Research Article - (2026) Volume 11, Issue 1
Plasma Chemerin may predict Type-2 Diabetes Remission after Bariatric Surgery
2Bariatric Surgery Departments, Centro Medico Nacional “20 de Noviembre”, ISSSTE, Mexico
3Laboratory of Experimental Metabolism and Clinical Research, Department of Clinical Research, Centro Medico Nacional “20 de Noviembre”, ISSSTE, Mexico
Received Date: Jan 07, 2026 / Accepted Date: Feb 17, 2026 / Published Date: Feb 25, 2026
Copyright: ©2026 Juan Antonio Suárez-Cuenca, et al. This is an openaccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Garduno-Perez, A. A., Montoya-Ramirez, J., Pena-Sosa, G. D. l., Vega-Moreno, K. D. L., Vazquez-Ramos, D. C., et al. (2026). Plasma Chemerin may predict Type-2 Diabetes Remission after Bariatric Surgery. Int J Diabetes Metab Disord, 11(1), 01-06.
Abstract
Bariatric surgery is an effective treatment for patients with severe obesity and type 2 diabetes mellitus (T2DM), leading to significant metabolic improvement and diabetes remission in a substantial proportion of cases. However, remission is not universal, and biological factors influencing postoperative metabolic response remain incompletely characterized. Adipokines such as chemerin and visfatin have been implicated in insulin resistance and adipose tissue inflammation and may contribute to heterogeneous outcomes after surgery.
Aim: To evaluate metabolic changes associated with T2DM remission after bariatric surgery and to assess the relationship between preoperative circulating adipokines and diabetes remission.
Methods: Prospective observational cohort study of 30 adults with T2DM and severe obesity undergoing bariatric surgery. Clinical, anthropometric, and biochemical variables were assessed at baseline and at 6 months postoperatively. Patients were classified according to complete T2DM remission status.
Results : At 6 months, 14 patients (46%) achieved complete T2DM remission. Remission was associated with significant reductions in fasting glucose and HbA1c, increased HDL-cholesterol, and reduced triglycerides. Higher preoperative chemerin levels were associated with lack of remission, while visfatin showed no significant association.
Conclusions: Elevated preoperative chemerin levels may identify patients at risk of persistent T2DM after bariatric surgery.
Keywords
Remission of Type 2 Diabetes Mellitus, Bariatric Surgery, Adipokines, Visfatin, Chemerin, Insulin resistance, Metabolic surgery, Glycemic control, Inflammation, Lipid profile, Predictive biomarkers, Metabolic outcomes
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder strongly associated with obesity and characterized by insulin resistance, progressive β-cell dysfunction, and increased cardiometabolic risk [1]. In individuals with severe obesity, conventional lifestyle and pharmacological interventions frequently fail to achieve durable glycemic control, leading to persistent hyperglycemia and diabetes-related complications [2].
In this context, bariatric surgery has emerged as the most effective therapeutic strategy for achieving substantial and sustained weight loss, as well as significant improvements in glucose metabolism [3].
Multiple randomized and observational studies have demonstrated that bariatric surgery can induce partial or complete remission of T2DM in a substantial proportion of patients, often within months after surgery and sometimes before maximal weight loss is achieved [4,5]. These observations support the concept that mechanisms beyond caloric restriction and adiposity reduction contribute to diabetes remission, including improvements in insulin sensitivity, enhanced incretin secretion, modulation of bile acid signaling, and changes in adipose tissue–derived inflammatory mediators [6,7]. Nevertheless, not all patients experience remission after bariatric surgery, highlighting the need to identify biological factors that influence metabolic responsiveness.
Adipose tissue functions as an active endocrine organ that secretes a wide range of adipokines involved in glucose homeostasis, lipid metabolism, and inflammation [8,9]. Among these, chemerin has gained increasing attention due to its strong association with obesity, insulin resistance, metabolic syndrome, and endothelial dysfunction [9,10]. Elevated circulating chemerin levels have been linked to impaired insulin signaling in skeletal muscle and liver, as well as to a pro-inflammatory adipose tissue phenotype, suggesting a potential role in limiting metabolic recovery after weight-loss interventions [11,12]. In contrast, visfatin, another adipokine originally described as preferentially secreted by visceral adipose tissue, has shown inconsistent associations with insulin sensitivity and glycemic outcomes, and its role in predicting metabolic improvement remains controversial [13,14].
Identifying adipokines that are associated with T2DM remission after bariatric surgery may improve preoperative risk stratification and contribute to a more personalized approach to metabolic surgery. Therefore, the aim of the present study was to evaluate metabolic changes associated with T2DM remission after bariatric surgery and to explore the relationship between preoperative circulating adipokines, specifically chemerin and visfatin, and diabetes remission at 6 months of follow-up.
Methods
Design and Population
Prospective, observational cohort study, conducted in adults who were candidates for bariatric surgery and have concomitant T2DM. Patients enrolled were older than 18 years old, diagnosed with morbid obesity (Body Mass Index [BMI] higher than 40 kg/ m2 or BMI higher than 35 kg/m2 experiencing obesity-related health conditions, such as t2DM, hypertension or obstructive sleep apnea/hypopnea), as well as T2DM (ADA standardized criteria: HbA1c 6.5% and fasting plasma glucose higher than 126 mg/ dL). Patients were excluded if they had received weight-reducing therapy during 6 months previous to the enrollment, second bariatric surgery, presence of inflammatory diseases, severe renal and/or hepatic disease, active malignancy, pregnancy or evidence of history of cardiovascular disease, considered if self-reported or diagnostic evidence of ischemic heart disease, coronary artery disease, myocardial structural abnormalities, cardiac interventions or being under treatment for any of such conditions. Participants were programmed for bariatric surgery between January 2016 and December 2024 at C.M.N. “20 de Noviembre”, ISSSTE at Mexico City. All females in the study sample were pre-menopausal. The study was designed and performed according to ethical guidelines of the 1975 Declaration of Helsinki, and approved by the Local Committees of Research, Ethics in Research and Biosafety of the Centro Médico Nacional ‘20 de Noviembre’ ISSSTE, Mexico City (Protocol ID No. 386.2013). All participants provided written informed consent.
Clinical Characteristics
BMI was calculated as weight/height2. Waist circumference was measured halfway between the lowest rib margin and the iliac crest at the end of a normal expiration. Blood pressure was obtained while the patient was in a seated position and was considered to be the mean of three readings obtained 5 min apart using an aneroid sphygmomanometer (Welch Allyn Inc.; Skaneateles Falls, NY, USA).
Remission of T2DM after Bariatric Surgery
All participants were followed after bariatric surgery, with standardized criteria to define remission of T2DM. Complete remission was defined as HbA1c < 6.0% and fasting plasma glucose < 100 mg/dL, without antidiabetic medications. Likewise, partial remission was considered; which was defined as HbA1c 6.0–6.4% and fasting plasma glucose 100-125 mg/dL without pharmacologic therapy. Both metabolic responses should be maintained for at least 1 year.
Statistical Analyses
Normality of data distribution was performed with Kolmogórov- Smirnov test. Continuous variables were expressed as the media ± SD; qualitative data were shown as n (%). For comparative analyses, U-Mann Whitney or non-paired T-test, as well as Fisher Exact Test, were used as appropriate. All statistics were performed using software GraphPad Prism software (v.7) as well as SPSS (v.23 SPSS Inc., Chicago, IL, USA). Statistical significance was considered if p ≤ 0.05.
Results
Study population was constituted by 30 patients who lived with T2DM, candidates for bariatric surgery, mean aged 49 years old, one third male gender, mean weight 126 kg, BMI 48 kg/m2 with elevated waist circumference and low number of additional metabolic abnormalities, including elevated fasting glucose, HbA1c and plasma insulin, consistent with insulin resistance. Clinical-demographic characteristics are shown in Table 1.
|
Craete table |
|
|
Age (years) |
49.4 ± 5.8 |
|
Male, n (%) |
11 (33.3) |
|
Height (m) |
1.70 ± 0.09 |
|
Weight (kg) |
126.5 ± 24.5 |
|
Body mass index (BMI), kg/m² |
48.1 ± 9.7 |
|
Waist circumference (cm) |
144.1 ± 21.1 |
|
Systolic blood pressure (mmHg) |
119.3 ± 7.5 |
|
Diastolic blood pressure (mmHg) |
73.7 ± 6.8 |
|
Fasting glucose (mg/dL) |
119 ± 23.6 |
|
Hemoglobin A1c (%) |
6.95 ± 1.4 |
|
Insulin (µIU/mL) |
23.92 ± 13.7 |
|
Total cholesterol (mg/dL) |
154.8 ± 27.4 |
|
HDL cholesterol (mg/dL) |
36.3 ± 8.1 |
|
LDL cholesterol (mg/dL) |
98.6 ± 23.3 |
|
Triglycerides (mg/dL) |
107.2 ± 41.8 |
|
AST (U/L) |
42.1 ± 32.5 |
|
ALT (U/L) |
41.5 ± 25.5 |
|
Creatinine (mg/dL) |
0.70 ± 0.16 |
|
Quantitative variables are expressed as mean ± standard deviation (SD). Categorical variables are shown as n (%). |
|
Table 1 : Study population (n = 30)
After bariatric surgery, patients were followed-up to identify remission of T2DM. Fourteen patients (46%) achieved remission of T2DM, who were characterized by significant reduction of fasting glucose and HbA1c levels as early as 6 months after bariatric surgery; as well as a notable increase in plasma HDL- cholesterol and a reduction of triglycerides concentrations after bariatric surgery, as compared with patients without remission of T2DM (Table 2).
|
|
No remission (n = 16) |
Remission (n = 14) |
p-value |
|
Age (years) |
51.2 ± 4.6 |
44.2 ± 6.4 |
0.05 |
|
Weight (kg) Baseline 6 months |
130.5 ± 18.3 99.8 ± 14.5 |
124.5 ± 33.3 95.3 ± 15.39 |
0.70 0.42 |
|
Baseline BMI (kg/m²) Baseline 6 months |
50.4 ± 10.4 38.6 ± 8.4 |
45.4 ± 9.4 32.5 ± 7.8 |
0.41 0.22 |
|
Systolic blood pressure (mmHg) Baseline 6 months |
117.3 ± 10.6 115.5 ± 33.3 |
119.5 ±9.3 108.3 ± 11.7 |
0.63 0.62 |
|
Diastolic blood pressure (mmHg) Baseline 6 months |
77.3 ± 5.8 75.4 ± 6.2 |
74.7 ± 8.4 73.8 ± 5.1 |
0.58 0.55 |
|
Fasting glucose (mg/dL) Baseline 6 months |
119.3 ± 30.2 107.7 ± 27.5 |
118.7 ±17.8 78.7 ± 4.3 |
0.96 0.02 |
|
Hemoglobin A1c Baseline 6 months |
6.98 ± 0.98 6.11 ± 0.73 |
6.91 ±1.84 5.22 ± 0.13 |
0.92 0.01 |
|
Insulin Baseline 6 months |
19.2 ± 15.2 9.9 ± 8.8 |
26.66 ± 11.3 9.7 ± 6.5 |
0.24 0.99 |
|
Total Cholesterol Baseline 6 months |
147.5 ± 22.6 158.3 ± 32.9 |
143.3 ± 23.2 150.3 ± 22.8 |
0.75 0.59 |
|
HDL cholesterol (mg/dL) Baseline 6 months |
40.5 ± 6.1 39.3 ± 7.0 |
41.9 ± 4.4 49.5 ± 4.3 |
0.10 0.03 |
|
LDL cholesterol (mg/dL) Baseline 6 months |
91.2 ± 22.1 104.2 ± 26.8 |
105.95 ±23.8 103.5 ±17.1 |
0.29 0.96 |
|
Triglycerides (mg/dL) Baseline 6 months |
149.33 ±70.34 118.83 ± 14.26 |
137 ±39.61 102.67 ± 29.61 |
0.71 0.02 |
|
AST (U/L) Baseline |
40.17 ±39.63 |
44 ±27.06 |
0.84 |
|
6 months |
37.8 ±32.07 |
25 ±9.46 |
0.41 |
|
ALT (U/L) Baseline 6 months |
40.17 ±39.63 26.5 ±6.16 |
44 ±27.06 36.33 ±10.16 |
0.84 0.06 |
|
Creatinine (mg/dL) Baseline 6 months |
0.68 ±0.13 0.67 ±0.15 |
0.72 ±0.19 0.67 ±0.18 |
0.67 0.95 |
|
Quantitative variables are expressed as mean ± standard deviation (SD). U-Mann Whitney or non-paired T-test were used as appropriate for comparative analyses. |
|||
Table 2 : Metabolic features and T2DM remission after bariatric surgery (n = 30)
Likewise, higher plasma concentrations of chemerin preceeded a lack of T2DM remission of T2M after bariatric surgery; while non- significant changes were observed for plasma visfatin (Figure 1).
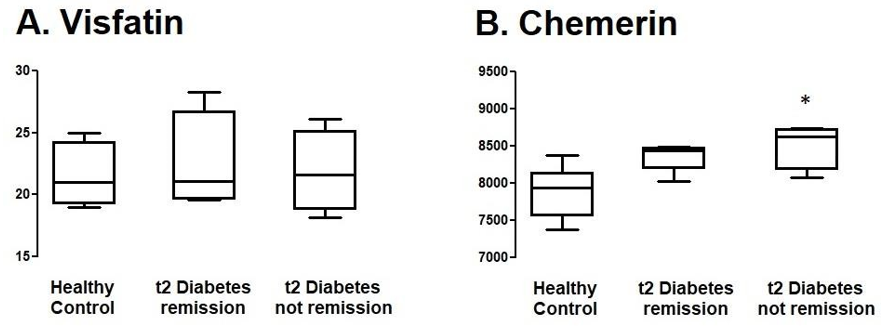
Figure 1: Adipokines and T2DM remission after bariatric surgery. The figure shows baseline plasma concentration of A. visfatin and B. chemerin. (*) p<0.05 U-mann whithney.
Discussion
In this prospective cohort of patients with severe obesity and T2DM undergoing bariatric surgery, nearly half achieved complete diabetes remission at 6 months, reinforcing the major glycemic benefits of metabolic surgery. This finding aligns with randomized evidence showing that bariatric procedures can outperform intensive medical therapy for glycemic control and can induce remission in a substantial proportion of patients with T2DM [14].
Patients who achieved remission had significantly lower fasting glucose and HbA1c at 6 months, together with a more favorable lipid profile (higher HDL-cholesterol and lower triglycerides). These postoperative changes are consistent with established mechanisms proposed for diabetes improvement after bariatric surgery, including enhanced insulin sensitivity, improved β-cell function, and shifts in gut-derived and metabolic signaling that influence glucose and lipid handling [5].
A clinically relevant finding of this study is that higher preoperative plasma chemerin was associated with lack of T2DM remission. Chemerin is an adipokine implicated in the interface between adipose inflammation and metabolic dysfunction; early human work identified chemerin as a marker linked to obesity and metabolic syndrome, supporting its role as a biomarker of metabolically unhealthy adiposity [7,9,16]. In line with this, mechanistic and translational literature connects chemerin to insulin resistance and inflammatory pathways that could plausibly blunt metabolic responsiveness to surgical intervention [10,18]. Thus, an adverse preoperative adipokine milieu may reflect a more entrenched inflammatory phenotype that is less likely to normalize glycemia after surgery.
In contrast, visfatin did not show a significant association with remission in this cohort. Visfatin was originally described as a visceral adipose–derived factor and has shown variable associations with insulin sensitivity and metabolic outcomes across studies, which may explain its weaker predictive value here [13,19]. This divergence between chemerin and visfatin emphasizes that adipokines are heterogeneous in biology and clinical utility, and that not all inflammatory mediators perform equally as predictors of remission after bariatric surgery.
Overall, these results support the concept that T2DM remission after bariatric surgery is shaped not only by weight loss and baseline glycemic burden, but also by underlying inflammatory/adipokine- related biology. Incorporating chemerin into preoperative phenotyping could improve risk stratification and help identify patients who may benefit from closer metabolic monitoring or adjunctive strategies to address residual insulin resistance [20].
Author Contributions: Contribution of A.A.G-P, J.A.S-C and J.M-R included conceptualization, funding acquisition and formal analysis. G.DLP-S, K.DLV-M, D.C.V-R, A.R-C C.R.Z-A, E.V-G, A.H-P and S.G performed investigation, methodology, data curation and validation.
Data Availability: The datasets generated and analyzed during the current study are not publicly available due to privacy policies of the hospital and patients information; but are available from the corresponding author on reasonable request.
Acknowledgement: The authors thank financial support from Programa Presupuestal E-015. Investigación y Desarrollo Tecnológico en Salud.
References
- DeFronzo, R. A. (2015). Pathogenesis of type 2 diabetes mellitus. International textbook of diabetes mellitus, 371-400.
- Prospective, D. U. (1998). Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet, 352(9131),837.
- Sjöström, L. (2013). Review of the key results from the Swedish Obese Subjects (SOS) trial–a prospective controlled intervention study of bariatric surgery. Journal of internal medicine, 273(3), 219-234.
- Mingrone, G., Panunzi, S., De Gaetano, A., Guidone, C., Iaconelli, A., Leccesi, L., ... & Rubino, F. (2012). Bariatric surgery versus conventional medical therapy for type 2 diabetes. New England Journal of Medicine, 366(17), 1577- 1585.
- Schauer, P. R., Kashyap, S. R., Wolski, K., Brethauer, S. A.,Kirwan, J. P., Pothier, C. E., ... & Bhatt, D. L. (2012). Bariatric surgery versus intensive medical therapy in obese patients with diabetes. New England Journal of Medicine, 366(17), 1567-1576.
- Rubino, F., Cohen, R. V., Mingrone, G., Le Roux, C. W., Mechanick, J. I., Arterburn, D. E., ... & Cummings, D. E. (2020). Bariatric and metabolic surgery during and after the COVID-19 pandemic: DSS recommendations for management of surgical candidates and postoperative patients and prioritisation of access to surgery. The Lancet Diabetes & Endocrinology, 8(7), 640-648.
- Thaler, J. P., & Cummings, D. E. (2009). Hormonal and metabolic mechanisms of diabetes remission after gastrointestinal surgery. Endocrinology, 150(6), 2518-2525.
- Kershaw, E. E., & Flier, J. S. (2004). Adipose tissue as an endocrine organ. The Journal of Clinical Endocrinology & Metabolism, 89(6), 2548-2556.
- Bozaoglu, K., Bolton, K., McMillan, J., Zimmet, P., Jowett, J., Collier, G., ... & Segal, D. (2007). Chemerin is a novel adipokine associated with obesity and metabolic syndrome. Endocrinology, 148(10), 4687-4694.
- Ernst, M. C., & Sinal, C. J. (2010). Chemerin: at the crossroads of inflammation and obesity. Trends in Endocrinology & Metabolism, 21(11), 660-667.
- Takahashi, M., Okimura, Y., Iguchi, G., Nishizawa, H.,Yamamoto, M., Suda, K., ... & Takahashi, Y. (2011). Chemerin regulates β-cell function in mice. Scientific reports, 1(1), 123.
- Zylla, S. (2020). Assoziationen zwischen der Plasmachemerinkonzentration und verschiedenen inflammatorischen, metabolischen, renalen und kardiovaskulären Phänotypen–Eine umfassende Untersuchung in der allgemeinen Bevölkerung– (Doctoral dissertation).
- Fukuhara, A., Matsuda, M., Nishizawa, M., Segawa, K., Tanaka, M., Kishimoto, K., ... & Shimomura, I. (2005). RETRACTED: Visfatin: A Protein Secreted by Visceral Fat That Mimics the Effects of Insulin. Science, 307(5708), 426- 430.
- De Luis, D. A., Sagrado, M. G., Conde, R., Aller, R., Izaola, O., Castro, M. J., & Romero, E. (2010). Lack of effect of a moderate hypocaloric diet on visfatin levels in morbid obese patients: relationship with insulin resistance. Eur Rev Med Pharmacol Sci, 14(12), 1031-1036.
- Mingrone, G., Panunzi, S., De Gaetano, A., Guidone, C., Iaconelli, A., Leccesi, L., ... & Rubino, F. (2012). Bariatric surgery versus conventional medical therapy for type 2 diabetes. New England Journal of Medicine, 366(17), 1577- 1585.
- Cho, Y. M. (2014). A gut feeling to cure diabetes: potential mechanisms of diabetes remission after bariatric surgery. Diabetes & metabolism journal, 38(6), 406.
- Bozaoglu, K., Bolton, K., McMillan, J., Zimmet, P., Jowett, J., Collier, G., ... & Segal, D. (2007). Chemerin is a novel adipokine associated with obesity and metabolic syndrome. Endocrinology, 148(10), 4687-4694.
- Rourke, J. L., Dranse, H. J., & Sinal, C. J. (2013). Towards an integrative approach to understanding the role of chemerin in human health and disease. Obesity Reviews, 14(3), 245-262.
- Chang, Y. C., Chang, T. J., Lee, W. J., & Chuang, L. M. (2010). The relationship of visfatin/pre–B-cell colony- enhancing factor/nicotinamide phosphoribosyltransferase in adipose tissue with inflammation, insulin resistance, and plasma lipids. Metabolism, 59(1), 93-99.
- Still, C. D., Wood, G. C., Benotti, P., Petrick, A. T., Gabrielsen, J., Strodel, W. E., ... & Argyropoulos, G. (2014). Preoperative prediction of type 2 diabetes remission after Roux-en-Y gastric bypass surgery: a retrospective cohort study. The lancet Diabetes & endocrinology, 2(1), 38-45.



