Research Article - (2024) Volume 1, Issue 2
Pelvic Scarring: A Result of Gravidity and Parity, or Simply Evidence of Biological Potential?
2The Kent and Medway Medical School, Pears Building, Parkwood Road, Canterbury, Kent, UK and Birthrights, Lise Meitner Research Group, Max Planck Institute for Evolutionary Anthropology, Leipzig, Germany
3The School of Chemistry and Forensic, Science at the University of Kent, Canterbury, Kent, UK
Received Date: Jul 16, 2024 / Accepted Date: Aug 23, 2024 / Published Date: Sep 25, 2024
Copyright: ©�2024 Georgina Ives, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ives, G., Johns, S. E., Deter, C. (2024). Pelvic Scarring: A Result of Gravidity and Parity, or Simply Evidence of Biological Potential. Archives Biol Life Sci, 1(2), 01-11.
Abstract
Background: Despite extensive research in recent decades, the association between pelvic scarring and obstetric events remains contentious, with discrepancies exacerbated by sample and methodological inconsistencies. This study revisits the investigation of a potential link between gravidity (pregnancy) and parity (childbirth) events and commonly observed scar sites on the modern pelvis using standardised analysis.
Method: A known sample from the Texas State Donated Skeletal Collection (TXSTDSC), comprising 169 females and 51 males, was utilised in the morphometric analysis of four key scar features around the pubic and auricular areas of the pelvis. Associations between each scar feature and obstetric events were examined within mixed-sex and female-only samples. Cross-tabulation and Chi-square analyses were utilised to assess simple scar occurrence, while potential associations with scar dimensions underwent Kendall’s tau-B testing.
Results; Combined-sex analyses revealed significant associations between gravidity and parity, and all scar features but pubic tubercle extension (p = <0.001 – 0.003). However, associations decreased upon the removal of male samples, with statistical significance remaining for only the preauricular sulcus (gravidity: p = 0.022; parity: p = 0.047) and superior interosseous cavity (gravidity: p = 0.002; parity: p = 0.004).
Conclusion: Detailed analysis of results highlights that while the sulcus development is influenced by obstetric events, biological sex plays a more significant role in presence and severity. The superior cavity appears to be most influenced by the biomechanical stress caused by pregnancy and vaginal birth – thus making this feature of particular interest and warranting further investigation with consideration of clinical practice and osteological study.
Keywords
Osteology, Pelvic Scarring, Parturition, Gravidity, Obstetrics
Introduction
In recent decades, osteologists have focused on the theory that obstetric events can cause macroscopic alterations around the pubic and auricular joints, including the dorsal pubic surface, pubic tubercle, and both the preauricular and retroauricular areas of the ilium [1-4]. Early studies first highlighted the potential obstetric association as an explanation for observed pelvic scar dimorphism [5-8]. Further studies have since supplied evidence of dimorphic scar presentation, with multiple studies indicating obstetric events, most frequently parity, to be the cause of this variation [3, 7-9, 11, 18-20]. However, considerable research has cautioned against definitive obstetric association, suggesting alternative causes such as general variation in pelvic shape and size, or dimorphic pelvic flexibility [1, 3, 4, 14, 16, 18, 21-23].
The Basis of Obstetric Pelvic Scar Theory
Musculoskeletal strain during pregnancy can be directly related to foetal growth, impacting maternal posture and skeletal load- bearing, and placing stress on associated muscles and their attachment sites [24-26]. Studies reveal an increase in pelvic pain with pregnancy, particularly in the last trimester [27-31]. As the foetus grows, the anterior abdominal and pelvic floor muscles endure significant strain leading to potential osteological evidence of gravidity, with areas around the pubis serving as anchor sites for these muscles [32-34]. Pelvic girdle stability is primarily maintained by the pubic and sacroiliac joints [35]. Hormone-induced softening of associated ligaments leads to joint instability, microtrauma, and joint inflammation potentially resulting in cortical changes. Late pregnancy significantly strains the sacroiliac joint due to uterine shift and increased lower back and posterior pelvis pressure [36-38].
The birth process causes further acute stress, with significant pelvic changes required for successful vaginal delivery [36, 39]. Joint diastasis, particularly at the pubis, can result in traumatic joint distortion, while sacroiliac joint nutation increasing the diameter of the pelvic outlet further strains the sacroiliac joint [40-43]. Post-natal ligament tightening can lead to persistent malalignment of the sacroiliac joint, resulting in permanently increased ligament tension [44]. Additionally, the pelvic floor muscles undergo remarkable stretching during delivery, causing significant strain at the bone-muscle interface and potential for tearing and detachment [45-47].
Osteological Research
Ullrich [8] suggested an association between pubic pitting and parity (vaginal birth) history, but subsequent studies express caution in linking the two [1, 6, 8, 18, 19, 21, 48]. McArthur et al. and Snodgrass & Galloway partially supported Ullrich's findings, with Snodgrass & Galloway concluding that parity incidence and count were associated with pubic pitting in females under 50 [11, 23]. McArthur et al. observed a strong association for pubic pitting, but only with parity incidence. Waltenberger et al. noted positive pitting associations with parity count until age 40-45, although the predictive potential was limited [3, 4]. Initial preauricular studies by Houghton [7], Ullrich, and Kelley noted associations between sulcus development severity and parturition, with Igarashi et al. later confirming associations with both gravidity and parity up to age 40 [7, 8, 18, 20]. However, many studies have failed to replicate these findings consistently [1, 4, 11, 14. 19, 22].
Waltenberger et al., Ullrich Cox & Scott and Snodgrass & Galloway investigated the association between pubic tubercle extension and obstetric history [3, 4, 19, 23]. Ullrich suggested its utility in obstetric history interpretation, while Waltenberger et al. found associations with parity up to age 50 [3, 8]. Cox & Scott reported associations with parity incidence and count, later disputed by Snodgrass & Galloway and Waltenberger et al. [4, 19, 23]. Houghton posited parturition as a potential cause of bone loss at the site of interosseous ligament insertion, with Waltenberger et al.’s study detailing an association with parity at this site until age 50. However, other studies have found it unreliable as an obstetric indicator [1, 3, 4, 7].
Methodological Considerations
Early investigations by Houghton and Ullrich utilised samples lacking biological records - thus reliant on the accuracy associated with any osteological estimation methods used to identify biological groups [7, 8]. Some studies have since relied on information obtained from archaeological records for at least part of their sample, presenting the risk of factual inaccuracy [14, 19]. Similarly, some studies utilised modern anonymous autopsied samples, requiring the estimation of parity status through soft tissue assessment alone [1, 18, 48]. Canty used a subsample of scans from a CT repository, while Waltenberger et al. conducted an entirely scan-based study [3, 4, 14]. While these benefit from associated complete medical data, it should be noted that pelvic scarring can be difficult to identify radiographically [49, 50]. Furthermore, McFadden observed considerable methodological variation in pelvic scar studies, commenting on the difference in the number of scar sites assessed in each study and a variation in analysis methods across key sites [51]. This can hinder full cross-study comparison or even intra-study conclusions where different methods are utilised for individual scar sites.
Study Aims
To date, the association between obstetric events and osteological evidence lacks clarity, thus highlighting the need for further research addressing the methodological limitations of previous studies. Therefore, this research uses a modern skeletal sample with a comprehensive known biological history to apply uniform analysis methods across multiple key pelvic scar sites, adapting existing methods where necessary. This study will facilitate the development of more accurate and insightful conclusions regarding the aetiology of pelvic scarring, thereby contributing to our knowledge of obstetric biomechanics and refining osteological investigation techniques.
Materials and Methods
Skeletal Sample and Data Collection
This study involved the analysis of 169 adult females and 51 adult males from the Texas State Donated Skeletal Collection (TXSTDSC), following ethical approval from the University of Kent (ID: 2022164607329563). This collection provides access to a large modern American population with detailed personal data, including the number of pregnancies and births. In roughly one-third of cases, personal information is provided by the donor upon registration, with the remaining two-thirds reliant on next- of-kin knowledge [52]. However, the Forensic Anthropology Center at Texas State (FACTS) ensures high accuracy by accepting only family donations from close family members proven not to be estranged from the deceased [52].
Skeletal samples were excluded where bilateral pubic symphyseal or sacroiliac fusion was present, or skeletal trauma impeded the observation of all pelvic scar sites (n = 18). Data was typically collected from the left os coxa, with the right assessed where features on the left were too damaged to analyse or the associated pelvic elements were absent. The final sample included all 169 curated adult females that met the necessary criteria alongside a smaller sample of 51 adult males, to avoid complete sex bias in this study. This also allowed for more even sampling across positive and negative obstetric groups, with only 34.1% of females having never been pregnant. Unfortunately, ancestral variation was limited, with almost 90% of the individuals in the sample being white - representative of the ancestry bias of the skeletal collection highlighted by Gocha et al. [52]. Table 1 below presents the number of individuals in each obstetric subsample group.
Table 1: Obstetric Group Demographics
|
Sample Group |
N |
|
Full sample |
220 |
|
Gravid |
126 |
|
Nulligravid (full sample) |
94 |
|
Nulligravid (female-only) |
43 |
|
Parous |
121 |
|
Nulliparous (full sample) |
99 |
|
Nulliparous (female-only) |
48 |
|
Comparative male sample |
51 |
Pelvic Scar Analysis
Dorsal Pubic Pitting
When assessing pitting on the dorsal pubic surface, the process outlined by Waltenberger et al. was used. Maximum pit width was recorded as the widest point of indentation perpendicular to the symphyseal surface. Meanwhile, depth was measured using the calliper rod as the maximum distance from the dorsal pubic surface to the bottom of the deepest pit [3, 4, 16]. Pitting length was recorded as the full expansion of the pit parallel to the symphysis (see Figure 1). In the case of multiple independent pits, length was recorded across all pits, as well as the longest pit separately.
Figure 1: The Dorsal Pubic Surface of the Left os Coxa Featuring a Single Large Pit. The White Line Represents Pit Length, and the Yellow Line Indicates Width.
Preauricular Sulcus
Width and depth measurements of the preauricular sulcus were recorded as outlined by Maass [2]. Width was measured at the widest point perpendicular to the anterior edge of the auricular surface, and sulcus depth was recorded as the difference between the unaffected preauricular surface and the deepest point of the sulcus floor. As first detailed in the research of Ives et al. an additional length measurement was taken of the depression as it runs parallel to the auricular surface (see Figure 2) [17]. Note that where the depression of the preauricular sulcus is exceptionally shallow, it may be difficult to perceive through visual observation. However, the perimeter should still be palpable and traceable to facilitate measurement.
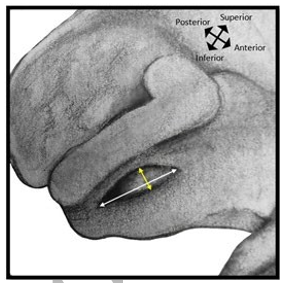
Figure 2: The Auricular Surface of the Left Os Coxa Featuring a Preauricular Sulcus. The White Line Represents Length, and the Yellow line Indicates Width.
Pubic Tubercle Extension
The methodology developed by Snodgrass & Galloway was followed with minimal deviations in the analysis of pubic tubercle extension [23]. A single measurement was recorded as the maximum distance of the centre of the tubercle extension apex from the line of the natural pubis curve (see Figure 3). However, in contrast to most previous research, this study preserves metric readings and avoids using generalised descriptive categories.
Figure 3: Anterior View of The Left Pubis Featuring A Large Pubic Tubercle. The Black Dashed Line Indicates the Natural Pubis Curve from Which the White Line Presents the Maximum Extension Measurement to The Tubercle Apex.
Interosseous Cavitation
The study of interosseous scarring typically involves the assessment of the width and depth of the full attachment site of the interosseous ligament [2, 4, 7, 8, 16]. However, this research focuses solely on the cavity formed by auricular surface lipping at the dorsal edge(s) (see Figure 4) as conducted by Ives et al. [17]. Measurement of cavity length reflects Maass’ method, by accounting for the entire length of the inferior ramus cavity. In this study, length measurement was also taken along the superior dorsal edge in cases where cavitation continued. Note that where this does occur, it always follows a small hiatus [17]. In terms of cavity depth and width, both are determined by the auricular lipping. Even in minor cases, cavity width can be ascertained by measuring the space between the retroauricular floor and auricular lipping. Meanwhile, depth is defined as the measurement from the edge of the auricular lip to the deepest point of the cavity (see Figure 4). When cavitation is particularly shallow or narrow, minor interruptions in cavitation along the inferior or superior lengths may occur, but for the purpose of simplifying maximum length measurements, these were disregarded.
Figure 4: Diagrams Showing Cavitation on The Left Auricular Surface - Medial View (Left) And Inferior View (Right). The Left Illustration Indicates Superior and Inferior Length Measurements, Represented by White Lines. The Adjacent Illustration Shows the Inferoposterior View of The Inferior Cavity, From the Base of The Auricular Demiface Apex. This Image Indicates Width (Yellow) And Depth (Blue) Measurement
Statistical Analyses
All analyses were conducted using IBM SPSS Statistics version 28, and results were considered significant at p <0.05. Although inter-observer study was not possible as part of this research due to time and logistical constraints, diligent intra- observer testing was conducted. This involved analysis of the first ten percent of the skeletal sample one week after the initial recording before continuing analysis for the remaining 200 cases. Secondary intraobserver measures were taken blind to the original results, and the difference between the two sets was assessed using Cronbach’s Alpha internal consistency test [53]. Phi was interpreted based on the criteria established by Rea & Parker as follows: negligible (<0.099), weak (0.1 - 0.199), moderate (0.2 – 0.399), moderately strong (0.4 – 0.599), strong (0.6 – 0.799), and very strong (>0.8). Data was cross-tabulated, and Chi-square (χ2) with Phi (φ) analyses were performed to quantify associations between binary obstetric group variables and scar feature presence. Kendall’s Tau-b (τb) testing was used to investigate relationships between obstetric events and scar measurements, with significant correlations classified as strong at 0.3+ [54, 55]. The data satisfied all necessary test assumptions for each analysis.
Results
Intra-Observer Testing
Repeat measure testing for all scar variables across all 20 cases produced a maximum pair difference of 6.15mm but an average difference of just 0.06mm. A test of internal consistency subsequently produced a high Cronbach’s Alpha (α = 0.999), reflecting excellent consistency between test pairs and high measurement precision [56]. This is further evident in Table S1, which presents the intra-observer results as mean values for each scar measurement variable.
Descriptive Statistics
Table 2 provides the descriptive statistics for the scar feature measurements within each obstetric group, including gravid, nulligravid (mixed-sex and female-only), parous, and nulliparous (mixed-sex and female-only). Please note that standard minimum value data is absent, as this value is consistently 0.0mm across all subgroup measurement variables - except for pubic tubercle extension in nulligravid and nulliparous female groups, at 0.62mm.
Table 2: Scar Measurement Data for Obstetric Groups, Including Mixed-Sex and Female-Only Results for Negative Groups and The Comparative Male Sample
|
Mea- sure- ment variable |
Gravid (n = 126) |
Nulligravid (mixed- sex) (n = 94) |
Nulligravid (females) (n = 43) |
Parous (n = 121) |
Nulliparous (mixed- sex) (n = 99) |
Nulliparous (females) (n = 48) |
Males only (n = 51) |
||||||||||||||
|
Max (mm) |
Mean (mm) |
Std. dev |
Max (mm) |
Mean (mm) |
Std. dev |
Max (mm) |
Mean (mm) |
Std. dev |
Max (mm) |
Mean (mm) |
Std. dev |
Max (mm) |
Mean (mm) |
Std. dev |
Max (mm) |
Mean (mm) |
Std. dev |
Max (mm) |
Mean (mm) |
Std. dev |
|
|
Pit length – Single |
18.78 |
1.82 |
4.021 |
15.97 |
0.55 |
2.193 |
15.97 |
0.75 |
2.605 |
18.78 |
1.82 |
4.021 |
15.97 |
0.61 |
2.27 |
15.97 |
0.84 |
2.692 |
9.45 |
0.44 |
1.878 |
|
Pitting length – Multi |
26.36 |
2.42 |
5.281 |
15.97 |
0.64 |
2.389 |
15.97 |
0.69 |
2.534 |
26.36 |
2.45 |
5.281 |
15.97 |
0.69 |
2.458 |
15.97 |
0.79 |
2.632 |
9.45 |
0.55 |
2.211 |
|
Pitting width |
6.47 |
0.91 |
1.743 |
3.01 |
0.21 |
0.632 |
3.01 |
0.32 |
0.778 |
6.47 |
0.92 |
1.743 |
4.29 |
0.24 |
0.741 |
4.29 |
0.38 |
0.939 |
2.43 |
0.11 |
0.462 |
|
Pitting depth |
4.54 |
0.47 |
0.927 |
1.91 |
0.12 |
0.38 |
1.91 |
0.19 |
0.472 |
4.54 |
0.48 |
0.927 |
1.91 |
0.13 |
0.394 |
1.91 |
0.2 |
0.485 |
1.34 |
0.07 |
0.267 |
|
Sulcus length |
52.52 |
25.29 |
9.647 |
54.06 |
13.79 |
11.75 |
54.06 |
20.4 |
11.75 |
52.52 |
25.35 |
9.647 |
54.06 |
14.3 |
11.7 |
54.06 |
20.75 |
11.25 |
26.23 |
8.23 |
8.462 |
|
Sulcus width |
18.75 |
6.6 |
2.888 |
12.13 |
3.45 |
3.388 |
12.13 |
5.75 |
3.525 |
18.75 |
6.6 |
2.888 |
12.13 |
3.6 |
3.429 |
12.13 |
5.82 |
3.474 |
5.26 |
1.52 |
1.594 |
|
Sulcus depth |
4.76 |
1.62 |
1.01 |
3.55 |
0.89 |
0.872 |
3.55 |
1.28 |
0.902 |
4.76 |
1.63 |
1.01 |
3.55 |
0.92 |
0.874 |
3.55 |
1.31 |
0.883 |
3.54 |
0.55 |
0.695 |
|
Superior cavity length |
29.24 |
4.61 |
6.678 |
22.66 |
1.58 |
4.535 |
22.66 |
2.28 |
6.044 |
29.24 |
4.72 |
6.678 |
22.66 |
1.6 |
4.456 |
22.66 |
2.26 |
5.774 |
11.27 |
0.99 |
2.605 |
|
Superior cavity width |
4.61 |
0.7 |
1.047 |
2.87 |
0.24 |
0.618 |
2.87 |
0.28 |
0.733 |
4.61 |
0.7 |
1.047 |
2.87 |
0.26 |
0.634 |
2.87 |
0.32 |
0.747 |
1.90 |
0.2 |
0.505 |
|
Superior cavity depth |
4.37 |
0.61 |
1.022 |
2.41 |
0.16 |
0.435 |
1.47 |
0.14 |
0.384 |
4.37 |
0.62 |
1.022 |
2.41 |
0.17 |
0.437 |
1.47 |
0.16 |
0.396 |
2.41 |
0.18 |
0.477 |
|
Inferior cavity length |
36.62 |
20.61 |
7.683 |
36.44 |
17.21 |
9.765 |
36.44 |
20.8 |
8.291 |
36.62 |
20.84 |
7.683 |
36.44 |
17.11 |
9.779 |
36.44 |
20.22 |
8.644 |
32.18 |
14.18 |
9.959 |
|
Inferior cavity width |
7.51 |
2.76 |
1.265 |
6.04 |
1.75 |
1.15 |
6.04 |
2.5 |
1.18 |
7.51 |
2.79 |
1.265 |
6.04 |
1.77 |
1.165 |
6.04 |
2.42 |
1.203 |
2.40 |
1.16 |
0.715 |
|
Inferior cavity depth |
7.94 |
2.96 |
1.754 |
6 |
1.64 |
1.434 |
6 |
2.58 |
1.47 |
7.94 |
3.01 |
1.754 |
6 |
1.65 |
1.426 |
6 |
2.52 |
1.46 |
2.86 |
0.84 |
0.773 |
|
Pubic tubercle exten- sion |
8.31 |
3.07 |
1.695 |
9.18 |
3.72 |
1.642 |
9.18 |
3.4 |
1.862 |
8.31 |
3.11 |
1.695 |
9.18 |
3.65 |
1.636 |
9.18 |
3.28 |
1.8 |
7.52 |
4 |
1.392 |
Non-Metric Analysis
Tables 3 and 4 provide data on the presence of each scar feature in association with gravidity and parity across the complete sample and the female-only sample. Analysis indicated multiple significant moderate associations between feature presence and both obstetric events in the combined sex sample, with the exclusion of pubic tubercle presence, which was not statistically significant. The preauricular sulcus presence was most strongly associated with gravidity (χ2(1) = 29.777, φ = 0.368), present in 96% of gravid individuals, but also 69.1% of nulligravid individuals (including biological males). The same feature also presented the strongest relationship with parity (χ2(1) = 26.381, φ = 0.346), appearing in 95.9% of parous individuals and 70.1% of nulliparous.
The number and strength of associations between feature presence and obstetric events were considerably reduced upon the removal of the male sample. In the female-only sample, only preauricular sulcus and superior cavity presence were significantly associated with gravidity and parity. Sulcus presence and gravidity for the female group were weakly associated (χ2(1) = 5.253, φ = 0.176). 86% of nulligravid females had evidence of a preauricular sulcus, compared to 96% of gravid females. The association between sulcus presence and parity was further reduced (χ2(1) = 3.954, φ = 0.153). Where almost 96% of parous females presented with a preauricular sulcus, so did 87.5% of nulligravid females. Notably, these results demonstrated a weaker relationship than observed between obstetric events and any of the significantly associated features for the mixed-sex group.
Superior interosseous cavitation was most strongly related to obstetric events in females. The association between superior cavitation and female gravidity was moderate (χ2(1) = 9.579, φ = 0.238). This feature was present in 39.7% of gravid females and 14% of nulligravid females. Chi-square analysis of parity groups indicated a moderate but reduced association with superior cavitation presence (χ2(1) = 8.207, φ = 0.22). Parous individuals were, again, more likely to have the feature (39.7%) than nulliparous (16.7%), but the difference between positive and negative groups was reduced compared to female-sample gravidity analysis. Furthermore, despite presenting the strongest obstetric associations for the female-only groups, superior cavitation associations were weaker than two of four feature relationships (including superior cavitation) for the mixed-sex sample in both obstetric groups.
Metric Analysis
All scar feature measurements significantly correlated with gravidity and parity in the mixed-sex sample (see Table 5). Most correlations were at least moderate across both gravidity and parity analyses (τb >0.2), excluding single pit length, inferior cavity length, tubercle extension, and the addition of multi- pit length in the assessment of parity. All preauricular sulcus measurements and all but the length measurement for the inferior interosseous cavity were strongly associated with both obstetric variables – with the maximum sulcus length value providing the strongest correlation across both (gravidity = τb 0.399; parity = τb 0.387). However, the number of significant correlations was again reduced for the female-only sample for both gravidity and parity analyses. Of those that remained statistically significant, correlations were classified as low to moderate in strength. Only the superior cavity depth values proved to be moderately correlated with gravidity and parity status in the female-only sample (τb 0.226 and 0.216, respectively).
Table 3: Cross-Tabulated Data for Individual Feature Occurrence – Presenting Full and Female-Only Results for Obstetric Groups Alongside the Comparative Male-Only Sample
|
|
Gravidity |
Parity |
Male-only Sample (n = 51) |
||||||||
|
Full Sample (n = 220) |
Female-only Sample (n = 169) |
Full Sample (n = 220) |
Female-only Sample (n = 169) |
||||||||
|
Pelvic scar feature |
Feature absent |
Feature present |
Feature absent |
Feature present |
Feature absent |
Feature present |
Feature absent |
Feature present |
Feature absent |
Feature present |
|
|
Dorsal pubic pitting |
Negative Group* |
84 (89.4%) |
10 (10.6%) |
36 (83.7%) |
7 (16.3%) |
88 (88.9%) |
11 (11.1%) |
40 (83.3%) |
8 (16.7%) |
n/a |
|
|
Positive Group* |
92 (73%) |
34 (27%) |
92 (73%) |
34 (27%) |
88 (72.7%) |
33 (27.3%) |
88 (72.7%) |
33 (27.3%) |
|||
|
Total |
176 (80%) |
44 (20%) |
128 (75.7%) |
41 (24.3%) |
176 (80%) |
44 (20%) |
128 (75.7%) |
41 (24.3%) |
48 (94.1%) |
3 (5.9%) |
|
|
Correctly Predicted |
118 (53.6%) |
70 (41.4%) |
121 (55%) |
73 (43.2%) |
n/a |
||||||
|
Preauricular sulcus |
Negative Group* |
29 (30.9%) |
65 (69.1%) |
6 (14%) |
37 (86%) |
29 (29.3%) |
70 (70.7%) |
6 (12.5%) |
42 (87.5%) |
n/a |
|
|
Positive Group* |
5 (4%) |
121 (96%) |
5 (4%) |
121 (96%) |
5 (4.1%) |
116 (95.9%) |
5 (4.1%) |
116 (95.9%) |
|||
|
Total |
34 (15.5%) |
186 (84.5%) |
11 (6.5%) |
158 (93.5%) |
34 (15.5%) |
186 (84.5%) |
11 (6.5%) |
158 (93.5%) |
23 (45.1%) |
28 (54.9%) |
|
|
Correctly Predicted |
150 (68.2%) |
127 (75.1%) |
145 (65.9%) |
122 (72.2%) |
n/a |
||||||
|
Superior interosseous cavity |
Negative Group* |
80 (85.1%) |
14 (14.9%) |
37 (86%) |
6 (14%) |
83 (83.8%) |
16 (16.2%) |
40 (83.3%) |
8 (16.7%) |
n/a |
|
|
Positive Group* |
76 (60.3%) |
50 (39.7%) |
76 (60.3%) |
50 (39.7%) |
73 (60.3%) |
48 (39.7%) |
73 (60.3%) |
48 (39.7%) |
|
|
|
|
Total |
156 (70.9%) |
64 (29.1%) |
113 (66.9%) |
56 (33.1%) |
156 (70.9%) |
64 (29.1%) |
113 (66.9%) |
56 (33.1%) |
43 (84.3%) |
8 (15.7%) |
|
|
Correctly Predicted |
130 (59.1%) |
87 (51.5%) |
131 (59.5%) |
88 (52.1%) |
n/a |
||||||
|
Inferior interosseous cavity |
Negative Group* |
12 (12.8%) |
82 (87.2%) |
2 (4.7%) |
41 (95.3%) |
12 (12.1%) |
87 (87.9%) |
2 (4.2) |
46 (95.8%) |
n/a |
|
|
Positive Group* |
2 (1.6%) |
124 (98.4%) |
2 (1.6%) |
124 (98.4%) |
2 (1.7%) |
119 (98.3%) |
2 (1.7%) |
119 (98.3%) |
|||
|
Total |
14 (6.4%) |
206 (93.6%) |
4 (2.4%) |
165 (97.6%) |
14 (6.4%) |
206 (93.6%) |
4 (2.4%) |
165 (97.6%) |
10 (19.6%) |
41 (80.4%) |
|
|
Correctly Predicted |
136 (61.8%) |
126 (74.6%) |
131 (59.5%) |
121 (72%) |
n/a |
||||||
|
Pubic tubercle extension |
Negative Group* |
0 (0%) |
94 (100%) |
0 (0%) |
43 (100%) |
0 (0%) |
99 (100%) |
0 (0%) |
48 (100%) |
n/a |
|
|
Positive Group* |
3 (2.4%) |
123 (97.6%) |
3 (2.4%) |
123 (97.6%) |
3 (2.5%) |
118 (97.5%) |
3 (2.5%) |
118 (97.5%) |
|||
|
Total |
3 (1.4%) |
117 (98.6%) |
3 (1.8%) |
163 (98.2%) |
3 (1.4%) |
117 (98.6%) |
3 (1.8%) |
163 (98.2%) |
0 (0%) |
51 (100%) |
|
|
Correctly Predicted |
123 (55.9%) |
123 (72.8%) |
118 (53.6%) |
118 (69.8%) |
n/a |
||||||
|
†Groups: Gravidity (negative = nulligravid; positive = gravid), Parity (negative = nulliparous; positive = parous) |
|||||||||||
Table 4: Chi-Square and Phi Strength of Association Results for Each Scar Feature Occurrence, Comparing Full and Female-Only Results Across Gravidity and Parity Groups
|
Pelvic scar feature |
Gravidity |
Parity |
||||||||||
|
Full Sample (n = 220) |
Female-only Sample (n = 169) |
Full Sample (n = 220) |
Female-only Sample (n = 169) |
|||||||||
|
Chi- square value (χ2) |
Strength of asso- ciation (φ) |
Test signif- icance (p) |
Chi- square value (χ2) |
Strength of asso- ciation (φ) |
Test sig- nificance (p) |
Chi- square value (χ2) |
Strength of asso- ciation (φ) |
Test sig- nificance (p) |
Chi- square value (χ2) |
Strength of asso- ciation (φ) |
Test sig- nificance (p) |
|
|
Dorsal pu- bic pitting |
8.99 |
.202 |
.003 |
1.999 |
.109 |
.157 |
8.889 |
.201 |
.003 |
2.104 |
.112 |
.147 |
|
Preauricular sulcus |
29.777 |
.368 |
<.001 |
5.253 |
.176 |
.022 |
26.381 |
.346 |
<.001 |
3.954 |
.153 |
.047 |
|
Superior interosseous cavity |
16.037 |
.27 |
<.001 |
9.579 |
.238 |
.002 |
14.587 |
.257 |
<.001 |
8.207 |
.22 |
.004 |
|
Inferior interosseous cavity |
11.29 |
.227 |
<.001 |
1.302 |
.088 |
.254 |
10.014 |
.213 |
.002 |
.94 |
.075 |
.332 |
|
Pubic tubercle extension |
2.269 |
-.102 |
.132 |
1.042 |
-.079 |
.307 |
2.488 |
-.106 |
.115 |
1.212 |
-.085 |
.271 |
Table 5: Kendall’s Tau-B Correlation Results for All Scar Feature Measurements, Comparing Full and Female-Only Sample Gravidity and Parity Group Associations
|
|
Gravidity |
Parity |
||||||
|
Full Sample (n = 220) |
Female-only Sample (n = 169) |
Full Sample (n = 220) |
Female-only Sample (n = 169) |
|||||
|
Variable measure- ments (mm) |
Correlation (τb) |
sig. (p) |
Correlation (τb) |
sig. (p) |
Correlation (τb) |
sig. (p) |
Correlation (τb) |
sig. (p) |
|
Maximum pit length – single |
0.198 |
0.002 |
0.118 |
0.104 |
0.196 |
0.002 |
0.118 |
0.106 |
|
Maximum pit length – multi |
0.2 |
0.002 |
0.121 |
0.097 |
0.199 |
0.002 |
0.122 |
0.094 |
|
Maximum pit width |
0.207 |
0.001 |
0.122 |
0.094 |
0.204 |
0.001 |
0.121 |
0.096 |
|
Maximum pit depth |
0.204 |
0.002 |
0.117 |
0.107 |
0.202 |
0.002 |
0.119 |
0.102 |
|
Maximum sulcus length |
0.399 |
<.001 |
0.168 |
0.008 |
0.387 |
<.001 |
0.168 |
0.008 |
|
Maximum sulcus width |
0.382 |
<.001 |
0.083 |
0.189 |
0.37 |
<.001 |
0.087 |
0.167 |
|
Maximum sulcus depth |
0.329 |
<.001 |
0.122 |
0.055 |
0.315 |
<.001 |
0.116 |
0.068 |
|
Maximum superi- or cavity length |
0.256 |
<.001 |
0.192 |
0.007 |
0.253 |
<.001 |
0.191 |
0.007 |
|
Maximum superi- or cavity width |
0.248 |
<.001 |
0.198 |
0.005 |
0.237 |
<.001 |
0.184 |
0.01 |
|
Maximum superi- or cavity depth |
0.258 |
<.001 |
0.226 |
0.001 |
0.251 |
<.001 |
0.216 |
0.002 |
|
Maximum inferi- or cavity length |
0.136 |
0.014 |
-0.011 |
0.857 |
0.147 |
0.008 |
0.014 |
0.826 |
|
Maximum inferi- or cavity width |
0.328 |
<.001 |
0.068 |
0.282 |
0.335 |
<.001 |
0.094 |
0.137 |
|
Maximum inferi- or cavity depth |
0.324 |
<.001 |
0.074 |
0.24 |
0.331 |
<.001 |
0.101 |
0.109 |
|
Maximum pubic tubercle extension |
-0.161 |
0.004 |
-0.048 |
0.45 |
-0.134 |
<.001 |
-0.017 |
0.79 |
Discussion
Combined Sex Analyses
In the initial non-metric analysis including both sexes, a significant association emerged between obstetric events and the presence of all scar features except pubic tubercle extension. The preauricular sulcus result proved most robust, accurately identifying the highest percentage of gravidity and parity groups, followed by the superior and then inferior interosseous cavities. However, these features were often observed in comparable percentages of positive and negative obstetric groups, echoing previous findings, thereby diminishing the predictive potential of scar occurrence analysis for obstetric events [1, 14, 21, 22, 48]. Significant correlations were also evident across all scar feature measurements. The preauricular sulcus displayed the strongest relationships with obstetric events, except for sulcus depth in parity assessment. In the case of parity groups, width and depth measurements of the inferior interosseous cavity produced higher correlations. These findings align with previous studies, emphasising obstetric-related strain around the sacroiliac joint and providing potential osteological evidence of posterior pelvic stress associated with pregnancy and birth [7, 8, 18, 20]. The remaining positive correlations at least partially corroborate additional prior obstetric investigations, all of which indicate scar severity increase at varying sites in connection with obstetric events [3, 8, 11, 19]. The sole exception is pubic tubercle extension, where our results revealed a negative correlation with both gravidity and parity, contradicting the positive correlation observed by Waltenberger et al. and Cox & Scott [3, 19].
Female-only Analyses
The predictive power of binary scar analysis diminished across all scar features in the female-only group. Significance was retained only for the preauricular sulcus and superior interosseous cavity, with the latter presenting the strongest relationship - although this remains lower than evident in the full sample group. This indicates a primary relationship between all scar feature variables and biological sex. However, the persistent significance of the preauricular sulcus and superior interosseous cavity with obstetric events indicates further marginal obstetric influence. Similarly, metric analysis revealed a reduction in the strength and significance of all results across both gravidity and parity groups, with only preauricular sulcus and superior interosseous cavity measurements remaining significantly associated with obstetric events. Studies conducted previously by Andersen, Waltenberger et al., Stewart, Kelley, Cox & Scott, Suchey et al., Snodgrass & Galloway, and Holt provide support for the observed decline in significance across all other metric scar variables. Interestingly, these scar sites that are no longer significant correspond with key muscle attachment sites of the pelvis, suggesting that obstetric events may not exert sufficient tension to yield prolonged cortical changes [1, 3, 6, 18, 19, 21, 23, 48].
Biological Sex or Obstetric Events?
The consistent reduction in result significance between the full and female-only samples indicates a greater influence of biological sex across all scar variables. Specifically, dorsal pubic pitting, pubic tubercle extension, and inferior interosseous cavitation showed no significant association with obstetric events in the female-only sample, subsequently indicating an association with biological sex. Previous studies support this conclusion regarding dorsal pitting, the full attachment site of the interosseous ligament [1, 10, 12], and pubic tubercle extension [1, 10-12, 15, 17]. The latter studies also explain the inverse correlation with tubercle extension, noted in our complete sample results. The development of the preauricular sulcus also appears to be primarily influenced by biological sex, as supported by numerous studies [1, 7, 9, 10, 13-15, 17]. However, our findings suggest that obstetric events may increase the likelihood or severity of the preauricular sulcus in biological females, albeit to a lesser extent than biological sex.
As the only other study to analyse interosseous cavitation, specifically, Ives et al. also identified a strong association with biological sex. However, according to Ives et al. [17], inferior cavitation was more significant in the estimation of biological sex than superior cavitation - in support of general interosseous groove studies - although both were found to be positively correlated with female sex. Comparatively, our research presents an increase in superior cavity correlation beyond that identified in Ives et al.’s biological sex study, with particular emphasis on superior cavity depth. These results therefore indicate increased strain on the superior margin of the interosseous ligament during obstetric events, with the higher superior interosseous correlation values observed in the mixed-sex sample likely a product of biological sex and the more significant obstetric influence in combination [17].
Conclusion and Recommendations
Our comprehensive study highlights the relationships between pelvic scarring, biological sex, and obstetric events. This study identified significant links between obstetric events and most scar features in both sexes. However, when focusing on females, the predictive power of scar analysis decreased – remaining significant for only the preauricular sulcus and superior interosseous cavity. Further investigation concluded that the preauricular sulcus primarily reflects sexual dimorphism, but obstetric events may exacerbate its severity in females. The superior interosseous cavity shows a stronger correlation with obstetric events than biological sex, making it an area of potential interest in further osteological obstetric research.
These insights underscore the importance of considering both obstetric history and biological sex when interpreting pelvic scarring patterns. Through the observable impact on pelvic morphology, this research has identified the sacroiliac joint as the most significant area of biomechanical stress associated with pregnancy and birth, with emphasis on superior interosseous cavitation. However, it would be beneficial to investigate variation in pelvic scarring across multiple populations and any additional biological factors that may further influence scar morphology. Continuing these endeavours will enhance our understanding of pelvic biomechanics, assisting in the development of osteological practice across a range of contexts
Data Availability Statement
The primary data relevant to the key results presented in this research is available by request made to the first author directly.
References
- Andersen, B. C. (1986). Parturition scarring as a consequence of flexible pelvis architecture [Doctoral dissertation, Simon Fraser University]. Burnaby, Canada.
- Maass, P. (2012). The bony pelvis: Scars of parturition and factors influencing their manifestation [Master’s dissertation, University of Cape Town]. Cape Town, South Africa.
- Waltenberger, L., Rebay-Salisbury, K., & Mitteroecker,P. (2022). Age dependent changes in pelvic shape during adulthood. Anthropologischer Anzeiger, 79(2), 143-156.
- Waltenberger, L., Rebay-Salisbury, K., & Mitteroecker,P. (2022). Are parturition scars truly signs of birth? The estimation of parity in a well-documented modern sample. International Journal of Osteoarchaeology, 32(3), 619-629.
- Angel, J. (1969). The bases of paleodemography. American Journal of Physical Anthropology, 30(3), 427-437.
- Stewart, T. D. (1970). Personal identification in mass disasters. National Museum of Natural History, Smithsonian Institution.
- Houghton, P. (1974). The relationship of the pre-auricular groove of the ilium to pregnancy. American Journal of Physical Anthropology, 41, 381–390.
- Ullrich, H. (1975). Estimation of fertility by means of pregnancy and childbirth alterations at the pubis, the ilium, and the sacrum. Ossa, 2(1), 23-39.
- Novak, L., Schultz, J. J., & McIntyre, M. (2012). Determining sex of the posterior ilium from the Robert J. Terry and William M. Bass collections. Journal of Forensic Sciences, 57(5), 1155–1160.
- Maass, P., & Friedling, L. J. (2016). Scars of parturition? Influences beyond parity. International Journal of Osteoarchaeology, 26(1), 121–131.
- McArthur, T. A., Meyer, I., Jackson, B., Pitt, M. J., & Larrison, M. C. (2016). Parturition pit: The bony imprint of vaginal birth. Skeletal Radiology, 45(9), 1263–1267.
- Mahadevappa, R. G., & Shivalingaiah, N. (2017). Sex determination by post auricular sulcus in South Karnataka. Indian Journal of Forensic and Community Medicine, 4(3), 176–180.
- Karsten, J. K. (2018). A test of the preauricular sulcus as an indicator of sex. American Journal of Physical Anthropology, 165(3), 604–608.
- Canty, S. E. (2020). The preauricular sulcus in relation to sexual dimorphism, pregnancy and parturition in humans [Doctoral dissertation, Liverpool John Moores University]. Liverpool, England.
- Praxmarer, E. M., Tutkuviene, J., & Kirchengast, S. (2020). Metric and morphological analysis of pelvic scars in a historical sample from Lithuania: Associations with sex, age, body size and pelvic dimensions. International Journal of Osteoarchaeology, 30(5), 629–641.
- Waltenberger, L., Pany-Kucera, D., Rebay-Salisbury, K., & Mitteroecker, P. (2021). The association of parturition scars and pelvic shape: A geometric morphometric study. American Journal of Physical Anthropology, 174(3), 519– 531.
- Ives, G., Johns, S. E., & Deter, C. (2024). Sexual dimorphism of pelvic scarring: A new method of adult biological sex estimation. Journal of Forensic Sciences.
- Kelley, M. A. (1970). Parturition and pelvic changes. American Journal of Physical Anthropology, 51(4), 541- 545.
- Cox, M., & Scott, A. (1992). Evaluation of the obstetric significance of some pelvic characters in an 18th century British sample of known parity status. American Journal of Physical Anthropology, 89(4), 431-440.
- Igarashi, Y., Shimizu, K., Mizutaka, S., & Kagawa, K. (2019). Pregnancy parturition scars in the preauricular area and the association with the total number of pregnancies and parturitions. American Journal of Physical Anthropology, 171(2), 1-15.
- Suchey, J. M., Wiseley, D. V., Green, R. F., & Noguchi, T.T. (1979). Analysis of dorsal pitting in the Os pubis in an extensive sample of modern American females. American Journal of Physical Anthropology, 51(4), 517–539.
- Spring, D. B., Lovejoy, C. O., Bender, G. N., & Duerr,M. (1989). The radiographic preauricular groove: Its non- relationship to past parity. American Journal of Physical Anthropology, 79(2), 247–252.
- Snodgrass, J. J., & Galloway, A. (2002). Utility of dorsal pits and pubic tubercle height in parity. Journal of Forensic Sciences, 48(6), 1226-1230.
- MacEvilly, M., & Buggy, D. (1996). Back pain and pregnancy: A review. Pain, 64(3), 405-414.
- Talbot, L., & Maclennan, K. (2016). Physiology of pregnancy. Anaesthesia & Intensive Care Medicine, 17(7), 341-345.
- Gharib, M., & Aglan, A. (2018). Changes in skeletal system during pregnancy. Interventions in Gynaecology and Women’s Healthcare, 2(1), 121-123.
- Daly, J., Frame, P., & Rapoza, P. (1991). Sacroiliac subluxation: A common, treatable cause of low-back pain in pregnancy. Family Practice Research, 11(2), 149-159.
- McIntyre, I., & Broadhurst, N. (1996). Effective treatment of low back pain in pregnancy. Australian Family Physician, 25(9), 65-67.
- Wu, W. H., Meijer, O. G., Uegaki, K., Mens, J. M. A., Van Dieën, J. H., Wuisman, P. I. J. M., & Östgaard, H. C. (2004). Pregnancy-related pelvic girdle pain (PPP), I: Terminology, clinical presentation, and prevalence. European Spine Journal, 13(7), 575-589.
- Kovacs, F. M., Garcia, E., Royuela, A., González, L., & Abraira, V. (2012). Prevalence and factors associated with low back pain and pelvic girdle pain during pregnancy. Spine, 37(17), 1516-1533.
- Keskburun, S., Güzelküçük, Ü., Fidan, U., Demir, Y., Ergün, A., & Tan, A. K. (2018). Musculoskeletal pain and symptoms in pregnancy: A descriptive study. Therapeutic Advances in Musculoskeletal Disease, 10(12), 229-234.
- Theodorsen, N., Strand, L., & Bø, K. (2019). Effect of pelvic floor and transversus abdominis muscle contraction on inter-rectus distance in postpartum women: A cross- sectional experimental study. Physiotherapy, 105(3), 315-320.
- Veljovic, F., Straus, S., Karabdic, I., & Masic, I. (2019). Spinal column and abdominal muscles loading in pregnant women dependent on working postures. Acta Informatica Medica, 27(1), 54-57.
- Netter, F. H. (2017). Atlas of human anatomy (7th ed.).Elsevier Health Sciences.
- Cunningham, C., Scheuer, L., & Black, S. (2016). Developmental juvenile osteology (2nd ed.). Elsevier Science.
- Fiani, B., Sekhon, M., Doan, T., Bowers, B., Covarrubias, C., Barthelmass, M., De Stefano, F., & Kondilis, A. (2021). Sacroiliac joint and pelvic dysfunction due to symphysiolysis in postpartum women. Cureus, 13(10).
- Pany-Kucera, D., Spannagl-Steiner, M., Desideri, J., & Rebay-Salisbury, K. (2021). Indicators of motherhood? Sacral preauricular extensions and notches in identified skeletal remains. International Journal of Osteoarchaeology, 31(1), 1-11.
- Ritchie, J. (2003). Orthopedic considerations during pregnancy. Clinical Obstetrics and Gynecology, 46(2), 456- 466.
- Dehghan, F., Haerian, B. S., Muniandy, S., Yusof, A., Dragoo, J. L., & Salleh, N. (2014). The effect of relaxin on the musculoskeletal system. Scandinavian Journal of Medicine & Science in Sports, 24(4), 220–229.
- Morino, S., Ishihara, M., Umezaki, F., Hatanaka, H., Yamashita, M., & Aoyama, T. (2019). Pelvic alignment changes during the perinatal period. PLoS ONE, 14(10).
- Seidman, A., Brossy, K., Faulkner, A., & Taylor, J. (2019). Case reports in orthopedics. Traumatic pelvic ring injury following childbirth with complete pubic symphysis.
- Pany-Kucera, D., Spannagl-Steiner, M., Argeny, S., Maurer- Gesek, B., Weninger, W. J., & Rebay-Salisbury, K. (2019). Sacral preauricular extensions, notches, and corresponding iliac changes: New terms and the proposal of a recording system. International Journal of Osteoarchaeology, 29(6), 1013-1021.
- Kiapour, A., Joukar, A., Elgafy, H., Erbulut, D. E., Agarwal,A. K., & Goel, V. K. (2020). Biomechanics of the sacroiliac joint: Anatomy, function, biomechanics, sexual dimorphism, and causes of pain. International Journal of Spine Surgery, 14, S3-S13.
- Williams, P. (1995). Gray's anatomy: The anatomical basis of medicine and surgery (38th ed.). Churchill Livingstone.
- Ashton-Miller, J., & DeLancey, J. (2009). On the biomechanics of vaginal birth and common sequelae. Annual Review of Biomedical Engineering, 11, 163-176.
- Rostaminia, G., Peck, J. D., Van Delft, K., Thakar, R., Sultan, A., & Shobeiri, S. A. (2016). New measures for predicting birth-related pelvic floor trauma. Female Pelvic Medicine & Reconstructive Surgery, 22(5), 292-296.
- Shek, K., & Dietz, H. (2019). Vaginal birth and pelvic floor trauma. Current Obstetrics and Gynecology Reports, 8(2), 15-25.
- Holt, C. A. (1978). A re-examination of parturition scars on the human female pelvis. American Journal of Physical Anthropology, 49(1), 91–94.
- Young, J. (1940). Relaxation of the pelvic joints in pregnancy: Pelvic arthropathy of pregnancy. Journal of Obstetrics and Gynaecology of the British Empire, 47(5), 493-524.
- Walker, P. L. (2005). Greater sciatic notch morphology: Sex, age, and population differences. American Journal of Physical Anthropology, 127(4), 385–391.
- McFadden, C. (2020). Parturition markers and skeletal sex estimation. In Sex estimation of the human skeleton: History, methods, and emerging techniques (pp. 131-146). Elsevier Science.
- Gocha, T. P., Mavroudas, S. R., & Wescott, D. J. (2022). The Texas State Donated Skeletal Collection at the Forensic Anthropology Center at Texas State. Forensic Sciences, 2(1), 7-19.
- Cronbach, L. J. (1951). Coefficient alpha and the internal structure of tests. Psychometrika, 16(3), 297-334.
- Rea, L. M., & Parker, R. A. (2014). Designing and conducting survey research: A comprehensive guide (4th ed.). Jossey-Bass.
- Sharma, R. D. (2017). Tests of significance for the ordinal level of variables. In Elements of statistics: A hands-on primer (pp. 144–157). Cambridge Scholars Publishing.
- Zeller, R. A. (2005). Measurement error, issues and solutions. Encyclopedia of social measurement, 665-676.



