Research Article - (2024) Volume 9, Issue 2
Nephroprotective Effect of Hydroalcoholic Extract of Manikara Zapota on Cisplatin Induced Nephrotoxicity in Male Wistar Rats
Received Date: Aug 23, 2024 / Accepted Date: Nov 05, 2024 / Published Date: Nov 19, 2024
Copyright: ©©2024 Maheshwari Reddy B, et al . This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Redd, B. M., Praveena Devi, C. B., Taj, S. K. S. (2024). Nephroprotective Effect of Hydroalcoholic Extract of Manikara Zapota on Cisplatin Induced Nephrotoxicity in Male Wistar Rats. J Pharmaceut Res, 9(2). 01-05.
Abstract
Nephrotoxicity is known to be a major complication during cisplatin chemotherapy in cancer patients. Cisplatin is a highly effective chemotherapeutic agent; its clinical use is severely limited by serious side effects as nephrotoxicity. The aim of this study is to evaluate the nephroprotective activity of hydroalcoholic extract of Manikara zapota (MZ) against cisplatin (CP)- induced renal damage using albino rats.
Keywords
Manikara Zapota, Nephrotoxicity, Cisplatin, HydroalcoholicIntroduction
The Kidneys play an important role in the maintenance of our endocrine, acid-base balance, blood pressure and erythropoiesis. The main functions of the kidney are formation of urine, maintenance of water and electrolyte balance as well as production of hormones and enzymes. Nephrotoxicity is renal dysfunction that arises as a direct result of exposure to external agents such as drugs and environmental chemicals.
Cisplatin (cis-diammine dichloroplatinum (II)) is effective against a variety of cancer conditions such as solid tumors, haematological malignancies, lymphoma, osteosarcoma, bladder, oesophageal, gastric, pulmonary, testicular, ovarian, head and neck cancers [1]. CP is a potent chemotherapeutic agent, which causes acute kidney injury (AKI) by inducing the oxidative stress, inflammation, and cell apoptosis. Although cisplatin therapy is able to reduce tumor burden effectively, it exhibits a narrow and unfavourable therapeutic index and can lead to severe tissue damage [2]. Due to the higher benefit to risk ratio, CP is still an integral part of a combination therapeutic treatment regimen used against various cancers [3]. Presently, there is a paucity of an effective treatment to prevent renal damage caused by cisplatin [4]. Therefore, there is an unmet need for a novel therapeutic treatment regimen to combat the development of nephrotoxicity during cisplatin chemotherapy.
Nephroprotective agents are material that has potential to minimize the effects of nephrotoxic agents. Medicinal plants have curative properties due to the presence of various complex chemical substances [5]. Ethnomedicinal plants from the traditional system of medicine Ayurveda and Unani, which are acclaimed by the Ayurvedic and Unani physicians to have nephroprotective properties and commonly used to treat various renal disorders, have been extensively investigated for their significant nephroprotective effects [6].
Manilkara zapota (family Sapotaceae) is widely distributed plant in tropical India. It is found in lowland, riverine and ground water forest. It is a tree that grows up to 14 m or more and has dark brown bark. The leaf is characterized by the presence of reddish-brown hairs underside. The fruits are edible. The fruit and stem bark are used as spice for cooking. The plant is used as in traditional medicine for treatment of cardiovascular disorders [7]. Although some plants have shown good results against nephrotoxicity-induced cisplatin to date, no approved drug is available on the market to regenerate the renal tubular cells after cisplatin damage. Further, there are no literature reports on beneficial effects of Manilkara zapota in experimentally induced kidney damage. Taking all these facts into consideration, we thought it worthwhile to undertake a detailed study employing drug discovery strategies such as in-vivo experiments to demonstrate the effectiveness of Manilkara zapota against cisplatin-induced renal toxicity [8]. The objective of the present work was to investigate the therapeutic potential of hydroalcoholic extract of Manilkara zapota (MZ) against cisplatin-induced renal damage in a rat model.
Materials and Methods
All chemicals used in this study, were of analytical grade. Cisplatin was purchased from Sigma Chemical Company, St. Louis, Missouri, USA. The fresh leaves of Manilkara zapota were collected from the outskirts of Telangana (India). The powder (830 g) was macerated at room temperature with 5 L of ethanol-water (8:2 v/v) for 72 h. The filtrate was evaporated under vacuum at 45 °C by a rotary evaporator (Rotavapor Buchi R100). The yield of hydroalcoholic extract of Manilkara zapota (MZ) was 14.15%.
Experimental Animals
An ethical approval of this experimental study was obtained from the Institutional Animal Ethical Committee of Malla Reddy College of pharmacy, Hyderabad with Reg. No 1217/PO/ Re/S/08/CPCSEA. Twenty-four albino rats with an average body weight from 150 to 250 g were utilized in this study. They were procured from Teena labs, Plot no 41, SV cooperative industrial estates, Bachupally (V), Quthbullapur. The rats were housed in polypropylene cages and maintained under standard conditions (12 h light and dark cycles at 25 ± 3°C and 35-60 % humidity). Standard pelletized feed and tap water were provided ad-libitum. A total of 24 animals were randomized based on their body weight into four groups, comprising of six rats in each group. Group I (Normal control): Normal saline was administered orally for 10 days; Group II (Cisplatin control): Normal saline was administered orally for 10 days and a single dose of cisplatin (7.5 mg/kg, i.p) dissolved in normal saline was administered on the 5th day; Group III (MZ 200 mg/kg): Rats were treated with MZ (200 mg/kg/day) orally for 10 days and a single dose of cisplatin (7.5 mg/kg, i.p) was administered on the 5th day, 1 h after the MZ dose; Group IV (MZ 400 mg/kg): Rats were treated with MZ (400 mg/kg/day) orally for 10 days and a single dose of cisplatin (7.5 mg/kg, ip) on the 5th day, 1 h after dosing the extract.
The body weight of the rats was measured daily during treatment. On Day 11 (5 days post cisplatin administration), urine samples were collected by keeping the animal on a plastic plate and awaiting spontaneous micturition. The urine samples were collected and centrifuged for 5 min at 1500 rpm and the clear supernatant was stored at −20 °C and thawed just before use for the biochemical analysis. After urine collection, animals were anesthetized with ether, and blood samples were collected from the orbital sinus in centrifuge tubes and allowed to clot for 20 min at room temperature. The samples were centrifuged at 4000 rpm for 10 min at 4 °C, and the resultant serum was separated and stored at −20 °C and thawed just before use. Rats were euthanized by an over dose of thiopentone sodium and kidney tissues were dissected and weighed. The left kidney was fixed in 10% neutral buffered formalin solution for histopathology and the right kidney was used for biochemical estimations.
Biochemical Analysis
The samples were allowed to clot and centrifuged at 3000 rpm at 30°c for 15 min and the separated serum was used for the following biochemical estimations using commercially available kits: total protein (Erba Mannheim, Ltd., India), albumin (Precision Biomed Ltd., India), creatinine (Erba Mannheim, Ltd., India), and urea (Erba Mannheim, Ltd., India). The 24 h urine was used for the estimation of creatinine clearance levels (Erba Mannheim, Ltd., India). All assays were performed according to the manufacturers’ instructions.
Histopathology of Rat Kidney
After blood sampling, all the animals were sacrificed and subjected to a complete necropsy followed by histopathology. The rat kidneys were identified and carefully dissected out for histopathological examination. After rinsing in normal saline, sections were taken from each harvested kidney, fixed in 10% formalin, dehydrated in gradual ethanol (50–100%), cleared in xylene, and embedded in paraffin wax. The 5–6 μm sections were prepared using a rotary microtome and stained with haematoxylin and eosin dye for microscopic observation of the histopathological changes.
Statistical Analysis
All the data were expressed as mean ± SD. One-way analysis of variance (ANOVA) was used to determine significant intergroup differences of each parameter. Dunnet’s test was used for individual comparisons after significant ANOVA results followed by “Dunnett’s multiple comparison test.
Results
Body Weight Changes
The administration of CP resulted in significant (P < 0.001) loss in body weight as compared to normal control group. However, MZ administration attenuated CP induced loss of body weight.
|
Groups |
Day 0 |
Day 5 |
Day 10 |
|
Normal Control |
146.43±11.34 |
142.24±11.54 |
144.12±13.62 |
|
Cisplatin |
138.32±12.23# |
135.26±12.34# |
132.12±12.58# |
|
MZ (200mg/kg) |
146.22±11.12* |
142.21±11.56* |
144.11±11.56* |
|
MZ (400mg/kg) |
144.43±10.43* |
141.18±11.51* |
142.16±11.63* |
Table 1: Effect of Aqueous Extract of Manilkara Zapota on Body Weight in Cisplatin Induced Renal Damage
Values are expressed as Mean±SEM of six animals. Statistical significance test for comparisons was done by one-way ANOVA, followed by “Dunnett’s multiple comparison test.” Comparisons were done between Cisplatin group compared with control (P#<0.001), MZ extract (200 and 400 mg/kg.b.w) in comparison with Cisplatin group (P*<0.01 and P**<0.001). 3.2. Serum Biochemical Markers After a single dose injection of cisplatin, serum levels of creatinine, urea, urine volume, and BUN in the cisplatin-control group were significantly increased (P#< 0.001) compared to the normal control group, while the levels of total protein (TP), albumin, creatinine clearance and kidney weight were significantly (P# < 0.01) decreased. These elevated levels of renal function parameters as a result of cisplatin treatment confirmed the induction of nephrotoxicity. However, pre-treatment with different doses of MZ extract (200 and 400 mg/kg) resulted in significant (P* < 0.01) reduction in kidney function biomarkers such as creatinine, urea, urine volume, and BUN. In addition, the protective effect of MZ extract was also confirmed by significantly (P** < 0.001) restored levels of total protein, albumin and kidney weight was restored as compared to Cisplatin-control group.
|
Groups |
Serum total protein (g/dl) |
Serum albumin (g/dl) |
Creatinine clearance (ml/min) |
GSH (µmol/mg Protein) |
TBARS (nM/mg Protein) |
|
Normal Control |
11±2.0 |
3.6±0.7 |
0.86±0.5 |
11.23 ± 0.023 |
146.36 ± 0.336 |
|
Cisplatin |
7.0±1.2# |
1.7±0.7# |
0.30±0.6# |
6.45 ± 0.004# |
386.09 ± 0.064# |
|
MZ (200mg/kg) |
8.9±1.6* |
2.6±0.8* |
0.75±0.3* |
7.39 ± 0.003* |
284.46 ± 0.500* |
|
MZ (400mg/kg) |
9.4±1.2** |
3.1±0.3** |
0.82±0.2** |
7.80 ± 0.002** |
236.81 ± 0.426** |
Table 2: Effect of Aqueous Extract of Manilkara Zapota on Body Weight in Cisplatin Induced Renal Damage
Values are expressed as Mean±SEM of six animals. Statistical significance test for comparisons was done by one-way ANOVA, followed by “Dunnett’s multiple comparison test.” Comparisons were done between Cisplatin group compared with control (P#<0.001), MZ extract (200 and 400 mg/kg.b.w) in comparison with Cisplatin group (P*<0.01 and P**<0.001).
|
Groups |
Urea |
Urine volume (ml) |
Creatinine (mg/dl) |
Blood urea nitrogen |
|
Normal Control |
38.45±0.5 |
5.5 ± 0.5 |
0.8±0.6 |
18.0±0.4 |
|
Cisplatin |
335±1.0# |
15.5 ± 0.5# |
1.7±0.5# |
41.7±0.3# |
|
MZ (200mg/kg) |
160.0 ± 18.05* |
9.0 ± 1.0* |
1.1±0.3* |
21.7±0.2* |
|
MZ (400mg/kg) |
53.14 ± 14.04** |
6.0 ± 0.7** |
0.9±0.4** |
18.6±0.2** |
Table 3: Effect of Aqueous Extract of Manilkara Zapota in Cisplatin Induced Renal Damage
Values are expressed as Mean±SEM of six animals. Statistical significance test for comparisons was done by one-way ANOVA, followed by “Dunnett’s multiple comparison test.” Comparisons were done between Cisplatin group compared with control (P#<0.001), MZ extract (200 and 400 mg/kg.b.w) in comparison with Cisplatin group (P*<0.01 and P**<0.001).
Oxidative-Stress Parameters
Cisplatin-challenged rats exhibited significantly (P# < 0.001) higher levels of tissue MDA (a lipid peroxidation marker) and reduction in both tissue and serum antioxidants such as GSH, SOD and Catalase respectively as compared to normal rats. Pre¬treatment with both the doses of MZ extract showed significantly (P** < 0.001; P* < 0.01) altered levels of enzymatic and non-enzymatic endogenous antioxidant levels compared to cisplatin-control group.
Histopathological Studies
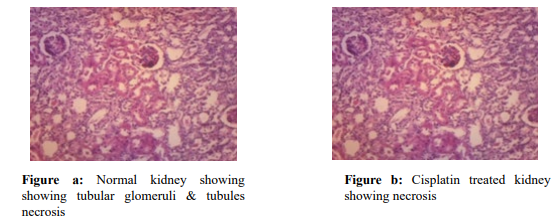
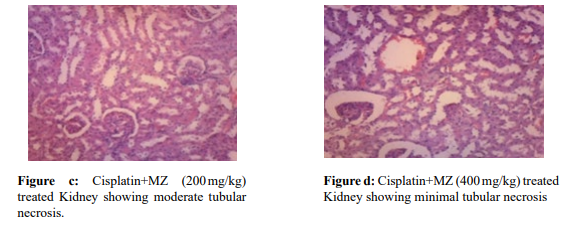
Histopathological slides of the normal group, Cisplatin alone group, and two used dosages of CP and MZ are presented in Fig 1. The kidney tissue of normal rats showed no visible signs of degeneration or necrosis and was previously confirmed from biochemical and antioxidant results (Fig. a). Extensive kidney tissue degeneration along with tubular necrosis was noted in the CP alone-treated rats (Fig. b). The rats which received 200 mg/ kg optimal Hydroalcoholic extract of Manikara zapota showed a tubular pattern through reasonable necrosis and degranulation (Fig. c), whereas the rats which received 400 mg/kg Hydroalcoholic extract of Manikara zapota exhibited very minimal swelling, necrosis, and cellular desquamation (Fig. d).
Discussion
Cisplatin is the first-line drug in platinum-based chemotherapy for the treatment of various cancers [9]. Cisplatin exposure is usually associated with nephrotoxicity, caused by activation of stress, inflammation and apoptosis via generation of reactive oxygen species (ROS). Therefore, strategies to target multiple pathophysiological processes, including the suppression of oxidative stress, inflammation and renal cryoprotection to prevent cisplatin-induced nephrotoxicity. Based on traditional medicine uses , chemical compositions, antioxidant and anti-inflammatory properties of Manikara zapota, form the basis for the present study conducted to examine the protective effects of MZ against cisplatin (CP)-induced toxicity and kidney injury in Rats. Favorably, the study reveals that MZ extract has no cytotoxic effect. However, CP-treated rats depicted typical clinical and pathological symptoms such as decreased relative kidney weight, altered kidney function parameters such as creatinine, urea, urine volume, total protein and albumin [10]. Further, increased levels of creatinine, urea, BUN and urine volume in animals treated with cisplatin indicated a reduction in glomerular filtration rate. Our results are similar to previous findings on cisplatin induced nephrotoxicity [11]. Treatment with MZ extract resulted in marked amelioration of these altered parameters. Thus, the models of CP-induced kidney injury in Rat were successfully established and the results confirm the nephroprotective activity of MZ extract.
Previous reports suggest that cisplatin induced nephrotoxicity is by initiation of lipid peroxidation and depletion of cellular thiols [12].
Cisplatin inhibits the activity of antioxidant enzymes (Glutathione and lipid peroxidase) in rat kidneys suggesting that cisplatin cytotoxicity results from the generation of reactive oxygen species (ROS). The results obtained in this study correlate with previous reports that lipid peroxidation contributes to cisplatin induced nephrotoxicity [13].
In the present study, Cisplatin administration resulted in severe nephropathy, with impaired histologic features of the kidneys and loss of body weight. Histological examination by H&E staining showed cisplatin-induced lesions and damage as evident by moderate to severe hydropic degeneration and necrosis of both proximal and distal renal tubules. Severe interstitial congestion and edema in the cortical and medullar areas have also been noted. Evaluations of animals in the MZ extract (400 mg/kg) treated group indicate that the extract combats deleterious effects of Cisplatin and kidney histology revealed near to normal histological architecture. Histological findings agree with those of biochemical data and are consistent with previous studies.
Conclusion
To conclude, our studies have shown that the root of plant Manikara zapota possesses marked nephroprotective activity with minimal toxicity and could have a promising role in the treatment of acute renal injury induced by nephrotoxins, especially cisplatin. Further work envisages evaluating its nephroprotective activity in chronic renal failure models.References
- Abdellatief, S. A., Galal, A. A., Farouk, S. M., & Abdel-Daim, M. M. (2017). Ameliorative effect of parsley oil on cisplatin-induced hepato-cardiotoxicity: a biochemical, histopathological, and immunohistochemical study. Biomedicine & Pharmacotherapy, 86, 482-491.
- Chtourou, Y., Aouey, B., Aroui, S., Kebieche, M., & Fetoui,H. (2016). Anti-apoptotic and anti-inflammatory effects of naringin on cisplatin-induced renal injury in the rat. Chemico-Biological Interactions, 243, 1-9.
- Mahgoub, E., Kumaraswamy, S. M., Kader, K. H., Venkataraman, B., Ojha, S., Adeghate, E., & Rajesh, M. (2017). Genipin attenuates cisplatin-induced nephrotoxicity by counteracting oxidative stress, inflammation, and apoptosis. Biomedicine & Pharmacotherapy, 93, 1083-1097.
- Abd El Motteleb, D. M., & Elshazly, S. M. (2013). Renoprotective effect of sitagliptin against hypertensive nephropathy induced by chronic administration of L-NAME in rats: role of GLP-1 and GLP-1 receptor. European journal of pharmacology, 720(1-3), 158-165.
- Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical biochemistry, 72(1-2), 248-254.
- Hemsl, J. H. (1963). About The family sapotacae. Kew Bull, 17, 171-172.
- Lorke, D. (1989). A new approach to practical acute toxicity testing. Archives of toxicology, 54, 275-287.
- Singh, M. P., Chauhan, A. K., & Kang, S. C. (2018). Morin hydrate ameliorates cisplatin-induced ER stress, inflammation and autophagy in HEK-293 cells and mice kidney via PARP-1 regulation. International immunopharmacology, 56, 156-167.
- Kumar, M., Dahiya, V., Kasala, E. R., Bodduluru, L. N., & Lahkar, M. (2017). The renoprotective activity of hesperetin in cisplatin induced nephrotoxicity in rats: Molecular and biochemical evidence. Biomedicine & Pharmacotherapy, 89, 1207-1215.
- Jamshidzadeh, A., Abdoli, N., Niknahad, H., Azarpira, N., Mardani, E., Mousavi, S., ... & Heidari, R. (2017). Taurine alleviates brain tissue markers of oxidative stress in a rat model of hepatic encephalopathy. Trends in pharmaceutical sciences, 3(3), 181-192.
- Olschwang, D., Bassene, E., & Colonna, J. P. (1991). Tradition africaine et analyse scientifique: l'utilisation du kinkéliba (Combretum micranthum G. Don) en Afrique de l'Ouest. Epistème, 2, 74-82.
- Zhen, F., Tian, J., & Li, L. S. (1992). Mechanisms and therapeutic effect of Cordyceps sinensis (CS) on aminoglycoside induced acute renal failure (ARF) in rats. Zhongguo Zhong xi yi jie he za zhi Zhongguo Zhongxiyi Jiehe Zazhi= Chinese Journal of Integrated Traditional and Western Medicine, 12(5), 288-91.
- Sadzuka, Y., Shoji, T., & Takino, Y. (1992). Effect of cisplatin on the activities of enzymes which protect against lipid peroxidation. Biochemical pharmacology, 43(8), 1872-1875.


