Research Article - (2025) Volume 10, Issue 2
Pilot Human Test on the Effect of SugarLock® Peanut Skin Extract in Stabilizing Postprandial Blood Glucose Fluctuations
2Research & Design Center, TCI CO., Ltd., Taipei, Taiwan
Received Date: Sep 13, 2025 / Accepted Date: Oct 20, 2025 / Published Date: Oct 27, 2025
Copyright: ©Ã??Ã?©2025 Chi-Fu Chiang,, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Lin, Y. K., Lin, Y. H., Chan, S. T., Chiang, C. F. (2025). Pilot Human Test on the Effect of SugarLock
Abstract
Postprandial glucose fluctuation contributes to oxidative stress, inflammation, and metabolic disorders. Natural plant-derived ingredients that modulate carbohydrate digestion offer a safe nutritional strategy for glycemic control. SugarLock® peanut skin extract, rich in polyphenols and proanthocyanidins, may inhibit α-amylase and α-glucosidase activities and reduce glucose absorption. This pilot human test evaluated the efficacy of peanut skin extract in stabilizing postprandial blood glucose in healthy adults. A self-controlled crossover study was conducted in nine healthy subjects (aged ≥18 years). Each subject completed four test sessions-control (Taiwanese braised pork rice), SugarLock® (400 mg), mulberry leaf extract (250 mg), and white kidney bean extract (500 mg)—with a one-week washout interval between sessions. Capsules were taken 15 minutes before the meal, and glucose levels were continuously monitored for 3 hours using a CGM system. The results showed that pre-meal intake of peanut skin extract led to a lower postprandial glucose peak (113.8 mg/dL vs. 127.1 mg/dL for control) and a smaller percentage increase (24.1% vs. 34.9%) in AUC0-3h. SugarLock® peanut skin extract attenuates postprandial glucose elevation and stabilizes glycemic response, suggesting potential as a functional ingredient for maintaining metabolic health.
Keywords
Peanut skin extract, postprandial glucose, SugarLock
Introduction
Postprandial blood glucose fluctuation has become a major focus in the prevention of metabolic and degenerative diseases. Excessive glucose excursions after meals accelerate the production of reactive oxygen species (ROS), trigger systemic inflammation, and impose metabolic stress on pancreatic β-cells [1]. Over time, these processes contribute to insulin resistance, vascular endothelial dysfunction, and the development of type 2 diabetes, cardiovascular disease, and obesity [2]. In addition, unstable glucose supply to the brain may lead to post-meal drowsiness, reduced cognitive performance, and, in the long term, neurodegenerative changes [3]. Elevated and fluctuating glucose levels also promote the formation of advanced glycation end products (AGEs), which accelerate tissue aging and impair skin elasticity [4]. Therefore, maintaining stable postprandial glucose levels is essential for metabolic homeostasis, vascular protection, and overall health longevity.
Although pharmacological agents such as metformin and α-glucosidase inhibitors effectively manage hyperglycemia, their long-term use can cause gastrointestinal discomfort and is generally unsuitable for preventive or nutritional applications in healthy populations [5]. As a result, there is growing interest in natural plant-based ingredients that can gently modulate carbohydrate metabolism and postprandial glucose responses through physiological rather than pharmacological mechanisms [6]. Such ingredients work by inhibiting carbohydrate-hydrolyzing enzymes, slowing glucose absorption, or improving insulin sensitivity, providing a safe, food-grade approach to glycemic balance. Among existing botanical ingredients, white kidney bean (Phaseolus vulgaris) and mulberry leaf (Morus alba) extracts are the most extensively studied natural options for managing postprandial blood glucose [7]. White kidney bean extract acts primarily through α-amylase inhibition, thereby reducing the breakdown of dietary starch into glucose [8]. Mulberry leaf extract, rich in 1-deoxynojirimycin (DNJ), inhibits α-glucosidase, delaying the conversion of disaccharides into monosaccharides in the small intestine [9]. Both ingredients have been shown to blunt the postprandial glucose rise and smooth glycemic variability. Building upon this concept, a novel food-grade ingredient derived from peanut skin (Arachis hypogaea L.), named SugarLock®, was provided by TCI Co., Ltd., an underutilized agricultural byproduct rich in polyphenols, especially proanthocyanidins [10]. The proanthocyanidins are oligomeric flavan-3-ols that possess potent antioxidant and anti-inflammatory activities, as well as the ability to inhibit key digestive enzymes involved in carbohydrate metabolism [11]. Several studies have demonstrated that proanthocyanidins suppress α-amylase and α-glucosidase activity, leading to slower starch digestion and reduced postprandial glucose elevation [12]. Additionally, proanthocyanidins modulate intestinal glucose transporters (such as SGLT1 and GLUT2), thereby further stabilizing glucose uptake [13]. Their high antioxidant capacity also counteracts oxidative stress generated by transient hyperglycemia, offering protection to pancreatic β-cells and vascular tissues [14]. Previous in vitro assays and preclinical data have supported the ability of peanut skin extract to inhibit starch digestion and attenuate glucose absorption [15]. However, limited human evidence exists regarding its efficacy in regulating postprandial glucose response.
Therefore, the present pilot human test aimed to evaluate whether SugarLock® peanut skin extract, which is rich in proanthocyanidins, can effectively stabilize postprandial blood glucose in healthy adults. Furthermore, the study sought to compare its performance with two well-established commercial botanical ingredients— white kidney bean extract and mulberry leaf extract under standardized dietary conditions. To achieve this, a continuous glucose monitoring (CGM) system was utilized to capture glucose fluctuations for three hours after meal ingestion, providing detailed quantitative insights into glycemic variability.
Material and Method
The manufacturing process of peanut skin extract
The SugarLock® peanut skin extract is produced using a standardized water extraction process, with the skin of Arachis hypogaea L. as the raw material. In brief, dried peanut skins are first ground into a fine powder, followed by water extraction under controlled temperature and time conditions to obtain a polyphenol- and proanthocyanidin-rich solution. The extract is then filtered to remove insoluble residues and concentrated under reduced pressure. The concentrated extract undergoes pasteurization to ensure microbial safety, and is subsequently spray-dried to yield a stable powder. The final product is packaged in sealed containers and stored in a cool, dry environment until use
Clinical trial design
A pilot human test adopted a self-controlled crossover design, in which each subject participated in four separate test sessions spaced at least 48 hours apart to minimize carry-over effects. Each subject completed four experimental conditions: (1) Control: one serving of Taiwanese braised pork rice; (2) Test 1: SugarLock® peanut skin extract capsule 400 mg taken 15 minutes before the meal plus one serving of braised pork rice; (3) Test 2: mulberry leaf extract capsule 250 mg taken 15 minutes before the meal plus one serving of braised pork rice; and (4) Test 3: white kidney bean extract capsule 500 mg taken 15 minutes before the meal plus one serving of braised pork rice. Nine healthy adults (aged ≥18 years) with normal fasting blood glucose levels were enrolled in this pilot test. All subjects provided informed consent before inclusion. Subjects were instructed to fast overnight after 10:00 p.m. prior to each test day. On the morning of each test, subjects were equipped with a continuous glucose monitoring (CGM) system (FreeStyle Libre, Abbott, USA) [16]. The CGM device automatically recorded interstitial glucose concentrations every 15 minutes for a total of 3 hours following meal consumption. During each session, subjects either consumed the control meal alone or ingested one of the test capsules 15 minutes before the standardized meal, with the sequence of test conditions randomized for each subject. Postprandial glucose responses were evaluated using three key indicators: maximum postprandial glucose level (mg/ dL), amplitude of glucose variation (ΔGlucose), and area under the curve (AUCâ??â??â??â??) within 3 hours after the meal.
Statistical analysis
The experimental data analysis was conducted using two-sample t-test and paired t-test for statistical analysis.
Results
SugarLock® peanut skin extract stabilizes blood glucose fluctuations
As shown in Figure 1A, the control meal (Taiwanese braised pork rice) resulted in a rapid rise in interstitial glucose, reaching a peak value of 127.1 mg/dL after ingestion. In contrast, pre- meal consumption of peanut skin extract (400 mg) significantly attenuated the postprandial glucose elevation, with a lower peak level of 113.8 mg/dL, representing a 10.4% reduction compared with the control meal. Among the comparative treatments, mulberry leaf extract (250 mg) exhibited a moderate effect, with a peak glucose value of 128.4 mg/dL, showing minimal improvement compared to the control. white kidney bean extract (500 mg) demonstrated the least favorable performance, with the highest peak of 136.6 mg/dL, indicating a 7.5% increase above the control condition. In addition, when postprandial glucose levels were normalized to baseline and expressed as percentage changes, a similar trend was observed (Figure 1B). The control meal alone induced the greatest increase in blood glucose, with a maximum rise of 34.9%, whereas the peanut skin extract group showed the smallest increase, at only 24.1%. The mulberry leaf extract group demonstrated a moderate improvement, with a peak increase of 32.7%, while the white kidney bean extract group exhibited the highest elevation (41.6%), indicating a weaker regulatory effect on postprandial glucose.
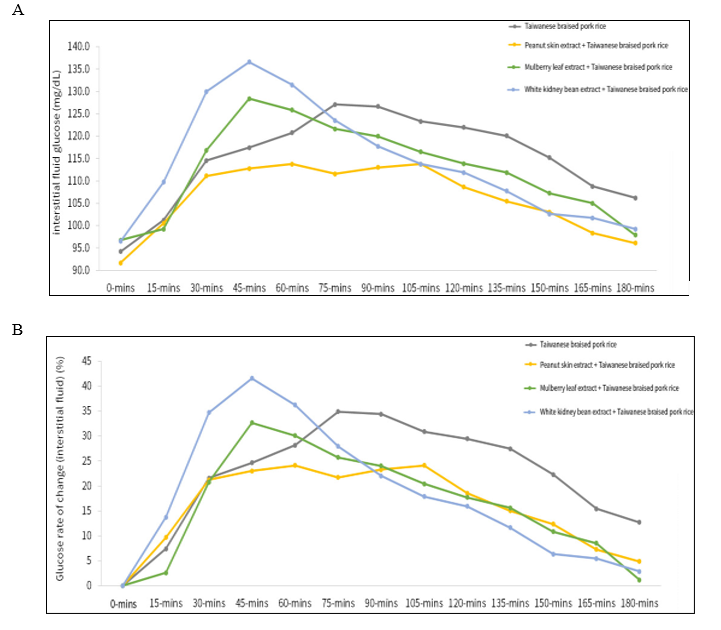
Figure 1: Postprandial glucose response after pre-meal supplementation with SugarLock® peanut skin extract
Subjects consumed one serving of Taiwanese braised pork rice alone (control) or the meal preceded by a single oral dose of pea- nut skin extract (400 mg), mulberry leaf extract (250 mg), or white kidney bean extract (500 mg) 15 minutes before eating. Data rep- resent mean values recorded every 15 minutes over 3 hours (n = 9). (A) Interstitial fluid glucose concentrations measured by continu- ous glucose monitoring (CGM) for 180 minutes after consumption of a standardized Taiwanese braised pork rice meal, with or with- out pre-meal supplementation. (B) Percentage change in intersti- tial glucose relative to baseline, representing the rate of glucose elevation after meal ingestion.
SugarLock® reduces three-hour postprandial glucose el- evation
Furthermore, analysis of the area under the curve (AUC0-3h) for postprandial glucose response demonstrated that pre-meal intake of peanut skin extract resulted in the lowest overall gly- cemic exposure among all test groups (Figure 2). Within 3 hours after meal consumption, the AUC value for the peanut skin ex- tract group was 0.70-fold relative to the control, representing a 30% reduction in glucose elevation. In comparison, the mulberry leaf extract group showed a smaller reduction (0.80-fold, 20% de- crease), while the white kidney bean extract group exhibited only a minimal improvement (0.93-fold, 7% decrease). These findings indicate that peanut skin extract not only blunts the postprandial glucose peak but also effectively decreases the total glucose expo- sure over time, confirming its superior performance in maintaining stable blood glucose levels compared with other botanical ingre- dients.
Figure 2. Area under the curve (AUC0-3h) for postprandial interstitial glucose response after pre-meal supplementation with Sug- arLock® peanut skin extract.
Subjects consumed a standardized Taiwanese braised pork rice meal alone (control) or 15 minutes after ingestion of peanut skin extract (SugarLock®, 400 mg), mulberry leaf extract (250 mg), or white kidney bean extract (500 mg). Interstitial glucose levels were continuously recorded using a CGM system for 180 minutes, and the incremental area under the curve (AUC0-3h) was cal- culated relative to the control meal.
Discussion
The present pilot human test demonstrated that pre-meal supple- mentation with SugarLock® peanut skin extract effectively stabi- lized postprandial blood glucose levels in healthy adults. Com- pared with the control meal, SugarLock® significantly reduced the postprandial glucose peak and decreased total glycemic exposure within three hours after consumption. Continuous glucose moni- toring showed a smoother and flatter postprandial glucose curve, indicating that SugarLock® modulated the rate of starch digestion and glucose absorption, thereby preventing rapid glycemic excur- sions.
The digestion of starch in humans is a multistep enzymatic pro- cess involving both α-amylase and α-glucosidase. Initially, sali- vary and pancreatic α-amylase hydrolyze complex polysaccha- rides into maltose, maltotriose, and small oligosaccharides [17]. These intermediates are subsequently converted to glucose by α-glucosidase enzymes on the intestinal brush border, after which glucose is absorbed via SGLT1 and GLUT2 transporters [18]. In- hibition of either α-amylase or α-glucosidase can therefore slow glucose release and absorption, attenuating postprandial spikes [19]. The functional activity of peanut skin extract is attributed to its abundant polyphenols and proanthocyanidins, which exhibit high binding affinity toward digestive enzymes [20]. Polyphenols can interact with the catalytic residues of α-amylase and α-gluco- sidase through hydrogen bonding and hydrophobic interactions, effectively reducing enzymatic activity [21]. Proanthocyanidins, oligomeric flavan-3-ols present in peanut skin, possess particularly strong inhibitory potency [22]. They can form non-covalent com- plexes with α-amylase, blocking the access of starch substrates to the active site. Additionally, their powerful antioxidant capacity protects pancreatic β-cells from oxidative stress caused by tran- sient hyperglycemia, while improving insulin sensitivity through modulation of oxidative and inflammatory pathways [23].
Although several botanical extracts exhibit carbohydrate-hy- drolyzing enzyme inhibition, their mechanisms of action differ in both molecular structure and target specificity. White kidney bean extract contains glycoprotein inhibitors collectively known as phaseolamin, which act as competitive inhibitors of α-amylase by physically occupying the enzyme’s active site [8]. These pro- tein-based inhibitors are large in size and are relatively sensitive to pH and temperature changes, which may limit their in vivo sta- bility and consistency [24]. Mulberry leaf extract, in contrast, con- tains 1-deoxynojirimycin (DNJ), an imidazole-type alkaloid that primarily inhibits α-glucosidase rather than α-amylase [9]. DNJ mimics the natural carbohydrate substrate and forms a reversible complex at the enzyme’s catalytic site, delaying the conversion of disaccharides into monosaccharides [25]. In comparison, peanut skin extract exerts a broader and dual mechanism: its polyphenolic compounds, especially proanthocyanidins, inhibit both α-amylase and α-glucosidase, while also modulating intestinal glucose trans- porters and mitigating oxidative stress. This multi-target effect allows SugarLock® to regulate glucose metabolism at multiple levels-digestion, absorption, and cellular utilization-resulting in a more comprehensive and sustained glycemic control. In contrast to the single-enzyme inhibition seen with white kidney bean or mulberry leaf extracts [26], the broader biochemical spectrum of peanut skin polyphenols may explain the superior stabilization of postprandial glucose observed in this study.
In the control group, starch digestion followed a gradual process in which gastric emptying was relatively slow and continuous. Starch was hydrolyzed by α-amylase to maltose and subsequently converted to glucose by α-glucosidase, leading to a dispersed glu- cose release and a moderate postprandial glycemic response that peaked at approximately 75 minutes (≈127 mg/dL). This pattern represents the physiological baseline of carbohydrate digestion and absorption under normal gastric kinetics. In contrast, both the white kidney bean and mulberry leaf groups exhibited accelerat- ed glucose excursions despite being administered in capsule form. The pre-prandial ingestion of capsules (15 min before the meal) induced gastric mechanoreceptor reflexes and gastrin secretion, thereby promoting premature gastrointestinal motility and advanc- ing gastric emptying. In the white kidney bean group, the active compound phaseolamin-a protein-based α-amylase inhibitor— was susceptible to degradation by gastric acid and pepsin, result- ing in reduced inhibitory activity in the early digestive phase [27]. Consequently, the main meal entered the proximal small intestine more rapidly, allowing pancreatic α-amylase to efficiently hydro- lyze starch into maltose and glucose [28]. As SGLT1 and GLUT2 transporters are highly expressed in this region, a concentrated and rapid glucose uptake occurred, producing an earlier and sharper glycemic peak at around 45 minutes [13]. Similarly, in the mul- berry leaf group, 1-deoxynojirimycin (DNJ) inhibits α-glucosidase at the disaccharide-to-monosaccharide step but exerts minimal in- fluence on the initial starch-to-maltose conversion [29]. Because gastric emptying was likewise accelerated, a large accumulation of maltose occurred in the upper intestinal tract, which was then rapidly converted to glucose in the distal small intestine, yielding a higher and steeper glucose peak [30].
These findings indicate that both treatments-though mechanistical- ly inhibitory-paradoxically induced a “front-loaded digestion” due to premature gastric emptying and temporally misaligned enzyme inhibition, resulting in an earlier yet higher glycemic response. By contrast, the peanut skin extract demonstrated a distinct modula- tory pattern. Although administered via capsules, its rich content of polyphenols and proanthocyanidins exhibited mild astringent and gastric-retentive properties that delayed gastric emptying and facilitated short-term co-mixing of the extract with the starch bo- lus in the stomach [31]. This allowed for more uniform enzymatic inhibition throughout the digestive process. Importantly, peanut skin polyphenols exerted dual-target inhibition, simultaneously suppressing pancreatic α-amylase and intestinal α-glucosidase [21]. Thus, the conversion from polysaccharides to disaccharides and subsequently to monosaccharides was continuously moderat- ed from the outset of digestion, rather than being interrupted only at the terminal step. Furthermore, the antioxidative components of the extract likely contributed to epithelial stability, reducing rapid membrane translocation of glucose transporters SGLT1 and GLUT2 and consequently limiting the rate of glucose influx into the bloodstream [32].
Nevertheless, this study has several limitations. As a pilot human test with a small sample size (n = 9) and short-term evaluation, the results should be considered preliminary. Although the Sugar- Lock® peanut skin extract group showed a clear trend toward im- proved postprandial glucose stability compared with the control, the differences did not reach statistical significance due to inter-in- dividual variability and limited sample size. All subjects were healthy adults with normal fasting glucose; thus, the magnitude of effect may differ in individuals with impaired glucose tolerance or diabetes. Additionally, this study focused on acute postprandi- al responses rather than long-term glycemic control or hormonal changes. Future clinical trials involving larger populations, ex- tended duration, and comprehensive biochemical assessments
Conclusion
SugarLock® peanut skin extract effectively stabilizes postprandial blood glucose through α-amylase inhibition, delayed starch diges- tion, and modulation of glucose absorption. These results highlight SugarLock® as a promising, safe, and multifunctional natural in- gredient for supporting glycemic stability and metabolic health.
References
- Blaak, E. E., Antoine, J. M., Benton, D., Björck, I., Bozzetto, L., Brouns, F., ... & Vinoy, S. (2012). Impact of postprandial glycaemia on health and prevention of disease. Obesity re- views, 13(10), 923-984.
- Lunde, M. S., Hjellset, V. T., Holmboe-Ottesen, G., & Høst- mark, A. T. (2011). Variations in postprandial blood glucose responses and satiety after intake of three types of bread. Journal of nutrition and metabolism, 2011(1), 437587.
- Han, R., Liang, J., & Zhou, B. (2021). Glucose metabolic dysfunction in neurodegenerative diseases—new mechanistic insights and the potential of hypoxia as a prospective therapy targeting metabolic reprogramming. International journal of molecular sciences, 22(11), 5887.
- Zhang, Y., Zhang, Z., Tu, C., Chen, X., & He, R. (2025). Ad- vanced Glycation End Products in Disease Development and Potential Interventions. Antioxidants, 14(4), 492.
- Vieira, R., Souto, S. B., Sánchez-López, E., Lopez Macha- do, A., Severino, P., Jose, S., ... & Souto, E. B. (2019). Sug- ar-lowering drugs for type 2 diabetes mellitus and metabolic syndrome—Review of classical and new compounds: Part-I. Pharmaceuticals, 12(4), 152.
- Mela, D. J., Alssema, M., Hiemstra, H., Hoogenraad, A. R., & Kadam, T. (2024). Effect of low-dose mulberry fruit ex- tract on postprandial glucose and insulin responses: a random- ized pilot trial in individuals with type 2 diabetes. Nutrients, 16(14), 2177.
- Przeor, M., & Ahmed, N. M. (2024). Technological process- ing of Phaseolus vulgaris and Morus alba leaves to create a new nutritional food product for individuals with diabetes. Scientific Reports, 14(1), 28686.
- Yan, J., Zhao, J., Naizemuding, P., Zhao, W., Sun, J., Wang,Y., ... & Cao, H. (2025). Unraveling the Anti-Obesity Poten- tial of White Kidney Bean α-Amylase Inhibitors: Mechanistic Insights From Enzyme Kinetics to Gut Microbiota Modula- tion. Food Science & Nutrition, 13(10), e71043.
- Kojima, Y., Kimura, T., Nakagawa, K., Asai, A., Hasumi, K., Oikawa, S., & Miyazawa, T. (2010). Effects of mulberry leaf extract rich in 1-deoxynojirimycin on blood lipid profiles in humans. Journal of clinical biochemistry and nutrition, 47(2), 155-161.
- Sorita, G. D., Leimann, F. V., & Ferreira, S. R. S. (2023). Phe- nolic fraction from peanut (Arachis hypogaea L.) by-prod- uct: Innovative extraction techniques and new encapsulation trends for its valorization. Food and bioprocess technology, 16(4), 726-748.
- Wang, Y., Zhang, L., Xiao, H., Ye, X., Pan, H., & Chen, S. (2024). Revisiting dietary proanthocyanidins on blood glu- cose homeostasis from a multi-scale structural perspective. Current Research in Food Science, 9, 100926.
- Williamson, G. (2013). Possible effects of dietary polyphenols on sugar absorption and digestion. Molecular nutrition & food research, 57(1), 48-57.
- Koepsell, H. (2020). Glucose transporters in the small intes- tine in health and disease. Pflügers Archiv-European Journal of Physiology, 472(9), 1207-1248.
- Gerber, P. A., & Rutter, G. A. (2017). The role of oxidative stress and hypoxia in pancreatic beta-cell dysfunction in dia- betes mellitus. Antioxidants & redox signaling, 26(10), 501- 518.
- Cordeiro-Massironi, K., Soares-Freitas, R. A. M., Sampaio,G. R., Pinaffi-Langley, A. C. D. C., Bridi, R., de Camargo,A. C., & Torres, E. A. F. (2023). In vitro digestion of peanut skin releases bioactive compounds and increases cancer cell toxicity. Antioxidants, 12(7), 1356.
- Cappon, G., Vettoretti, M., Sparacino, G., & Facchinetti, A. (2019). Continuous glucose monitoring sensors for diabetes management: a review of technologies and applications. Dia- betes & metabolism journal, 43(4), 383.
- Peyrot des Gachons, C., & Breslin, P. A. (2016). Salivary am- ylase: digestion and metabolic syndrome. Current diabetes reports, 16(10), 102.
- Gyimesi, G., Pujol-Giménez, J., Kanai, Y., & Hediger, M. A. (2020). Sodium-coupled glucose transport, the SLC5 fam- ily, and therapeutically relevant inhibitors: from molecular discovery to clinical application. Pflügers Archiv-European Journal of Physiology, 472(9), 1177-1206.
- Gong, L., Feng, D., Wang, T., Ren, Y., Liu, Y., & Wang, J. (2020). Inhibitors of α-amylase and α-glucosidase: Potential linkage for whole cereal foods on prevention of hyperglyce- mia. Food science & nutrition, 8(12), 6320-6337.
- Jantip, P., Singh, C. K., Klu, Y. A. K., Ahmad, N., & Bolling,B. W. (2025). Peanut skin polyphenols inhibit proliferation of leukemia cells in vitro, and its -type procyanidins selectively pass through a Caco-2 intestinal barrier. Journal of Food Sci- ence, 90(2), e70018.
- CorkoviC, I., Gašo-SokaC D., Pichler, A., ŠimunoviC, J., & Kopjar, M. (2022). Dietary polyphenols as natural inhibitors of α-amylase and α-glucosidase. Life, 12(11), 1692.
- Zhang, H., Yerigui, Yang, Y., & Ma, C. (2013). Structures and antioxidant and intestinal disaccharidase inhibitory activities of A-type proanthocyanidins from peanut skin. Journal of ag- ricultural and food chemistry, 61(37), 8814-8820.
- Eguchi, N., Vaziri, N. D., Dafoe, D. C., & Ichii, H. (2021). The role of oxidative stress in pancreatic β cell dysfunction in diabetes. International journal of molecular sciences, 22(4), 1509.
- Mosca, M., Boniglia, C., Carratù, B., Giammarioli, S., Nera, V., & Sanzini, E. (2008). Determination of α-amylase inhib- itor activity of phaseolamin from kidney bean (Phaseolus vulgaris) in dietary supplements by HPAEC-PAD. Analytica chimica acta, 617(1-2), 192-195.
- Tricase, A. F., Cavalluzzi, M. M., Catalano, A., De Bellis, M., De Palma, A., Basile, G., ... & Lentini, G. (2025). Insights into the Activities and Usefulness of Deoxynojirimycin and Morus alba: A Comprehensive Review. Molecules, 30(15), 3213.
- Li, Q., Wang, C., Liu, F., Hu, T., Shen, W., Li, E., ... & Zou,Y. (2020). Mulberry leaf polyphenols attenuated postprandial glucose absorption via inhibition of disaccharidases activity and glucose transport in Caco-2 cells. Food & function, 11(2), 1835-1844.
- Micheli, L., Lucarini, E., Trallori, E., Avagliano, C., De Caro, C., Russo, R., ... & Di Cesare Mannelli, L. (2019). Phaseolus vulgaris L. extract: Alpha-amylase inhibition against metabol- ic syndrome in mice. Nutrients, 11(8), 1778.
- Sensoy, I. (2021). A review on the food digestion in the di- gestive tract and the used in vitro models. Current research in food science, 4, 308-319.
- Yatsunami, K., Ichida, M., & Onodera, S. (2008). The rela- tionship between 1-deoxynojirimycin content and α-glucosi- dase inhibitory activity in leaves of 276 mulberry cultivars (Morus spp.) in Kyoto, Japan. Journal of natural medicines, 62(1), 63-66.
- Leiper, J. B., Maughan, R., & Murray, R. (2001). Gastric emp- tying and intestinal absorption of fluids, carbohydrates, and electrolytes (pp. 89-128). Boca Raton, FL: CRC Press.
- Camilleri, M. (2019). Gastrointestinal hormones and regula- tion of gastric emptying. Current Opinion in Endocrinology, Diabetes and Obesity, 26(1), 3-10.
- Abbasi, N. N., Purslow, P. P., Tosh, S. M., & Bakovic, M. (2016). Oat β-glucan depresses SGLT1-and GLUT2-mediated glucose transport in intestinal epithelial cells (IEC-6). Nutri- tion Research, 36(6), 541-552.



