Research Article - (2021) Volume 6, Issue 1
Ongoing Research for Managing Stress Urinary Incontinence in Women with a FDA Approved Device, The FemCap
2Director of Life Span Medical Institute San Diego, CA, USA
3Midwife in Falun Sweden, USA
Received Date: Dec 09, 2020 / Accepted Date: Dec 20, 2020 / Published Date: Jan 28, 2021
Copyright: ©Shihata Alfred. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Shihata Alfred, Brody Steven A, Linderoth Birgit. (2021). Ongoing Research for Managing Stress Urinary Incontinence in Women with a FDA Approved Device, The FemCap. Int J Women's Health Care, 6(10), 95-99.
Abstract
Background: The previous successful pilot study led the inventor to expand into Sweden and increase the number of participants. We also expanded the research comparing the FemCap to other Pessaries on the market. Pessary treatment is conservative with significant limitations. These limitations include displacement, erosion, ulceration, and urethral obstruction. Stress urinary incontinence (SUI) is prevalent particularly among menopausal women. The first line of SUI treatment is the ring pessary however, a variety of different pessaries have been introduced into the market with hope of achieving better results.
Objectives: To provide women with a safer, more effective device to treat SUI.
Materials and Methods: The FemCap combines the features of the ring and space- occupying pessaries into one device, which makes it more successful. The Bowl of the Dome of the FemCap covers the cervix and prevents it from prolapsing. The Rim fits snugly into the vaginal fornices that supports the bladder neck. The Brim flares outward pushing against the cystocele and urethrocele anteriorly to restore the anatomy of the urethra and the bladder. We recruited 118 women who had significant SUI and asked them to compare their experience for one week before using the FemCap and one week after. The FemCap was self-inserted and removed by the participants.
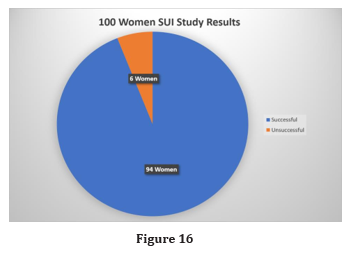
Results: Only 100 women completed the study 94 were completely dry after one week, while 6 women were not satisfied with the results. No side effects were reported by the participants and pelvic examinations did not show any erosion or ulceration of the vagina.
Conclusion: The FemCap is safe and effective in restoring the anatomy of the bladder, and urethra, which could make it ideal for the treatment and prevention of mild to moderate SUI. More studies are warranted to further prove the utility of the FemCap to manage SUI.
Keywords
FemCap, FemContinence, Stress Urinary Incontinence
Introduction
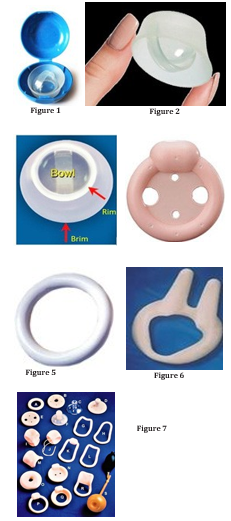
Stress urinary incontinence (SUI) is prevalent among women of all ages, particularly menopausal women. SUI is under-reported by women as well as under-diagnosed and treated by doctors. A woman using the FemCap for contraception reported that she had suffered from stress urinary incontinence, but when using the Fem-Cap (Figure 1,2,3), her SUI subsided [1-8]. Thankfully, she shared her experience, which led me to investigate a new usage for the FemCap as an SUI pessary called FemContinence. The first line of treatment for SUI is pelvic floor muscle Kegel exercises and vaginal pessaries (Figure 4,5,6,7). The most popular is the ring pessary (Figure 5), however, a variety of shapes and sizes are now becoming available in hopes of achieving better results (Figure 6, 7). [9-10].
It should be noted that pessaries are NOT a cure but are safe and effective to treat mild to moderate SUI.
Pessaries are conservative alternatives to surgical repair which is done with a sling for pelvic organ prolapse (POP) and SUI and (Figure 8)
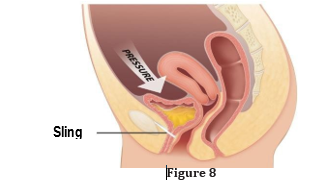
The success rate of the surgical sling procedure is 80% at best, with 30% risk of reoperation.
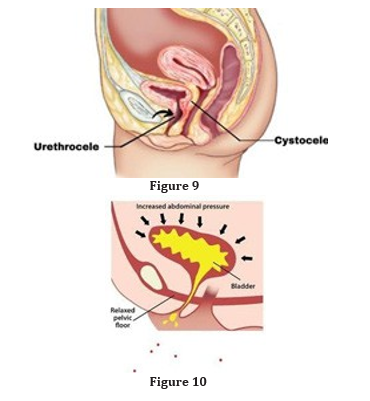
The main causes of SUI include pregnancy, vaginal delivery and pelvic floor muscles weakening, which in turn cause pelvic organs to prolapse. When the bladder prolapses (cystocele) it stretches, widens, and kinks the urethra, which leads to distortion of the ure-thral sphincters and renders them incompetent (Figure 9).
Therefore, anything that increases the abdominal pressure such as coughing, sneezing, bending over, lifting, or laughing, can put pressure on your bladder and cause urine leakage (Figure 10). Oth-er factors that may worsen SUI include age, menopause, chronic coughing, obesity, jumping, etc. SUI causes emotional distress, and embarrassment. It can disrupt work, social activities, relation-ships and even a woman’s sex life
Materials
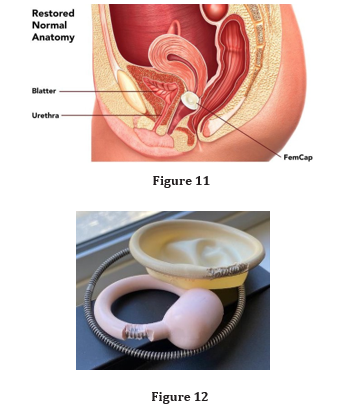
The FemCap combines the features of the ring and space-occupy-ing pessary into one device, which makes it much more successful. Our investigation started by looking at the similarities between the FemCap and the ring pessary with support. The FemCap shows marked similarity to the ring pessary and the space-occupying pessary. The Rim of the FemCap is similar in shape and function to the ring pessary that supports the bladder neck. The outward flaring of the brim pushes the cystocele forward which restores the anatomy of the bladder and urethra (Figure 11).
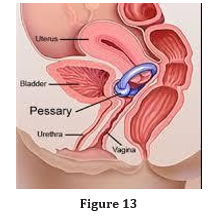
The bowl of the FemCap supports the cervix and prevents it from descending, which provides further support (Figure 3). The cur-rent pessary has many side effects including erosion and ulceration of the vagina. These side effects are due to the rigid wire in the rim that holds its shape (Figure 12). Some pessaries have a hard knob designed to press over the urethra (Figure 13). Unfortunately, this knob can rotate completely missing the target or push heavily against the urethra and obstruct the flow of urine. The FemCap is soft and flexible with no metal wire or any knobs. The Brim provides uniform gentle pressure over the urethra even with any rotation (Figure 3).
The FemCap has been in use for 20 years without any significant reportable side effects
Methods
We recruited 118 women to be participants in a feasibility study for the management SUI. 14 women did not complete the study and 4 were lost to follow up. 100 women completed the protocol using the FemCap, to control their SUI for one week. 94 had suc-cessful outcomes and 6 did not.
We conducted an earlier pilot clinical trial to check the feasibility of the FemCap in controlling stress urinary incontinence [11].
The Protocol has the following
Inclusion Criteria: a) women to be aged 18 -75 old and b) they must have at least 2 episodes of stress incontinence per day c) they must be capable of inserting and removing the FemCap on their own, and d) would be able to fill the Case Record Forms (CRF) Exclusion Criteria: Women who had stage 3 or 4 prolapse, over-active bladder, overflow incontinence or pelvic Infection or ulcer-ation of the vagina
Study Procedures
Enrollment Visit
The investigator or his or her assistant explained to the enrollee, the purpose of the study and how to fill the case record forms. If the enrollee is eligible for the study the investigator performed a pelvic exam to rule out any contraindications such as cancer, in-fection, or ulceration. If there is no reason for exclusion the inves-tigator showed her the FemCap and taught her how to insert and remove it. The investigator provided the CRF card and asked her to record any and every episode of SUI as well as any side effects or problem.
Second or Final Visit
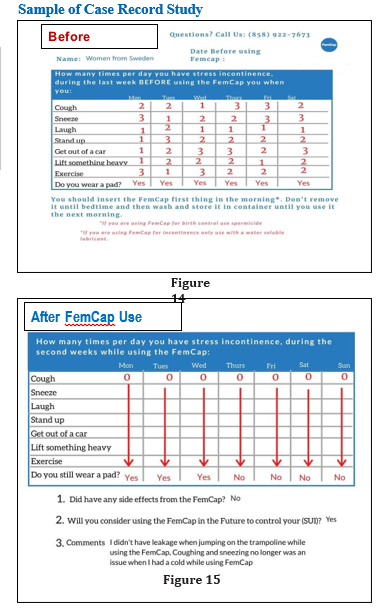
Interview the participant and checked the case records forms be-fore using the FemCap (Figure 14) and after using the FemCap (Figure 15) to compare it to the previous CRF.
Risk Analysis:
We did not anticipate any risk related to or from using the FemCap however, we ask all participants to report any issues they think may be related.
Results
Of the 100 women, 94 were completely dry after one week using the FemCap, while 6 women were not satisfied. No side effects were reported by the participants and pelvic examinations did not show any erosion or ulceration of the vagina (Figure 16).
Conclusion
Currently stress urinary incontinence is an unspoken condition that causes shame and embarrassment. The FemCap is safe and effec-tive alternative to the current pessaries. It successfully restores the anatomy of the bladder, and urethra, which could make it ideal for the treatment and prevention of mild to moderate SUI. More studies are warranted to further prove the utility of the FemCap to manage SUI [12-15]
Acknowledgements
This paper was prepared at the request of Lillian Grace Editorial Assistant, of International Journal of Women’s Health Care, we are indebted to the woman who brought this new usage informa-tion to our attention, to the women who consented to participate in this research study. To Birgit Linderoth, Midwife in Sweden, and to Dr. Steven Brody who co-authored and edited this manuscript. No funding was requested or received from any institution. The authors are responsible for the content of this article.
References
- Alfred A Shihata MD, James Trussell (1991) New Female In-travaginal Barrier Contraceptive Device Preliminary Clinical Trial, Contraception 44: 11-19.
- C K Mauck, J M Baker, S P Barr, W Johanson, D F Archer (1997) Phase I study of FemCap used with and without sper-micide: Postcoital testing. Contraception 56: 111-115.
- C Mauck, M Callahan, D H Weiner, R Dominik (1999) A comparative study of the safety and efficacy of FemCap,’I:Ma new vaginal barrier contraceptive, and the Ortho All-Flex® diaphragm. Contraception 60: 71-80.
- Carcio H, Clarke Secor M, Koenige Donohue R (2010) Ad-vanced Health Assessment of Women: Clinical Skills and Pro-cedures Chapter 15 The FemCap. Springer Publishing Com-pany 2010: 271-278.
- Koeniger-Donohue R (2006) The FemCap a Non-HormonalContraceptive, Women’s Health Care NPWH 5: 79-91.
- Shoupe D, Kjos S (2006) The Handbook of Contraception, Barrier Contraceptives Chapter 10 Humana Press 2006: 147-17
- Shihata A (1998) The FemCap™, a new contraceptive choice. Eur J Contracept and Reprod. Health Care 3: 160-166.
- Shihata A (1998) The FemCap™, a new contraceptive choice. Eur J Contracept and Reprod. Health Care 3: 160-166.
- Shihata, Alfred MD, Brody Steven (2020) Multipurpose, Reusable, Female Contraceptive Device That Enhances the Effectiveness of Fertility Awareness Methods and Controls Stress Incontinence, August 2020 Medical Research Archives 8: 1-9.
- G Willy Davila (2009) Introl™ Bladder Neck Support Pros-thesis: A Nonsurgical Urethropexy Journal o fEndourology 1996: 293-296.
- Jones, K, Harmanli O (2010) Pessary Use in Pelvic Organ Prolapse and Urinary Incontinence. Reviews in Obstetrics & Gynecology.
- Shihata A (2004) New FDA approved woman-controlled, la-tex-free barrier contraceptive device “FemCap™”. Fertil Ster-il. International Congress Series 1271: 303-330
- Viera AJ, Larkins-Pettigrew M (2000) Practical use of the pessary [published correction appears in Am Fam Physician 2002. Am Fam Physician 61: 2719-2729.
- Al Shaikh G, Syed S, Osman S, Bogis A, Al Badr A (2018) Pessary use in stress urinary incontinence: a review of advan-tages, complications, patient satisfaction, and quality of life. Int J Womens Health 10: 195-201.
- Danny Lovatsis, Mount Sinai Hospital (2017) Canada A Ran-domized Controlled Trial of the Uresta Continence Pessary (SURE) ClinicalTrials.gov Identifier: NCT01284244 Recruit-ment Status.


