Research Article - (2023) Volume 8, Issue 1
Microwave Thermal Ablation of Uterine Fibroids during Laparoscopic Surgery for Endometriosis
2Obstetric and Gynecologic Department of San Raffaele Hospital – Milano, Italy
Received Date: Feb 06, 2023 / Accepted Date: Feb 14, 2023 / Published Date: Feb 22, 2023
Copyright: ©Bardi Massimo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Bardi, M., Rosaschino, P., Pezzetti, G., Chiesa, A., Gangarossa, G., et al. (2023). Microwave thermal ablation of uterine fibroids during laparoscopic surgery for endometriosis. Int J Women's Health Care, 8(1), 15-20.
Abstract
Endometriosis is defined as the presence of endometrial glands or stroma outside the uterus, commonly diagnosed in women of childbearing age frequently with subfertility. Endometriosis is associated with the presence of uterine fibroids, which are benign tumors of the smooth muscle cells of the uterus, in 20-25% of cases. It is unclear whether fibroids interfere with fertility and pregnancy. The association of the two pathologies makes us ask the question of which is the best therapy to use. Sometimes medical therapy is not effective or has contraindications, surgery can be burdened with secondary effects that can impair fertility. In recent years, new techniques have been introduced to integrate conventional therapies, which involve the use of different forms of energy. Among these energies, an important place is occupied by microwaves (MW). MW creates an electromagnetic field that transforms kinetic energy into heat that induces cell death by coagulation necrosis. We have chosen to use MW thermal ablation to treat fibroids during laparoscopic surgery for endometriosis due to the fact that laparoscopic removal of myomas significantly lengthens the surgical time and increases possible complications.
Keywords
Uterine fibroids, microwaves thermal ablation, endometriosis, laparoscopic surgery
Background
The use of MW requires very short intervention times (4-5 min-utes) and involves minimal bleeding. We had no intra- or post-surgical complications. The technique used and the follow-up at 6 months are shown. There is an improvement in hyper-dysmen-orrhea and blood count. Fibroids shrink in volume and their echo structure tends to conform to that of the surrounding myometrium. Our experience confirms what is present in the literature. Micro-wave thermal ablation is a minimally invasive, rapid, effective, and safe technique.
Introduction
Uterine fibroids are benign tumors of the smooth muscle cells of the uterus and are the most common benign tumors in women during their reproductive years. The etiopathogenesis is multifac-torial but it is mainly estrogen and progesterone that induce its formation and growth. The most frequent symptoms range from abnormal genital bleeding (menorrhagia, metrorrhagia, hyper-dys-menorrhea, and intermenstrual spottings) and abdominal-pelvic pain [1]. Endometriosis is associated with the presence of uterine fibroids in 20-25% of cases [2]. Endometriosis is defined as the presence of endometrial glands or stroma outside the uterus and is diagnosed most commonly in women of childbearing age.
Both endometriosis and uterine fibroids impact, albeit with vary¬ing degrees of severity, the fertile capacity of women. The purpose of our work is to evaluate how to improve female fertility, as well as the specific symptoms of the two pathologies.
Endometriosis and Fertility
There is a higher prevalence of endometriosis in women with sub¬fertility (up to 50%) than in women with proven fertility (5-10%) and a reduced monthly fertility rate in women with endometriosis (2-10%) compared to fertile couples (15-20%) [3].
Several factors have been implicated as contributing to the infer¬tility of patients with endometriosis. Mechanical factors play an important role both in the initial stages of the disease (tubal oc¬clusions, peritoneal adhesions) and in the advanced stages when severe anatomical alterations significantly modify the pelvic anat¬omy [4]. Seyhan et al. reported significantly altered serum AMH levels in patients with endometriosis compared to healthy controls, indicating a lower follicular reserve [5]. Patients with stage III-IV endometriosis have fewer recoverable oocytes, fewer fertilizable oocytes, and therefore fewer still low-quality embryos, compared to the group of patients with stage endometriosis. I-II or without endometriosis [6]. The increase of the aromatase enzyme in the granulosa cells determines elevated levels of estradiol and re-sistance to progesterone, altering the eutopic endometrium with ca onsequent impediment to the implantation of the embryo [7]. Impairment of oocyte quality, which plays an important role in decreasing fertility rates in patients with endometriosis, has also been shown [8]. Finally, immune and inflammatory factors lead to the production of high levels of inflammatory cytokines, angiogen-ic factors, and growth factors which have toxic effects on sperm, oocytes, gamete transport, and implantation, increasing rates of pregnancy termination. [9].
Uterine fibroids and Fertility
There are still conflicting views on the interference fibroids can have with fertility and the course of pregnancy. Fibroids are traditionally believed to exert negative effects by de-forming the endometrial cavity, thinning the endometrium, and altering blood flow. They can also obstruct tubal hosts and the cervical canal by interfering with the transport of seminal fluid. However, their effect is more complex as it is related to a biochem-ical change in the myometrium-endometrium environment [10, 11]. Fibroids, through the production of extracellular cells of the matrix, cytokines, and growth factors, cause endometrial inflam-mation, which, with an altered local hormonal environment, can prevent embryonic implantation. Richlin demonstrated a cyclical altered regulation of plasma glycodelin levels which can compro-mise the implantation of the embryo [12]. Ikhena demonstrates that fibroids, in particular submucosis, induce decidualization by decreasing the release of cytokines and growth factors essential for embryo implantation [13].
In summary, it can be defined that submucosal myomas reduce fertility and increase spontaneous abortions, intramural myomas with a diameter greater than 4 cm, even without distortion of the cavity, can decrease fertility, with little evidence, and can increase abortion and subserosal myomas have no impact on fertility [14].
The Surgery of Uterine Fibroids
The experience of many authors underlines how the problems re-lated to fibromatous disease are important for a woman’s health but also for her fertility. The question, therefore arises as to which is the best therapeutic treatment to use. In the event that medical therapy has not been effective in resolving symptoms or has not been able to be implemented due to specific contraindications, sur-gery or alternative treatments should be sought.
The type of treatment depends on the location, size and number of uterine fibroids and whether their removal can improve symptoms. Conventional surgery includes laparotomic, hysteroscopic or lapa¬roscopic myomectomy up to vaginal, laparotomic or laparoscopic hysterectomy.
Laparoscopic myomectomy is a fairly difficult operation, which sometimes takes a long time to complete and can have major com¬plications. One of the most important complications is intraoper¬ative bleeding, mainly due to the number, location and size of the fibroid. The lack of precision in the suture, the loose knots and the inexperience of the surgeon must also be taken into account. It is important to underline that the most frequent intraoperative complications occur in the presence of adenomyosis/adenomyoma [15].
Morcellation is often necessary during laparoscopic myomectomy. This technique can involve major complications such as vascu¬lar lesions or abdominal viscera and can sometimes lead to the spread of leiomyoma fragments that can develop into “parasitic” leiomyomas [16]. In 2014, morcellation was performed on an un-diagnosed leiomyosarcoma with the dissemination of the disease in the pelvic and abdominal areas and a consequent reduction in survival [17]. As a result of this incident, the FDA discouraged the use of electric morcellators, stressing the need for a review of uter¬ine tissue extraction techniques. The FDA then indicated that the prevalence of unsuspected uterine sarcoma in patients undergoing hysterectomy or myomectomy for presumed benign leiomyoma is 1 in 352 and the prevalence of unsuspected uterine leiomyosarco¬ma is 1 in 498. More recent studies have shown that the percentage of uterine sarcomas in the literature varies from 1: 498 to 1: 1.851 on all uterine myoma operations, confirming however that morcel¬lation is a potentially harmful technique for the patient and must always be performed with the protection of an endo-bag [18, 19].
Pregnancy planner
It is also important to evaluate what problems may arise when a woman, with a previous myomectomy, faces pregnancy and child¬birth.
An increasing trend of uterine rupture after laparoscopic surgery of the uterus has been observed. Xiaoyi Wu reports ten cases of uterine rupture that occurred during the third trimester of pregnan¬cy after laparoscopic myomectomy [20].
The author identifies as the causes of the ruptures the overuse of electro hemostasis and multilayer suturing was not adequately performed (20). A recent study of Gimovsky included 34,002 women of which 367 had a prior myomectomy versus 33,635 controls. The rate of in¬traoperative transfusion in the prior myomectomy group was 1.4% vs. 0.4% (131/33,635) in the control group [21].
The prior myomectomy group had a higher incidence of post-partum transfusion rate (2.5% vs. 1.2%), uterotonic usage (5.4% vs.3.5%), bowel injury (0.5% vs. 0.0%) and cesarean hysterecto¬my (1.4% vs. 0,22%). Neonatal outcomes were not different be¬tween groups.
Uterine fibroids and thermoablation
In recent years, new therapeutic techniques have been introduced to replace or integrate conventional surgical and medical therapies of fibroids that involve the use of different forms of energy to de¬stroy, modulate or regenerate biological tissues [22].
We remember the uterine embolization, the High-intensity focused ultrasound (HIFU), the Magnetic resonance imaging guided high intensity focused ultrasound (MRgFUS) and the thermal ablation with radiofrequency and microwaves.
Microwaves are radiations that are placed in the electromagnetic spectrum between radio waves and infrared radiations, with fre-quencies between 300 MHz and 30 GHz and a wavelength between 1 cm and 1 m. The creation of an electromagnetic field induces a continuous oscillation of dipolar molecules. The oscillation causes frequent molecular collisions and the clutch transforms the kinetic energy into heat. The obtained heating induces cell-death by coag¬ulation necrosis [23].
Yang in 2014 used microwave ablation to treat fibroids and had ab¬lation rates of up to 89%. Furthermore, after 3, 6 and 12 months of treatment, fibroids decreased by an average of 61%, 78% and 93%. Anemia caused by excessive genital bleeding was also reduced or disappeared in 99% of patients [24]. A multicenter study of 311 women with uterine fibroids in China showed that fibroid volume decreases after ablation with microwave treatment, with a reduc¬tion rate of 78% after 6 months [25]. Both of these authors con¬clude that microwave ablation is effective and safe for treatment of uterine fibroids. Also in consideration of these conclusions, we decided to intervene with microwave ablation on symptomatic uterine fibroids that were found in patients who were undergoing surgery for endometriosis.
The choise of the use of MW thermoablation
The choice to use a thermos-ablative treatment is due to the fact that a myomectomy performed during surgery for endometriosis would have greatly lengthened the surgical time and there is an increase of intra-operative complications
The use of microwaves requires very short time and does not in-volve bleeding, if not minimal. No author has reported any surgi-cal or post-surgical complications due to the use of MW [24-26].
It is therefore desirable to have a careful surgical intervention that can simultaneously cure endometriosis and uterine fibroid. It is certainly important to solve the clinical problems that the two pathologies cause, such as dysmenorrhea, menorrhagia, chronic pelvic pain, etc., but it must be remembered that our surgery inter¬venes on young patients, often with reduced fertile capacity. It is therefore necessary to use minimally invasive techniques that also tend to improve the fertile capacity of the woman who is affected by both diseases.
Our experience
To support our surgical activity we use the AMICA System of HS Hospital Service Spa, which is a programmable radiofrequency and microwave generator for thermal ablation treatments.
RF&MW AMICA is structured with a simple and intuitive graphic interface with touch screen display to set the procedure parame¬ters that are automatically identified and continuously monitored during ablation. The time, the power delivered and the energy stored in the patient, the temperature and the reflected power are checked.
MicroWave AMICA has some specific characteristics of which the most interesting are the coagulation performance, adaptable through an adequate setting of the energy delivery mode, the pow¬er and duration of the treatment and the monitoring of the tempera¬ture in a deep target, by means of a probe with interstitial thermo-couple and temperature reader.
The MW AMICA probe, which we will define as “antenna”, is a disposable applicator for microwave thermal ablation and is pat¬ented by the CNR, granted exclusively by HS Hospital Service. The antenna is a medical cannula in stainless steel, has a 14 G gauge and a length of 27 cm, with endocirculation of the coolant that allows the use of high power placed in a 2 cm tip. The antenna is equipped with a mini-choke for trapping reflected microwaves and eliminating the effects of back-heating.
We classified the fibroids in relation to the indications of the FIGO classification system (PALM-COEIN) of Munro and Coll. [27]. We started by performing thermal ablation of G5-G6 fibroids to avoid causing technical damage to the endometrium. We then worked on G4 fibroids. Our next step will be to termablate the G3 fibroids as well.
The protocol for this experience admits patients with uterine my¬omas already scheduled for surgery for ovarian endometriosis. In a first moment we limited ourselves to treating fibroids with a di¬ameter of less than 5 cm, subsequently we have treated fibroids with a diameter until 7 cm, using a double simultaneous antenna or repeating the insert.
The surgical ways to treat uterine fibroids with microwaves are percutaneous, transvaginal and hysteroscopic but, by surgically intervening on endometriosis, we used the laparoscopic one. At the end of the operation for endometriosis we place a transvaginal ultrasound probe to have a guide that allows the MW Amica anten¬na to adequately reach the inside of the fibroid without getting too close to the endometrium.
Immediately prior to ablation we perform a biopsy with an 18G 25cm disposable automatic needle. We insert the microwave an¬tenna inside the fibroid, trying to get the tip closer to its distal edge. We will wait for the time that will be established with MW AMICA, based on the position and size of the fibroid (on average 4-5 minutes), partially retracting the antenna halfway through the established time and then completely extracting it (Figure: 1).
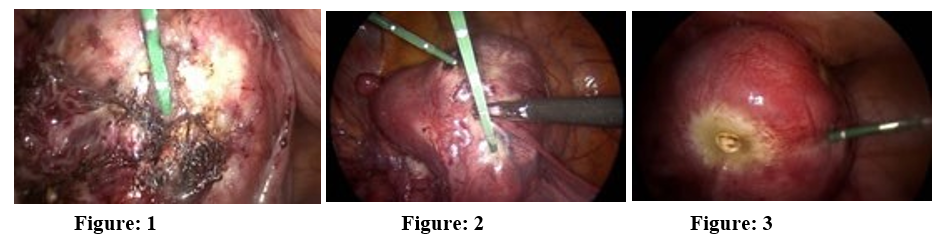
In two cases we have not respected the admission protocol and we have treated fibroids slightly larger than 5 cm in diameter. We used, at the same time, two antennas inserted through two different trocars Figure 2. In a woman who had a fibroid with a diameter of 6 cm, after performing the thermal ablation with two contempo¬rary antennas, we inserted a third antenna to obtain a more com¬plete myolysis Figure: 3.
Post-surgical morbidity did not occur in any of the operated wom¬en and all were discharged after 48 hours with indication for an ul¬trasound checkup and a blood count check 3 months after surgery. Further ultrasound checks will then be agreed at 6 and 12 months. For the time being, we have performed microwave thermal abla¬tion in 8 women with endometriosis and uterine myomas at the same time.
The first patient treated had a G5 fibroid of only 2 cm in diameter associated with peritoneal and left adnexal endometriosis with in¬fertility. This is the only case, for the moment, to which we have had an ultrasound check after one year from the operation: the fi¬broid has almost completely disappeared Figure 4,5.
Figure: 4 (fibroid after 3 months) Figure: 5 (fibroid after one year)
In the five women with myomas less than 5 cm in diameter, the ultrasound examination after six months showed a slight decrease in volume but a significant echo-structural alteration. In the two women with large fibroids the ultrasound examination was per¬formed after three months and showed an initial modification of the structure and an initial reduction in the volume of fibroids.
To evaluate the benefits of thermal ablation on symptomatology, we were able to rely only on changes in the amount of menstrual blood because dysmenorrhea and dyspareunia are symptoms that can be confused with those of endometriosis. The amount of men¬strual blood was assessed using the “menstrual pictogram” taken from the publication of Magnay [28].
In all cases treated there was a reduction in menstrual flow as early as the third month after surgery to stabilize at a normal level in the sixth month.
Hemoglobin had risen from a mean value of 106 g / L (8.2 - 12.2) to 121 g / L (10.8-12.6); hematocrit from 33% (26 - 45) to 41% (35 - 48); the Mean Corpuscular Volume from 78.3 fL (72.6 - 90.1) to 86.4 fL (80.8 - 88.3).
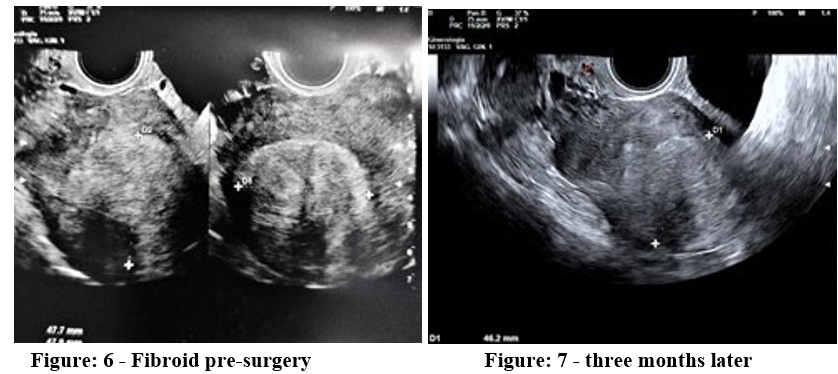
Of course, the reduction of myoma volume is important but we believe that the structural modification and the healing process are more important. In some cases we have seen the thermos-ablat-ed myoma that no longer shows its boundaries separating it from the surrounding myometrium. The ultrasound examination, a few months after the thermal ablation, shows us a myoma with ultra¬sound characteristics similar to those of the healthy myometrium Figure 6,7. This aspect leads us to think of a more suitable recov¬ery for the establishment of a future pregnancy.
Conclusions
It is necessary to solve the clinical problems that endometriosis causes and those fibroids can worsen. Laparoscopic surgery for endometriosis is performed on young women suffering from pa-thologies that undermine the fertile capacity. Endometriosis in ad¬vanced stages requires long-lasting and sometimes at least partially demolishing interventions. It is important to perform operations on associated myoma that do not further lengthen the surgical time, that does not worsen the bleeding, and that do not increase the risk of complications. Thermal ablation with microwaves is simple to perform, takes a very short time, and does not involve bleeding, if not minimal. There are no significant side effects or complications in the bibliography, and our experience confirms it.
In conclusion, we can confirm what Zhang Bing-Song in 2015 claims [25]:
“Microwave Ablation is an effective minimally invasive therapy to treat uterine fibroids, specifically aimed at those women who wish to maintain their reproductive capacity. Microwave Ablation has the additional benefit of rapid postoperative course and a signifi¬cant improvement in the quality of life of patients”.
Authors Contributions
All Authors have given substantial contributions to the conception or the design of the manuscript. All authors have participated in drafting the manuscript. The first author A revised it critically. All authors read and approved the final version of the manuscript.
Congresses
This paper was presented as free communication at the 16° SEGi National Congress. Congress that was held in Rome on 19-22 June 2022
Authors Contributions
All Authors have given substantial contributions to the conception or the design of the manuscript. All authors have participated in drafting the manuscript. The first author A revised it critically. All authors read and approved the final version of the manuscript.
Congresses
This paper was presented as free communication at the 16° SEGi National Congress. Congress that was held in Rome on 19-22 June 2022
References
- Parker, W. H. (2007). Etiology, symptomatology, and diagnosis of uterine myomas. Fertility and sterility, 87(4), 725-736.
- Maclaran, K., Agarwal, N., & Odejinmi, F. (2014). Co-ex-istence of uterine myomas and endometriosis in women undergoing laparoscopic myomectomy: risk factors and surgi-cal implications. Journal of Minimally Invasive Gynecology,21(6), 1086-1090.
- Practice Committee of the American Society for Reproductive Medicine. (2004). Endometriosis and infertility. Fertility and sterility, 82, 40-45.
- Holoch KJ, Lessey BA: Endometriosis and infertility. Clin Obstet Gynecol. (2010) 53:429–38.
- Seyhan, A., Ata, B., & Uncu, G. (2015, November). The impact of endometriosis and its treatment on ovarian reserve. In Seminars in reproductive medicine (Vol. 33, No. 06, pp. 422-428). Thieme Medical Publishers.
- Máté, G., Bernstein, L. R., & Török, A. L. (2018). Endometriosis is a cause of infertility. Does reactive oxygen damage to gametes and embryos play a key role in the pathogenesis of infertility caused by endometriosis?. Frontiers in endocrinol-ogy, 9, 725.
- Brosens, I., Brosens, J. J., & Benagiano, G. (2012). The eutopic endometrium in endometriosis: are the changes of clinical significance?. Reproductive BioMedicine Online, 24(5), 496-502.
- Simon, C., Gutiérrez, A., Vidal, A., de los Santos, M. J., Tarín,J. J., Remohí, J., & Pellicer, A. (1994). Outcome of patients with endometriosis in assisted reproduction: results from in-vitro fertilization and oocyte donation. Human reproduction, 9(4), 725-729.
- Eleftheriou, G., Butera, R., & Manzo, L. (2012). Holoprosen-cephaly in clomiphene-induced pregnancy: a possible association? A case report and literature review. Clinical and Experimental Obstetrics & Gynecology, 39(4), 535-536.
- Zepiridis, L. I., Grimbizis, G. F., & Tarlatzis, B. C. (2016). Infertility and uterine fibroids. Best Practice & Research Clinical Obstetrics & Gynaecology, 34, 66-73.
- Taylor, H. S. (2018). Fibroids: when should they be removed to improve in vitro fertilization success?. Fertility and Sterility, 109(5), 784-785.
- Richlin, S. S., Ramachandran, S., Shanti, A., Murphy, A. A., & Parthasarathy, S. (2002). Glycodelin levels in uterine flush-ings and in plasma of patients with leiomyomas and polyps: implications for implantation. Human Reproduction, 17(10),2742-2747.
- Ikhena, D. E., & Bulun, S. E. (2018). Literature review on the role of uterine fibroids in endometrial function. Reproductive Sciences, 25, 635-643.
- Tanos, V., Berry, K. E., Frist, M., Campo, R., & DeWilde,R. L. (2018). Prevention and management of complications in laparoscopic myomectomy. BioMed research international, 2018.
- Vlahos, N. F., Theodoridis, T. D., & Partsinevelos, G. A. (2017). Myomas and adenomyosis: impact on reproductive outcome. BioMed Research International, 2017.
- American Cancer Society. Survival rates for uterine sarcoma, by stage. Atlanta, GA American Cancer Society; 2014.
- Food and Drug Administration. (2014). Quantitative assessment of the prevalence of unsuspected uterine sarcoma in women undergoing treatment of uterine fibroids: summary and key findings. Silver Spring (MD): FDA, 2014.
- Ricci, S., Stone, R. L., & Fader, A. N. (2017). Uterine leiomyosarcoma: Epidemiology, contemporary treatment strategies and the impact of uterine morcellation. Gynecologic oncology, 145(1), 208-216.
- Juhasz-Böss I, Gabriel L, Bohle R et al.: Uterine Leiomyosarcoma. Oncol Res Treat 2018;41:680-686.
- Wu, X., Jiang, W., Xu, H., Ye, X., & Xu, C. (2018). Characteristics of uterine rupture after laparoscopic surgery of the uterus: clinical analysis of 10 cases and literature review. Journal of International Medical Research, 46(9), 3630-3639.
- Gimovsky, A. C., Frangieh, M., Phillips, J., Vargas, M. V., Quinlan, S., Macri, C., & Ahmadzia, H. (2020). Perinatal outcomes of women undergoing cesarean delivery after prior myomectomy. The Journal of Maternal-Fetal & NeonatalMedicine, 33(13), 2153-2158.
- Bardi M, Pezzetti G, Chiesa A et Al: Which is the best fertility-sparing treatment for uterine fibroids? Review of literature. MOJ Wom Health 2020; 9: 84-90.
- Simon, C. J., Dupuy, D. E., & Mayo-Smith, W. W. (2005). Microwave ablation: principles and applications. Radiographics, 25(suppl_1), S69-S83.
- Yang, Y., Zhang, J., Han, Z. Y., Yu, M. A., Ma, X., Zhou, H.Y., ... & Ge, H. L. (2014). Ultrasound-guided percutaneous microwave ablation for submucosal uterine fibroids. Journal of Minimally Invasive Gynecology, 21(3), 436-441.
- Liu, H., Zhang, J., Han, Z. Y., Zhang, B. S., Zhang, W., Qi, C. S., ... & Xu, R. F. (2016). Effectiveness of ultrasound-guided percutaneous microwave ablation for symptomatic uterine fibroids: a multicentre study in China. International Journal of Hyperthermia, 32(8), 876-880.
- Munro, M. G., Critchley, H. O., Fraser, I. S., FIGO Menstrual Disorders Committee, Haththotuwa, R., Kriplani, A.,... & Warner, P. (2018). The two FIGO systems for normal and abnormal uterine bleeding symptoms and classification of causes of abnormal uterine bleeding in the reproductive years: 2018 revisions. International Journal of Gynecology & Obstetrics, 143(3), 393-408.
- Magnay, J. L., O’Brien, S., Gerlinger, C., & Seitz, C. (2020). Pictorial methods to assess heavy menstrual bleeding in research and clinical practice: a systematic literature review. BMC women’s health, 20(1), 1-15.
- Bing-Song, Z., Jing, Z., Zhi-Yu, H., Chang-Tao, X., Rui-Fang, X., Xiu-Mei, L., & Hui, L. (2016). Unplanned pregnancy after ultrasound-guided percutaneous microwave ablation of uterine fibroids: A follow-up study. Scientific Reports, 6(1), 1-5.


