the generated pollutants according to the type of animal production and its functional characteristics such as breed and welfare needs, as well as local climate, management actions or rearing, typology of building/shed and environmental control actuators, and analysis of the data collected through continuous monitoring. Mitigation procedures must consider all those factors and data for successful implementation.
According to our literature review, the most cited production variables that lead to changes in the characteristics of IAQ are the rearing system, indoor microclimate, and manure management. Although these three variables tend to be quite interrelated in a systemic view, a specific view of each variable is essential when selecting mitigating actions.
The production of pollutant gases derives from complex chemical reactions that strongly depend on the availability and quantity of specific compounds and on the conditions in the manure and its environment such as temperature, moisture content, presence of oxygen, surrounding air flow, physical characteristics of the deposits (porosity, compaction, specific surface, adsorption potential, etc.). Hence, the quantity of emissions is not just a direct function of the volume of manure generated by the livestock, but also of where and how it is generated, including microclimate conditions (T and RH) of the manure storage sites, mixture with other components such as bedding or water, the dynamics of air flow through the building, and the effects of gas dispersion.
Solid particulate matter and microorganisms, attached or not to PM, also depend on all three factors (rearing, microclimate, manure). Fresh air intake for renewal and cleaning is also a source of PM in the indoor building. Microorganisms, on the other hand, depend on the organic matter and environmental conditions to reproduce and are linked to both the hygiene condition of the sheds and the health condition of the herds.
Herd management, bedding composition, and building typology and design (spaces for movement of the livestock, feeders and drinkers, manure storage areas for short and long periods, etc.) are also factors that influence qualitatively and quantitatively the production of air pollutants.
Manure management, especially bedding turning, removal, storage, and manure-bedding mixing operations, causes important concern about emissions in indoor livestock production. Most mitigating solutions aim at the physical-chemical treatment of manure, especially when they are deposited outside the shed. However, certain herding practices and manure-bedding mixtures can result in lower environmental impact even inside the building.
The microclimate conditions of the shed are decisive for the development and propagation of indoor air emissions. Temperature and air flow are the predominant, interdependent microclimate variables, as ventilation conditions are primarily determined by the livestock’s sensitivity to adverse thermal conditions, both high and low. These variables exert a significant influence on the production of pollutant emissions from manure, depending on the rearing system in place. A ventilation program in an animal building that does not consider emissions and their buildup could lead to two undesirable results: 1) dispersion of contaminants from stored manure throughout the house, and 2) in cold climates, elevated indoor contaminant levels through the drastic reduction of ventilation to minimize heat loss. Both outcomes may noticeably affect the health of animals and workers.
A new trend to collaborate in the hard work of mitigating emissions in animal buildings may be emerging when designing livestock sheds with low environmental impact. Investments in renewed barns can be associated with innovation and sustainable thinking. The relationship between sustainability and well-being issues seems to be a path that highlights the care with the IAQ in animal buildings that is certainly worth exploiting [245].
Conclusion
Animal by-products generated in high-efficiency, intensive farming systems in closed and controlled buildings, lead to the generation of significant amounts of waste per unit area of animal occupation. In addition to the solid waste that is produced in large volumes, the production of livestock in walled buildings results in a similarly important volume of aerial pollutant emissions, such as gases, solid particles, and microorganisms. The aerial pollutant emissions affect the confined animals, the users responsible for handling the herd, and the natural environment outside the shed due to the significant exhalation of dirty air from inside the building that occurs constantly due to the necessary air renewal [246-273].
The type and amount of emissions for any livestock class will be strongly conditioned by 1) the indoor environment; 2) the rearing system applied, mainly the management of the herd and the typology of the shed; and 3) the manure management, especially when stored for a long time inside the building.
These three sets of variables could be analyzed separately to verify their specific impact on the production and level of each type of pollutant. However, all three sets are usually interrelated, and therefore a joint analysis is desirable in order to account for synergies and cancellations. Mitigating solutions for low IAQ in animal buildings should thus be also based on a systemic relational study of the variables considered here.
Declarations
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflicts of Interest/Competing Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Availability of Data and Material
(Data Transparency) Nothing to declare. Code Availability (Software Application or Custom Code) Nothing to declare.
Authors' Contributions
Idea for the review article: Leonardo de Brito Andrade; César Martín-Gómez Literature search and data analysis: Leonardo de Brito Andrade; Arturo H. Ariño
Drafted and revised the work: César Martín-Gómez; Amaia Zuazua-Ros; Arturo H. Ariño
Acknowledgements
The authors would like to thank their respective university centers, the Federal University of Santa Catarina, and the Universidad de Navarra, for encouragement and support
References
- FAO,. (2020). FAOSTAT, Statistics Division. In: Food AgricOrgan. United Nations.
- Herrero, M., Henderson, B., Havlík, P., Thornton, P. K., Conant, R. T., Smith, P., ... & Stehfest, E. (2016). Greenhouse gas mitigation potentials in the livestock sector. Nature Climate Change, 6(5), 452-461.
- FAO. (2017). The State of Food and Agriculture. Rome.
- Leip, A., Billen, G., Garnier, J., Grizzetti, B., Lassaletta, L., Reis, S., & Westhoek, H. (2015). Impacts of European livestock production: nitrogen, sulphur, phosphorus and greenhouse gas emissions, land-use, water eutrophication and biodiversity. Environmental Research Letters, 10(11), 115004.
- Ramankutty, N., Mehrabi, Z., Waha, K., Kremen, C., Herrero, M., & Rieseberg, L. H. (2018). Trends in global agricultural land use: implications for environmental health and food security.
- Mallin, M. A., McIver, M. R., Robuck, A. R., & Dickens, A.K. (2015). Industrial swine and poultry production causes chronic nutrient and fecal microbial stream pollution. Water, Air, & Soil Pollution, 226(12), 1-13.
- Ni, J. Q. (2015). Research and demonstration to improve air quality for the US animal feeding operations in the 21st centu-ry–A critical review. Environmental pollution, 200, 105-119.
- Zhao, Y., Shepherd, T. A., Li, H., & Xin, H. (2015). Environmental assessment of three egg production systems–Part I: Monitoring system and indoor air quality. Poultry Science, 94(3), 518-533.
- Chai, L., Zhao, Y., Xin, H., Wang, T., & Soupir, M. L. (2018). Mitigating airborne bacteria generations from cage-free layer litter by spraying acidic electrolysed water. Biosystems Engineering, 170, 61-71.
- Ni J-Q, Chai L, Chen L, et al (2012a) Characteristics of ammonia, hydrogen sulfide, carbon dioxide, and particulate matter concentrations in high-rise and manure-belt layer hen houses. Atmos Environ 57:165–174.
- Wang K, Wei B, Zhu S, Ye Z (2011) Ammonia and odour emitted from deep litter and fully slatted floor systems for growing-finishing pigs. Biosyst Eng 109:203–210.
- Amon, B., Kryvoruchko, V., Amon, T., & Zechmeister-Bol-tenstern, S. (2006). Methane, nitrous oxide and ammonia emissions during storage and after application of dairy cattle slurry and influence of slurry treatment. Agriculture, ecosys-tems & environment, 112(2-3), 153-162.
- Blanes-Vidal, V., Hansen, M. N., Pedersen, S., & Rom, H.B. (2008). Emissions of ammonia, methane and nitrous oxide from pig houses and slurry: Effects of rooting material, animal activity and ventilation flow. Agriculture, Ecosystems & Environment, 124(3-4), 237-244.
- Cortus, E. L., Jacobson, L. D., Hetchler, B. P., Heber, A. J., & Bogan, B. W. (2015). Methane and nitrous oxide analyzer comparison and emissions from dairy freestall barns with manure flushing and scraping. Atmospheric Environment, 100, 57-65.
- Olivier, J. G. J., Bouwman, A. F., Van der Hoek, K. W., & Berdowski, J. J. M. (1998). Global air emission inventories for anthropogenic sources of NOx, NH3 and N2O in 1990. Environmental pollution, 102(1), 135-148.
- Cederberg, C. (2009). Greenhouse gas emissions from Swedish production of meat, milk and eggs 1990 and 2005. SIK Institutet för livsmedel och bioteknik.
- Vergé, X. P. C., Dyer, J. A., Desjardins, R. L., & Worth, D. (2009). Long-term trends in greenhouse gas emissions from the Canadian poultry industry. Journal of Applied Poultry Research, 18(2), 210-222.
- Lesschen, J. P., van den Berg, M., Westhoek, H. J., Witzke, H. P., & Oenema, O. (2011). Greenhouse gas emission profiles of European livestock sectors. Animal Feed Science and Technology, 166, 16-28.
- Liang, L., Lal, R., Du, Z., Wu, W., & Meng, F. (2013). Esti-mation of nitrous oxide and methane emission from livestock of urban agriculture in Beijing. Agriculture, ecosystems & environment, 170, 28-35.
- Koerkamp, P. G., & Uenk, G. H. (1997). Climate conditions and aerial pollutants in and emissions from commmercial animal production systems in the Netherlands. In Ammonia and Odour Emissions from Animal Production Facilities (pp. 139-144).
- Sharpe, R. R., Harper, L. A., & Simmons, J. D. (2001). Methane emissions from swine houses in North Carolina. Chemosphere-Global Change Science, 3(1), 1-6.
- Gallmann, E., Hartung, E., & Jungbluth, T. (2003, June). Long-term study regarding the emission rates of ammonia and greenhouse gases from different housing systems for fattening pigs–final results. In Proceedings of the International Symposium on Gas and Odor Emissions from Animal Production, Horsens, Denmark. CIGR (pp. 122-130).
- Guarino M., Claudio F., Navarotto P., et al (2003) Ammonia, methane and nitrous oxide emissions and particulate matter concentrations in two different buildings for fattening pigs. Proc Int Symp Gaseous Odour Emiss from Anim Prod Facil 140–149.
- Nicks B., Laitat M., Vandenheede M., .(2003). Emissions of ammonia, nitrous oxide, methane, carbon dioxide and water vapor in the raising of weaned pigs on straw-based and sawdust-based deep litters. Anim Res 52:299–308.
- Dong, H., Zhu, Z., Shang, B., Kang, G., Zhu, H., & Xin, H. (2007). Greenhouse gas emissions from swine barns of various production stages in suburban Beijing, China. Atmospheric Environment, 41(11), 2391-2399.
- Stinn, J. P. (2014). Environmental assessment and control towards improved swine breeding-gestation-farrowing operation in the Midwestern United States (Doctoral dissertation, Iowa State University).
- Richardson, C. M., Baes, C. F., Amer, P. R., Quinton, C., Hely, F., Osborne, V. R., ... & Miglior, F. (2020). Estimating the environmental impact of dairy cattle breeding programs through emission intensity. Animal, 15(1), 100005.
- G Gustafsson, G., Jeppsson, K. H., Hultgren, J., & Sannö, J.O. (2005). Techniques to reduce the ammonia emission from a cowshed with tied dairy cattle. Agricultural Engineering International: CIGR Journal.
- Weiske, A., Vabitsch, A., Olesen, J. E., Schelde, K., Michel, J., Friedrich, R., & Kaltschmitt, M. (2006). Mitigation of greenhouse gas emissions in European conventional and organic dairy farming. Agriculture, ecosystems & environment, 112(2-3), 221-232.
- Starmans, D. A., & van der Hoek, K. W. (Eds.). (2007). Ammonia: the case of the Netherlands. Wageningen Academic Publishers.
- Stinn, J. P., Xin, H., Shepherd, T. A., Li, H., & Burns, R. T. (2014). Ammonia and greenhouse gas emissions from a modern US swine breeding-gestation-farrowing system. Atmospheric Environment, 98, 620-628.
- EPA USEPA. (2009). Inventory of U.S. Greenhouse GasEmissions and Sinks: 1990 – 2007.
- EPA USEPA .(2010). Environmental Mandatory Reporting of Greenhouse Gases: Injection and Geologic Sequestration of Carbon Dioxide. Fed Regist 75:75060–75089.
- Sun, F., Yun, D. A. I., & Yu, X. (2017). Air pollution, food production and food security: A review from the perspective of food system. Journal of integrative agriculture, 16(12), 2945-2962.
- Gobierno de España. (2019). Informe de Inventario Nacional de Gases de Efecto Invernadero (GEI). España, Informe In-ventarios GEI 1990-2018 (Edición 2020).
- Koerkamp, P. G., Metz, J. H. M., Uenk, G. H., Phillips, V.R., Holden, M. R., Sneath, R. W., & Wathes, C. M. (1998). Concentrations and emissions of ammonia in livestock buildings in Northern Europe. Journal of Agricultural Engineering Research, 70(1), 79-95.
- Ni, J. Q., Chai, L., Chen, L., Bogan, B. W., Wang, K., Cortus,E. L., ... & Diehl, C. A. (2012). Characteristics of ammonia, hydrogen sulfide, carbon dioxide, and particulate matter concentrations in high-rise and manure-belt layer hen houses. Atmospheric Environment, 57, 165-174.
- Ni, J. Q., Heber, A. J., Lim, T. T., Hanni, S. M., & Diehl, C.(2017). Laboratory Evaluation of a Manure Additive for Mitigating Gas and Odor Releases from Layer Hen Manure.Aerosol and Air Quality Research, 17(10), 2533-2541.
- Aneja, V. P., Roelle, P. A., Murray, G. C., Southerland, J., Erisman, J. W., Fowler, D., & Patni, N. (2001). Atmospheric nitrogen compounds II: emissions, transport, transformation, deposition and assessment. Atmospheric Environment, 35(11), 1903-1911.
- EPA USEPA. (2008). CERCLA/EPCRA administrative reporting exemption for air releases of hazardous substances from animal waste at farms.
- Blunden, J., Aneja, V. P., & Westerman, P. W. (2008). Measurement and analysis of ammonia and hydrogen sulfide emissions from a mechanically ventilated swine confinement building in North Carolina. Atmospheric environment, 42(14), 3315-3331.
- Méda, B., Hassouna, M., Lecomte, M., Germain, K., Dour-mad, J. Y., & Robin, P. (2015). Influence of season and outdoor run characteristics on excretion behaviour of organic broilers and gaseous emissions. Biosystems Engineering, 139, 35-47.
- Costantino, A., Fabrizio, E., Villagrá, A., Estellés, F., & Calvet, S. (2020). The reduction of gas concentrations in broiler houses through ventilation: Assessment of the thermal and electrical energy consumption. Biosystems Engineering, 199, 135-148.
- Shen, D., Wu, S., Li, Z., Tang, Q., Dai, P., Li, Y., & Li, C.(2019). Distribution and physicochemical properties of particulate matter in swine confinement barns. Environmental Pollution, 250, 746-753.
- Nicholson, F. A., Chambers, B. J., & Walker, A. W. (2004). Ammonia emissions from broiler litter and laying hen manure management systems. Biosystems Engineering, 89(2), 175-185.
- Rotz, C. A. (2004). Management to reduce nitrogen losses in animal production. Journal of animal science, 82(suppl_13), E119-E137.
- Wathes, C. M., Phillips, V. R., Holden, M. R., Sneath, R. W., Short, J. L., White, R. P. P., & Linke, S. (1998). Emissions of aerial pollutants in livestock buildings in Northern Europe: Overview of a multinational project. Journal of Agricultural Engineering Research, 70(1), 3-9.
- Von Essen, S., & Donham, K. (1999). Illness and injury in animal confinement workers. Occupational Medicine (Philadelphia, Pa.), 14(2), 337-350.
- Lee, C., Giles, L. R., Bryden, W. L., Downing, J. L., Owens,P. C., Kirby, A. C., & Wynn, P. C. (2005). Performance and endocrine responses of group housed weaner pigs exposed to the air quality of a commercial environment. Livestock Production Science, 93(3), 255-262.
- Rule, A. M., Chapin, A. R., McCarthy, S. A., Gibson, K. E., Schwab, K. J., & Buckley, T. J. (2005). Assessment of an aerosol treatment to improve air quality in a swine concentrated animal feeding operation (CAFO). Environmental science & technology, 39(24), 9649-9655.
- Amon, B., Kryvoruchko, V., Fröhlich, M., Amon, T., Pöllinger, Mösenbacher, I., & Hausleitner, A. (2007). Ammonia and greenhouse gas emissions from a straw flow system for fattening pigs: Housing and manure storage. Livestock Science, 112(3), 199-207.
- Banhazi, T. M., Seedorf, J., Laffrique, M., & Rutley, D. L. (2008). Identification of the risk factors for high airborne particle concentrations in broiler buildings using statistical modelling. biosystems engineering, 101(1), 100-110.
- Susanna, G., & Banks, D. E. (2009). Life-long exposures on the farm, respiratory symptoms, and lung function decline. Chest, 136(3), 662-663.
- Ulens, T., Millet, S., Van Ransbeeck, N., Van Weyenberg, S., Van Langenhove, H., & Demeyer, P. (2014). The effect of different pen cleaning techniques and housing systems on indoor concentrations of particulate matter, ammonia and greenhouse gases (CO2, CH4, N2O). Livestock Science, 159, 123-132.
- Xu, W., Zheng, K., Meng, L., Liu, X., Hartung, E., Roelcke, M., & Zhang, F. (2017). Concentrations and emissions of particulate matter from intensive pig production at a large farm in North China. Aerosol and Air Quality Research, 16(1), 79-90.
- FAO. (2011). World Livestock 2011 Livestock in food security.
- Stetzenbach, L. D., Buttner, M. P., & Cruz, P. (2004). Detection and enumeration of airborne biocontaminants. Current opinion in biotechnology, 15(3), 170-174.
- Zhai, Y., Li, X., Wang, T., Wang, B., Li, C., & Zeng, G. (2018).A review on airborne microorganisms in particulate matters: composition, characteristics and influence factors. Environment international, 113, 74-90.
- Ebner, M. R., Haselwandter, K., & Frank, A. (1992). Indoor and outdoor incidence of airborne fungal allergens at low-and high-altitude alpine environments. Mycological Research, 96(2), 117-124.
- Takahashi, T. (1997). Airborne fungal colony-forming units in outdoor and indoor environments in Yokohama, Japan. Myco-pathologia, 139(1), 23-33.
- Zuraimi, M. S., Fang, L., Tan, T. K., Chew, F. T., & Tham, K.W. (2009). Airborne fungi in low and high allergic prevalence child care centers. Atmospheric Environment, 43(15), 2391-2400.
- Rajasekar, A., & Balasubramanian, R. (2011). Assessment of airborne bacteria and fungi in food courts. Building and Environment, 46(10), 2081-2087.
- Dueker, M. E., O’Mullan, G. D., Weathers, K. C., Juhl, A. R., & Uriarte, M. (2012). Coupling of fog and marine microbial content in the near-shore coastal environment. Biogeoscienc-es, 9(2), 803-813.
- Lee, S. A., & Liao, C. H. (2014). Size-selective assessment of agricultural workers’ personal exposure to airborne fungi and fungal fragments. Science of the total environment, 466, 725-732.
- Vittal, B. P. R., & Glory, A. L. (1985). Airborne fungus spores of a library in India. Grana, 24(2), 129-132.
- Lacey, J., & Crook, B. (1988). Fungal and actinomycete spores as pollutants of the workplace and occupational allergens. The Annals of occupational hygiene, 32(4), 515-533.
- Hanhela, R., Louhelainen, K., & Pasanen, A. L. (1995). Prevalence of microfungi in Finnish cow barns and some aspects of the occurrence of Wallemia sebi and Fusaria. Scandinavian journal of work, environment & health, 223-228.
- Seedorf, J., Hartung, J., Schröder, M., Linkert, K. H., Phillips,V. R., Holden, M. R., ... & Wathes, C. M. (1998). Concentrations and emissions of airborne endotoxins and microorganisms in livestock buildings in Northern Europe. Journal of agricultural engineering research, 70(1), 97-109.
- Huijskens, E. G. W., Smit, L. A. M., Rossen, J. W. A., Heed-erik, D., & Koopmans, M. (2016). Evaluation of patients with communityâ?Â├é┬Éacquired pneumonia caused by zoonotic pathogens in an area with a high density of animal farms. Zoonoses and public health, 63(2), 160-166.
- Geornaras, I., De Jesus, A. E., Van Zyl, E., & Von Holy, A. (1996). Bacterial populations associated with poultry processing in a South African abattoir. Food Microbiology, 13(6), 457-465.
- Ellerbroek, L. (1997). Airborne microflora in poultry slaughtering establishments. Food Microbiology, 14(6), 527-531.
- Whyte, P. J. D. C., Collins, J. D., McGill, K., Monahan, C., & O’mahony, H. (2001). Distribution and prevalence of airborne microorganisms in three commercial poultry processing plants. Journal of Food Protection, 64(3), 388-391.
- Lues, J. F. R., Theron, M. M., Venter, P., & Rasephei, M.H. R. (2007). Microbial composition in bioaerosols of a high-throughput chicken-slaughtering facility. Poultry science, 86(1), 142-149.
- De Reu, K., Grijspeerdt, K., Heyndrickx, M., Zoons, J., De Baere, K., Uyttendaele, M., & Herman, L. (2005). Bacterial eggshell contamination in conventional cages, furnished cages and aviary housing systems for laying hens. British poultry science, 46(2), 149-155.
- De Reu, K., Messens, W., Heyndrickx, M., Rodenburg, T. B., Uyttendaele, M., & Herman, L. (2008). Bacterial contamination of table eggs and the influence of housing systems. World’s poultry science journal, 64(1), 5-19.
- Vuèemilo, M., Vinkoviæ, B., Matkoviæ, K., Štokoviæ, I., Jakšiæ, S., Radoviæ, S., & Graniæ, K. (2010). The influence of housing systems on the air quality and bacterial eggshell contamination of table eggs. Czech Journal of Animal Science, 55(6), 243-249.
- Hannah, J. F., Wilson, J. L., Cox, N. A., Cason, J. A., Bouras-sa, D. V., Musgrove, M. T., & Buhr, R. J. (2011). Comparison of shell bacteria from unwashed and washed table eggs harvested from caged laying hens and cage-free floor-housed laying hens. Poultry science, 90(7), 1586-1593.
- Ahmed, M. F. M., Schulz, J., & Hartung, J. (2013). Survival of Campylobacter jejuni in naturally and artificially contaminated laying hen feces. Poultry Science, 92(2), 364-369.
- WHO, G. (1990). Selected mycotoxins: ochratoxins, trichothecenes, ergot.
- Donnelly, P. (1994). Take my word for it: Trust in the context of birding and mountaineering. Qualitative Sociology, 17(3), 215-241.
- Eisel, W. G., Linton, R. H., & Muriana, P. M. (1997). A survey of microbial levels for incoming raw beef, environmental sources, and ground beef in a red meat processing plant. Food microbiology, 14(3), 273-282.
- Shale, K. (2004). The Prevalence of Meat-borne and Airborne Staphylococci in Deboning Area of Low-and High-through-put Red Meat Abattoirs (Doctoral dissertation, Central University of Technology, Free State).
- Zhao, Y., Zhao, D., Ma, H., Liu, K., Atilgan, A. T. I. L. G. A.N., & Xin, H. (2016). Environmental assessment of three egg production systems–Part III: Airborne bacteria concentrations and emissions. Poultry science, 95(7), 1473-1481.
- Hellmann, B., Zelles, L., Palojarvi, A., & Bai, Q. (1997). Emission of climate-relevant trace gases and succession of microbial communities during open-windrow composting. Applied and environmental microbiology, 63(3), 1011-1018.
- Moss, A. R., Jouany, J. P., & Newbold, J. (2000, May). Methane production by ruminants: its contribution to global warming. In Annales de zootechnie (Vol. 49, No. 3, pp. 231-253). EDP Sciences.
- Amon, B., Amon, T., Boxberger, J., & Alt, C. (2001). Emissions of NH3, N2O and CH4 from dairy cows housed in a farmyard manure tying stall (housing, manure storage, manure spreading). Nutrient cycling in Agroecosystems, 60(1), 103-113.
- Olesen, J. E., Schelde, K., Weiske, A., Weisbjerg, M. R., As-man, W. A. H., & Djurhuus, J. (2006). Modelling greenhouse gas emissions from European conventional and organic dairy farms. Agriculture, ecosystems & environment, 112(2-3), 207-220.
- Monteny, G. J., Bannink, A., & Chadwick, D. (2006). Greenhouse gas abatement strategies for animal husbandry. Agriculture, Ecosystems & Environment, 112(2-3), 163-170.
- Philippe, F. X., & Nicks, B. (2015). Review on greenhouse gas emissions from pig houses: Production of carbon dioxide, methane and nitrous oxide by animals and manure. Agriculture, Ecosystems & Environment, 199, 10-25.
- Kebreab, E., Clark, K., Wagner-Riddle, C., & France, J. (2006). Methane and nitrous oxide emissions from Canadian animal agriculture: A review. Canadian Journal of Animal Science, 86(2), 135-157.
- Hegarty, R. S., Goopy, J. P., Herd, R. M., & McCorkell, B. (2007). Cattle selected for lower residual feed intake have reduced daily methane production. Journal of animal science, 85(6), 1479-1486.
- Rotz, C. A. (2018). Modeling greenhouse gas emissions from dairy farms. Journal of dairy science, 101(7), 6675-6690.
- Külling, D. R., Menzi, H., Kröber, T. F., Neftel, A., Sutter,F., Lischer, P., & Kreuzer, M. (2001). Emissions of ammonia, nitrous oxide and methane from different types of dairy manure during storage as affected by dietary protein content. The Journal of Agricultural Science, 137(2), 235-250.
- Snell, H. G. J., Seipelt, F., & Van den Weghe, H. F. A. (2003). Ventilation rates and gaseous emissions from naturally ventilated dairy houses. Biosystems Engineering, 86(1), 67-73.
- Zhang, G., Strøm, J. S., Li, B., Rom, H. B., Morsing, S., Dahl, P., & Wang, C. (2005). Emission of ammonia and other contaminant gases from naturally ventilated dairy cattle buildings. Biosystems engineering, 92(3), 355-364.
- Webb, J., Sommer, S. G., Kupper, T., Groenestein, K., Hutch-ings, N. J., Eurich-Menden, B., & Amon, B. (2012). Emissions of ammonia, nitrous oxide and methane during the management of solid manures. In Agroecology and strategies for climate change (pp. 67-107). Springer, Dordrecht.
- Kumari, S., Fagodiya, R. K., Hiloidhari, M., Dahiya, R. P., & Kumar, A. (2020). Methane production and estimation from livestock husbandry: A mechanistic understanding and emerging mitigation options. Science of the Total Environment, 709, 136135.
- Mathot, M., Decruyenaere, V., Stilmant, D., & Lambert, R. (2012). Effect of cattle diet and manure storage conditions on carbon dioxide, methane and nitrous oxide emissions from tie-stall barns and stored solid manure. Agriculture, Ecosystems & Environment, 148, 134-144.
- Xie, Q., Ni, J. Q., & Su, Z. (2017). Fuzzy comprehensive evaluation of multiple environmental factors for swine building assessment and control. Journal of Hazardous Materials, 340, 463-471.
- Alonso-Moreno, C., Garde, J. J., Zafrilla, J. E., Cana-les-Vázquez, J., Gueddari, A., & García-Yuste, S. (2018). The Carbon Dioxide-Rumen Fermentation Processes-strategy, a proposal to sustain environmentally friendly dairy farms. Journal of Cleaner Production, 204, 735-743.
- Liang, Y., Xin, H., Wheeler, E. F., Gates, R. S., Li, H., Zajaczkowski, J. S., & Zajaczkowski, F. J. (2005). Ammonia emissions from US laying hen houses in Iowa and Pennsylvania. Transactions of the ASAE, 48(5), 1927-1941.
- McGovern, R. H., Feddes, J. J. R., Zuidhof, M. J., Hanson,J. A., & Robinson, F. E. (2001). Growth performance, heart characteristics and the incidence of ascites in broilers in response to carbon dioxide and oxygen concentrations. Canadian Biosystems Engineering, 43, 4-1.
- Gerritzen, M., Lambooij, B., Reimert, H., Stegeman, A., & Spruijt, B. (2007). A note on behaviour of poultry exposed to increasing carbon dioxide concentrations. Applied Animal Behaviour Science, 108(1-2), 179-185.
- Olanrewaju, H. A., Dozier, W. A., Purswell, J. L., Branton,S. L., Miles, D. M., Lott, B. D., ... & Thaxton, J. P. (2008).Growth performance and physiological variables for broiler chickens subjected to short-term elevated carbon dioxide concentrations. International Journal of Poultry Science, 7(8),738-742.
- Jeppsson, K. H. (2000). SE—structure and environment: carbon dioxide emission and water evaporation from deep litter systems. Journal of agricultural engineering research, 77(4), 429-440.
- Wolter, M., Prayitno, S., & Schuchardt, F. (2004). Greenhouse gas emission during storage of pig manure on a pilot scale. Bioresource technology, 95(3), 235-244.
- EPA USEPA. (2003). Toxicological Review of Hydrogen Sul-fide (CAS No. 7783-06-4).
- Oesterhelweg, L., & Püschel, K. (2008). Death may come on like a stroke of lightening. International journal of legal medicine, 122(2), 101-107.
- Lee, C., Morris, D. L., Lefever, K. M., & Dieter, P. A. (2020). Feeding a diet with corn distillers grain with solubles to dairy cows alters manure characteristics and ammonia and hydrogen sulfide emissions from manure. Journal of dairy science, 103(3), 2363-2372.
- Kandylis, K. (1984). Toxicology of sulfur in ruminants. Journal of dairy science, 67(10), 2179-2187.
- Watt, A. C., Aziz, S. M., & Banhazi, T. M. (2010, June). Air pollution and livestock production. In Proceedings of the 17th World Congress of the International Commission of Agricultural and Biosystems Engineering (CIGR 2010) (pp. 1-12). International Commission of Agricultural and Biosystems Engineering (CIGR).
- Bjerg B., Norton T., Banhazi T., (2013). Modelling of ammonia emissions from naturally ventilated livestock buildings. Part 1: Ammonia release modelling. Biosyst Eng 116:232–245.
- EPA USEPA. (2001). Emissions from animal feeding operations.
- Carew, R. (2010). Ammonia emissions from livestock industries in Canada: Feasibility of abatement strategies. Environmental Pollution, 158(8), 2618-2626.
- NRC. (2003). Air Emissions from Animal Feeding Operations: Current Knowledge, Future Needs. National Academies Press, Washington, D.C.
- Hallquist, M., Wenger, J. C., Baltensperger, U., Rudich, Y., Simpson, D., Claeys, M., & Wildt, J. (2009). The formation, properties and impact of secondary organic aerosol: current and emerging issues. Atmospheric chemistry and physics, 9(14), 5155-5236.
- EEA. (2017). Air quality in Europe.
- Webb, J., Menzi, H., Pain, B. F., Misselbrook, T. H., Däm-mgen, U., Hendriks, H., & Döhler, H. (2005). Managing ammonia emissions from livestock production in Europe. Environmental pollution, 135(3), 399-406.
- Oenema, O., Oudendag, D., & Velthof, G. L. (2007). Nutrient losses from manure management in the European Union. Livestock science, 112(3), 261-272.
- Seedorf, J., & Hartung, J. (1999). Survey of ammonia concentrations in livestock buildings. The Journal of Agricultural Science, 133(4), 433-437.
- Kristensen, H. H., Burgess, L. R., Demmers, T. G., & Wathes,C. M. (2000). The preferences of laying hens for different concentrations of atmospheric ammonia. Applied Animal Behaviour Science, 68(4), 307-318.
- Popescu, S. I. L. V. A. N. A., Stefan, R., Borda, C., Lazar, E, Sandru, C. D., & Spinu, M. A. R. I. N. A. (2010). The am-monia concentration in growing-finishing pig houses. LucrÃ├é??ri Ã├é??tiinÃ├é?£ifice MedicinÃ├é?? VeterinarÃ├é??, 43, 320-326.
- Barrasa, M., Lamosa, S., Fernandez, M. D., & Fernandez,E. (2012). Occupational exposure to carbon dioxide, ammonia and hydrogen sulphide on livestock farms in north-west Spain. Annals of Agricultural and Environmental Medicine, 19(1).
- Olson, D. K., & Bark, S. M. (1996). Health hazards affecting the animal confinement farm worker. AAOHN Journal, 44(4), 198-204.
- Hamilton, T. D. C., Roe, J. M., Hayes, C. M., & Webster, A.J. F. (1998). Effects of ammonia inhalation and acetic acid pretreatment on colonization kinetics of toxigenic Pasteurella multocida within upper respiratory tracts of swine. Journal of clinical microbiology, 36(5), 1260-1265.
- Mosquera, J., Monteny, G. J., & Erisman, J. W. (2005). Overview and assessment of techniques to measure ammonia emissions from animal houses: the case of the Netherlands. Environmental Pollution, 135(3), 381-388.
- Fabbri, C., Valli, L., Guarino, M., Costa, A., & Mazzotta,V. (2007). Ammonia, methane, nitrous oxide and particulate matter emissions from two different buildings for laying hens. Biosystems Engineering, 97(4), 441-455.
- Cornejo, C., & Wilkie, A. C. (2010). Greenhouse gas emissions and biogas potential from livestock in Ecuador. Energy for sustainable development, 14(4), 256-266.
- Monteny, G. J., Groenestein, C. M., & Hilhorst, M. A. (2001). Interactions and coupling between emissions of methane and nitrous oxide from animal husbandry. Nutrient Cycling in Agroecosystems, 60(1), 123-132.
- Monteny, G. J., & Erisman, J. W. (1998). Ammonia emission from dairy cow buildings: a review of measurement techniques, influencing factors and possibilities for reduction. Netherlands Journal of Agricultural Science, 225-247.
- Dekker, S. E. M., Aarnink, A. J. A., De Boer, I. J. M., & Koerkamp, P. G. (2011). Emissions of ammonia, nitrous oxide, and methane from aviaries with organic laying hen husbandry. Biosystems Engineering, 110(2), 123-133.
- Duxbury, J. M. (1994). The significance of agricultural sources of greenhouse gases. Fertilizer research, 38(2), 151-163.
- Jarvis, S. C., & Pain, B. F. (1994). Greenhouse gas emissions from intensive livestock systems: their estimation and technologies for reduction. In Climate change: Significance for agriculture and forestry (pp. 27-38). Springer, Dordrecht.
- Mosier AR. (1994). Nitrous oxide emissions from agriculturalsoils. Fertil Res 37:191–200.
- Van Amstel, A. R., & Swart, R. J. (1994). Methane and nitrous oxide emissions: an introduction. Fertilizer research, 37(3), 213-225.
- Khan, R. Z., Müller, C., & Sommer, S. G. (1997). Micromete-orological mass balance technique for measuring CH4 emission from stored cattle slurry. Biology and fertility of soils, 24(4), 442-444.
- Zhu, Z., Dong, H., Zhou, Z., Xin, H., & Chen, Y. (2011). Am-monia and greenhouse gases concentrations and emissions of a naturally ventilated laying hen house in Northeast China. Transactions of the ASABE, 54(3), 1085-1091.
- Fournel, S., Pelletier, F., Godbout, S., Lagacé, R., & Feddes, J. (2011). Greenhouse gas emissions from three cage layer housing systems. Animals, 2(1), 1-15.
- Alberdi, O., Arriaga, H., Calvet, S., Estellés, F., & Merino,P. (2016). Ammonia and greenhouse gas emissions from an enriched cage laying hen facility. Biosystems Engineering, 144, 1-12.
- Alberdi, O., Arriaga, H., Calvet, S., Estellés, F., & Merino,P. (2016). Ammonia and greenhouse gas emissions from an enriched cage laying hen facility. Biosystems Engineering, 144, 1-12.
- Zong, C., Li, H., & Zhang, G. (2015). Ammonia and greenhouse gas emissions from fattening pig house with two types of partial pit ventilation systems. Agriculture, Ecosystems & Environment, 208, 94-105.
- Jungbluth, T., Hartung, E., & Brose, G. (2001). Greenhouse gas emissions from animal houses and manure stores. Nutrient cycling in Agroecosystems, 60(1), 133-145.
- EEA. (2020). European Environment Agency’s home page — European Environment Agency. In: Eur. Environ.
- European Commission Publication. (1999). Directive 1999/30/EC of 22 April 1999 relating to limit values for sulphur dioxide, nitrogen dioxide and oxides of nitrogen, particulate matter and lead in ambient air C directive. Off J Eur Comm 41–60.
- Harry, E. G. (1978). Air pollution in farm buildings and meth-ods of control: a review. Avian Pathology, 7(4), 441-454.
- Cambra-López, M., Aarnink, A. J., Zhao, Y., Calvet, S., & Torres, A. G. (2010). Airborne particulate matter from livestock production systems: A review of an air pollution problem. Environmental pollution, 158(1), 1-17.
- Mostafa, E., & Buescher, W. (2011). Indoor air quality improvement from particle matters for laying hen poultry houses. Biosystems Engineering, 109(1), 22-36.
- Van Ransbeeck N, Van Langenhove H, Van Weyenberg S, et al. (2012). Typical indoor concentrations and emission rates of particulate matter at building level: A case study to setup a measuring strategy for pig fattening facilities. Biosyst Eng 111:280–289.
- Van Ransbeeck N, Van Langenhove H, Demeyer P. (2013). Indoor concentrations and emissions factors of particulate matBiosyst Eng 116:518–528.
- Wang-Li, L., Li, Q., & Byfield, G. E. (2013). Identification of bioaerosols released from an egg production facility in the southeast United States. Environmental Engineering Science, 30(1), 2-10.
- Mostafa, E., Nannen, C., Henseler, J., Diekmann, B., Gates, R., & Buescher, W. (2016). Physical properties of particulate matter from animal houses—empirical studies to improve emission modelling. Environmental Science and Pollution Research, 23(12), 12253-12263.
- Mostafa, E., Hoelscher, R., Diekmann, B., Ghaly, A. E., & Buescher, W. (2017). Evaluation of two indoor air pollution abatement techniques in forced-ventilation fattening pig barns. Atmospheric Pollution Research, 8(3), 428-438.
- Yao, Q., Yang, Z., Li, H., Buser, M. D., Wanjura, J. D., Downey, P. M., & Hapeman, C. J. (2018). Assessment of particulate matter and ammonia emission concentrations and respective plume profiles from a commercial poultry house. Environmental Pollution, 238, 10-16.
- Rundell, K. W. (2012). Effect of air pollution on athlete health and performance. British journal of sports medicine, 46(6), 407-412.
- Ivester, K. M., Smith, K., Moore, G. E., Zimmerman, N. J., & Couëtil, L. L. (2012). Variability in particulate concentrations in a horse training barn over time. Equine Veterinary Journal, 44, 51-56.
- Ivester, K. M., Couëtil, L. L., & Zimmerman, N. J. (2014). Investigating the link between particulate exposure and airway inflammation in the horse. Journal of veterinary internal medicine, 28(6), 1653-1665.
- Giles, L. V., & Koehle, M. S. (2014). The health effects ofexercising in air pollution. Sports Medicine, 44(2), 223-249.
- Millerickâ?Â├é┬ÉMay, M. L., Karmaus, W., Derksen, F. J., Berthold, B., & Robinson, N. E. (2015). Airborne particulates (PM 10) and tracheal mucus: A case–control study at an A merican Thoroughbred racetrack. Equine Veterinary Journal, 47(4), 410-414.
- Nazarenko, Y., Westendorf, M. L., Williams, C. A., & Maine-lis, G. (2018). The effects of bedding type in stalls and activity of horses on stall air quality. Journal of Equine Veterinary Science, 67, 91-98.
- Hafner, S. D., Howard, C., Muck, R. E., Franco, R. B., Mon-tes, F., Green, P. G., ... & Rotz, C. A. (2013). Emission of volatile organic compounds from silage: Compounds, sources, and implications. Atmospheric Environment, 77, 827-839.
- Douglas, P., Robertson, S., Gay, R., Hansell, A. L., & Gant, T.W. (2018). A systematic review of the public health risks of bioaerosols from intensive farming. International journal of hygiene and environmental health, 221(2), 134-173.
- Trabue, S., Kerr, B., Bearson, B., & Ziemer, C. (2011). Swine odor analyzed by odor panels and chemical techniques. Journal of Environmental Quality, 40(5), 1510-1520.
- Guffanti, P., Pifferi, V., Falciola, L., & Ferrante, V. (2018).Analyses of odours from concentrated animal feeding opera-tions: A review. Atmospheric Environment, 175, 100-108.
- Wang, Y. C., Han, M. F., Jia, T. P., Hu, X. R., Zhu, H. Q., Tong,Z., ... & Hsi, H. C. (2021). Emissions, measurement, and control of odor in livestock farms: A review. Science of The Total Environment, 776, 145735.
- Komilis, D. P., Ham, R. K., & Park, J. K. (2004). Emission of volatile organic compounds during composting of municipal solid wastes. Water research, 38(7), 1707-1714.
- Filipy, J., Rumburg, B., Mount, G., Westberg, H., & Lamb,B. (2006). Identification and quantification of volatile organic compounds from a dairy. Atmospheric Environment, 40(8), 1480-1494.
- Fang, J. J., Yang, N., Cen, D. Y., Shao, L. M., & He, P. J.(2012). Odor compounds from different sources of landfill: characterization and source identification. Waste Management, 32(7), 1401-1410.
- Conti, C., Guarino, M., & Bacenetti, J. (2020). Measurements techniques and models to assess odor annoyance: A review. Environment international, 134, 105261.
- Martin, R. S., Silva, P. J., Moore, K., Erupe, M., & Doshi, V.S. (2008). Particle composition and size distributions in and around a deep-pit swine operation, Ames, IA. Journal of Atmospheric Chemistry, 59(2), 135-150.
- Chung, M. Y., Beene, M., Ashkan, S., Krauter, C., & Hasson,S. (2010). Evaluation of non-enteric sources of non-meth-ane volatile organic compound (NMVOC) emissions from dairies. Atmospheric Environment, 44(6), 786-794.
- Howard, C. J., Kumar, A., Malkina, I., Mitloehner, F., Green,P. G., Flocchini, R. G., & Kleeman, M. J. (2010). Reactive organic gas emissions from livestock feed contribute significantly to ozone production in central California. Environmental Science & Technology, 44(7), 2309-2314.
- Hu, J., Howard, C. J., Mitloehner, F., Green, P. G., & Kleeman, M. J. (2012). Mobile source and livestock feed contributions to regional ozone formation in Central California. Environmental science & technology, 46(5), 2781-2789.
- Monks, P. S., Granier, C., Fuzzi, S., Stohl, A., Williams, M. L., Akimoto, H., & von Glasow, R. (2009). Atmospheric composition change–global and regional air quality. Atmospheric environment, 43(33), 5268-5350.
- ISO. (2008). ISO 5492:2008 - Sensory analysis — Vocabulary.
- Henshaw, P., Nicell, J., & Sikdar, A. (2006). Parameters for the assessment of odour impacts on communities. Atmospheric Environment, 40(6), 1016-1029.
- Aatamila, M., Verkasalo, P. K., Korhonen, M. J., Suominen,L., Hirvonen, M. R., Viluksela, M. K., & Nevalainen, A. (2011). Odour annoyance and physical symptoms among residents living near waste treatment centres. Environmental Research, 111(1), 164-170.
- Capelli, L., Sironi, S., Del Rosso, R., & Guillot, J. M. (2013). Measuring odours in the environment vs. dispersion model-ling: A review. Atmospheric Environment, 79, 731-743.
- Schiffman, S. S., Bennett, J. L., & Raymer, J. H. (2001). Quantification of odors and odorants from swine operations in North Carolina. Agricultural and Forest Meteorology, 108(3), 213-240.
- Domingo, J. L., & Nadal, M. (2009). Domestic waste composting facilities: A review of human health risks. Environment international, 35(2), 382-389.
- Palmiotto, M., Fattore, E., Paiano, V., Celeste, G., Colombo, A., & Davoli, E. (2014). Influence of a municipal solid waste landfill in the surrounding environment: Toxicological risk and odor nuisance effects. Environment international, 68, 16-24.
- Blanes-Vidal, V. (2015). Air pollution from biodegradable wastes and non-specific health symptoms among residents: direct or annoyance-mediated associations?. Chemosphere, 120, 371-377.
- Keck, M., Mager, K., Weber, K., Keller, M., Frei, M., Steiner, B., & Schrade, S. (2018). Odour impact from farms with animal husbandry and biogas facilities. Science of the Total Environment, 645, 1432-1443.
- Bibbiani, C., & Russo, C. (2012). Odour emission from intensive livestock production system: Approaches for emission abatement and evaluation of their effectiveness. Large Anim. Rev, 18(3), 135-138.
- Hayes, J. E., Stevenson, R. J., & Stuetz, R. M. (2014). The impact of malodour on communities: A review of assessment techniques. Science of the Total Environment, 500, 395-407.
- Hansen, M. N., Kai, P., & Møller, H. B. (2006). Effects of anaerobic digestion and separation of pig slurry on odor emission. Applied engineering in agriculture, 22(1), 135-139.
- Orzi, V., Riva, C., Scaglia, B., D’Imporzano, G., Tambone, F., & Adani, F. (2018). Anaerobic digestion coupled with di-gestate injection reduced odour emissions from soil during manure distribution. Science of The Total Environment, 621, 168-176.
- Akdeniz, N., Koziel, J. A., Ahn, H. K., Glanville, T. D., Crawford, B. P., & Raman, D. R. (2009). Air sampling and analysis method for volatile organic compounds (VOCs) related to field-scale mortality composting operations. Journal of agricultural and food chemistry, 57(13), 5658-5664.
- Akdeniz, N., Koziel, J. A., Ahn, H. K., Glanville, T. D., & Crawford, B. P. (2010). Field scale evaluation of volatile organic compound production inside biosecure swine mortality composts. Waste Management, 30(10), 1981-1988.
- Akdeniz, N., Koziel, J. A., Ahn, H. K., Glanville, T. D., Crawford, B. P., & Raman, D. R. (2010). Laboratory scale evaluation of volatile organic compound emissions as indication of swine carcass degradation inside biosecure composting units. Bioresource technology, 101(1), 71-78.
- Akdeniz, N., Koziel, J. A., Glanville, T. D., Ahn, H., & Crawford, B. P. (2011). Air sampling methods for VOCs related to field-scale biosecure swine mortality composting. Biore-source technology, 102(3), 3599-3602.
- Costa, T., & Akdeniz, N. (2019). A review of the animal disease outbreaks and biosecure animal mortality composting systems. Waste Management, 90, 121-131.
- Ellis, C. K., Stahl, R. S., Nol, P., Waters, W. R., Palmer, M. V., Rhyan, J. C., & Salman, M. D. (2014). A pilot study exploring the use of breath analysis to differentiate healthy cattle from cattle experimentally infected with Mycobacterium bovis. PloS one, 9(2), e89280.
- Mottram, T. T., Dobbelaar, P., Schukken, Y. H., Hobbs, P. J., & Bartlett, P. N. (1999). An experiment to determine the feasibility of automatically detecting hyperketonaemia in dairy cows. Livestock production science, 61(1), 7-11.
- Fend, R., Geddes, R., Lesellier, S., Vordermeier, H. M., Corner, L. A. L., Gormley, E., & Chambers, M. A. (2005). Use of an electronic nose to diagnose Mycobacterium bovis infection in badgers and cattle. Journal of clinical microbiology, 43(4), 1745-1751.
- Kumanan V., Nugen SR., Baeumner AJ., Chang YF,. (2009). A biosensor assay for the detection of Mycobacterium avium subsp. paratuberculosis in fecal samples. J Vet Sci 10:35–42.
- Shirasu, M., & Touhara, K. (2011). The scent of disease: volatile organic compounds of the human body related to disease and disorder. The Journal of Biochemistry, 150(3), 257-266.
- Peled, N., Ionescu, R., Nol, P., Barash, O., McCollum, M., VerCauteren, K., & Haick, H. (2012). Detection of volatile organic compounds in cattle naturally infected with Mycobacterium bovis. Sensors and Actuators B: Chemical, 171, 588-594.
- Ngwabie, N. M., Jeppsson, K. H., Gustafsson, G., & Nimmermark, S. (2011). Effects of animal activity and air temperature on methane and ammonia emissions from a naturally ventilated building for dairy cows. Atmospheric Environment, 45(37), 6760-6768.
- Huang, C. M., Yuan, C. S., Yang, W. B., & Yang, L. (2019).Temporal variations of greenhouse gas emissions and carbon sequestration and stock from a tidal constructed mangrove wetland. Marine Pollution Bulletin, 149, 110568.
- Gustafsson, G. (1999). Factors affecting the release and concentration of dust in pig houses. Journal of Agricultural Engineering Research, 74(4), 379-390.
- Puma, M. C., & Maghirang, R. G. (2000). A macroscopic model for predicting dust concentration distribution in swine buildings. Indoor and Built Environment, 9(3-4), 182-191.
- Kim, K. Y., Ko, H. J., Lee, K. J., Park, J. B., & Kim, C. N.(2005). Temporal and spatial distributions of aerial contaminants in an enclosed pig building in winter. Environmental Research, 99(2), 150-157.
- Nannen, C., & Büscher, W. (2006). Einfluss der Tieraktivität auf Staubfreisetzung und Partikelemissionen-Untersuchungen in Mastschweine-Ställen. Landtechnik, 61(1), 36-37.
- Bunney, P. E., Zink, A. N., Holm, A. A., Billington, C. J., & Kotz, C. M. (2017). Orexin activation counteracts decreas-es in nonexercise activity thermogenesis (NEAT) caused by high-fat diet. Physiology & behavior, 176, 139-148.
- Elfman, L., Wålinder, R., Riihimäki, M., & Pringle, J. (2011). Air quality in horse stables. In Chemistry, Emission Control, Radioactive Pollution and Indoor Air Quality. IntechOpen.
- Adhikari, A., Sen, M. M., Gupta-Bhattacharya, S., & Chanda,S. (2004). Volumetric assessment of airborne fungi in two sections of a rural indoor dairy cattle shed. Environment International, 29(8), 1071-1078.
- Herrero, B., & Zaldivar, P. (1997). Effects of meteorological factors on the levels of Alternaria and Cladosporium spores in the atmosphere of Palencia, 1990–92. Grana, 36(3), 180-184.
- Rong, L., Liu, D., Pedersen, E. F., & Zhang, G. (2014). Effect of climate parameters on air exchange rate and ammonia and methane emissions from a hybrid ventilated dairy cow building. Energy and Buildings, 82, 632-643.
- Koinakis, C. J. (2005). Combined thermal and natural ventilation modeling for long-term energy assessment: validation with experimental measurements. Energy and Buildings, 37(4), 311-323.
- Schulze, T., & Eicker, U. (2013). Controlled natural ventilation for energy efficient buildings. Energy and buildings, 56, 221-232.
- Ecim-Djuric, O., & Topisirovic, G. (2010). Energy efficiency optimization of combined ventilation systems in livestock buildings. Energy and Buildings, 42(8), 1165-1171.
- Ngwabie, N. M., Jeppsson, K. H., Nimmermark, S., Swens-son, C., & Gustafsson, G. (2009). Multi-location measurements of greenhouse gases and emission rates of methane and ammonia from a naturally-ventilated barn for dairy cows. Biosystems engineering, 103(1), 68-77.
- Arogo, J., Zhang, R. H., Riskowski, G. L., Christianson, L. L., & Day, D. L. (1999). Mass transfer coefficient of ammonia in liquid swine manure and aqueous solutions. Journal of Agricultural Engineering Research, 73(1), 77-86.
- Ye, Z., Zhang, G., Li, B., Strøm, J. S., & Dahl, P. J. (2008).Ammonia emissions affected by airflow in a model pig house: effects of ventilation rate, floor slat opening, and headspace height in a manure storage pit. Transactions of the ASABE, 51(6), 2113-2122.
- Saha, C. K., Zhang, G., Ni, J. Q., & Ye, Z. (2011). Similarity criteria for estimating gas emission from scale models. Biosystems engineering, 108(3), 227-236.
- KATAYAMA, Y., OIKAWA, M. A., YOSHIHARA, T., KU-WANO, A., & HOBO, S. (1995). Clinico-pathological effects of atmospheric ammonia exposure on horses. Journal of Equine Science, 6(3), 99-104.
- Curtis, L., Raymond, S., & Clarke, A. (1996). Dust and ammonia in horse stalls with different ventilation rates and bedding. Aerobiologia, 12(1), 239-247.
- Holcombe, S. J., Jackson, C., Gerber, V., Jefcoat, A., Berney, C., Eberhardt, S., & Robinson, N. E. (2001). Stabling is associated with airway inflammation in young Arabian horses.Equine veterinary journal, 33(3), 244-249.
- Kwiatkowska-Stenzel, A., SowiÃ├é??ska, J., & Witkowska, D. (2014). Analysis of noxious gas pollution in horse stable air. Journal of Equine Veterinary Science, 34(2), 249-256.
- Bøe, K. E., Dragsund, G., Jørgensen, G. H., & Fabian-Wheel-er, E. (2017). Air Quality in Norwegian Horse Stables at Low Outdoor Temperatures. Journal of Equine Veterinary Science, 55, 44-50.
- Clements, J. M., & Pirie, R. S. (2007). Respirable dust concentrations in equine stables. Part 1: validation of equipment and effect of various management systems. Research in Veterinary Science, 83(2), 256-262.
- Fleming, K., Hessel, E. F., & Van den Weghe, H. F. A. (2009). Gas and particle concentrations in horse stables with individual boxes as a function of the bedding material and the mucking regimen. Journal of animal science, 87(11), 3805-3816.
- Wålinder, R., Riihimäki, M., Bohlin, S., Hogstedt, C., Nord-quist, T., Raine, A., & Elfman, L. (2011). Installation of mechanical ventilation in a horse stable: effects on air quality and human and equine airways. Environmental health and preventive medicine, 16(4), 264-272.
- Cheng, T., Jiang, Y., Xu, Y., & Zhang, Y. (2002). Mathematical model for simulation of VOC emissions and concentrations in buildings. Atmospheric Environment, 36(32), 5025-5030.
- Young, P., Price, L., Berckmans, D., & Janssens, K. (2000). Recent developments in the modelling of imperfectly mixed airspaces. Computers and electronics in agriculture, 26(3), 239-254.
- Wu, W., Zhang, G., Bjerg, B., & Nielsen, P. V. (2012). An assessment of a partial pit ventilation system to reduce emission under slatted floor–Part 2: Feasibility of CFD prediction using RANS turbulence models. Computers and Electronics in agriculture, 83, 134-142.
- EPA USEPA. (2004). Risk assessment Evaluation for concentrated animal feeding operations.
- Cambra-López, M., Torres, A. G., Aarnink, A. J. A., & Ogink,N. W. (2011). Source analysis of fine and coarse particulate matter from livestock houses. Atmospheric Environment, 45(3), 694-707.
- Smets, W., Moretti, S., Denys, S., & Lebeer, S. (2016). Airborne bacteria in the atmosphere: Presence, purpose, and potential. Atmospheric Environment, 139, 214-221.
- Rylander, R., & Carvalheiro, M. F. (2006). Airways inflammation among workers in poultry houses. International archives of occupational and environmental health, 79(6), 487-490.
- Zucker, B. A., Trojan, S., & Müller, W. (2000). Airborne gramâ?Â├é┬Énegative bacterial flora in animal houses. Journal of Veterinary Medicine, Series B, 47(1), 37-46.
- Wales, A., Breslin, M., & Davies, R. (2006). Assessment of cleaning and disinfection in Salmonella-contaminated poultry layer houses using qualitative and semi-quantitative culture techniques. Veterinary Microbiology, 116(4), 283-293.
- Quarles, C. L., Gentry, R. F., & Bressler, G. O. (1970). Bacte-rial contamination in poultry houses and its relationship to egg hatchability. Poultry Science, 49(1), 60-66.
- Soupir, M. L., Mostaghimi, S., Yagow, E. R., Hagedorn, C., & Vaughan, D. H. (2006). Transport of fecal bacteria from poultry litter and cattle manures applied to pastureland. Water, Air, and Soil Pollution, 169(1), 125-136.
- Wang, X., Zhang, Y., Riskowski, G. L., & Ellis, M. (2002). SE—Structures and environment: Measurement and analysis of dust spatial distribution in a mechanically ventilated pig building. Biosystems engineering, 81(2), 225-236.
- O’Shaughnessy PT., Achutan C., Karsten AW., .(2002). Temporal Variation of Indoor Air Quality in an Enclosed Swine Confinement Building. J Agric Saf Health 8:349–364.
- Vandenput, S., Istasse, L., Nicks, B., & Lekeux, P. (1997). Airborne dust and aeroallergen concentrations in different sources of feed and bedding for horses. Veterinary Quarterly, 19(4), 154-158.
- Fleming, K., Hessel, E. F., & Van den Weghe, H. F. A. (2008). Generation of airborne particles from different bedding materials used for horse keeping. Journal of equine veterinary science, 28(7), 408-418.
- Arriaga, H., Pinto, M., Calsamiglia, S., & Merino, P. (2009). Nutritional and management strategies on nitrogen and phosphorus use efficiency of lactating dairy cattle on commercial farms: An environmental perspective. Journal of Dairy Science, 92(1), 204-215.
- Merino, P., Ramirez-Fanlo, E., Arriaga, H., Del Hierro, O., Artetxe, A., & Viguria, M. (2011). Regional inventory of methane and nitrous oxide emission from ruminant livestock in the Basque Country. Animal Feed Science and Technology, 166, 628-640.
- Nimmermark, S., Lund, V., Gustafsson, G., & Eduard, W. (2009). Ammonia, dust and bacteria in welfare-oriented systems for laying hens. Annals of Agricultural and Environmental Medicine, 16(1), 103-113.
- G Gustafsson, G., Jeppsson, K. H., Hultgren, J., & Sannö, J.O. (2005). Techniques to reduce the ammonia emission from a cowshed with tied dairy cattle. Agricultural Engineering International: CIGR Journal.
- Sebacher, D. I., Harriss, R. C., & Bartlett, K. B. (1983). Methane flux across the airâ?Â├é┬Éwater interface: Air velocity effects. Tellus B, 35(2), 103-109.
- Kaiser, E. A., Eiland, F., Germon, J. C., Gispert, M. A., Heinemeyer, O., Hénault, C., & Webster, C. (1996). What predicts nitrous oxide emissions and denitrification Nâ?Â├é┬Éloss from European soils?. Zeitschrift für Pflanzenernährung und Bodenkunde, 159(6), 541-547.
- Galama, P. J., Ouweltjes, W., Endres, M. I., Sprecher, J. R., Leso, L., Kuipers, A., & Klopic, M. (2020). Symposium review: Future of housing for dairy cattle. Journal of dairy science, 103(6), 5759-5772.
- Amon, B., Amon, T., Boxberger, J., & Alt, C. (2001). Emissions of NH3, N2O and CH4 from dairy cows housed in afarmyard manure tying stall (housing, manure storage, manure spreading). Nutrient cycling in Agroecosystems, 60(1), 103-113.
- EPA USEPA. (2001). Emissions from animal feeding operations.
- European Commission Publication. (2001). Directive 2001/81/EC on national emission ceilings for atmospheric pollutants. Off J Eur Communities 22–30.
- European Commission Publication. (2010). Directive 2010/75/EU Industrial Emissions. Off J Eur Union L334:17–119.
- European Commission Publication. (2007). Council Directive 2007/43/EC of 28 June 2007 laying down minimum rules for the protection of chickens kept for meat production. Off J Eur Union 19–28.
- Koerkamp, P. G. (1994). Review on emissions of ammonia from housing systems for laying hens in relation to sources, processes, building design and manure handling. Journal of Agricultural Engineering Research, 59(2), 73-87.
- Hansen, M. J., Adamsen, A. P., Jonassen, K., & Feilberg, A. (2012). The effect of pit ventilation on the emission of odorants from pig production. Chemical Engineering Transactions, 30, 229-234.
- Hansen, M. J., Kamp, J. N., Adamsen, A. P. S., & Feilberg,(2020). Low-emission slurry pits for pig houses with straw application. Biosystems Engineering, 197, 56-63.
- Eggleston, S., Buendia, L., Miwa, K., Ngara, T., & Tanabe, K. (2006). Agriculture, forestry and other land use. Eggleston S, Buendia L, Miwa K, Ngara T, Tanabe K, eds.
- Ivester, K. M., Couëtil, L. L., & Zimmerman, N. J. (2014). Investigating the link between particulate exposure and airway inflammation in the horse. Journal of veterinary internal medicine, 28(6), 1653-1665.
- Rubright, S. L. M., Pearce, L. L., & Peterson, J. (2017). Environmental toxicology of hydrogen sulfide. Nitric oxide: biology and chemistry, 71, 1.
- Ni, J. Q., & Heber, A. J. (2010). An on-site computer system for comprehensive agricultural air quality research. Computers and Electronics in Agriculture, 71(1), 38-49.
- Ni, J. Q., Heber, A. J., Lim, T. T., Hanni, S. M., & Diehl, C.(2017). Laboratory Evaluation of a Manure Additive for Mitigating Gas and Odor Releases from Layer Hen Manure. Aerosol and Air Quality Research, 17(10), 2533-2541.
- Ni, J. Q., Robarge, W. P., Xiao, C., & Heber, A. J. (2012). Volatile organic compounds at swine facilities: A critical review. Chemosphere, 89(7), 769-788.
- Nimmermark, S., & Gustafsson, G. (2005). Influence of temperature, humidity and ventilation rate on the release of odour and ammonia in a floor housing system for laying hens. Agricultural Engineering International: CIGR Journal.
- National Research Council. (2009). Emergency and Continuous Exposure Guidance Levels for Selected Submarine Contaminants: Volume 2 (Vol. 2). National Academies Press.
- Park, J., Kang, T., Heo, Y., Lee, K., Kim, K., Lee, K., & Yoon,C. (2020). Evaluation of short-term exposure levels on ammonia and hydrogen sulfide during Manure-Handling processes at livestock farms. Safety and health at work, 11(1), 109-117.
- Rong, L., Nielsen, P. V., & Zhang, G. (2009). Effects of airflow and liquid temperature on ammonia mass transfer above an emission surface: experimental study on emission rate. Bioresource technology, 100(20), 4654-4661.
- Saha, C. K., Zhang, G., Kai, P., & Bjerg, B. (2010). Effects of a partial pit ventilation system on indoor air quality and ammonia emission from a fattening pig room. Biosystems Engineering, 105(3), 279-287.
- Schneider, F. E., Engelhardt, T., & Wieser, P. H. (1998). Characterization of aerosol particles from animal husbandry with single particle analytic techniques. In 2001 ASAE Annual Meeting (p. 1). American Society of Agricultural and Biological Engineers.
- Andre, R. S., Kwak, D., Dong, Q., Zhong, W., Correa, D. S., Mattoso, L. H., & Lei, Y. (2018). Sensitive and selective NH3 monitoring at room temperature using ZnO ceramic nanofi-bers decorated with poly (styrene sulfonate). Sensors, 18(4), 1058.
- Solomon, S., Manning, M., Marquis, M., & Qin, D. (2007). Climate change 2007-the physical science basis: Working group I contribution to the fourth assessment report of the IPCC (Vol. 4). Cambridge university press.
- Takai, H., Nimmermark, S., Banhazi, T., Norton, T., Jacobson,L. D., Calvet, S., & Berckmans, D. (2013). Airborne pollutant emissions from naturally ventilated buildings: Proposed research directions. biosystems engineering, 116(3), 214-220.
- Tullo, E., Finzi, A., & Guarino, M. (2019). Environmental impact of livestock farming and Precision Livestock Farming as a mitigation strategy. Science of the total environment, 650, 2751-2760.
- Von Essen, S., & Donham, K. (1999). Illness and injury in animal confinement workers. Occupational Medicine (Philadelphia, Pa.), 14(2), 337-350.
- Xin, H., Gates, R. S., Green, A. R., Mitloehner, F. M., Moore Jr, P. A., & Wathes, C. M. (2011). Environmental impacts and sustainability of egg production systems. Poultry Science, 90(1), 263-277.
- Zhao, Y., Aarnink, A. J., De Jong, M. C., & Groot Koerkamp,P. W. (2014). Airborne microorganisms from livestock production systems and their relation to dust. Critical Reviews in Environmental Science and Technology, 44(10), 1071-1128.
- Zhao, Y., Zhao, D., Ma, H., Liu, K., Atilgan, A. T. I. L. G. A.N., & Xin, H. (2016). Environmental assessment of three egg production systems–Part III: Airborne bacteria concentrations and emissions. Poultry science, 95(7), 1473-1481.


