Research Article - (2025) Volume 4, Issue 1
Mitigation of Quick Decline Syndrome in Ancient and Monumental Olive Trees of Ostuni (Apulia, Italy) Positive to Xylella fastidiosa and other Phytopathogens
2Chemist, Multidisciplinary Scientific Committee, Italy
3Agronomist, Multidisciplinary Scientific Committee, Italy
4Plant Pathologist, Multidisciplinary Scientific Committee, Italy
5Soil Biologist, Multidisciplinary Scientific Committee, Italy
6Chemist and Environmental Engineer, Multidisciplinary Scientific Committee, Italy
7Soil/Plant Microbiologist, Institute of Crop Sciences, School of Advanced Studies Sant’Anna – Univer, Italy
Received Date: Apr 01, 2025 / Accepted Date: May 05, 2025 / Published Date: May 14, 2025
Copyright: ©Â©2025 Giorgio Doveri, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Nuti, M., Doveri, G., Pergolese, G., Sardella, E., Scortichini, M., et al. (2025). Mitigation of Quick Decline Syndrome in Ancient and Monumental Olive Trees of Ostuni (Apulia, Italy) Positive to Xylella fastidiosa and other Phytopathogens. World J Forest Res, 4(1), 01-09.
Abstract
The Olive Quick Decline Syndrome (OQDS) in Apulia region, south of Italy, has affected over the last decade more than 6.5 million of olive trees. The syndemic nature of this syndrome, which includes the phytopathogenic bacterium Xylella fastidiosa (Xf), has caused great difficulties in the control of OQDS, and both the insect vector control of Xf, and eradication of olive trees have proven to be of limited efficacy. In this on-farm case study a co-existence and mitigation approach is described, applied to thirty-eight monumental, or having the traits of monumentality, olive trees of the municipality of Ostuni. They were PCR-positive to Xf in 2021, showing random symptoms of desiccation, and the symptoms of generalized OQDS. The trees were affected by different bacterial and fungal pathogens, nematodes, and pests. These same trees, treated during three consecutive years with an agronomic protocol (Good Agronomic Practices including microbial biostimulants), in 2025 are alive, productive, still PCR-positive to Xf except one, and having milder symptoms associated to the syndrome. The results suggest that mitigation measures, and co-existence with Xf, might represent an alternative to the generalized eradication of olive trees affected by OQDS.
Keywords
Olive Quick Decline Syndrome, Olive Desiccation, Olive Syndemic Outbreak, Xylella fastidiosa, Mitigation
Introduction
The Olive Quick Decline Syndrome (OQDS) in Apulia region, south of Italy, has affected over the last decade millions of trees. Although a precise estimate to date of the number of dead trees is lacking, the data obtained with the analysis of satellite images, indicate approximately 6.5 millions diseased trees at the end of 2017, with a rapidly growing trend [1,2]. Although initially the spread of the disease was called OQDS, contrasting views were issued to explain the syndrome, one view attributing the death of such an important number of trees solely to the causal agent Xylella fastidiosa (Regione Puglia, 2024 http://www.emergenzaxylella.it/portal/portale_gestione_agricoltura), another view being that OQDS in Apulia could be identified as a syndemic outbreak, due to concurrent pathological events of biological, agronomic, and societal nature, requiring the development and application of risk mitigation measures [3-7]. Initially the contrast approaches were developed, targeting mainly at one of the causes of OQDS, namely the phytopathogenic bacterium Xylella fastidiosa subsp. pauca (Xfp) [8,9]. These treatments were successfully applied to 20-years-old (cv. Ogliarola salentina) and to 60-75 years old olive trees (cv. Ogliarola salentina and Cellina di Nardò), respectively. However, it was progressively recognized that the eradication of the bacterium is impossible and that attempts to eradicate the Xfp insect vector are of limited efficacy [10,11]. Moreover, in 2021-2022 Xfp was detected only in 3.21% of the sampled plants affected by the OQDS [12]. Consequently, the pathogenic bacterium was moved from the A1 to the A2 EPPO list due to its establishment in southern Europe [13]. Phytopathogens other than Xfp are found in desiccating olive trees, namely the agents of cercosporiosis, of leprosy Phlyctema vagabunda Desm., of olive leaf spot, of root and crown rot, root nematodes (https://ipm.ucanr. edu/agriculture/olive/nematodes/#gsc.tab=0), of olive tree scab due to Pseudomonas savastanoi pv. savastanoi, of verticilliosis [14-20]. Recently Neofusicoccum mediterraneum has been described as associated to the OQDS [21]. Overall considered, mitigation and co-existence with Xfp are retained as possibly the only effective strategies to control OQDS, along with the control of the other phytopathogens, within a holistic approach [12]. Here we describe an agronomic approach, applied to thirty-eight olive trees, monumental or having the monumentality traits, of the Municipality of Ostuni, aiming at (a) developing a sustainable mitigation strategy of OQDS to olive trees PCR-positive to Xylella fastidiosa, and (b) verifying at field level whether the application of the protocol allows to avoid the death or mandatory eradication measures of these trees of historical and naturalistic interest.
Materials and Methods
Study Location Area
Ostuni, 218 m a.s.l. and 10 Km from the Adriatic Sea, is one of the Municipalities of the Province of Brindisi (Apulia). Its surface is 22,417 Ha including 7,037 Ha cultivated with olive trees, several of them being centenary or millenary in protected areas. The soil of the Municipality is of reduced depth, with a calcareous crust on the surface, remarkably stony, and has been subjected to anthropic pressure since the last 50 years; it has high to very high salinity (750 wells are present in the territory), with progressive salinization of the aquifer [5]. The soil erosion risk assessment, according to PESERA model (https://www.fao.org/land-water/land/land- governance/land-resources-planning-toolbox/category/details/ en/c/1111230/), is 1-3 t/Ha/year. The residents in the Municipality at the date of January 31st, 2024 were 29,877 but thousands visitors during the year make it a very densely populated recreational area.
Olive Trees and Their Environment
Table 1 reports the geographic coordinates of each of the olive trees, monumental or having traits of monumentality (cv. Cellina di Nardò, Ogliarola, Toscanina, and Cima di Melfi Figure 1).
The date of first sampling was October 21st, 2021, the latter is reported in for all of them. The date of second sampling was October 21st, 2024, for all of them. The study ended in January 2025. The studied trees are within olive orchards, randomly spaced (Figure 2). Soil physicochemical characteristics of the orchards are: pH 7.8, EC 123 mS/cm-1, organic carbon 0.8% w/w, organic matter 1.5% w/w. The trees were rainfed, and no irrigation was carried out. The main climatic conditions, i.e. temperature and rainfall 2021-2024 are reported in Figure 3.
|
Olive tree ID |
Latitude |
Longitude |
Land Reg. sheet |
Land Reg. parcel |
|
1257489 |
40,72608697 |
17,50561063 |
134 |
215 |
|
1257485 |
40,72607236 |
17,50577193 |
134 |
215 |
|
1257442 |
40,72619162 |
17,5059434 |
134 |
215 |
|
1257471 |
40,72613572 |
1750588506 |
134 |
215 |
|
1249521 |
40,72652684 |
17,50593536 |
134 |
215 |
|
1249661 |
40,72682175 |
17,50591278 |
134 |
215 |
|
1251618 |
40,72691169 |
17,50641279 |
134 |
166 |
|
1251624 |
40,72687866 |
17,50642329 |
134 |
166 |
|
1251584 |
40,72683671 |
17,50648521 |
134 |
166 |
|
1251658 |
40,72678623 |
17,50656495 |
134 |
166 |
|
1253656 |
40,72685238 |
17,50672247 |
134 |
24 |
|
1251636 |
40,72678482 |
17,50649195 |
134 |
166 |
|
1251709 |
40,72672266 |
17,50657542 |
134 |
166 |
|
1251779 |
40,7267086 |
17,50645007 |
134 |
166 |
|
1251745 |
40,72667031 |
17,50657171 |
134 |
166 |
|
1251762 |
40,72665812 |
17,50651225 |
134 |
166 |
|
1251873 |
40,72665778 |
17,50645229 |
134 |
166 |
|
1253396 |
40,72660425 |
17,50650533 |
134 |
166 |
|
1253398 |
40,72659976 |
17,50657728 |
134 |
166 |
|
1252016 |
40,72654669 |
17,50651624 |
134 |
166 |
|
1253407 |
40,72655699 |
17,50658445 |
134 |
166 |
|
1251970 |
40,72649523 |
17,50647868 |
134 |
166 |
|
1253409 |
40,72649957 |
17,50658892 |
134 |
166 |
|
1253419 |
40,7265168 |
17,50672691 |
134 |
24 |
|
1253827 |
40,72637198 |
17,50667626 |
134 |
167 |
|
1253913 |
40,7263713 |
17,50686 |
134 |
25 |
|
- |
40,726492 |
17,506554 |
134 |
167 |
|
1255513 |
40,72638439 |
17,50643447 |
134 |
167 |
|
1255567 |
40,72635841 |
17,50648618 |
134 |
167 |
|
1255636 |
40,726236 |
17,50670426 |
134 |
167 |
|
1255704 |
40,72618711 |
17,50659071 |
134 |
167 |
|
1255798 |
40,72614337 |
17,5064912 |
134 |
167 |
|
1255820 |
40,72606366 |
17,50655314 |
134 |
167 |
|
1254068 |
40,72626984 |
17,50728737 |
134 |
241 |
|
1259408 |
40,72626474 |
17,50728737 |
134 |
241 |
|
1254096 |
40,72637354 |
17,50725699 |
134 |
241 |
|
1259380 |
40,72625701 |
17,50712022 |
134 |
241 |
|
1223144 |
40,7376708 |
17,51586594 |
83 |
51 |
|
1233189 |
40,73771999 |
17,51613204 |
83 |
148 |
Table 1: Geographic coordinates of the 39 olive trees under study (ID = identifier)

Figure 1: One monumental olive tree of this study (see Table 1 for coordinates of ID 1233189; see also the aerial map reported in Figure 2b, the monumental olive tree is on the right). The monumental olive tree before (left), and after (right) the mandatory pollarding, and the application of GAP, including the treatment with microbial biostimulants. The symptom severity (desiccation scale 0-4) was 0 in 2022 and remains 0 in 2025.

Figure 2: Aerial map: the olive orchards (a) and (b) are distant ca.1 km from each other; the trees of this study are the ones marked with the white placeholder icons
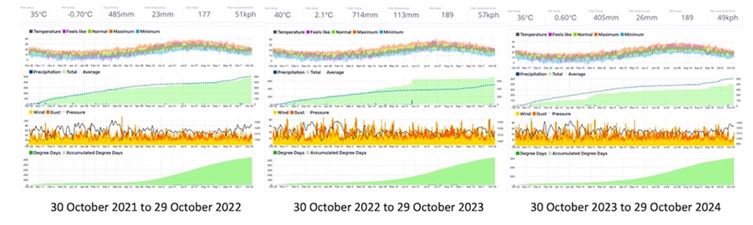
Figure 3: Climatic data throughout the study period. The upper line represents the maximum temperature, the total precipitation, the maximum daily precipitation, the rain days, and the maximum sustained wind, respectively, from October 30th, 2021 to October 29th, 2024
Agronomic Management
The studied trees were managed according to the cultivation techniques of the area, i.e. no regular pruning, no soil fertilization, no control of main pests and diseases, no herbicide treatment for most of them. According to the Regional Authorities provisions at the beginning of 2022, the trees were pollarded and grafted with the aim of minimizing the risk of diffusion of Xf. The tree 1255513, positive to Xf in 2021, within the same orchard and left without the treatment described in this case report during the entire period, was considered as a control. The trees were rainfed, and no irrigation was carried out. The first agronomic treatment was carried out in April, 2022 by spraying 150 L/ha (per 100 L water: 500 ml industrial bleach, 400 g wettable sulphur). Five days after the treatment, both the upper canopy and the tree base were sprayed with a biostimulant microbial consortium (containing Bacillus sp., Pseudomonas sp., and Trichoderma sp.) purchased from BEA, Galazzano, Republic of San Marino. A week after the olive trees had been pollarded and grafted between the end of May and June 2022, according to the provisions established by the Regional Administrative Court of Bari, a second treatment was carried out with 150 L/ha of a solution/suspension (per 100 L water: 100 g citric acid, 300 g Ergofito shark, 200 g Nemacontrol, 300 g technical urea, 200 g calcium nitrate, 100 g Ergofito boron) [22]. A third treatment was carried out in June 2023 with a solution/suspension [per 100 L water: 100 g citric acid, 250 g of ternary fertilizer 30-10-10, 300 g Ergofert start plus Bio, 200 g Nemacontrol 100 g Ergofito (Fe-Mg-Cu-Mo), 100 g Ergofito (Zn- Mn-Mg-Mo)]. The products for the second and third treatment were purchased from BEA, Galazzano, Republic of San Marino. In March 2024 the olive trees were sprayed with a solution of lime and wettable sulphur, 4-10 L per tree, followed by foliar zeolite suspension (350-400 L/ha). In June 2024 the pollards and cuts were disinfected where needed, and the tree were cleaned from occasional dry twigs. The severity of the desiccation symptom is expressed according to the scale 0-4 proposed by Scholten et al. [1].
Sampling and Analysis of Xfp by qPCR
The sampling was carried out in agreement with the European Union’s guidelines [23]. After visual inspection, sampling was made on branches at least one year old and according to the cardinal points plus between cardinal points. The twigs were mainly the symptomatic ones, 6-8 for each tree, cut by using a telescopic lopper. The latter was disinfected with hypochlorite at each sampling. Each twig was placed in a sterile plastic bag, numbered according to the tree identifier, and brought to the laboratory in a refrigerated portable container within 3 hours. The twigs from each of the plants reported in Table 1 were collected on October 21st, 2021, and October 21st, 2024, respectively. The presence of Xfp was determined by Polymerase Chain Reaction (PCR) confirmed by quantitative PCR (qPCR). Samples were finely chopped and then sonicated for 1 min, then incubated 15 min at room temperature before DNA extraction following the procedure described by Dupas et al. [24]. The occurrence of phytopathogenic fungi and nematodes was monitored on all the studied trees.
Results
Visual Inspection
The grafts obligatorily made at the beginning of 2022, according to the provisions of the Regional Authorities, were unsuccessful since 2023. In spring 2023 the new twigs and old branches emerging from the pollarded trees were vegetated. At the beginning of this case study in 2022 some olive trees showed random symptoms of desiccation (Figure 4a).The symptoms of generalized OQDS were visually present in all trees: scattered leaf scorching and twig death throughout the upper part of the canopy, or occasionally along with signs of olive tree scab (caused by Pseudomonas savastanoi; Figure 4b), caries (caused by Fomitiporia punctata; Figure 4c), root rot (caused by pathogenic oomycetes; Figure 4d), distorted roots (caused by nematodes; Figure 4e), internal galleries in the branches (caused by Zeuzera pyrina, the common yellow woodworm; Figure 4f), peacok eye (caused by Spilocaea oleaginea; Figure 4g), sooty mold and cochineal (Figure 4h). At the end of the study case in 2025, all the trees showed symptoms of agronomically not relevant desiccation (scale 0-2; usually the desiccated twigs are removed by pruning) except the trees ID 1249521, ID 1255567, and the untreated tree 1255513, which were at scale 3-4 (Figure 5).

Figure 4: The OQDS symptoms were exhibited by the olive trees, besides being positive to Xfp, at the start of the study in 2022. Left to right (upper part): 4a – random symptoms of desiccation; 4b - olive tree scab; 4c - caries; 4d – root rot; left to right (lower part): 4e – distorted roots; 4f – internal galleries; 4g – peacock eye; 4h - sooty mold and cochineal

Figure 5: Olive trees showing different levels of desiccation on the canopy (from left to right: ID 1259408 level 0, ID 1249661 level 1, ID 1253398 level 2, ID 1249521 level 3, and the untreated control tree ID 1255513 level 4) at the beginning of 2025. The frequency, i.e. the number of trees affected, of symptoms of desiccation is reported in Table 2
OQDS Monitoring and PCR Test
At the beginning of the case study, all the trees were PCR-positive to Xf. At the end of the case study they were still qRT-PCR-positive to Xf subsp. pauca, except one (ID 1223144, cv. Cima di Melfi).
Mitigation
After 3 years of agronomic management focusing on mitigation measures, all treated trees rebuilt their foliage and new vegetation, produced drupes, and were without or strongly reduced desiccation symptoms. They were visually healthy, but with signs of scattered OQDS symptoms. The level 0 desiccation affected 24 trees, level 1 affected 9 trees, level 2 affected 3 trees, level 3 affected 1 tree, and level 4 affected 2 trees. Table 2 enlists the symptoms for each individual tree affected to date by OQDS including desiccation symptoms, along with the productivity of drupes. The latter were in the majority within the range of 5-15 kg/tree. Figure 6 summarizes the symptomatology present on January 2025.
|
ID number |
Desiccation severity (level 0 to 4) |
Tree scab % |
Caries |
Root rot |
Aspecific nematodes |
Internal galleries |
Production of drupes (Kg / Tree) |
Additional symptoms/ Notes |
|
1257489 |
1 |
10 - 30 |
Extended |
Present |
Present |
0 |
B |
|
|
1257485 |
2 |
10 - 30 |
Extended |
Present |
0 |
0 |
B |
|
|
1257442 |
1 |
31 - 50 |
Extended |
Present |
Present |
Present |
B |
Wood-eating insect, presumed Armillaria |
|
1257471 |
1 |
10 - 30 |
Extended |
Present |
Present |
0 |
B |
Wood-eating insect |
|
1249521 |
3 |
10 - 30 |
Extended |
Extended |
Present |
Present |
B |
Phleotrib insect Mechanical damages on the roots |
|
1249661 |
1 |
31 - 50 |
Extended |
Extended |
Present |
Present |
B |
Sooty mold |
|
Peacock eye |
|
|
|
|
|
|
|
|
|
1251618 |
0 |
31 - 50 |
Extended |
Present |
Present |
Present |
B |
|
|
1251624 |
0 |
10 - 30 |
Extended |
Present |
Present |
Present |
B |
Cottony mealbug / leaf stress by heat |
|
1251584 |
0 |
10 -30 |
Extended |
Extended |
Present |
0 |
B |
|
|
1251658 |
0 |
10 - 30 |
Extended |
Present |
Present |
0 |
B |
|
|
1253656 |
0 |
< 10 |
Absent |
Present |
Present |
0 |
B |
|
|
1251636 |
0 |
0 |
Absent |
Present |
Present |
0 |
B |
|
|
1251709 |
1 |
10 - 30 |
Limited |
Present |
Present |
0 |
B |
|
|
1251779 |
0 |
10 - 30 |
Limited |
Extended |
Present |
0 |
B |
|
|
1251745 |
1 |
10 - 30 |
Limited |
Present |
Present |
0 |
B |
Sooty mold |
|
1251762 |
0 |
10 - 30 |
Extended |
Extended |
Present |
0 |
B |
|
|
1251873 |
0 |
10 - 30 |
Extended |
Present |
Present |
0 |
B |
|
|
1253396 |
0 |
10 - 30 |
Extended |
Present |
Present |
Present |
B |
Sooty mold |
|
1253398 |
2 |
10 - 30 |
Extended |
Extended |
Present |
0 |
B |
|
|
1252016 |
0 |
0 |
Extended |
Present |
Present |
Present |
B |
|
|
1253407 |
1 |
10 - 30 |
Extended |
Present |
Present |
0 |
B |
Sooty mold and cochineal |
|
1251970 |
0 |
10 - 30 |
Extended |
Extended |
Present |
0 |
B |
Sooty mold and cochineal |
|
1253409 |
1 |
< 10 |
Extended |
Present |
Present |
Present |
B |
|
|
1253419 |
0 |
0 |
Extended |
Present |
Present |
0 |
B |
Sooty mold and cochineal |
|
1253827 |
2 |
10 - 30 |
Extended |
Extended |
Extended |
Si |
B |
Sooty mold and cochineal |
|
1253913 |
1 |
0 |
Extended |
Present |
Present |
Si |
B |
Bark rot symptoms |
|
- |
|
|
|
|
|
|
|
|
|
|
0 |
> 50 |
Extended |
Present |
Present |
No |
B |
Sooty mold. Tree Xf-negative in 2021 to 2025 |
|
1255513 |
4 |
0 |
Extended |
Present |
Extended |
Extended |
A |
Sooty mold. Heavily pollarded tree. Not treated comparator tree |
|
1255567 |
4 |
0 |
Extended |
Present |
Present |
Extended |
A |
Sooty mold, heavily pollarded (>50%) tree |
|
1255636 |
0 |
< 10 |
Extended |
Present |
Present |
Present |
B |
Heavily pollarded (>50%) tree |
|
1255704 |
0 |
10 - 30 |
Extended |
Extended |
Extended |
Present |
B |
|
|
1255798 |
0 |
< 10 |
Extended |
Present |
Present |
0 |
B |
|
|
1255820 |
0 |
< 10 |
Extended |
Present |
Present |
Present |
B |
|
|
1254068 |
0 |
0 |
Extended |
Present |
Present |
Extended |
B |
|
|
1259408 |
0 |
< 10 |
Extended |
Present |
Present |
Extended |
C |
Sooty mold and heavy cochineal |
|
1254096 |
0 |
< 10 |
Extended |
Extended |
Present |
Present |
B |
Sooty mold and cochineal |
|
1259380 |
0 |
< 10 |
Extended |
Present |
Present |
Present |
C |
Sooty mold and cochineal |
|
1223144 |
0 |
< 10 |
Extended |
Present |
Present |
0 |
C |
Leprosy. Tree Xf + in 2021 and negative in 2025 |
|
1233189 |
0 |
< 10 |
Extended |
Present |
Present |
0 |
C |
Peacock eye |
Table 2: Symptoms of generalized OQDS and of desiccation of the trees at the date of january 2025. For productivity a = 0-5 kg/tree, b
= 5-15 kg/tree, c = 15-25 kg/tree

Figure 6: At the end of the monitoring period (January 2025) the trees showed some OQDS symptoms (for the description see Table 2)
Discussion
The olive trees monumental or having traits of monumentality of Ostuni offered the opportunity to confirm and expand our knowledge also considering the experimental approaches of other Authors on younger trees, because (a) these ancient trees were the rarest affected by OQDS, (b) they had never been approached to evaluate mitigation strategies, (c) they were present at the beginning of this case report in the former so-called “containment area”, i.e. the area where containment legal provisions included the removal of infected plants (Regione Puglia, https://press.regione. puglia.it/-/xylella-fastidiosa-aggiornata-la-zona-di-contenimento, accessed 29.10.2024). The olive trees of this report were pollarded at least 50% and then subjected to the agronomic treatments. In this case study a co-existence and mitigation protocol was applied, based on good agronomic practices, including the treatment with microbial biostimulants. Although some difficulties were encountered in field-testing new protocols due to stringent, and sometimes not well aligned, provisions established, Apulian Regional Authority, Regional Administrative Court of Bari, and international Authorities, the results obtained in the field for three consecutive years indicate that the protocol can be effective by mitigating the olive quick decline syndrome [11,22,25-29]. The olive trees were without or strongly reduced OQDS symptoms, resulting visually healthy, and PCR-positive to Xfp (except one, cv. Cima di Melfi, ID 1223144) which was Xfp-negative. Clearly, further investigation would be required for the trees Xfp-positive to understand whether they contain Xfp cells alive, since the PCR test only says that there is the DNA of Xfp. However, this finding confirms and extends the observation of other Aa. that OQDS symptomatology can occur also in the absence of Xfp. Up to 97% of Xfp PCR-negative vs. OQDS-positive trees was found in the areas monitored to reveal olive trees infected by Xfp. Contrary to expectations, there is a not significant effect of cultivar and interaction treatment x cultivar [12]. In our case, it was interesting to note that another olive tree (latitude 40,726492 longitude 17,506554), five meters from the tree ID 1255567 and negative to Xf, remained PCR-negative to Xf from 2021 to 2024, without being affected by OQDS. Most reports are focused on the inhibition of just one of the components of OQDS, namely the bacterium Xfp, and the conclusion by EFSA was that “based on the reviewed results, although several published experiments show some effects in reducing symptoms development, the tested control measures are not able to completely eliminate Xf from diseased plants, and that there is currently no control measure available to eliminate Xf from a diseased plant in open field conditions” [11]. Genomic analysis suggests that the pathogen arrived in southern Italy in 2008 on a coffee plant from Costa Rica [30]. It is interesting to note that previous studies on another crop, i.e. coffee, indicated that the deterioration of soil properties, damage of root-knot nematodes, and accumulation of soil fungi may exacerbate the coffee plants diseases, and that the gradual decline in rhizosphere microorganism diversity and imbalanced community structure, which enriches harmful bacteria, directly contributes to coffee diseases in long- standing continuous plantations [31]. Unfortunately measures to contrast the various components of OQDS in Apulian olive plantations (e.g. erratic agronomic management practices, salinization, pollution, erosion, decline of organic matter and biodiversity, misuse of the territory) have not been adopted on a scale larger than small field experiments. The organic carbon in Ostuni is actually 0.8%, while in the neighbouring Salento sub- region where severe desiccations have occurred, the organic carbon is 0.4%. Furthermore, to our knowledge, studies on how the olive rhizosphere and endophytic microbiome diversity and community structure can change in the presence of OQDS in Apulia are scanty. However, interest is recently growing on the relevance of olive microbiome for the olive tree biology, resilience, and health [32- 35]. A pioneering work on the olive-Xf pathosystem has recently shown that in susceptible plants there is a significant change in the associated microbiota with a drastic loss of beneficial genera [36]. Studies aiming at inhibiting in vitro the growth of one component of OQDS, i.e. Xf and the application of olive tree endophytes and species of Bacillus were recently reviewed [4,9]. The use of silver ultrananoclusters, and the management of mineral composition of host plants have also been proposed as a control strategy of the bacterium [37,38]. A protocol which promotes, supports, and restores new vegetation, flowers, fruits, and oil production of the treated olive plants affected by OQDS without losing susceptible olive plants has been recently proposed [39]. In this study the productivity (Kg of drupes per tree) was in the average of 5-15, while a productivity of 0-5 was observed in the trees where level 4 desiccation was present. In the case of olive trees affected by OQDS but Xfp-negative, (as well as in the case of OQDS-positive and Xfp-positive) other phytopathologies can be associated to the olive quick decline syndrome, including olive cercosporiosis, leprosy, olive wilt, olive leaf spot, peacock eye, root and crown rot, root nematodes, tree scab, verticilliosis. All these symptoms can lead to desiccation, and this could help understanding why Xf was not found in most desiccated trees in the surveillance area and in many desiccated trees of the infected area.
Conclusive Remarks
Our field case study on ancient olive trees affected or not hit by OQDS, but positive to Xfp, indicates that the co-existence with microbial phytopathogens including Xfp is possible without losing productivity or, even worst, without the elimination of the tree or the forced adoption of other drastic measures such as heavy pollarding and grafting, provided that careful application of good agronomic practices is made. These findings might be relevant to those farmers who prefer to adopt alternative, more sustainable risk mitigation measures, leading to the rescue of such an important tree crop in Apulia.
References
1. Scholten, R., Sanchez, L. M., Hornero, A., Navas-Cortes, J. A., Zarco-Tejada, P. J., & Beck, P. S. (2019). Monitoring the impact of Xylella on Apulia’s olive orchards using Sentinel-2 satellite data and aerial photographs. In Second European conference on Xylella fastidiosa, Ajaccio (France), October 30.
2. Hornero, A., Hernández-Clemente, R., North, P. R., Beck, P. S. A., Boscia, D., Navas-Cortes, J. A., & Zarco-Tejada, P. J. (2020). Monitoring the incidence of Xylella fastidiosa infection in olive orchards using ground-based evaluations, airborne imaging spectroscopy and Sentinel-2 time series through 3-D radiative transfer modelling. Remote Sensing of Environment, 236, 111480.
3. Dongiovanni, C., Di Carolo, M., Fumarola, G., Ciniero, A., Tauro, D., Palmisano, F., ... & Faretra, F. (2017). Evaluation of field treatments to reduce the impact of Xylella fastidiosa infections in olive trees. In European conference on Xylella 2017-Finding answers to a global problem-Book of Abstracts (p.16).
4. Del Grosso, C., Grandi, L., Lombardi, T., D’Attoma, G., Schmitt, N., De Michele, V. R., & Saponari, M. (2025). In vitro high-throughput screening of the antimicrobial activity of different compounds against Xylella fastidiosa subsp. pauca. Chemical and Biological Technologies in Agriculture, 12(1), 15.
5. Fidelibus, M. D., & Tulipano, L. (2004). Inquinamento Salino Ed Antropico Degli Acquiferi Costieri Della Murgia E Del Salento: Azioni Di Salvaguardia. Geologi e Territorio, Sup, 1, 95-104.
6. Giovannetti, G., Polo, F., Nutricato, S., Masoero, G., & Nuti, M. (2019). Efficacy of commercial symbiotic bio-fertilizer consortium for mitigating the Olive Quick Decline Syndrome (OQDS). Journal of Agronomy Research, 2(1), 1-21.
7. Nuti, M., Giovannetti, G., Scortichini, M., Pergolese, G., Saracino, M., & Doveri, G. (2021). The olive quick decline syndrome: A syndemic outbreak in the Apulia region, southern Italy. Journal of Agronomy Research, 3(3), 13-25.
8. Scortichini, M., Chen, J., De Caroli, M., Dalessandro, G., Pucci, N., Modesti, V., ... & Loreti, S. (2018). A zinc, copper and citric acid biocomplex shows promise for control of Xylella fastidiosa subsp. pauca in olive trees in Apulia region (southern Italy). Phytopathologia Mediterranea, 57(1), 48-72.
9. Bruno, G. L., & Tommasi, F. (2021). Gestione sostenibile di Xylella fastidiosa subsp. pauca su olivo: una mini review. Notiziario della Società Botanica Italiana, 5, 1-5.
10. Bruno, G. L., Cariddi, C., & Botrugno, L. (2021). Exploring a sustainable solution to control Xylella fastidiosa subsp. pauca on olive in the Salento Peninsula, Southern Italy. Crop Protection, 139, 105288.
11. EFSA Panel on Plant Health (EFSA PLH Panel), Bragard, C., Dehnenâ?Schmutz, K., Di Serio, F., Gonthier, P., Jacques, M. A., ... & Parnell, S. (2019). Effectiveness of in planta control measures for Xylella fastidiosa. Efsa Journal, 17(5), e05666.
12. Ciervo, M., & Scortichini, M. (2024). A decade of monitoring surveys for Xylella fastidiosa subsp. pauca in olive groves in Apulia (Italy) reveals a low incidence of the bacterium in the demarcated areas. Journal of Phytopathology, 172(1), e13272.
13. EPPO (European and Mediterranean Plant Protection Organization). (2020). Emergency measures, A2 Quarantine pest (Annex II B) EU Categorization XYLEFA.
14. Chliyeh, M., Touati, J., Selmaoui, K., Touhami, A. O., Filali-Maltouf, A., El Modafar, C., & Douira, A. (2014). Bibliographic inventory of the olive tree (Olea europaea L.) fungal diseases in the world. International Journal of Pure and Applied Biosciences, 2, 46-79.
15. Nigro, F., Ippolito, A., Gallone, P., Romanazzi, G., Carmignano, P., & Laccone, G. (2002). Cercosporiosis of olive in Apulia and attempts to control the disease. Acta
Horticulturae, 2, 773-776.
16. Romero, J., Raya, M. C., Roca, L. F., Agustíâ?Brisach, C., Moral, J., & Trapero, A. (2018). Phenotypic, molecular and pathogenic characterization of Phlyctema vagabunda, causal agent of olive leprosy. Plant pathology, 67(2), 277-294.
17. Buonaurio, R., Almadi, L., Famiani, F., Moretti, C., Agosteo, G. E., & Schena, L. (2023). Olive leaf spot caused by Venturia oleaginea: An updated review. Frontiers in Plant Science, 13, 1061136.
18. Cacciola, S., Faedda, R., Pane, A., & Scarito, G. (2011). Root and crown tot of olive caused dy Phytophthora spp. Olive diseases and Disorders, 2011-Transworld Research Network, 305-327.
19. Mitro, S., Hakim, T., Ameslek, O., Zanane, C., Mazigh, D., Elgoulli, M., ... & Zahir, H. (2024). Control of Olive Tuberculosis Trees with Olive Mill Wastewater: Inhibition of Pseudomonas savastanoi Adhesion. Tropical Journal of Natural Product Research, 8(6).
20. Barguigua, A., Zahir, I., Youss, S., Fikri, N., & Youss, B. (2021). Prevalence of olive tree phytopathologies of microbial origin in Fquih Ben Salah, Azilal and Beni Mellal (Morocco). Journal of Analytical Sciences and Applied Biotechnology, 3(1), 48-55.
21. Brunetti, A., Matere, A., Lumia, V., Pasciuta, V., Fusco, V., Sansone, D., ... & Pilotti, M. (2022). Neofusicoccum mediterraneum is involved in a twig and branch dieback of olive trees observed in Salento (Apulia, Italy). Pathogens, 11(1), 53.
22. TAR (Tribunale Amministrativo Regionale di Bari). (2023). Sezione III, Sentenza n. 546/2023.
23. EPPO (European and Mediterranean Plant Protection Organization). (2023). PM 7/24 (5) Xylella fastidiosa. EPPO Bulletin 53, 205–276.
24. Dupas, E., Briand, M., Jacques, M. A., & Cesbron, S. (2019). Novel tetraplex quantitative PCR assays for simultaneous detection and identification of Xylella fastidiosa subspecies in plant tissues. Frontiers in Plant Science, 10, 1732.
25. European Commission. (2020). Commission implementing regulation (EU) 2020/1201 of 14 August 2020 as regards measures to prevent the introduction into and the spread within the Union of Xylella fastidiosa (Wells et al.). Oficial Journal of the European Union.
26. European Parliament. (2024). Legislative resolution of 27 February 2024 on the draft regulation of the European Parliament and of the Council amending Protocol No 3 on the Statute of the Court of Justice of the European Union [07307/2022 – C9-0405/2022 – 2022/0906(COD)].
27. Regione Puglia. (2007). Legge n. 14 del 4 giugno “Tutela e valorizzazione del paesaggio degli ulivi monumentali della Puglia”. Bollettino Uficiale Regione Puglia 83 suppl. del 7-6-2007.
28. Regione Puglia. (2015). Delibera Giunta Regionale 16 febbraio 2015, n. 176, Approvazione del Piano Paesaggistico Territoriale della Regione Puglia (PPTR). Bollettino Uficiale Regione Puglia 40 del 23â?03â?2015 pag. 10080.
29. Regione Puglia. 2021. Legge n. 45 del 30.11.2021. Modifiche alla legge regionale 29 marzo 2017, n. 4 “Gestione della batteriosi da Xylella fastidiosa nel territorio della Regione Puglia). Bollettino Uficiale Regione Puglia. 150, 03/12/2021.
30. Sicard, A., Saponari, M., Vanhove, M., Castillo, A. I., Giampetruzzi, A., Loconsole, G., ... & Almeida, R. P. (2021). Introduction and adaptation of an emerging pathogen to olive trees in Italy. Microbial genomics, 7(12), 000735.
31. Sun, Y., Yan, L., Zhang, A., Yang, J., Zhao, Q., Lin, X., ... & Wang, X. (2024). Effects of Grafting on the Structure and Function of Coffee Rhizosphere Microbiome. Agriculture, 14(10), 1854.
32. Dias, M. C., Silva, S., Galhano, C., & Lorenzo, P. (2024). Olive tree belowground microbiota: Plant growth-promoting bacteria and fungi. Plants, 13(13), 1848.
33. Marques, M., Sierra-Garcia, I. N., Leitão, F., Martins, J., Patinha, C., Pinto, G., & Cunha, Â. (2024). Rhizosphere-xylem sap connections in the olive tree microbiome: implications for biostimulation approaches. Journal of Applied Microbiology, 135(7), lxae152.
34. Mechri, B., Tekaya, M., Guesmi, A., Hamadi, N. B., Khezami, L., Soltani, T., ... & Chehab, H. (2024). Enhancing olive tree (Olea europaea) rhizosphere dynamics: Co-inoculation effects of arbuscular mycorrhizal fungi and plant growth-promoting rhizobacteria in field experiments. Applied Soil Ecology, 202, 105596.
35. Thenappan, D. P., Thompson, D., Joshi, M., Mishra, A. K., & Joshi, V. (2024). Unraveling the spatio-temporal dynamics of soil and root-associated microbiomes in Texas olive orchards. Scientific Reports, 14(1), 18214.
36. Vergine M, Vita F, Casati P, Passera A, Ricciardi L, Pavan S, Aprile A, Sabella E, De Bellis L, Luvisi A. 2024. Characterization of the olive endophytic community in genotypes displaying a contrasting response to Xylella fastidiosa. BMC Plant Biology 24,337.
37. Orfei, B., Moretti, C., Scian, A., Paglialunga, M., Loreti, S., Tatulli, G., ... & Buonaurio, R. (2024). Combat phytopathogenic bacteria employing Argirium-SUNCs: limits and perspectives. Applied Microbiology and Biotechnology, 108(1), 357.
38. Navarrete, F., & De La Fuente, L. (2015). Zinc detoxification is required for full virulence and modification of the host leaf ionome by Xylella fastidiosa. Molecular Plant-Microbe Interactions, 28(4), 497-507.
39. Bruno, G. L. (2024). Coexistence between Xylella fastidiosa Subsp. pauca and Susceptible Olive Plants in the Salento Peninsula (Southern Italy). Agronomy, 14(9), 2119.




