Research Article - (2020) Volume 3, Issue 1
Phytochemical and Pharmacological Investigations of Crude Aqueous Extract of Momordica charantia
Received Date: Feb 24, 2020 / Accepted Date: Mar 03, 2020 / Published Date: Mar 23, 2020
Copyright: ©Copyright: ©2020 Muhammad Torequl Islam, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The search for new therapeutic remedies from natural sources is a continuous process. Momordica charantia under the family Cucurbitaceous is commonly known as tita korola in Bangladesh. Traditionally, it is used to treat many diseases, including diabetes and obesity, infections, cough, respiratory diseases, wounds and skin diseases, ulcer, gout and rheumatism, spasm, and cancer. This study aimed at the evalualtion of toxicity, anti-inflammatory, membrane stabilizing and thrombolytic activity of M. charantia aqueous leaf extract by applying a number of in vitro and ex vivo test systems. For this, toxicity analysis was done in Allium cepa, while anti-inflammatory, and membrane stabilizing and clotlysis capacity were tested by using egg albumin and human erythrocytes, respectively. Additionally, preliminary phytochemical analysis was also done. The results suggest that the crude aqueous crude extract of M. charantia possesses alkaloid, flavonoid, terpinoid, saponins, glycoside, tannins, and reducing sugars. The crude extract exhibited toxicity in A. cepa in a concentration and time-dependent manner. No adaptation capacity of DNA damage was seen during the exposure time (24-72 h). The extract at 0.03 – 0.14% (v/v) significantly (p <0.05, p <0.01, p <0.001) inhibited protein denaturation, membrane lysis and clot lysis capacity in a concentration-dependent manner. Of note, the extract exerted toxic effects at high concentrations, while protective effects against toxic and cytotoxic effects at low concentrations. M. charantia may be one of the potential sources of phytotherapeutic agents for various diseases, including oxidative stress, inflammatory diseases and cancer.
Keywords
Momordica charantia , Toxicity, Anti-Inflammatory, Membrane Stabilization, Thrombolysis
Introduction
Medicinal plants are commonly used in treating and preventing many ailments in the world [1]. According to the World Health Organization (WHO), medicinal plants can be used for therapeutic purposes as well as for the synthesis of many useful drugs. The World Health Organization (WHO) estimated that above 80% of people worldwide rely on herbal medicines for their primary health care. Around 21,000 plant species have been identified as a potential source of important therapeutic agents.
Momordica charantia (different parts of the plant: Figure 1), commonly called ‘bitter melon’ belongs to the family Cucurbitaceae and grows in tropical areas, including parts of the Amazon, East Africa, Asia, and the Caribbean, and is cultivated throughout South America as a food and medicine [2]. Most of the parts of M. charantia are used traditionally for the treatment of diabetes, and as a stomachic, laxative, anti-bilious, emetic, anthelmintic agent; for the treatment of cough, respiratory diseases, skin diseases, wounds, ulcer, gout, and rheumatism [3]. Scientific reports suggest that the herb contains many important chemicals, including triterpenes, proteins, steroids, steroids, saponins, insulin-like peptides, and alkaloids [4].

Figure 1: Different parts of Momordica charantia. [(A) Aerial parts (B) Leaf (C) Flower (D) Unripe fruit (E) Ripe fruit (F) Pulp and seeds].
This study aims at preliminary screening for phytochemical and pharmacological activities of the aqueous leaf extract of M. charantia by adopting some in vtiro and ex vivo test systems.
Materials and Methods
Plant collection and identification
Fresh leaves of M. charantia were collected from Gopalganj District, Bangladesh in August 2018. The plant material was then identified by the taxonomist, Forest Research Institute Herbarium (BFRIH #2491), Chittagong, Bangladesh.
Preparation of plant extract
The plant materials were washed with running tap water and crushed with the aid of a mortar and pestle to get 100% crude extract, which was then diluted with a little amount of distilled water.
Sources of reagents and chimicals
Analytical grade copper sulphate (CuSO4.5H2O) was purchased commercially from Merck, India. Acetyl salicylic acid was kindly provided by the Zenith Pharmaceuticals Ltd., Bangladesh. Streptokinase (Durakinase Powder for Injection, 1.5 million units) was purchased from Dong Kook Pharm. Ltd., Korea. Other reagents and chemicals were of analytical grade and purchased commercially from Merck, India.
Preliminary phytochemical screening
Different phytochemical tests were done to screen the presence of alkaloids, steroids, terpenoids, flavonoids, saponins, tannins, glycosides, and reducing sugars in aqueous crude extracts [5].
Evaluation of toxic effects in Allium cepa and determination of half-minimal inhibitory concentration (IC50)
The outer layers and budding parenchyma of the central crown of onions were carefully removed by making a small circular incision to facilitate root growth. The bulbs were then rinsed with tap water for 20 min and the root portion was soaked in distilled water in previously washed and clean glass containers (capacity: 15-20 mL) for the first 24 h at 25 ± 1 °C in the dark. Only, the onion with satisfactory root growth were transferred to the controls or sample containers (five for each concentration) for 24, 48 and 72 h of exposure. After the exposure period, the roots were counted and measured in mm. To determine the toxicity of the crude extract inhibition of root growth was calculated [6]. IC50 was also determined for the test sample. Distilled water and CuSO4.5H2O were used as negative and positive controls, respectively. CuSO4.5H2O was dissolved in distilled water to attain 6 µg/mL.
Evaluation of anti-inflammatory activity (egg albumin test)
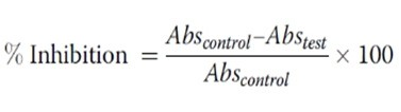
This test was carried out with a slight modification of Ullah et al.[7]. Briefly, the reaction mixture (5 mL) consisted of 0.2 mL of egg albumin (from fresh hen’s egg), 2.8 mL of phosphate buffered saline (PBS, pH 6.4) and 2 mL of varying concentrations of extract so that final concentrations become (0.03-0.14) % (v/v). A similar volume of distilled water served as negative control. Then the mixtures were incubated at (37 °C ± 2) in a BOD incubator (Lab line Technologies) for 15 min and then heated at 70 °C for 5 min. After cooling, their absorbance was measured at 660 nm (LABOCON MODEL: LUVS- 201 Monochromatic beam) by using the vehicle as blank. Acetyl salicylic acid at the final concentration 100 µg/mL was used as reference drug and treated similarly for determination of absorbance. The percentage inhibition of protein denaturation was calculated by using the following formula:
Membrane stabilization test (HRBC method)
Fresh whole blood (3 mL) collected from healthy volunteers into heparinized tubes was centrifuged at 3000 rpm for 10 min. A volume of normal saline equivalent to that of the supernatant was used to dissolve the red blood pellets. The volume of the dissolved red blood pellets obtained was measured and reconstituted as a 40% v/v suspension with an isotonic buffer solution (10 mM sodium phosphate buffer, pH 6.4). The buffer solution contained 3.1202 g of NaH2PO4, 1.7799 g of Na2HPO4 and 4.383 g of NaCl in 500 mL of distilled water. The reconstituted red blood cells (resuspended supernatant) were used as such [7].
Evaluation of anti-atherotrombosis activity (human blood counting method)
Experiments for clot lysis were carried as reported earlier [8]. In brief, 4 mL venous blood drawn from healthy volunteers and was distributed in Gr. I: negative control (distilled water); Gr. II: standard (streptokinase) and Gr.III-VII: for 5 different concentrations of the aqueous crude extract pre-weighed sterile micro centrifuge (alpin/ ephendorf’s) tubes (0.5 mL/tube) and incubated at 37 °C for 45 min. After clot formation, serum was completely removed without disturbing the clot and each tube having clot was again weighed to determine the clot weight (clot weight = weight of clot containing tube – weight of the tube alone).
To each micro-centrifuge tube containing pre-weighed clot, 100 μL (5 µg/µL) of crude extract of the plant were added separately. As a positive control, 100 μL of streptokinase (SK) and as a negative control, 100 μL of distilled water were separately added to the controls marked tubes. All the tubes were then incubated at 37 °C for 90 min and observed for clot lysis. After incubation, fluid released was removed and tubes were again weighed to observe the difference in weight after clot disruption. Difference obtained in weight taken before and after clot lysis was expressed as percentage of clot lysis. The experiment was duplicated at different days with the blood samples of 5 healthy volunteers (both male and female have no using contraceptives or anticoagulants).
Statistical analysis
Values are mean ± SD (standard deviation) and percentages. The data were analyzed by means of analysis of variance (ANOVA) followed by Tukey post-test using the Graph Pad Prism software (version 6.0) with 95% confidence interval at p <0.05.
Results
In our study, the preliminary phytochemical investigation is suggesting that, the M. charantia possesses alkaloids, glycosides, flavonoids, reducing sugar, tannins saponins and terpenoids.
In A. cepa test a concentration and time dependent toxic effect was observed. Highest root growth (RG) inhibition was observed at exposure time 72 h in 2.50% (v/v) concentration, while lowest at 0.625% (v/v) aq. extract of the herb. The half-minimal inhibitory concentrations (IC50s) calculated for 24, 48 and 72 h are 0.42 ± 0.03, 0.43 ± 0.08 and 0.48 ± 0.02, respectively (Table 1). In our study, the preliminary phytochemical investigation is suggesting that, the M. charantia possesses alkaloids, glycosides, flavonoids, reducing sugar, tannins saponins and terpenoids. In A. cepa test a concentration and time dependent toxic effect was observed. Highest root growth (RG) inhibition was observed at exposure time 72 h in 2.50% (v/v) concentration, while lowest at 0.625% (v/v) aq. extract of the herb. The half-minimal inhibitory concentrations (IC50s) calculated for 24, 48 and 72 h are 0.42 ± 0.03, 0.43 ± 0.08 and 0.48 ± 0.02, respectively (Table 1).
Table 1: Toxic effects of test sample and the controls on Allium cepa
|
Treatments |
Root length in cm |
% inhibition of root growth |
IC [CI; R2] 50 |
|||||
|
24 h |
48 h |
72 h |
24 h |
48 h |
72 h |
|
||
|
NC |
14.54 ± 0.96 |
19.34 ± 0.10 |
26.50 ± 0.13 |
- |
- |
- |
- |
|
|
PC (6 µg/mL) |
5.02 ± 0.99 |
6.00 ± 0.18 |
12.36 ± 0.17 |
65.47 |
68.97 |
53.35 |
- |
|
|
Aqueous extract of M. charantia (%v/v) |
0.625 |
6.38 ± 0.28 |
7.74 ± 0.52 |
5.22 ± 0.60 |
56.12 |
59.98 |
60.30 |
At 24 h: 0.42 ± 0.03 [0.19-0.99; 0.99] |
|
1.25 |
3.98 ± 0.37 |
4.54 ± 0.36 |
4.42 ± 1.42 |
72.63 |
76.53 |
83.32 |
At 48 h: 0.43 ± 0.08 [0.04-4.24; 0.96] |
|
|
2.50 |
3.12 ± 0.30 |
2.02 ±0 .49 |
3.60 ± 0.32 |
78.54 |
89.56 |
90.42 |
At 72 h: 0.48 ± 0.02 [0.29-0.80; 0.99] |
|
Values are mean ± SD (n = 5); NC: negative control (distilled water); PC: CuSO4.5H2O; IC50: concentration required to inhibit 50% of the root growth; CI: confidence of interval; R2: coefficient of determination
In egg albumin (in vitro) test, the crude aqueous extract of the plant showed an anti-inflammatory effect in a concentration-dependent manner. The extract exhibited highest inhibition of albumin denaturation at 0.14% (v/v). The standard ASA at 100 µg/mL exhibited better anti-inflammatory effect than the test sample. The EC50 calculated for the extract was 0.16 ± 0.01% (v/v). Multiple comparison study between the concentrations and standard for each test suggests that the extract at 0.14 to 0.09% ( v/v) produced significant effects in comparison to the 0.06 and 0.03% (v/v) groups. ASA exerted a significant anti-inflammatory effect when compared to all the test concentrations (Table 2).
The membrane stabilization assay performed in HRBC (ex vivo) method suggests that the extract concentration dependently inhibited heat-induced hemolysis, where the highest inhibition capacity was seen in 0.14% (v/v) extract. The EC50 calculated for the extract was 0.09 ± 0.05% (v/v). ASA exerted a significant anti-inflammatory effect when compared to all the test concentrations (Table 2).
Table 2: Anti-inflammatory and membrane stabilization activities of aqueous crude extract of M. charantia
|
Treatments |
Egg albumin test |
Hemolysis test |
|||
|
TIPD |
EC50 [CI; R2] |
TIHL |
EC50 [CI; R2] |
||
|
NC |
- |
- |
- |
- |
|
|
ASA (100 µg/mL) |
72.78 ± 0.07a-e |
- |
89.83 ± 0.02a-e |
- |
|
|
Aqueous crude extract (v/v) |
0.03% |
12.19 ± 0.01 |
0.16 ± 0.01 [0.14 – 0.17; 0.82] |
6.17 ± 0.01 |
0.09 ± 0.02 [0.07 – 0.11; 0.91] |
|
0.06% |
16.18 ± 0.01 |
27.41 ± 0.01a |
|||
|
0.09% |
25.08 ± 0.08ab |
34.39 ± 0.01a |
|||
|
0.12% |
32.01 ± 0.03a-c |
52.57 ± 0.03a-c |
|||
|
0.14% |
44.02 ± 0.03a-d |
71.58 ± 0.04a-d |
|||
Values are mean ± SD (n = 3) One-way ANOVA with Tukey post-test with multiple comparisons between the test concentrations and standard group; p <0.05 when compared to the a0.03%, b0.06, c0.09, d0.12, and e0.14 group; NC: Negative control (distilled water); ASA: Acetyl salicylic acid; TIPD: Total inhibition of protein denaturation; TIH: Total inhibition of hemolysis; EC50: concentration that gives half-maximal response; CI: Confidence interval; R2: Co-efficient of determination.
The crude aqueous extract of the plant showed a clot lysis capacity in a concentration-dependent manner. Highest clot lysis (53.62 ± 2.96) was observed in 0.14% extract. The standard, SK produced clot lysis 81.08 ± 0.01%, (v/v) while negative control (NC) group exhibited a negligible clot lysis capacity (1.94 ± 0.49) (v/v). The clot lysis capacity, when compared between the test groups and SK group, suggests that both the extract and the SK group produced significant (p <0.05, p <0.01, p <0.001) clotlysis effects in comparison to the NC group. The extract at 0.14% showed a significant clot lysis capacity as compared to the 0.03 – 0.12% extract groups, while at 0.12% (v/v) (p <0.001) in comparison to the 0.03 – 0.09% (v/v); 0.10% (p <0.001) in comparison to the 0.03 and 0.06% (v/v) groups, and 0.06% (p <0.01) in comparison to the lowest concentration (0.03% v/v) extract group. Both the extract at (0.06% v/v) and SK group exhibited significant (p <0.05) clot lysis capacity when compared to the NC group (Table 3).
Table 3. Thrombolytic effects of aqueous crude extract of M. charantia
|
Treatments |
|
% clotlysis |
EC50 (%v/v) [CI (%v/v); R2] |
|
NC |
|
1.94 ± 0.49 |
- |
|
SK (100 μl (30,000 I.U.)) |
|
81.08 ± 0.01***a-e |
- |
|
Aqueous extract of M. charantia (%v/v) |
0.03 |
9.09 ± 2.06* |
0.11 ± 0.02 [0.10 – 0.13; 0.96] |
|
0.06 |
22.07 ± 3.74**a |
||
|
0.09 |
32.02 ± 3.19***a,b |
||
|
0.12 |
39.89 ± 4.51***a-c |
||
|
0.14 |
53.62 ± 2.96***a-d |
Values are mean ± SEM (n = 3); One-way ANOVA with Tukey post-test with multiple comparisons; *p <0.05; **p <0.01; ***p <0.001 compared to the NC, a0.03%, b0.06%, c0.09%, d0.12% v/v, and d0.14%v/v group; DW (100 µL): Distilled water (vehicle); SK (100 μL (30,000 I.U.)): Streptokinase (positive control); EC : concentration that gives half-maximal response; CI: Confidence of interval; R2: 50 Co-efficient of determination.
Pearson correlation
Pearson’s correlation study suggests that the herb’s aq. extract mediated anti-inflammatory, anti-atherothrombosis and membrane stabilizing activities are correlated. According to Figure 2A, an increase in membrane stabilizing capacity has been linked to the increase in anti- inflammatory effect gradually till 0.09% (v/v) concentration. However, a rapid change of both activities was seen at 0.12 - 0.14 %v/v concentration. A similar correlation was also seen in case of membrane stabilizing and anti-atherothrombosis effect in Figure 2C. On the other hand, no sharp changes were observed in the case of anti-atherothrombosis vs. Anti-inflammatory effect correlation as shown in Figure 2B.
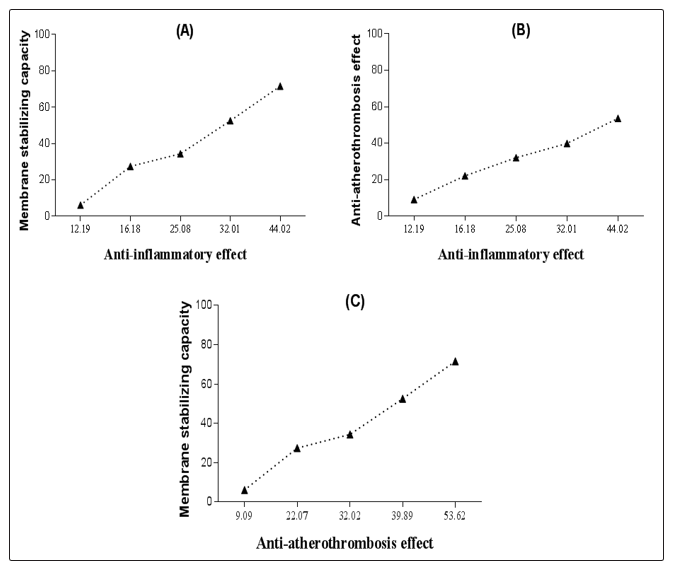
Figure 2: Pearson’s correlation [(A) Membrane stabilizing capacity vs Anti-inflammatory effect; (B) Anti-atherothrombosis effect vs Anti-inflammatory effect; and (C) Anti-atherothrombosis effect vs Membrane stabilizing capacity].
Discussion
Analysis of cytogenotoxic effects of a wide variety of substances in A. cepa is a widely known test modility [9]. According to Qin et al. [10]. Toxic substance can accumulate in roots of A. cepa and inhibit root growth, results in chromosomal aberrations (e.g., C-mitosis, chromosomal bridges, chromosomal tack and micronuclei). It may be due to the accumulation of toxic agents at the meristematic cell site of the root tip and substantially impaired the microtubule arrangements in this eukaryotic test system. Therefore, the toxicity and cytotoxic effects that cause an inhibition in root growth in A. cepa are generally related to the cell cycle elongation during differentiation, apical meristematic activity, and inhibition of protein synthesis [11-13]. Secondary metabolites, especially alkaloids, glycosides and the polyphenols possess anti-oxidant capacity [14]. Strong antioxidants at high concentration may act as pro-oxidants and can induce cytotoxic effects in eukaryotic test systems [15]. Both oxidative and anti-oxidative stress capable to cause damage to the cell macromolecules, such as protein, carbohydrates, lipids and genetic materials (e.g., DNA and RNA) [16,17]. In this study, aqueous M. charantia leaves extract was found to exert cytotoxic effects within 0.625 - 2.25% (v/v), which is equivalent to 6 - 225 mL/kg for experimental animals.
Each biological system has its own repair system that can act against oxidative stress and cytogenetic alterations caused by toxicants coming from various sources [18]. Cytotoxicity can be attained by several ways, including ROS-induced oxidative stress and mitochondrial dysfunctions, DNA damage, apoptosis, necrosis, cell cycle arrest, chromosomal aberrations, and so on [19]. Certain protective agents, especially the antioxidants can act against these types of detrimental effects [20]. There are reports that plant-based antioxidants (e.g., alkaloids, glycosides, flavonoids) can act against DNA damage [21]. These results are an agreement with the previous reports, demonstrated antioxidant and anti-inflammatory effects of the plant [19,22].
Risk assessment for any substance prior to install in any biological system is an important issue In essence, checking of the inflammatory response to a substance and the effects of it on cell membrane stability are two major consequences [23,24]. Plants containing secondary metabolites, especially the polyphenols and flavonoids alkaloids having antioxidant capacity may impart anti-inflammatory effects in biological systems [25,26,27]. Generally, inflammation is a type of protective response, but at severity level, it may cause serious problems in our body, including cardiovascular and neurological diseases and disorders [28,29].
Previous scientific reports also suggest that the plant has antioxidant, anti-inflammatory and membrane stabilizing capability. Examples of plant extract containing antioxidant as well as anti-inflammatory and membrane stabilizing capacity are numerous [22,30-32]. In this study, the crude extract at low concentration showed anti- inflammatory and membrane stabilizing capacity.
Furthermore, herbal drugs are a good choice for cardio-vascular diseases, especially for the management of atherothrombosis [33]. Generally, these kinds of medications are inexpensive and readily available, possess less side effects than the synthetic drugs [34]. Nowadays, edible plants and those are traditionally used by the various ethnic groups are in the spotlight for plant-based drug discovery and development [1,35]. Plant-based polyphenols, alkaloids, glycosides are already known to possess clot lysis capacity [36-39] Scientific reports suggest that substances acting against oxidative stress and inflammation are capable to resist formation of clot inside the blood vessels [40-42]. Therefore, the crude extract of M. charantia mediated anti-inflammatory and the previous study mediated antioxidant capacity may link with its anti-atherothrombosis effect [22,30].
Conclusion
The aqueous crude extract of the herb revealed the presence of alkaloids, tannins, saponins, steroids, glycoside, flavonoids, and reducing sugars. In A. cepa test, the extract exerted concentration and time-dependent toxic effect. No adaptation capability concerning to the DNA damage was observed. In egg albumin (in vitro) test, the crude aqueous extract of the plant showed significant anti-inflammatory effect in a concentration-dependent manner. Additionally, the extract also exhibited a significant membrane stabilizing capacity in HRBC test. Moreover, the crude aq. extract of the plant showed a clot lysis capacity in a concentration-dependent manner. Although in all cases the extract exhibited lower effects than the respective standards, but the activities were significant in comparison to the negative control groups. Pearson’s correlation study suggests that the herb’s aq. extract mediated anti-inflammatory, anti-atherothrombosis and membrane stabilizing activities are correlated to each other. As the plant contains important phytoconstituents such as alkaloids, flavonoids, etc., and these compounds may be the responsible active moieties for the observed biological activities. Further studies are needed to isolate, characterize and investigate the exact mechanism of action for each biological effects in test systems.
Acknowledgements
We are very much thankful to the Department of Pharmacy, BSMRSTU, Gopalganj (Bangladesh) for the laboratory facilities to conduct this project.
References
- Jaradat N, Zaid AN, Hussein F, Zaqzouq M, Aljammal H, et al. (2017) Anti-Lipase Potential of the Organic and Aqueous Extracts of Ten Traditional Edible and Medicinal Plants in Palestine; a Comparison Study with Orlistat. Medicines (Basel) 8: 1-13.
- Das DR, Sachan, AK, Imtiyaz M, Shuaib M (2015) Momordica charantia as a Potential Medicinal Herb: An Overview. J Med Plant Studies 3: 23-26.
- Wang L, Waltenberger B, Pferschy-Wenzig EM, Blunder M, Liu X, et al. (2014) Natural product agonists of peroxisome proliferator-activated receptor gamma (PPARγ): a review. Biochem Pharmacol 92: 73-89.
- Ayeni MJ, Oyeyemi SD, Kayode J, Peter GP (2015) Phytochemical, Proximate and Mineral Analyses of the Leaves of Gossypium hirsutum L and Momordica charantia L. Department of Plant Science, Ekiti State University, Ado-Ekiti 36001, Nigeria. Journal of Natural Sciences Research 5: 99-107.
- Trease GE (1992) A text book of Pharmacognosy. 13th ed. London: Baillier, Tindal and Caussel.
- Konuk M, Liman, R, Cigerci IH (2007) Determination of genotoxic effect of boron on Allium cepa root meristematic cells. Pak J Bot 39: 73-79.
- Ullah HMA, Zaman S, Juhara F, Akter L, Tareq SM, et al. (2014) Evaluation of antinociceptive, in-vivo & in-vitro anti-inflammatory activity of ethanolic extract of Curcuma zedoaria rhizome. BMC Compl Alt Med 14: 346.
- Prasad S, Kashyap RS, Deopujari JY, Purohit HJ, Taori GM, et al. (2006) Development of an in vitro model to study clot lysis activity of thrombolytic drugs. Thromb J 4: 14.
- Adeyemo OA, Farinmade AE (2013) Genotoxic and cytotoxic effects of food flavor enhancer, monosodium glutamate (MSG) using Allium cepa assay. Afric J Biotechnol 12: 1459-1466.
- Qin R, Wang C, Chen D, Bj€orn LO, Li S (2015) Copper- induced root growth inhibition of Allium cepa var. agrogarum L. involves disturbances in cell division and DNA damage. Environ Toxicol Chem 34: 1045-1055.
- Fusconi A, Repetto O, Bona E, Massa N, Gallo C, et al. (2006) Effect of cadmium on meristem activity and nucleus ploidy in roots of Pisum sativum L. CV, Frisson seedlings. Enviro Exp Bot 58: 253-260.
- Webster PL, Macleod RD (1996) The root apical meristem and its margin. In: Waishel Y, Eshel A, Kafkafi U (Eds.), Plant Roots. The Hidden Half, 2nd ed. Marcel Dekker, New York, 51-76.
- Seth CS, Chaturvedi PK, Misra V (2007) Toxic effect of arsenate and cadmium alone and in combination on Giant Duckweed (Spirodela polyrrhiza L.) in response to its accumulation. Environ Toxicol 22: 539-549.
- Kumar Singh S, Patra A (2018) Evaluation of phenolic composition, antioxidant, anti-inflammatory and anticancer activities of Polygonatum verticillatum (L.). J Integr Med 16: 273-282.
- Melo PS, Arrivetti LOR, Alencar SM, Skibsted LH (2016) Antioxidative and prooxidative effects in food lipids and synergism with α-tocopherol of açaí seed extracts and grape rachis extracts. Food Chem 213: 440-449.
- Zhou J, Chao G, Li Y, Wu M, Zhong S, et al. (2016) Activation of NRF2/ARE by isosilybin alleviates Aβ25-35-induced oxidativestress injury in HT-22 cells. Neurosci Lett 632: 92-97.
- Zhou Y, Sun Y, Li P, Qin G, Cheng Q, et al. (2018) Monoside antagonizes triptolide-induced hepatocyte apoptosis via the anti-oxidativestress pathway. Nan Fang Yi Ke Da Xue Bao 38: 949-955.
- Kim DC, Kang M, Biswas A, Yang CR, Wang X, et al. (2017) Effects of low dose ionizing radiation on DNA damage-caused pathways by reverse-phase protein array and Bayesian networks. J Bioinform Comput Biol 15: 1750006.
- Sharma R, Kumar R, Kodwani R, Kapoor S, Khare A, et al. (2015) A Review on Mechanisms of Anti-Tumor Activity of Chalcones. Anticancer Agents Med Chem 16: 200-211.
- Sevcovicova A, Bodnarova K, Loderer D, Imreova P, Galova E, et al. (2014) Dual activities of emodin--DNA protectivity vs mutagenicity. Neuro Endocrinol Lett 35: 149-154.
- Pistollato F, Cano S, Elio I, Vergara M, Giampieri F, et al. (2015) Plant-Based and Plant-Rich Diet Patterns during Gestation: Beneficial Effects and Possible Shortcomings. Adv Nutr 6: 581-591.
- Umukoro, Ashorobi (2006) Evaluation of anti-inflammatory and membrane stabilizing property of aqueous leaf extract of Momordica charantia in rats. Afric J Biomed Res 9: 119-124.
- Árvay J, Šnirc M, Hauptvogl M, Bilcková J, Bobková A, et al. (2018) Concentration of Micro- and Macro-Elements in Green and Roasted Coffee: Influence of Roasting Degree and Risk Assessment for the Consumers. Biol Trace Elem Res 190: 226-233.
- Bag A, Kumar Bhattacharyya S, Kumar Pal N, Chattopadhyay R (2013) Anti-inflammatory, anti-lipid peroxidative, antioxidant and membrane stabilizing activities of hydroalcoholic extract of Terminalia chebula fruits Pharm Biol 51: 1515-1520
- Karaogan ES, Albayrak A, Kutlu Z, Bayir Y (2018) Gastroprotective and antioxidanteffects of Eremurus spectabilis Bieb. Methanol extract and its isolated component isoorientin on indomethacin induced gastric ulcers in rats. Acta Cir Bras 33: 609-618.
- Sdayria J, Rjeibi I, Feriani A, Ncib S, Bouguerra W, et al. (2018) Chemical Composition and Antioxidant, Analgesic, and Anti- Inflammatory Effects of Methanolic Extract of Euphorbia retusa in Mice. Pain Res Manag 2018: 4838413.
- Majinda RRT (2018) An Update of Erythrinan Alkaloids and Their Pharmacological Activities. Prog Chem Org Nat Prod 107: 95-159.
- Golia E, Limongelli G, Natale F, Fimiani F, Maddaloni V, et al. (2014) Inflammation and cardiovascular disease: from pathogenesis to therapeutic target. Curr Atheroscler Rep 16: 435
- Khansari PS, Sperlagh B (2012) Inflammation in neurological and psychiatric diseases. Inflammopharmacology 20: 103-107.
- Semiz A, Sen A (2007) Antioxidant and chemoprotective properties of Momordica charantia L. (bitter melon) fruit extract. Afric J Biotechnol 6: 273-277.
- Sen S, Chakraborty R, Rekha B, Revathi D, Ayyanna SC, et al. (2013) Anti-inflammatory, analgesic, and antioxidant activities of Pisonia aculeata: folk medicinal use to scientific approach. Pharm Biol 51: 426-432.
- Arawwawala M, Thabrew I, Arambewela L, Handunnetti S (2010) Anti-inflammatory activity of Trichosanthes cucumerina Linn. In rats. J Ethnopharmacol 131: 538-543.
- Li P, Yang Y, Liu M (2011) Xuezhikang, extract of red yeast rice, inhibited tissue factor and hypercoagulable state through suppressing nicotinamide adenine dinucleotide phosphate oxidase and extracellular signal-regulated kinase activation. J Cardiovasc Pharmacol 58: 307-318.
- Mahmood ZA, Sualeh M, Mahmood SB, Karim MA (2010) Herbal treatment for cardiovascular disease the evidence based therapy. Pak J Pharm Sci 23: 119-124.
- Balunas M J, Kinghorn A D (2005) Drug discovery from medicinal plants. Life Sci 78: 431-441.
- Ogston D, Lea AG, Langhorne P, Wilson SB (1985) The influence of the polyphenols of cider on plasmin and plasminogen activators. Br J Haematol 60: 705-713.
- Booyse FM, Pan W, Grenett H E, Parks D A, Darley-Usmar VM, et al. (2007) Mechanism by which alcohol and wine polyphenols affect coronary heart disease risk. Ann Epidemiol 17: S24-S31.
- Olatunji LA, Michael OS, Adeyanju OA, Areola ED, Soladoye AO (2017) Anti-inflammatory and antithrombotic effects of nicotine exposure in oral contraceptive-induced insulin resistance are glucocorticoid-independent. Pharmacol Rep 69: 512-519.
- Nan ZD, Zeng KW, Shi SP, Zhao MB, Jiang Y, et al. (2013) Phenylethanoid glycosides with anti-inflammatory activities from the stems of Cistanche deserticola cultured in Tarim desert. Fitoterapia 89: 167-174
- Hugenholtz GC, Macrae F, Adelmeijer J, Dulfer S, Porte RJ, et al. (2016) Procoagulant changes in fibrin clot structure in patients with cirrhosis are associated with oxidative modifications of fibrinogen. J Thromb Haemost 14: 1054-1066.
- Sambola A, Ruiz-Meana M, Barba I, Del Blanco BG, Barrabés JA, et al. (2017) Glycative and oxidative stress are associated with altered thrombus composition in diabetic patients with ST-elevation myocardial infarction. Int J Cardio 243: 9-14.
- Bester J, Matshailwe C, Pretorius E (2018) Simultaneous presence of hypercoagulation and increased clotlysis time due to IL-1β, IL-6 and IL-8. Cytokine 110: 237-242.


