Research Article - (2025) Volume 8, Issue 3
New Understanding of Atomic Structure
Received Date: Sep 02, 2025 / Accepted Date: Oct 09, 2025 / Published Date: Oct 16, 2025
Copyright: ©Â©2025 Chun Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Wang, C. (2025). New Understanding of Atomic Structure. Adv Envi Man Rec, 8(3), 01-18.
Abstract
This paper is based on the Rutherford-Bohr shell atomic model and combines famous atomic experiments in history to propose a new understanding of atomic structure. The positive electric field of the nucleus and the negative electric field of the electron are marked on the atomic structure. The repelling events between electrons in the same shell led to the formation of polyhedral symmetries in atomic sub-orbits (p, d, f). Electron spin and the configurations of paired and unpaired electrons are introduced in the model. Pauli’s four quantum numbers (n, l, m, s) correspond to the new atomic model: n-main shells, l-sub-orbits by repelling events, m-different orientations of the same sub-orbits detected by Zeeman effect, s-paired and unpaired electrons.
The new atomic model can provide new understanding of chemical single bond, double bond, triple bond, lone pair of electrons, and metallic bond, thereby distinguishing the electrical and thermal conductivities of materials at the atomic level. The work also extends to discovering the nature of superconductivity and the root cause of the Hall effect. By introducing atomic structure into the Stern-Gerlach experiment, the space-quantization of atomic spins was reinterpreted.
By studying the secondary characteristic radiation energy of elements in detail, the author confirmed that Bohr's principal shell atomic model is correct and proposed an improved Siegbahn notation for elemental analysis with X-rays and electron beams.
Electrons must revolve in atoms, at a high speed of 106 m/s (three times around the Earth in one minute), while the speed of the electric current of copper is 1 m/s.
Keywords
Bohr Model, Sub-Orbits, Orbital Magnetism, Pauli Four Quantum Numbers, Thermal and Electrical Conductivities, Single, Double and Triple Bonds
Introduction
People’s understanding of atoms is acquired step by step. Atoms are the basic building blocks of matter and can absorb and emit visible light [1-3]. Under the action of an external magnetic field, the atomic spectral lines are split [4-6]. After this early period, research on atoms made great progress in the early twentieth century.
Thomson (1897) [7] and Millikan (1913) [8] experiments have determined the electron charge (e =1.6021773•10-19C) and electron mass (me = 9.109390•10-31 kg).
Rutherford experiment (1911) [9] confirmed that the positive charge and mass of the atom are concentrated in the center, and the nucleus is very small comparing to the size of an atom (1:10000). In order to form a stable atom, the electrons with charge and mass must revolve around the nucleus at ultrafast speed of about 2.2x106 m/s to balance the electrostatic attraction of the nucleus as described in Bohr’s model (1913) [10, 11]. Later, the discovery of electron spin by Uhlenbeck and Goudsmit in 1925 [12, 13] led to the recognition that electron is a charged, self-rotating, tiny magnet. Pauli cited this new understanding and summarized in 1925 the behavior of electrons in atoms and proposed the exclusion principle, namely the four quantum numbers [14]. He established the rule that no two electrons in an atom can have the same set of quantum numbers. He perfectly distinguished the electrons in the atom. His discovery preceded Schrödinger's quantum theory proposed in 1926. Therefore, the four quantum numbers have nothing to do with Schrödinger’s atomic orbitals.
The advantage of Bohr model is that electrons can only switch to these discrete orbits by absorbing or releasing energy, which explains the observation of atomic spectra. The shortcoming of Bohr model is that different shapes and orientations of the sub- orbits cannot be explained.
The Schrödinger atomic orbitals in 1926 [15-18] explained the shapes and orientations of the sub-orbits, but it cannot explain the reversible process of absorption and emission spectra. A fatal flaw in Schrödinger’s model is that the dumbbell orbitals intersect the positively charged nuclei. His model was based on the assumption that electrons possess both wave and particle duality, similar to the idea of Louis de Broglie. However, de Broglie's wave-particle du- ality refers to the creation of X-rays by an electron beam impacting a solid (the Roentgen experiment). He proposed the relationship between the electron beam velocity and the wavelength of the X-rays. New insights into wave-particle duality will be presented in another paper.
Both atomic structures currently appear in textbooks. Atoms are real objects, and all relevant experimental evidence, proposed models, and derived parameters should be highly consistent. They cannot vary from person to person.
Now we can “see” atoms with atomic force microscope (AFM), scanning tunneling microscope (STM) and high-resolution trans- mission electron microscope (HRTEM). They are the main instru- ments used in my research work all the year round. Achieving di- rect observation of atoms is the final limit of “seeing” in science. We cannot “see” the electrons in the atoms. This requires indirect methods to understand them, to recognize their configurations in atoms and to comprehend their roles in various physical and chemical changes.
This paper first conducts an in-depth study of the Bohr and Schrödinger models, then proposes a new atomic model that pro- vides new explanations for concepts in fundamental physics and chemistry. This new model proposes a single-bond structure that is fundamentally different from the Schrödinger orbital, suggests an intrinsic connection between covalent and metallic bonds, and dis- tinguishes the fundamental difference between thermal and electrical conductivities at the atomic level.
The new atomic model provides new interpretation for superconductivity, the Hall effect, and the space-quantization of atomic spins proposed by the Stern-Gerlach experiment.
The final section reviews the researches conducted a century ago by Siegbahn, Moseley, and Barkla on the secondary characteristic X-rays radiation of elements. These studies confirmed the correctness of Bohr's principal shell atomic model. This article proposes a modified Siegbahn notation for X-rays and electron beams in elemental analysis.
Methodology
The proposed new understanding of atomic structure is rooted in a rethinking of existing experimental facts and a new interpretation of electron behavior. This approach connects fundamental principles of physics and chemistry with experimental evidence. A strict chain of reasoning is maintained throughout the proposal. Once a new idea is proposed, it should not be adopted immediately. Instead, it should be repeatedly verified and refuted until it is proven correct from several aspects. Many structures under various situations were drawn by using Power Point Presentation.
The author has been working in the fields of chemistry, physics and materials science for many years and is familiar with various experimental instruments. Due to the comprehensive integration of knowledge, the author was able to develop a logical and reason- able understanding of some of the confusing concepts presented in currently published scientific articles and in textbooks.
Results & Discussion
Bohr Shell Atomic Model and His Calculation
Based on Rutherford’s work, and by combining the knowledge of abundant atomic spectra, Bohr proposed a shell atomic model in 1913, commonly known as Rutherford-Bohr model. Before Bohr’s work, Kirchhoff and Bunsen [3], Balmer [19, 20], Rydberg [21] et al. had obtained a great deal of information from the visible light emission and absorption spectra of atoms and had summarized some rules, such as the Rydberg constant, the Balmer series and so on. Bohr’s contemporary scientists Planck [22], Einstein [23] and others had confirmed that some energies were discontinuous, which was the initial introduction of quantum concept. Bohr com- bined this new knowledge with the classical laws of physics and asserted: ''Electrons travel in several circular orbits around the nucleus. These orbits have quantized dimensions and quantized energies.”.
Figure 1 (a) was the Bohr’s original presentation of the shell atomic model [24]. If the electrons in the atom gain additional energy, such as the thermal energy, the impact energy of the particles, the radiant energy etc. it will jump to the adjacent higher energy orbit, or so to speak that the electron is excited. At this point, the electron is metastable. In the process of falling back to the ground state, the electron emits a piece of energy. If this energy is in the visible light range, we can see a bright line at a specific position in the visible light atomic spectrum. When irradiating a cold atomic gas, the transmitted light through a glass prism shows discrete dark lines. The emission and absorption spectra of the same element have exactly the same pattern. So, this is a reciprocal process, as shown in Figure 1 (b).
Figure 1: (a). Bohr’s Original Presentation of The Shell Atomic Model [24]. (b). Discrete Energies in Atomic Visible Light Emission and Absorption Spectra [internet].
This energy is equal to hν, called photon (h-Planck constant, ν-frequency of light = speed of light/wavelenghth, ν = c/λ). Each atom has its own specific series of ν-values, just like a human fingerprint. Thus, we can identify various elements from the spectra. Bohr has achieved great success in many aspects with this model.
More than just the shell atomic model, there are still some inner relationships hidden in the atoms. In an atom, the nucleus has an This formula was equivalent to the Rydberg’s empirical formula presented in 1888 to describe the spectral bright lines. integer number of positive unit charges, while each electron has only one negative unit charge. In the absence of external interfer- ence, the nucleus and its electrons are always arranged in a regular manner, namely the closed n-shell (full) always contains n2 pairs of electrons or n2 orbits. Bohr made a series of calculations based on two principles. First, the electrostatic attractive force between electron and nucleus is equal to the centrifugal force of the electron that circles around the nucleus. Second, the orbits allowed to exist are those that the angular momentum of the electrons are in accordance with quantum values. They were expressed as following formula
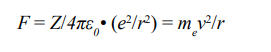
(F force, Z atomic number, ε0 electric constant, e unit charge, r shell radius, me electron mass, v speed of electron).
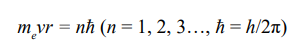
The n-shell radius (rn) of a given atom (Z) was expressed as following:
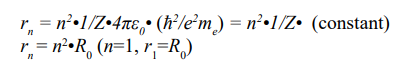
The total energy (kinetic energy plus potential energy) in the n-shell (En) was described in the formula below:
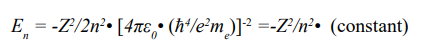
The energy difference between two neighbor shells could be calculated as following:
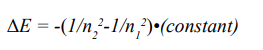
This formula was equivalent to the Rydberg’s empirical formula presented in 1888 to describe the spectral bright lines.
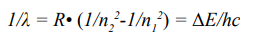
(λ wavelength of the electromagnetic radiation, R Rydberg’s constant, c speed of light). The result means that the Bohr’s calculation is very correct. Subsequently, the velocity of an electron in a certain shell and the circumference of an n-shell can be deduced.
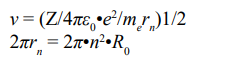
The most important points of the Bohr atomic model are as following:
(1) The neutral atom is built up by capturing and binding electrons one by one.
(2) If the electrons do not change orbits, there is no energy change in the system.
(3) As the electron jumps up or falls down into other orbits, the atom gains or loses energy in discontinuous value.
This model contains charge, mass, discrete energies, reciprocal process of emission and absorption of atomic spectra. However, it cannot explain the existence of s-, p-, d-, and f-sub-orbits of different shapes and different energies within the same shell. For example, even a small atom like boron, with only five electrons, has already problem. The first shell is filled with two electrons. The second shell with three, and one of the electrons is in a non- circular orbit, resulting in a slightly higher energy than the second shell. This is not to mention larger and more complex atoms. Bohr made many attempts, such as introducing celestial orbits and studying atomic spectra under external electric fields, but none of them yielded a significant breakthrough.
Therefore, modern science believes that Bohr’s model is outdated. Instead, some quantum theories, especially the Schrödinger orbital theory, have spread.
Schrödinger Wave Function of Atomic Model
Schrodinger’s atomic orbitals are the mathematical solutions (wave functions) to the Schrödinger equation, which treats electrons as wave-like particles [15-18, 25].
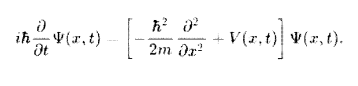
Here ψ (x, t) is a wave function to assign a complex number to each point x at each time t. The parameter m is the mass of the particle, and V (x, t) is the potential energy function that represents the environment in which the particle exists. The constant i is the imaginary unit, and h is the reduced Planck constant.
Here we only present Schrödinger's results. When n = 1, one spherical orbital; n = 2, one spherical orbital and three dumbbell shaped orbitals perpendicular to each other; n = 3, one spherical orbital, three dumbbell orbitals and five crossed double dumbbell orbitals, evenly distributed in space. Each orbital holds up to two electrons. The numbers of the s-, p-, d-, and f-sub-orbitals are 1, 3, 5, 7, as shown in Figure 2 and Figure 3.
These orbitals have different shapes and directions, which com- pensated for the inadequacy of the Bohr model to a certain extent. This arrangement of electrons happens to coincide with the order of electrons entering an atom summarized by previous scientists. The orientations of the dumbbell shaped sub-orbitals can also be reflected in the chemical reaction.
Figure 2: Schrödinger’s One s- and Three p-Orbitals
Figure 3: Schrödinger’s Five d-Orbitals
The serious problem is how electrons move in non-spherical orbitals. In particular, the five crossed double dumbbell sub-orbitals ac- tually have one of them in the shape of a stick penetrated a doughnut. Schrödinger appointed that the orbitals were the probabilities to find any electron in an atom in any specific region around the nucleus.
First of all, the first step of Schrödinger's theory (1926) is unreasonable. Long before Schrödinger, Millikan (1913) experimentally determined that the charge range of the electron is the same as the charge range of the hydrogen nucleus. The Coulomb attraction:
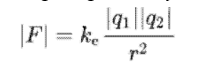
If electrons appear around the nucleus according to probability, you can imagine how unstable the system would be.
Secondly, a large number of facts show that atoms can absorb and emit light, which is a reversible process. How can Schrödinger's electron cloud show this characteristic of atoms?
Thirdly, according to probability, one in ten thousand chances can also happen. If an electron once appears in the position of the nu- cleus, it will definitely be sucked into the nucleus.
Scientists have long pointed out: “Quantum physics; basic idea is not clear, why it can deduce right results”. Many scientists now believe that quantum mechanics are incomprehensible and inexplicable, so they simply put it aside, regardless of whether it is sweet grapes or sour grapes.
Although Schrödinger's theory has been modified numerous times since then, extending it to molecular orbital theory and density functional theory. The results are that the electrons in the π-bond must be paired up, completely eliminating the double bond's activity, and the electronic structure of the simple C6H6 benzene molecule comes from tiling of the correlated 126-dimensional wave function. Should we fundamentally consider whether Schrödinger's theory is wrong?
New Proposal for Atomic Structure
One of the greatest Greece’s mathematicians and thinkers Pythag- oras said that “All things are numbers”. If we look at the periodic table of elements, we can see that the basic properties of the el- ements are decided by the numbers of the positive unit charges (number of protons Z+). Despite the existence of isotopes (dif- ferent numbers of neutrons), the difference of properties between them is very small. The entire material world is actually construct- ed only by two sorts of oppositely charged particles through differ- ent arrangements and different combinations, as shown in Figure 4.
Figure 4: The Entire Material World Is Actually Composed Only Two Sorts of Oppositely Charged Particles Through Different Arrangements and Different Combinations.
In everyday matter, atomic nuclei are stable. Various chemical and physical changes in matter are primarily related to the behavior of electrons. Therefore, understanding electron is the key to understand materials. To understand the atomic structure is to have a reasonable understanding of the arrangement of electrons outside the nucleus.
The electron motion should obey the general laws of nature like least action, symmetry, harmony, exactness, perfection and lowest energy.
In order to highlight the key points, three acceptable and reasonable simplifications have been made in this work.
(1) The nucleus is regarded as a whole of the positive charge carriers (do not care about the internal structure of the nucleus composed of neutrons and protons).
(2) The nuclei are stable (do not study radioactive nuclei).
(3) The nuclei have no magnetism (do not concern nuclear spin magnetism of the isotopes of certain elements, like 13C, 29Si etc.).
An overlooked fact in materials is that the electric field of an electron has the same magnitude as that of a hydrogen nucleus, but the mass ratio of the two is 1:1800, as shown in Figure 5. Electrons must revolve around the nucleus at ultra-high speed, otherwise they will be sucked into the nucleus. None of the atomic models have taken this important factor into account.
In 1925, Uhlenbeck and Goudsmit [12, 13] discovered that in addition to mass and charge, an electron also had a self-rotating, magnetic, intrinsic property, called electron spin. Later we can see how important this property is to understand materials.
Figure 5: The Electric Field of An Electron Has the Same Magnitude as That of a Hydrogen Nucleus, But the Mass Ratio of The Two is 1:1800. Electrons Must Revolve Around the Nucleus at Ultra-High Speed, Otherwise They Will Be Sucked into The Nucleus.
Figure 6 summarizes the fundamental characteristics of an elec- tron and an atomic nucleus. An electron possesses mass (me), spin orientation (±½), spin magnetism (µs) and unit charge (e). A nucle- us possesses mass, charge, spin orientation, but most of the nuclei have no magnetism. The radius of a hydrogen nucleus is about 1.0•10 -15 m and the radius of a hydrogen atom is about 1.0•10-11 m, so we can imagine how large this empty space in an atom is. But we should notice here, that the empty space is not really empty, it is fully filled with electric fields and sometimes also filled with magnetic fields, because electrons and nuclei are charged parti- cles. The electron is circling at an ultra-high speed of 2.2•106 m/s around the nucleus at a distance of 10.000 times the radius of the nucleus. This speed means that an electron can travel around the earth three time in a single minute. At such a high speed, the mov- ing path of the electron around the nucleus looks like a shell. This is the basic structure of the atom. If this shell is treated as a solid, it will lead to astray.
Figure 6: A summary of the Basic Characteristics of Electrons and Atomic Nucleia
When an atom consists of many electrons, the electrons are arranged in several shells. A closed n-shell (full) always contains 2•n2 electrons or n2 orbits.
People have long been puzzled why the electrons don’t squeeze into one shell. In fact, this is the natural law to reach lowest energy. Electrostatic repulsive force exists between electrons, so electrons cannot be regarded as mathematical points. This is what scientists did not realize before. The most important step in my work is to mark the electric and magnetic fields on the atomic structure. The repulsive force between the electrons limits the number of electrons in each shell. The range of this electric field is much larger than the size of the electron itself. This repulsive force is also the supporting force for constructing several shells in an atom.
Bohr’s model implies another message. If we divide the circum- ference of a closed n-shell by the number of the orbits in the same shell, no matter which shell, the lengths of the segments are always the same as the circumference of the first shell. Therefore, it is reasonable to believe that all orbits in an atom are equivalent, as shown in Figure 7.
Figure 7: Bohr’s Model Implies A Message That All Orbits in An Atom Are Equivalent
Logically, the orbits in the same shell should of course have the same energy. However, the experimental evidences do show sub- orbits with different energies in the same shell, for instance, 4s, 4p, 4d and 4f, as shown in Figure 8.
In the Bohr’s model, the electric fields of the nucleus and its electrons were not depicted. If two electrons in the same shell, but in different orbits, run into the same place, one of them must be repelled a little away from the main shell and acquires a little more energy. The more electrons in the same shell, the more the repelling events will occur. Statistically, we can find out the regularities of the repelling events and classify them into s-, p-, d-, and f- sub- orbits according to their different shapes and different energies, as shown in Figure 9. These orbits are fundamentally different from Schrödinger’s dumbbell-shaped orbitals.
Figure 8: Atomic Energy Diagram [Internet] And the Circumferences of The Various S-Orbits
Figure 9: Repulsion Between Electrons in The Same Shell but In Different Orbits Give Rise to Sub-Orbits with Polyhedral Symmetry
Further, it is necessary to introduce electron spin and orbital magnetism into Bohr’s model. When Bohr proposed his atomic model, the electron spin was not known. Unpaired electron creates whirl-shaped magnetic field around its orbit, which has been confirmed by nuclear magnetic resonance method (NMR); while paired electrons with anti-parallel spins cancel out the induced orbital magnetic fields and lower the entire system energy, as shown in Figure 10 (a) and 10 (b).
Figure 10: (a). Unpaired Electron Produces Whirl-Shaped Magnetic Field Around Its Orbit. (b). Paired Electrons with Antiparallel Spins Cancel Out the Induced Magnetic Fields and Lower the Entire System Energy.
Bohr noticed that some atoms were magnetic, but he didn’t know that there were paired and unpaired electrons. He had already pointed out that the magnetic moment produced by electron circling around nucleus was in the same way as the one produced by the circular current in a wire. The magnetic moment produced by an electron in an atom was called a Bohr magneton, which was usually marked with an arrow on the atom. After that, many theoretic studies used the Bohr magneton as a constant. In fact, Bohr magneton is a variable, otherwise we cannot understand the strong diamagnetism of matter. Unpaired electrons are active; paired electrons are relatively stable. Later, we can see that the unpaired electrons play a key role in electron transport property and chemical reactivity.
Now, we have the modified Bohr shell atomic model, which allows us to give real meaning to Pauli's four quantum numbers.
Real Meaning of Pauli’s Four Quantum Numbers
Pauli studied a large number of atomic spectra. The position of each line in the spectrum represents an energy level. These spectral lines have certain rules to follow. Shortly after the discovery of electron spin, Pauli introduced the new knowledge into his work and in 1925 proposed the four quantum numbers describing the electrons in the atom [14]. These are also known as the Pauli Exclusion Principle, which means that there are no any same electrons in an atom. He perfectly marked the electrons in the atom. These four quantum numbers were named as following:
(1) Principal quantum number (n, natural number)
(2) Azimuthal quantum number (l = 1, 2,…n-1)
(3) Magnetic quantum number [m = -l, -(l-1)…0…+(l-1), +l]
(4) Spin quantum number (s = ±½)
Pauli’s four quantum numbers discovered in 1925, which related to spectral energies and have nothing to do with Schrödinger’s wave function atomic orbitals (1926).
Based on the fact that the electrons repel each other at the same position, based on the proposal that electron spin aligns itself along the external magnetic field line, based on the natural laws of least action, symmetry, harmony, lowest energy, and combined with the improved Bohr’s shell atomic model, we can give the four quantum numbers the real meanings.
The principal quantum number (n) divides the electrons in an atom into n groups. That is to distribute the electrons into the n shells of the Bohr atomic model. That is also corresponding to the nth period of the element table.
The azimuthal quantum (l) indicates the different shapes and different energies of the orbits in the same shell due to the repelling events. Non-circular orbits have directionality. By n=1, l=n-1=0, there is only a spherical orbit. By n=2, l=0, 1, there are two sorts of orbits with different energies marked 0, 1. By n=3, l=0, 1, 2. In the third shell, there are three sorts of orbits with different energies and so on.
The magnetic quantum number (m) tells us that the same sub-orbits of the same energy, for example, the five d-orbits, have different directions, which can be detected by applying an external magnetic field, which gives the orbits different energies due to the different angles with the magnetic field. (Zeeman effect, [4, 5],1897).
The spin quantum number (s) describes that each orbit can only contain up to two electrons with anti-parallel spins.
How many orbits are there in an nth shell?
For example, n=3, there are three types of orbits with three energy levels in the third shell, l= 0, 1, 2. When l=0, it is a single spherical orbit. When l=1, m=-1, 0, +1, there are three sub-orbits with same energy, but with different orientations. When l=2, m=-2, -1, 0, +1, +2, In the third type of sub-orbits, there are five sub-orbits in different directions. There is a total of nine orbits in the third shell (Comparing Fig. 7.).
The four quantum numbers describing the electrons in an atom are consistent with the previous conclusion that a closed n-shell always contains 2•n2 elecrons. These laws cannot be found out by electron cloud model of Schrödinger probability theory. All of these is controlled by the influence of the quantized nuclear electric field on the quantized electron electric field, the electrostatic repulsive and the attractive forces, the orientation of the electron spin etc.
The motion of the electrons in an atom looks very complicated, but the circular motion is the basic manner.
Covalent Bond, Double Bond and Metallic Bond
A new understanding of atomic structure led directly to a new understanding of covalent bonds. The covalent bond (σ-bond, or single bond) is the most important bonding in nature. It involves the sharing of two electrons between two adjacent atoms. But how to share?
The most accepted model for covalent bond at present is the over- lap of Schrödinger atomic orbitals. The region of maximum elec- tron cloud density is considered to be the probability to find two electrons with antiparallel spins. That is another reason why the Schrödinger atomic model is thought to be superior to the Bohr shell model. This model is based on the maximum electron cloud density. As analyzed above, the electron cloud is irrational. In contrast, in this work, the positive electric field of the nucleus is taken as the basis, and the movement of electrons changes with the nucleus.
Electrons can be attracted by any positive electric field. When two atoms get closer, the higher-energy valence electron (chemical name of unpaired electron) in one atom can also be attracted by the other nucleus, and vice versa. The maximum superimposed electric field of two identical nuclei is in the fixed ring, as shown in Figure 11 (a). Once the Coulomb attractive forces between the nuclei and the electrons, the repulsive force between nucleus-1 and nucleus-2, as well as the centrifugal forces of the circling electrons are balanced, the two unpaired electrons from the two atoms enter the fixed ring region with antiparallel spins, which cancel out the original orbital magnetism and lower the system energy. After that, the two electrons travel in the new orbit and form a new covalent bond. This should be a reasonable structure of a covalent bond. In fact, the positive electric fields determine how electrons move. Electrons cannot form electron clouds and be discovered by probability. The correctness of the new covalent bond model lies in its ability to explain the one-electron σ-bond, as shown in Figure 11 (b)
Figure 11: (a). The Formation of a Two-Electrons Covalent σ-bond. (b). The One-Electron Covalent σ-bond. The Electrons Circle in The Maximum Superimposed Electric Field of Two Positively Charged Carbons. (Bond length 2.92 Å [27]).
In 2024, T. Shimajiri et al. published a paper in Nature “Direct evidence for a carbon–carbon one-electron σ-bond” [27]. The C1+ and C2+ superimposed electric field can enter a single electron or enter two electrons with antiparallel spins. This fact shows that the σ-bond is not formed by the overlap of two p-orbitals. However, single-electron covalent bonds are unstable, and the covalent bonds discussed below are all two-electrons covalent bonds.
Similarly, the π-bond in a double bond is not formed by overlapping p-orbitals perpendicular to the σ-bond. Instead, the p-electrons are attracted by two positive electric fields and repelled by the electrons in the σ-bond, forming two parallel elliptical orbits, as shown in Figure 12 (a). This structure is also supported by NMR experiment, as shown in Figure 12 (b). It can be seen that the electric fields parallel to the paper plane can still accommodate two π-electrons, thus forming a triple bond structure.
The proposed new model of covalent bonding also finds support from the gradually changing properties of the carbon group of
elemental solids, as shown in Figure 1.
Figure 12: (a). New Proposal of Double Bond. Two Parallel Unpaired Electrons Describe Two π-orbits Above and Below The σ-bond.
The Two Unpaired Electrons Make the Double Bond Magnetic. (b). 1H- NMR Confirmed Double Bond Structure
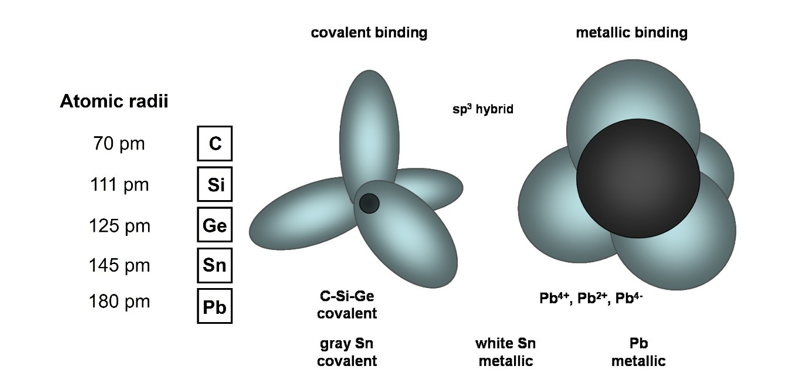
Figure 13: The Gradual Changing Properties of The Carbon Group of Elemental Solids. The Larger the Ion Cores, The More Difficult to Share the Electrons and The More Metallic Properties. Grey Tin and White Tin Are Just at The Critical Point Between Covalent Bonding and Metallic Bonding.
Their inner shells are fulfilled to form stable closed shells, and the four electrons in the outermost shell are sp3 hybridized in the positively charged crystal lattice due to the attraction of the adjacent positive charges. This is completely different from Schrödinger's dumbbell p-orbitals. The sp3 elliptical orbits have no intersection with the nucleus. As the atomic radius increases, this group of elements changes from being insulator to semiconductors and eventually conductors. The atomic connection changes from covalent bonds to metallic bonds. The larger the ion core, the more difficult it is to share the unpaired electrons and the more metallic properties
Thermal and Electrical Conductivities
Re-understanding covalent bonds and metallic bonds can help us clarify the vague concepts of electrical conductivity and thermal conductivity in solid-state physics, and thus correctly understand the superconductivity phenomenon that has always puzzled people.
Figure 14: (a). The Carbon-Carbon σ-Bond in Diamond Is Like A Strong Spring. The Springs Are Connected into Regular Chains. If the Vibrational Motion of The Atoms Is Along the Zigzag Way, There Is No Blockage from The Perpendicular Direction. Heat Can Then Be Transferred in Diamond at Ultra-High Speed. (b). The Jumping Motion of Unpaired Electron Orbits Circling Around the Ion Core of The Atom Forms an Electric Current.
It is generally believed that a good electrical conductor is naturally a good thermal conductor and vice versa. Diamond is a typical insulator without unpaired electrons. It has the highest electrical resistance value of any material (with a band gap of 5.47 eV), but its thermal conductivity (2300 W•m-1•K-1) is thousands of times higher than that of a good electrical conductor like copper (386 W•m-1•K-1). In fact, these two conductivities have very different mechanisms. Heat is transferred through the vibrational motion of atoms, and electricity is transmitted through the jumping motion of electron orbits.
Figure 14 (a) illustrates schematically the vibration motion of the atoms in diamond through covalent bonds. The carbon-carbon σ-bond in diamond is like a strong spring. The springs are con- nected into regular chains. If the vibrational motion of the atoms is along the zigzag way, there is no blockage from the perpendicular direction. Heat can then be transferred in diamond at ultra-high speed. The thermal conductivities of all materials are directional or anisotropic, and the heat transfer is obstructed at the interface between two different crystals. These phenomena can be well explained by the newly proposed heat transfer mechanism.
Figure 14 (b) illustrates the jumping motion of the unpaired electron orbits in metals to form electric current. According to the currently widely accepted model, free electrons are shared by positively charged ionic cores in the crystal lattice, just like molecular gases in air. Under an external electric field, the random motion of free electrons drifts toward one direction to form a current flow. The speed of charge drift in common copper wires is about 1 m/s. However, the electrons have to travel at superfast speeds of 106 m/s to balance the electrostatic attraction of the nucleus. Otherwise, the electrons could be sucked into the nuclei and the metal would get very much heavier.
If we change our mind and believe that each “free electron” is bound to each atom with orbital motion, then many properties of metals can be better understood.
• Metals should have unpaired electrons in their atomic outermost shells.
• Unpaired electrons circle rapidly around their nuclei.
• The electric current is formed by the jumping motion of the unpaired electron orbits. In this way, the electrical conductivity of some amorphous metals can also be explained.
The good thermal conductivity of most metals is also related to the regular chains in certain directions, because the close-packed spheres cause the connection of adjacent atoms by gravitational force. Amorphous solids such as rubbers and glasses have irregular atomic arrangement and cannot propagate the vibrational motion of the atoms properly, so they are thermal insulators. Instead, the electric current can be transferred through amorphous metals and can pass through the interface of two different metallic crystals, by the orbital motion of unpaired electrons.
Atomic Structure and Superconductor
This section does not discuss in depth the mechanism of supercon- ductivity, but only discusses the influence of atomic structure on the superconductivity of certain materials. Figure 15 briefly introduces the Mott-insulator, normal conductor, and superconductor. The distance between the orbits decides their electrical properties.
Figure 15: The Distance Between the Unpaired Electron Orbits Determines the Matter from Mott-Insulator to Superconductor
In the new atomic structure section, we mentioned that p-, d-, and f-sub-orbits are generated by electron repelling events. Atomic energy diagram clearly shows that the 4s sub-orbit is lower in energy than the 3d sub-orbits. Figure 16 shows the electronic configuration of copper atoms in a solid.
Figure 16: Electron Configuration of Cu Atom. The Single Unpaired Electron In 4s Orbit Can Jump Over to Adjacent Atom with Certain Resistance, But The 4s Orbits Cannot Get in Contact Due to The Screen of the 3d Orbits.
higher than the 4s orbit. The first, second and third shell are all filled with electrons with antiparallel spins. These electrons are stable and cannot shift for electric current. Only one unpaired electron in the 4s orbit can jump over to adjacent atom with certain resistance for normal conductivity. The fully filled five quatrefoil 3d orbits prevent the unpaired electrons in the 4s orbits from making contact, so pure copper will not be a superconductor. Similarly, gold and silver will not show superconductivity.
Why are pure white tin and lead superconductors, but pure grey tin not? The answer is clear and direct from Figure 13. Grey tin has covalent bonds without unpaired electrons and white tin has metallic bonds with unpaired electrons.
Atomic Structure and Hall Effect
Until now, the Hall effect [28] has not been explained at the atomic level. If we take the newly discovered atomic structure into account, we see that only electrons are shifting in the system. The holes are formed in the positive electric field where electrons are missing and do not flow. Figure 17 (a) shows the setup of the Hall experiment. Figure 17 (b) shows the unique unpaired electron orbit in gold atom, which has the electron configuration [Xe] 4f14 5d10 6s1. The electron spin is oriented along the magnetic field lines, with the orbital plane perpendicular to the magnetic field. The movement of the electron orbit should be parallel to the sample surface.
When an electron in the orbit is moving along or against the external electric field (E), the velocities of the electron on the left and right parts of the orbit are different. The net Lorentz force on the orbit is then not zero. It is this force difference ΔF that drives the orbit to drift laterally. Finally, more electrons are accumulated on one edge, which establish a Hall voltage.
Figure 17: Ordinary Hall Effect. (a). Hall Experiment Setup [28]. (b). Microscopic Mechanism of Hall Effect
Atomic Structure and Stern-Gerlach Effect
The Stern-Gerlach effect was believed to be very important for revealing the space quantization of atomic spins (magnetic moments). The silver atom has the electron configuration [Kr] 4d10 5s1 with a single unpaired electron in the round s-orbit. They believed that a magnetic field gradient caused the magnetic moment of the atoms to reverse, but this conclusion was erroneous because they failed to consider the atomic structure, as shown in Figure 18.
The most commonly used neutral atoms in the Stern-Gerlach experiment reported in the literatures were Ag, H, Na, K, Cu and Au. Why? The commonality of these atoms is that they all contain only one unpaired electron, running in their round s-orbits. The orbital magnetism of the unpaired electron (one Bohr magneton) is much weaker than the external magnetic field (ca. 1 Tesla) and can be neglected. The electron spin aligns itself along the magnetic field line, so that the orbital plane is perpendicular to the direction of the magnetic field line. The force exerted by the magnetic field on the moving charge is as shown in Figure 19 (a).
F = qvxB
F: Lorentz force, q: electron charge, v: speed of electron, B: local magnetic field.
Since the linear densities of the magnetic field on the left and right sides of the orbit is different, the net magnetic force on the entire orbit is not zero.
ΔF=qvx(B1-B2)
It’s Just this force difference that causes the atoms to deviate from the beam path of the silver atoms.
When viewed in front of the instrument, the small arrows in Figure 19 (b) indicate the net magnetic forces in the orbital planes and also indicates the distribution of the atoms in the entire beam path.
Figure 18: Stern-Gerlach Experimental Setup [29].
Figure 19: (a). The Mechanism at Atomic Level. (b). Viewed in Front of The Instrument. The Small Arrows Indicate the Net Magnetic Forces in The Orbital Planes and Also Indicate the Distribution of The Atoms in The Entire Beam Path
Figure 20 (a-d) show the gradual deflection of the atoms in the inhomogeneous magnetic field. The corresponding positions a, b, c, and d were marked in the previous sketch of the device. During the long journey, the s-orbits constantly adjust their directions to maintain the perpendicular posture with the magnetic field lines.
Once leaving the magnetic field, the atoms keep their previous directions of movement to aim at the screen with inclined angles.
Thus, the deposited pattern is the result of the trajectories of the atoms; it is not the space quantization of the atomic spins.
Figure 20: The Gradual Deflection of The Atoms in The Inhomogeneous Magnetic Field. The Corresponding Positions a, b, c, and dWere Marked in The Previous Sketch of The Device
The above examples show how important it is to have a correct understanding of atomic structure. The facts cannot be explained by Schrödinger's electron cloud density model.
Deep Understanding of The Atomic Structure
Siegbahn had mentioned that the arrangement of electrons of heavy atoms is the same in the inner shells [30, 31], which is consistent with the Bohr atomic model. However, due to differences in the number of positive unit charges, the binding forces of the nuclei on electrons are different.
When irradiated with X-rays [32], the atoms emit secondary radiation that contains the elemental signature. This part of the research is only helpful for the theoretical understanding of atoms, and is not very helpful for the colorful chemical and physical changes because the active electrons are all in the outer shells.
By applying Bragg X-rays diffraction spectroscopy [33], Siegbahn was able to determine the wavelengths of the characteristic radiations of the K-series and L-series of many elements, and he also discovered the lower-energy M-series. He summarized the wavelengths of the secondary radiations from oxygen (8) to uranium (92) into the following chart. Clearly, there were three energy levels, which corresponded to three atomic shells, as shown in Figure 2
1.
Figure 21: Siegbahn’s Secondary Characteristic Radiations of Elements. Atomic Numbers Vs Wavelengths [30]
In 1922, Dolejšek [34, 35] found the N-series in soft X-rays spectra of the elements Bi, Th and U. These elements have over 5 shells. This shows that the three series Siegbahn discovered are not yet complete.
Moseley [36, 37] converted the atomic number Z versus wavelength λ into Z to frequency ν½. It turned out to be straight lines, as shown in Figure 22. The result was in perfect agreement with Bohr's theoretical model.
Figure 22: Moseley’s Diagram of Atomic Number Vs Square Root of Secondary Radiation Frequencies [36].
The energy difference between Bohr neighbor shells is as following:
ΔEn,n-1 = -Z2 [1/n2-1/(n-1)2] • (constant) = hν = hc/λ
Z = constant • {[1/n2-1/(n-1)2]}-½ • ν½
Then, Z vs ν½ is straight line. In the same atom, the closer the electrons to the nucleus, the harder they are to be excited. In the same series, the higher the atomic number, the harder it is for the inner electrons to be excited. Bohr’s atomic model was proved to be very correct.
Siegbahn’s notation is used in X-rays spectroscopy to name the spectral lines that are characteristic to elements. He thought that the notation was the complete electron excitation energy levels. Until now, it is generally believed that the K-series corresponded to 1s-shell. All elements have 1s-shells, but Siegbahn did not get K-radiations from the elements smaller than oxygen.
However, Barkla did find characteristic radiations [38-41] from the elements smaller than oxygen, but at higher frequencies than the K series. He named it the J-series. The J-series was only a small fraction and was of close similarities to K- and L-series in the whole process involved, so it was not possible that it was emitted from the nucleus. Barkla arranged the secondary radiations into the sequence of J-, K-, L- and M-series, but the J-series found no place in the Siegbahn notation. Barkla’s finding has been ignored so far, but he might well be right. The introduction of the J-series gave us a very reasonable understanding of the famous Compton effect [42, 43]: a very good experiment, but a very controversial theory of billiard ball collisions.
Why is the J-series so hard to spot? We can understand the fact by using Bohr's formula to calculate the energy difference between two adjacent shells.
ΔEn,n-1 = -Z2 [1/n2-1/(n-1)2] • (constant) = hν=hc/λ
ΔE2,1= -Z2 (1/22-1/12) • (constant) (0,750) (2s shell to 1s shell)
ΔE3,2= -Z2 (1/32-1/22) • (constant) (0.139) (3s Shell to 2s shell)
ΔE4,3 = -Z2 (1/42-1/32) • (constant) (0.047)
ΔE5,4= -Z2 (1/52-1/42) • (constant) (0.023)
ΔE6,5 = -Z2 (1/62-1/52) • (constant) (0.012)
ΔE7,6 = = -Z2 (1/72-1/62) • (constant) (0.007) (7s shell to 6s shell)
When these energy differences are represented graphically, it can be clearly seen that there is a large energy difference between the first shell and the second shell, making it difficult for electrons in the first shell to be excited into the second shell, as shown in Figure 23 (a). The Siegbahn notation must be corrected, as shown in Figure 23 (b).
Taking the Barkla J-series into account, the Siegbahn and Moseley diagrams can be easily explained.
Figure 23: (a). Energy Levels Between Shells of Bohr’s Atomic Model. (b). Modified Siegbahn Notation by introducing Barkla J-Series [41]
The second shell contains two kinds of electrons, 2s and 2p, which mainly produce two characteristic radiation lines, Kα and Kβ. The third shell contains three kinds of electrons, 3s, 3p and 3d, which mainly produce three radiation lines of L-series. During the en- tire excitation process, several shells should expand and contract simultaneously. That is the reciprocal process of absorption and emission of energies by atoms, similar to visible light atomic spectra.
When we do elemental analysis with X-rays and electron beam, we just use Bohr’s shell atomic model to explain the results and never use the Schrödinger’s wave function atomic model.
Postscript
Ultimately, everyday matter is made up of positively charged atomic nuclei and negatively charged electrons. Normally, the nuclei are stable, providing the basis for the movement of electrons. Electrons can exist in six states within materials. (1) Paired electrons in atomic orbit, no magnetism. (2) Paired electrons in molecule, covalent bond, no magnetism. (3) Unpaired electron in atom, with magnetism. (4) Unpaired electron in molecule, radical, with magnetism. (5) Unpaired two π-electrons with parallel spins in double bond, with magnetism. (6) Unpaired two lone pair of electrons with parallel spins in hybrid orbits, with magnetism. The basic motion of electrons in atoms is to move in circular and elliptical orbits at ultra-high speeds.
Conclusion
The Bohr shell atomic model is correct and not outdated, but it does have its shortcomings. Introducing electron spin and charge would further improve the atomic structure. The existence of s-, p-, d-, and f-sub-orbits is believed to be caused by the repulsive forces between electrons orbiting in different orbits within the same shell. This modified Bohr model can give true meaning to Pauli's four quantum numbers. The fundamental causes of quantum phenomena can be traced back to the unit charge of the nucleus, the unit charge of the electron, and the circular motion of electrons around the nucleus.
New structures of basic chemical concepts such as single bonds, double bonds, triple bonds, metallic bonds and lone pairs of electrons were proposed, and the mechanisms of thermal and electrical conductivities were elucidated.
This work could provide new insights into phenomena like super- conductivity, Hall effect, space quantization of atomic spins.
Acknowledgement
The author would like to thank Dr. Thomas VON LARCHER, Se- nior Editor, Research Publishing, Springer Nature. He took great effort to provide the author with a series of guidance, which en- abled the author to comprehensively improve her ability to com- plete scientific papers.
References
- Wollaston, W. H. (1802). XII. A method of examining refractive and dispersive powers, by prismatic reflection. Philosophical transactions of the Royal Society of London, (92), 365-380.
- Fraunhofer, J. (1817). Determination of the refractive and color dispersion power of various types of glass, with reference to the improvement of achromatic telescopes. Annalen der Physik , 56 (7), 264–313.
- Kirchhoff, G., & Bunsen, R. (1860). Chemische Analyse durch Spektralbeobachtungen. Annalen der Physik und Chemie 2nd Series, 110 (6), 161-189.
- Zeeman, P. (1897). On the influence of magnetism on the nature of the light emitted by a substance. Philosophie Magazin 5th Series, 43(262), 226-239.
- Zeeman, P. (1897). The effect of magnetisation on the natureof light emitted by a substance. Nature, 55(1424), 347.
- Zeeman, P. (1897). VII. Doublets and triplets in the spectrum produced by external magnetic forces. The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science, 44(266), 55-60.
- Thomson, J. J. (1897). Cathode Rays. Philosophie Magazin 5th Series, 44(269), 293-316.
- Millikan, R. A. (1913). On the Elementary Electrical Charge and the Avogadro Constant. Physical Review, 2(2), 109-143.
- Rutherford, E. (1911). The scattering of α and β particles by matter and the structure of the atom. Philosophie Magazin 6th Series, 21(125), 669-688.
- Bohr, N. (1913). On the constitution of atoms and molecules, Part I. Philosophie Magazin 6th Series, 26(151),1-25.
- Bohr, N. (1913). On the constitution of atoms and molecules, Part II. Systems Containing Only a Single Nucleus. Philosophie Magazin 6th Series, 26(151), 476-502.
- Uhlenbeck, G. E., & Goudsmit, S. (1925). Ersetzung der Hypothese vom unmechanischen Zwang durch eine Forderung bezüglich des inneren Verhaltens jedes einzelnen Elektrons. Die Naturwissenschaften, 13(47), 953-954.
- Uhlenbeck, G. E., & Goudsmit, S. (1926). Spin electrons andthe structure of spectra. Nature, 117(2938), 264-265.
- Pauli, W. (1925). Über den Zusammenhang des Abschlusses der Elektronengruppen im Atom mit der Komplexstruktur der Spektren. Zeitschrift für Physik, 31(1), 765-783.
- Schrödinger, E. (1926). Quantisierung als EigenwertproblemI. Annalen der Physik, 79(6), 361-376.
- Schrödinger, E. (1926). Quantisierung als EigenwertproblemII. Annalen der Physik, 79(6), 489-527.
- Schrödinger, E. (1926). Quantisierung als EigenwertproblemIII. Annalen der Physik, 80(13), 437-490.
- Schrödinger, E. (1926). Quantisierung als EigenwertproblemIV. Annalen der Physik, 81(18), 109-139.
- Balmer, J. J. (1885). Notiz über die Spectrallinien desWasserstoffs. Annalen der physik, 261(5), 80-87.
- Balmer, J. J. (1897). Eine neue Formel für Spectralwellen. Verhandlungen der Naturforschenden Gesellschaft in Basel, 11(3), 448-463.
- Rydberg, J. R. (1890). XXXIV. On the structure of the line- spectra of the chemical elements. The London, Edinburgh, and Dublin philosophical magazine and journal of science, 29(179), 331-337.
- Planck, M. (1978). Über das gesetz der energieverteilung im normalspektrum. In Von Kirchhoff bis Planck: Theorie der Wärmestrahlung in historisch-kritischer Darstellung (pp. 178- 191). Wiesbaden: Vieweg+ Teubner Verlag.
- Einstein, A. (1989). Uber einen die Erzeugung und Verwandlung des Lichtes Betreffenden Heuristischen Gesichtspunkt. Collected Papers of Albert Einstein, 2, 150- 166.
- Bohr, N. (1923). The Structure of the Atom1. Nature, 112(2801), 29-44.
- Schrödinger, E. (1926). An undulatory theory of the mechanics of atoms and molecules. Physical Review, 28(6), 1049-1070.
- Liu, Y., Kilby, P., Frankcombe, T. J., & Schmidt, T. W. (2020). The electronic structure of benzene from a tiling of the correlated 126-dimensional wavefunction. Nature Communications, 11(1), 1210-1214.
- Shimajiri, T., Kawaguchi, S., Suzuki, T., & Ishigaki, Y. (2024). Direct evidence for a carbon–carbon one-electron σ-bond.Nature, 634(8033), 347-351.
- Hall, E. H. (1879). On a new action of the magnet on electric currents. American Journal of Mathematics, 2(3), 287-292.
- Gerlach, W., & Stern, O. (1989). Der experimentelle nachweis der richtungsquantelung im magnetfeld. In Walther Gerlach (1889–1979) Eine Auswahl aus seinen Schriften und Briefen (pp. 26-29). Berlin, Heidelberg: Springer Berlin Heidelberg.
- Siegbahn, M. (1962). X-ray Spectroscopy. In Fifty Years of X-Ray Diffraction: Dedicated to the International Union of Crystallography on the Occasion of the Commemoration Meeting in Munich July 1962 (pp. 265-276). Boston, MA: Springer US.
- Siegbahn, M. (1916). Relations between the K and L series of the High-Frequency Spectra. Nature, 96, 676.
- Röntgen, W. C. (1896). On a new kind of rays. Science, 3(59), 227-231.
- Bragg, W. H., & Bragg, W. L. (1913). The Reflexion of X-rays by Crystals. Proceedings of the Royal Society of London A, 88(605), 428-438.
- Dolejšek, V. (1922). On the N-Series in X-Bay Spectra.Nature, 109(2740), 582-582.
- Dolejšek, V. (1922). Über die N-Serie der Röntgenspektren.Zeitschrift für Physik, 10(1), 129-136.
- Moseley, H. G. J. (1913). XCIII. The high-frequency spectra of the elements. The London, Edinburgh, and DublinPhilosophical Magazine and Journal of Science, 26(156), 1024-1034.
- Moseley, H. G. J. (1914). LXXX. The high-frequency spectra of the elements. Part II. The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science, 27(160), 703-713.
- Barkla, C. G. (1905). Secondary röntgen radiation. Nature, 71(1845), 440-440.
- Barkla, C. G. (1906). Polarisation in secondary Röntgen radiation. Proceedings of the Royal Society of London. Series A, Containing Papers of a Mathematical and Physical Character, 77(516), 247-255.
- Barkla, C. G., & Sadler, C. A. (1907). Homogeneous Secondary Röntgen Radiations. Proceedings of the Physical Society of London, 21(1), 336.
- Barkla, C. G. (1923). The “J” phenomena and X-ray scattering.Nature, 112(2820), 723-724. [42]
- Compton, A. H. (1922). Secondary Radiations produced by X-rays, and some of their applications to physical problems. Bulletin of the National Research Council, 4(20), 1-56.
- Compton, A. H. (1923). A Quantum Theory of the Scattering of X-rays by Light Elements. Physical Review, 21(5), 483- 502.