Research Article - (2020) Volume 2, Issue 4
Micropercutaneous Laser Lithotripsy: How to Improve Stone Free Rate
2Department of Urology, Federal Medical-Biological Agency of Russian Federation, Moscow, Russia
3Department of Urology, Saint Luca St Petersburg Public Hospital, Moscow, Russia
Received Date: Aug 22, 2020 / Accepted Date: Aug 28, 2020 / Published Date: Oct 14, 2020
Copyright: ©Â©2020 Olga A Plekhanova, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Micropercutaneous Nephrolithotomy is a novel technique with the creation of a 4.85-8F working channel. The advantage of micro-PCNL is the possibility of kidney puncture under direct endoscopic control.
Materials and Methods: 99 patients aged 47,3 ± 16.9 years were included in the study. 78 patients had isolated kidney stones (78.8%). The stone size ranged from 8 to 38 mm (13.9 ± 5.4 mm). Group A included stones smaller than 15 mm (57 patients, 57,6%) and Group B included stones larger than 15 mm (42 patients, 42,4%). A 4.85 F percutaneous sheath was used in 38 (38,4%) patients, 8 F sheath was used in 61 (61,6%) patients. A holmium laser for lithotripsy was used in 46.5% cases, the thulium fiber laser in 53.5%.
Results: The average surgery time was 39,9 ± 13.7 min (from 13 to 75 min). A stone-free rate (SFR) for patients of both groups was 88.8%. A Double-J stent was placed in 39 (39.4%) patients. In one case conversion to the mini-PCNL was done. In four cases (4%) retrograde stone manipulations were performed through ureteral access sheath. 5 (5.1%) patients required the second micro-PCNL tract. 3 (3%) patients required stenting postoperatively. In 7.1% of cases the episode of acute pyelonephritis developed. In 7 cases (7,1%), Extracorporeal Shock Wave Lithotripsy was required due to residual stones.
Conclusions: We recommend using the laser micro-PCNL for the category of patients with severe comorbidities, including coagulopathy. The micro-PCNL could be safely performed in patients with kidney stones larger than 1.5 cm.
Abbreviations Used
PCNL – Percutaneous Nephrolithotomy
Micro-PCNL - Micropercutaneous Nephrolithotomy
ESWL – Extracorporeal Shock Wave Lithotripsy
Mini-PCNL- Minipercutaneous Nephrolithotomy
Ultra-Mini-PCNL- Ultraminipercutaneous Nephrolithotomy
Introduction
Since its controversial introduction in 1976, percutaneous nephrolithotomy (PCNL) has remained one of the most prevalent methodologies of surgical intervention in patients with large kidney stones [1]. PCNL has been widely used in a myriad of urological clinics due to its minimally invasive nature, low associated complication rate and high efficiency in removing stone burden [1]. The complex yet succinct technique of PCNL is closely associated with scientific and technological progress, especially in the last decade. Several major breakthroughs in the improvement of medical instrumentation, optical systems, and alongside the invention of new methods for stone disintegration, has allowed the technique of PCNL to flourish and evolve.
For many years, the European Urological Association (EAU) has considered PCNL as the first-line method and gold standard of treatment for kidney stones greater than 2 cm in size [1]. For stones of smaller diameter, extracorporeal shock wave lithotripsy (ESWL) remains the preferred and least invasive method of treatment, as well as retrograde intrarenal surgery, including the use of a flexible ureteroscope [2]. However, in high-density stones, ESWL failure even after several extensive sessions is well described, which significantly lengthens the duration of treatment, and increases related costs and risk of complications development [3]. For example, the risk of developing a perirenal hematoma is drastically increased after consecutive ESWL sessions. A steinstrasse complication may cause renal colic, requiring repeated hospitalizations and additional procedures, such as Double-J stenting or a nephrostomy tube [4]. Retrograde intrarenal surgery in addition to the high cost of equipment and disposals, is primarily limited by the need for a constant irrigation flow. Moreover, pressurized irrigation is often required to prevent the deterioration of endoscopic visibility, which significantly increases a risk of septic complications due to an acute increase in the intrarenal pressure, especially in sheath less procedures [5]. Considering these limitations, a new, more efficient and safer treatment method for the elimination of kidney stones is urgently needed to improve and hasten patient outcomes.
The technique of PCNL involves several key steps, but the most important remains that of obtaining percutaneous access or the establishment of a working channel between the skin and collecting system to facilitate stone removal. However, as mentioned above, the reason to improve upon this procedure still remains actual, particularly with the goal to diminish adverse damage progressively decreasing the size of the working tools to create an access in addition to the use of smaller size of optical scope. Standard sheaths of size 24-30 Ch have proved to be quite effective in case of large, particularly staghorn, stones that involve multiple calculi. At the same time, large-bore access is associated with a high risk of both intra- and postoperative bleeding, the rate of which one could reach up to 18% [6]. Despite the fact that the renal puncture is usually directed along the Brodel’s line, the bleeding is most often caused by a traumatic consecutive dilatation of the tract, as well as damage to the caliceal infundibulum during placement and movements of the nephroscope [6]. In this regard, there is a need to search for some new and gentler methods for performing percutaneous access to the kidney.
Finally, the miniaturization of surgical instruments has become possible due to the accumulation of experience in performing minimally invasive interventions in both adults and children [7, 8]. Thus, mini-percutaneous nephrolithotomy (mini-PCNL) with an access size of 14-22 Ch and ultra-mini-percutaneous nephrolithotomy (ultra-mini-PCNL) of size 9-13 Ch were introduced into clinical practice. In both variations of the procedure, stone fragments can be passively flushed by means of the Bernoulli principle [9, 10].
Further technological development of percutaneous interventions has led to the appearance of micropercutaneous nephrolithotomy (micro-PCNL); the first description was made by Bader et al. in 2011 [11]. The distinct advantage to this method is the possibility of kidney puncture not only under X-ray or ultrasound control, but also under direct endoscopic control, which significantly reduces the potential risk for damage to large vessels and neighboring structures. The size of the puncture access is 4.85-8 F; significantly smaller than that of a mini-PCNL or ultra-mini-PCNL, thereby drastically limiting the patient’s exposure. Therefore, the aim of this study was to thoroughly analyze the capabilities, efficiency and safety of micro-PCNL and to determine its potential role in the combined treatment of kidney stones.
Materials and Methods
A total of 99 patients, 43 males and 56 females aged 47,3 ± 16.9 years, who had undergone micro-PCNL during the period from January 2017 to May 2019, were included in the study. All procedures were performed in the City Clinical Hospital named after D.D. Pletnev, Moscow and Public Hospital of Saint Luca, St. Petersburg. All data was gathered into a single database for analysis according to research protocols and guidelines. All patients underwent standard comprehensive clinical and laboratory examination, in addition to the clinical history-taking and physical examination. Laboratory studies included the following: complete blood count, serum biochemistry panel, urinalysis, blood coagulation test and urine culture and antimicrobial sensitivity tests. Ultrasound, X-ray and computed tomography (CT) were performed in order to assess the localization and size of stones. CT-scan allowed to more accurately determine the location of the stone and its density. Postoperatively, all patients underwent follow-up laboratory tests, an ultrasound and kidney X-ray. In addition, follow-up helical computed tomography was performed one month after surgery to assess the presence of residual fragments.
The majority of patients had isolated kidney stones (n = 78, 78.8%). In the remaining cases multiple stones were found in different localizations. 10 patients had renal pelvis and lower pole stones, 5 - renal pelvis and middle pole stones, 3 - renal pelvis and upper pole stones, 2 - upper and lower pole stones and in one case there was a combination of a lower pole stone and stone obstructing the ureteropelvic junction. Among 78 patients with isolated stones, pelvis stones predominated (n = 42, 53.8%), followed by lower pole stones (n = 27, 34,6%). Moreover, eight patients with a single middle pole stone and one patient with a single upper pole stone were included in study. In addition to localization of stone, its size and density often influenced on the duration of the procedure. The largest transverse size of the stone was measured and recorded. In case of multiple stones, a total size was summarized. Thus, the study included patients with kidney stones from 8 to 38 mm (13.9 ± 5.4 mm) in size. Stone density ranged from 350 to 1500 Hounsfield units (HU) (951.4 ± 269.8 HU). All patients were divided into two groups according to the stone size: Group A being stones smaller than 15 mm (57 patients, 57,6%) and Group B being stones larger than 15 mm (42 patients, 42,4%) (Tabl.1).
Our Technique of Micro-PCNL
All procedures were performed under general anesthesia in the operating room with C-arm. At the beginning an urethrocystoscopy was performed. For the purpose of passive flushing of stone fragments during the lithotripsy, in most patients with a stone size greater than 1.5 cm, a ureteral access sheath (UAS) with a diameter of 10/12 F was placed under x-ray control. In total, the UAS was used in 59 patients (59.6%). In the remaining cases, only a ureteral catheter or an external Mono-J stent with a diameter of 5-7 F were placed. The presence of retrograde catheters allowed for the injection of contrast into the kidney and, if necessary, to dilate the collecting system and to wash out small fragments (up to 3 mm) through the UAS. Most surgical interventions were performed in the prone position (62 patients, 62.6%). A supine position was used in other 37 cases (37.4%).
Micro-PCNL was performed using the semi-rigid optical system MICROPERC (PolyDiagnost® GmbH) (Figure 1). The micro-PCNL set includes two types of percutaneous working sheaths sized of 4.85 and 8 F. A third 10.5 F sheath is present, though is only used for ultra-mini-PCNL, therefore was not utilized during these procedures. The 4.85 F percutaneous working sheath is compatible with a standard puncture needle, which in combination with semi-rigid optics and a fixation device, constitutes the so-called “all-seeing needle”. A perfusion tube with a three-way adapter allows to use irrigation during the puncture and, if necessary, to inject contrast into the collecting system. Semi-rigid fiber-optic system has a thickness of 0.9 mm, 0 degrees’ direct view, 120 degrees viewing angle and a resolution of 10000 pixels. Kidney puncture was performed under combined X-ray, ultrasound and direct endoscopic control (Figure 2). The optical and irrigation system were directly connected to the puncture needle with the 4.85 F sheath. Considering the limited maneuverability inside of the kidney with a semi-rigid instrument with a 4.85 F sheath, puncture was done whenever possible directly on the stone (38 patients, 38,4%). The combination of endoscopic imaging with X-ray or ultrasound guidance allowed for the creation of an access tract in the optimal manner. A successful puncture was confirmed by urine flushing through the inner lumen of the needle, as well as the endoscopic visualization of the collecting system or the stone. When 8 F sheaths were used (61 patients, 61,6%), especially in the situations when endoscopic examination of the collecting system was required due to the acute angle between the calyx axis and the access tract, a super-stiff guidewire with a straight or angled tip (Lunderquist) was advanced into collecting system with subsequent tract dilation up to 8 F. After the removal of the needle, a special three-port adapter was attached to the instrument tube for connecting the irrigation tube and advancing the laser fiber and optics. Considering the need to actively maneuver via nephroscope inside the collecting system, particularly in cases involving stones larger than 1.5 cm, a rigid 8 F sheath was used instead (Figure 3). All patients with stones lager than 1.5 cm were operated with UAS and 8 F percutaneous sheath.
For stone disintegration, a 200 nm laser fiber was used in combination with: a 50 W holmium laser (Auriga XL, Boston Scientific, USA), 100 W holmium laser (Lumenis, Israel), as well as the Russian innovative thulium fiber laser FiberLase U2 (IRE-POLUS, Russia) were used. Technical characteristics of lasers are depicted in Table 2.
Thulium fiber laser is a brand-new technology in urology. The structure of Thulium fiber is fundamentally different from existing Ho:YAG, Tm:YAG, Greenlight, etc. Instead of typical YAG crystals, there are 10-30 meters of fiber inside, the last one is doped with Thulium ions and activated with diode lasers (Figure 4,5). The system does not require any water-cooling system; therefore, it can be implemented in a small cavity. The rationale for using the Tm fiber laser in lithotripsy is based on the major emission line of the Tm fiber laser (1940 nm) closely matching a strong absorption peak in water. It is hypothesized that water is a primary chromophore facilitating conversion of laser energy into mechanical and thermal energy during laser lithotripsy. The penetration depth of the laser is 0.2 mm, making all the soft tissues applications more precise. Moreover, such penetration is safe in terms of accidental tissue damaging during lithotripsy. A unique pulse shape with a possible fixed peak power is reducing retropulsion of the object and brings lithotripsy to the next level, providing availability of microdusting. As concerning the heating inside a potential cavity, it is studied, that heat level does not exceed existing technologies [12].
For the complete stone fragmentation with holmium (49 patients, 49.5%) or thulium (50 patients, 50.5%) laser, a combination of fragmentation and dusting techniques were used. For holmium laser, depending on the laser model, fragmentation was achieved with energy of 0.6-1.2 J and a frequency of 12-40 Hz, while an energy of 0.2-0.8 J and a frequency of 18-80 Hz was used for dusting. The respective setting for thulium fiber laser were 0.8–1.0 J and 6–10 Hz for fragmentation, and 0.025–0.5 J and 30–200 Hz for dusting. During the lithotripsy, the setting of pulse energy and frequency were changed in order to achieve maximum efficiency. During the procedure, small stone fragments (up to 3 mm) were spontaneously washed out through the ureteral access sheath (Figure 6). When 4.85 F percutaneous sheath was used, the purpose of lithotripsy was the complete dusting of the stone, without any need for evacuation of fragments. The unique feature of the thulium fiber laser is the minimal stone retropulsion during fragmentation. An exceedingly high efficacy of lithotripsy was achieved via the uniform distribution of peak power over the entire pulse length, which allowed for significantly increased efficiency of the laser beam and, accordingly, reduced operation time by over 30% [12]. These advantages of the thulium fiber laser make it the most optimal laser type to use in conjunction with the technique of micro-PCNL; thereby possibly expanding the potential role of micro-PCNL in the treatment of larger stone burdens.
The efficacy of fragmentation was evaluated intraoperatively by means of endoscopic inspection and X-ray control. All procedures were tubeless. In most cases, a ureteral catheter or external stent was left in the kidney. 39 (39.4%) patients required intraoperative stenting due to the large number of small residual fragments. Intra¬operative outcomes are summarized in Table 3.
Results
The effectiveness of the procedure was assessed by such criteria, as the duration of the surgery, the presence of residual fragments, the need for auxiliary procedures, associated complication rate, the duration of hospitalization, as well as the overall stone-free rate (Table 4). Considering the variability in the time required for the retrograde placement of ureteral catheters or UAS and the requirement to rotate a patient to the prone position in some cases, the duration of the procedure in the study was defined as beginning from the point of kidney puncture to the removal of the percutaneous sheath. The average duration of surgery in our group was 39,9 ± 13.7 min (from 13 to 75 min). The duration was significantly influenced by the stone size and density in each respective patient. The larger the size of the stone burden and the higher the density according to preoperative computed tomography, the more time was required for its fragmentation and inherent removal.
All patients underwent kidney X-ray and ultrasound examination on the first postoperative day in order to evaluate the effectiveness of the flushing of small fragments from the kidney. Based on these results, a decision was made to remove the ureteric catheter. In addition, the early stone-free rate was evaluated. Finally, the effectiveness of the procedure was assessed by means of a non¬contrast CT, performed one month after the surgery. An overall stone-free rate (SFR) for patients of both groups after one-session of micro-PCNL was 88.8% and it was 91,2% and 85,7%, respectively, in patients with stones sized less and more than 1.5 cm. A Double-J stent was placed in 39 (39.4%) patients, considering a large number of small residual fragments and the potential risk of the obstruction. In one case, conversion to the mini-PCNL technique was done due to optical breakage and disturbance. Due to the inaccessibility of the stone through the first percutaneous tract in four cases (4%) retrograde stone manipulations via flexible ureterorenoscope and nitinol baskets were done through UAS; 5 (5.1%) patients with multiple kidney stones required the second micro-PCNL tract to improve lithotripsy conditions.
3 (3%) patients required stenting due to renal colic caused by the migration of stone fragments into the ureter after micro-PCNL. One patient had a more severe incidence of renal colic requiring re-hospitalization; however, this incident was managed conservatively. Four patients (4%) had hematuria in the postoperative period, however, there was no marked decrease of hemoglobin or need for blood transfusion. In 7 patients (7.1%), a postoperative fever was observed, which resolved after a change of antibacterial therapy. No case of excessive bleeding was noted. In 7 cases (7,1%), ESWL was required due to residual stones one month after the PCNL treatment. In general, the minimally invasive, tubeless nature of micro-PCNL, as well as a small percentage of complications and additional interventions contributed to the early discharge of patients with subsequent and significantly rapid recovery rates; the average hospitalization time was 2.8 days. All stents were removed 1-3 weeks after surgery.
Discussion
The primary aim of modern percutaneous interventions for the management of kidney stones is achieving a stone-free status during one-session of treatment with minimal risk of concomitant complications. Progressive miniaturization of surgical instruments has led to the appearance of the least invasive technique for the percutaneous removal of kidney stones, namely micro-PCNL.
Currently, there are some publications in modern literature dedicated to micro-PCNL, with varying numbers of patients and different results. The technique of optical puncture of the collecting system using the “all-seeing needle” was first described by Bader et al. in 2011 [11]. Bader’s study included 15 patients who underwent an optical puncture of the kidney before a standard PCNL procedure. The use of the “all-seeing needle” allowed the surgeons to optimize the development of the percutaneous access tract, thereby reducing the risk of complication and limiting operative time drastically. In our study, all punctures were performed under combined ultrasound and endoscopic or X-ray and endoscopic guidance following the retrograde pyelography. A direct puncture into the stone-containing calyx in the patients with radiopaque stones allowed for the avoidance of additional instrumentation maneuvering inside the kidney, which is well documented to potentially cause a quality decline of endoscopic visibility due to hematuria. This remains a distinct advantage of the “all-seeing needle”, at the same time offering patients less postoperative complications. In most available modern literature, micro-PCNL was carried out in the prone position, however, successful micro-PCNL in the supine position has also been well documented [13]. In our study, more than one third of the interventions (37.4%) were performed in a supine position to extrapolate upon this point. According to our findings, there were no technical differences in the puncture or creating of access tract, depending on the patient position.
Upon its introduction into clinical practice, the standard belief was that micro-PCNL could not serve as an effective technique to combat large renal stones and was reserved in practice only for stones smaller than 1.5 cm [14-16]. However, accompanied by the use of 8 F percutaneous sheath, surgeons are now readily capable of performing a micro-PCNL in patients with larger stones [17]. There is some data in the current literature suggesting that during micro-PCNL performed through 8 F sheath it is possible to use ultrasonic and ballistic probes, stone baskets or grasps sized between 1.9-3 F [16, 18]. Penbegul et al. described highly successful micro-PCNL in children with the use of angiographic catheter of 6.6 F as a sheath [19]. In our study, 42,4% of patients were diagnosed with kidney stones larger than 1.5 cm based on preoperative data and imaging parameters. To achieve the maximum efficiency in these patients, a ureteral access sheath of 10/12 F was placed, permitting the passive evacuation of small fragments during lithotripsy. SFR with the use of UAS was significantly higher after the single procedure (p < 0.05). However, no statistically significant difference in stone free status was shown in cases of laser type preference and the use of the 8F percutaneous sheath (Table 5).
In the literature there are very few data concerning the use of UAS during micro-PCNL cases [20]. Nevertheless, an 8 F percutaneous sheath together with the UAS made it possible in our patients to achieve a stone-free status in 85,7% of patients with large stones (≥ 1,5 cm); a remarkable feat that lend credence to the statement that micro-PCNL may be in fact applicable for patients with larger stone burdens, contrary to previous settings.
According to the literature, micro-PCNL is generally a highly effective procedure for treatment of kidney stones. In one of their first publications, Desai et al. demonstrated stone-free rate 89% in 10 patients with an average stone size of 14.3 mm [21]. In larger studies, the effectiveness of micro-PCNL never stooped lower than 82%. The highest stone-free rates were described by Armagan (93%, n = 30), Karatag (95.7%, n = 70) and Sabnis (97.1%, n = 35) [17,22,23]. In our study, the overall stone-free rate across all patients was 88,8%, while in patients with a stone sized less than 1.5 cm, it was 91.2%. These results are not only in accordance with the published data but extrapolate upon the use of micro-PCNL for larger renal stones. Nevertheless, in the majority of current studies the average stone size does not exceed 1.5 cm, the use of micro-PCNL in this setting remained unstudied until now.
The most serious and threatening complication of PCNL is excessive bleeding, which can occur both intraoperatively and postoperatively. The number of tracts, access dilatation techniques and attempts number affect the bleeding complications rate as well as the tract size [24]. During micro-PCNL, one step dilatation of the access tract was performed, and the maximum diameter of percutaneous sheath was 8 F, therefore bleeding complications occurred extremely rarely, if not negligibly. However, intraoperative hematuria may occur more frequently, leading to a deterioration in vital endoscopic visibility. The hematuria is associated with the active movements of the instrument in the collecting system during stone detection. The direct access onto stone, especially in case of calyceal stones, prevented the development and onset of hematuria. Hatipoglu et al. described one case of severe bleeding that required blood transfusion, though this does not remain the norm [25]. According to the study of Sabnis et al., mild hematuria was observed in five patients who underwent micro-PCNL [23]. Two patients had intraoperative hematuria, which did not lead to serious complications.
The most common postoperative complication of micro-PCNL is the formation of a steinstrasse due to the migration of stone fragments into the ureter, which can lead to renal colic and acute pyelonephritis. Such situations often require additional procedures, such as Double-J stenting, nephrostomy tubes, ureteroscopy or ESWL. In order to prevent the development of these complications, Hatipoglu et al. recommended to place the stent at the end of the procedure in patients with large kidney stones (more than 2 cm), which also limits the risk of stone fragment migration [25]. In our study, only 3 patients required postoperative stenting and in seven cases ESWL was performed secondarily due to remaining stone fragments found in postoperative CT scans.
The combination of fragmentation and dusting modalities and, in particular, the use of thulium fiber laser and the placement of UAS together with 8 F percutaneous sheath with a nephroscope, significantly reduces the risk of missed large fragments of stone that would remain in the collecting system and their potential migration into the ureter. According to our study and in concordance with published data, micro-PCNL does not lead to serious complications. An overall complication rate in different studies was not more than 13-14%. In our study a total number of minor complications was 12.1%. The conversion of micro-PCNL to mini-PCNL due to intraoperative hematuria or the inability to visualize the stone are described [17, 21, 26, 27]. In our series, the conversion was performed only in one case due to the breakage of the optical system, rather than a failure of the technique.
Conclusions
Our results showed the high efficiency of laser micropercutaneous nephrolithotomy for the treatment of kidney stones of various sizes, including stones over 1.5 cm, with a low associated complications rate. Careful planning of percutaneous access with the possibility of puncture directly into the stone containing calyx greatly simplifies subsequent stone fragmentation. The high efficiency of laser micro-PCNL, which is virtually bloodless with minimal complications, allows us to recommend this method for patients with severe comorbidities, including coagulopathy. The placing of ureteral access sheath of size 10/12 F contributes to the passive flushing of fragments during lithotripsy and greatly improves SFR. The use of 8 F percutaneous working sheath is justified in cases of kidney stones larger than 1.5 cm. Thulium fiber laser FiberLase U2 provides the fragmentation of stones in a quick and effective fashion with ideal preservation of good endoscopic visibility and with the absence of stone retropulsion, hematuria and mucosal damage by laser pulses. On the other hand, several limitations still exist for this technique, including the inability to use an ultrasonic probe, restricted fragment extraction, and the necessary presence of forced irrigation, therefore micro-PCNL still requires further improvement. Overall, our study indicates that micro-PCNL may potentially play a very promising role in the management of kidney stones of various sizes, in contrast to previous statements, and therefore should be implemented more often in order to improve the issues of stone disease surgical treatment.
Table 1: A total of 99 patients, 43 males and 56 females aged 47,3 ± 16.9 years, who had undergone laser micro-PCNL during the period from January 2017 to May 2019, were included in the study.
|
Demographics |
|
|
Total Patients |
n=99 |
|
Males |
n=43 (43.4%) |
|
Female |
n=56 (56.6%) |
|
Average Age |
47,3 ± 16.9 years |
|
Stone Characterization |
|
|
Average Stone Size |
13.9 ± 5.4 mm |
|
Average Stone Density |
951,4 ± 269.8 HU |
|
Stone size ≤ 15 mm |
n=57 (57.6%) |
|
Stone size > 15 mm |
n=42 (42.4%) |
|
|
|
|
Multiple Kidney Stones |
n=21 (21,2%) |
|
|
Renal Pelvis and Lower Pole; n=10 (47.6%) |
|
|
Renal Pelvis and Middle Pole; n=5 (23.8%) |
|
|
Renal Pelvis and Upper Pole; n=3 (14.3%) |
|
|
Upper and Lower Pole; n=2 (9.5%) |
|
|
Lower Pole and UPJ; n=1 (4.8%) |
|
Isolated Kidney Stones |
n=78; (78.8%) |
|
|
Renal Pelvis; n=42 (53.8%) |
|
|
Lower Pole; n=27 (34.6%) |
|
|
Middle Pole; n=8 (10.3%) |
|
|
Upper Pole; n=1 (1.3%) |
Table 2: Technical characteristics of Ho-Yag and Tm Fiber lasers
|
Parameters |
Laser Type |
|
|
Tm Fiber |
Ho:YAG |
|
|
Wavelength, µm |
1.94 |
2.1 |
|
Power, W |
50 |
120 |
|
Min energy per pulse, J |
0.025 |
0.2 |
|
Max energy per pulse, J |
8 |
6 |
|
Max rep rate, Hz |
2000 |
80 |
Table 3: Analysis of the effectiveness of micro-PCNL in treating kidney stones. Intra-Operative Outcomes
|
Intra-Operative Outcomes |
|
|
Average Duration of Surgery |
33.9 ± 13.7 min (13-72 min) |
|
|
|
|
With UAS |
n=59 (59.6%) |
|
Without UAS |
n=40 (40.4%) |
|
|
|
|
Percutaneous sheath 4,85 F |
n=38 (38.4%) |
|
Percutaneous sheath 8 F |
n=61 (61.6%) |
|
|
|
|
Ho:YAG laser |
n=46 (46.5%) |
|
Tm Fiber laser |
n=53 (53.5%) |
|
|
|
|
Additional retrograde stone manipulations |
n=4 (4%) |
|
Second micro-PCNL tract |
n=5 (5.1%) |
|
Conversion to Mini-PCNL |
n=1 (1%) |
|
Occurrence of double J stent placement |
n=39 (39.4%) |
|
Incidence of excessive bleeding |
n=0 (0%) |
Table 4: Analysis of the effectiveness of micro-PCNL in treating kidney stones. Post-Operative Outcomes
|
Post-Operative Outcomes |
|
|
Average Duration of Hospitalization |
2.8 days |
|
Overall stone free rate |
89.1% |
|
Stenting due to renal colic |
n=3 (3%) |
|
Incidence of rehospitalization |
n=1 (1%) |
|
Incidence of hematuria |
n=4 (4%) |
|
Post-operative fever noted |
n=7 (7.1%) |
|
Follow up ESWL procedure required |
n=7 (7.1% |
Table 5: Results of Micro-PCNL depending on the stone size and the operative technique
|
Value |
SFR (pts) |
SFR (%) |
F, Fisher exact test, p < 0.05 |
Statistically significant difference |
|
Stone size, mm |
||||
|
Less than 15 mm |
52/57 |
91,2% |
0,52, OR = 1,73, ε2 = 0,74 |
insignificant |
|
More than 15 mm |
36/42 |
85,7% |
||
|
UAS |
||||
|
With UAS |
57/59 |
96,6% |
0,006, OR = 8,27, ε2= 8,81 |
significant |
|
Without UAS |
31/40 |
77,5% |
||
|
Sheath size |
||||
|
4,85 F |
31/38 |
81,6% |
0,099, OR = 0,31, ε2= 3,34 |
insignificant |
|
8 F |
57/61 |
93,4% |
||
|
Laser type |
||||
|
Ho:YAG |
39/46 |
84,8% |
0,337, OR = 0,45, ε2= 1,47 |
insignificant |
|
Tm fiber |
49/53 |
92,5% |
||
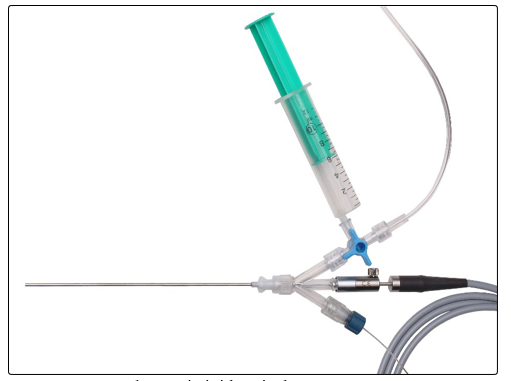
Figure 1: The semi-rigid optical system MICROPERC (PolyDiagnost® GmbH)
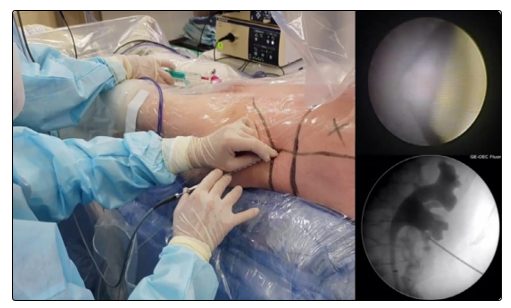
Figure 2: Kidney is punctured using “all-seeing needle” under combined X-ray and direct endoscopic control.
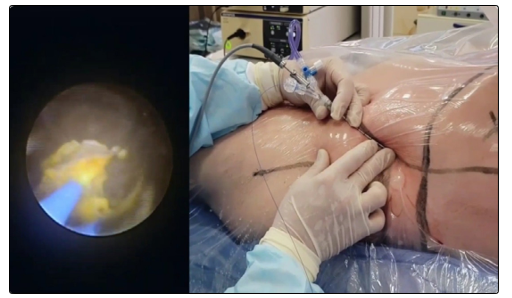
Figure 3: The use of the 8F percutaneous access sheath gives the way for active maneuver of the nephroscope inside the collecting system during lithotripsy with no damage in case of the acute angle between the calyx axis and the access tract.

Figure 4: Thulium fiber laser FiberLase U2 (IRE-POLUS, Russia)
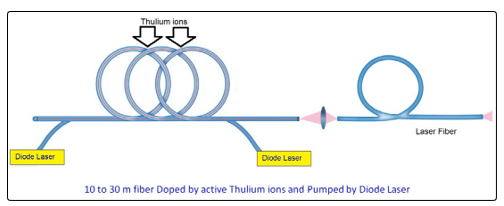
Figure 5: Structure of the Thulium Fiber Laser
Figure 6: Small stone fragments are spontaneously washed out through the ureteral access sheath during the procedure.
Research Involving Human Participants and Animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee. For this type of study, formal consent is not required. This article does not contain any studies with animals.
Informed Consent
Informed consent was obtained from all participants included in the study.
References
- C Türk, T Knoll, A Petrik, K Sarica, A Skolarikos, et al. (2015) EAU Guidelines on Urolithiasis 71.
- J Burr, H Ishii, N Simmonds, B Somani (2015) Is flexible ureterorenoscopy and laser lithotripsy the new gold standard for lower pole renal stones when compared to shock wave lithotripsy: Comparative outcomes from a University hospital over similar time period. Cent European J Urol 68: 183-186.
- El-Nahas AR, El-Assmy AM, Mansour O, Sheir KZ (2007) A prospective multivariate analysis of factors predicting stone disintegration by extracorporeal shock wave lithotripsy: the value of high-resolution noncontrast computed tomography. Eur Urol 51: 1693-1694.
- G Jung, J Jung, T Ahn, Joong Sub Lee, Sung Yong Cho, et al. (2015) Comparison of retrograde intrarenal surgery versus a single-session percutaneous nephrolithotomy for lower-pole stones with a diameter of 15 to 30 mm: A propensity score-matching study. Korean J Urol 56: 525-532.
- Doizi S, Letendre J, Bonneau C, Gil Diez de Medina S, Traxer O, et al. (2015) Comparative study of the treatment of renal stones with flexible ureterorenoscopy in normal weight, obese, and morbidly obese patients. Urology 85: 38-44.
- Michel MS, Trojan L, Rassweiler JJ (2007) Complications in percutaneous nephrolithotomy. Eur Urol 51: 899-906.
- Helal M, Black T, Lockhart J, Figueroa T (1997) The Hickman peel-away sheath: alternative for pediatric percutaneous nephrolithotomy. J Endourol 11: 171-172.
- Jackman SV, Domico SG, Cadeddu JA, Bishoff JT, Kavoussi LR, et al. (1998) The mini-perc technique: a less invasive alternative to percutaneous nephrolithotomy. World J. Urol 16: 371-374.
- Nagele U, Schilling D, Anastasiadis AG, U Walcher, K D Sievert, et al. (2008) Minimally invasive percutaneous nephrolitholapaxy (MIP). Urologe A 47: 1068-1073.
- De S, Autorino R, Kim F, Wilson R. Molina, Manoj Monga, et al. (2015) Percutaneous nephrolithotomy versus intrarenal surgery: a systematic review and meta-analysis Europ. Urology 67: 125-137.
- Bader MJ, Gratzke C, Seitz M, Sharma R, Stief CG, et al. (2011) The “all-seeing needle”: initial results of an optical puncture system confirming access in percutaneous nephrolithotomy. Eur Urol 59: 1054-1059.
- Martov AG, Ergakov DV, Guseynov MA, Andronov AS, Dutov SV, et al. (2018) First experience in clinical use of thulium lithotripsy in transurethral treatment of urolithiasis. Urologia 1: 112-121.
- Caione P, Dominicis MD, Collura G, Matarazzo E, Nappo SG, et al. (2015) Microperc for pediatric nephrolithiasis: technique in Valdivia-modified position. European journal of pediatric surgery: official journal of Austrian Association of Pediatric Surgery. Zeitschrift für Kinderchirurgie 25: 94-99.
- Preminger GM (2013) Micro-percutaneous nephrolithotomy (micro-PCNL) vs retrograde intra-renal surgery (RIRS): dealer’s choice? The devil is in the details. BJU Int 112: 280-281.
- Ganpule AP, Bhattu AS, Desai M (2015) PCNL in the twenty-first century: role of microperc, miniperc and ultraminiperc. World J Urol 33: 235-240.
- Sabnis RB, Ganesamoni R, Ganpule AP, Mishra S, Vyas J, et al. (2013) Current role of microperc in the management in the small renal calculi. Indian J Urol 29: 214-218.
- Armagan A, Tepeler A, Silay MS, Cevper Ersoz, Muzaffer Akcay, et al. (2013) Micropercutaneous nephrolithotomy in the treatment of moderate-size renal calculi. J Endourol 27: 177-181.
- Sabnis RB, Chhabra JS, Ganpule AP, Abrol S, Desai MR, et al. (2014) Current role of PCNL in pediatric urolithiasis. Curr Urol Rep 15: 1-8.
- Penbegul N, Bodakcy MN, Hatipoglu NK, Sancaktutar AA, Atar M, et al. (2013) Micro sheath for microperc: 14 gauge angoicath. J Endourol 27: 835-839.
- Popov SV, Orlov IN, Martov AG, Asfandiarov FR, Emelianenko AV (2018) Our experience with the Micro-Perc in the treatment of big stones. Experimental and Clinical Urology 4: 48-52.
- Desai MR, Sharma R, Mishra S et al. (2011) Single-step percutaneous nephrolithotomy (microperc): the initial clinical report. J Urol 186: 140-145.
- Karatag T, Tepeler A, Buldu I, Akcay M, Tosun M, et al. (2015) Is micro-percutaneous nephrolithotomy surgery technically feasible and efficient under spinal anesthesia? Urolithiasis 13: 1-6.
- Sabnis RB, Ganesamoni R, Doshi A, Ganpule AP, Jagtap J, et al. (2013) Micropercutaneous nephrolithotomy (microperc) vs retrograde intrarenal surgery for the management of small renal calculi: a randomized control trial. BJU Int112: 355-361.
- Kukreja R, Desai M, Patel S, Sharad Bapat, Mahesh Desai, et al. (2004) Factors effecting blood loss during percutaneous nephrolithotomy: prospective study. J Endourol 18: 715-722.
- Hatipoglu NK, Tepeler A, Buldu I, Atis G, Bodakci MN, et al. (2014) Initial experience of micropercutaneous nephrolithotomy in the treatment of renal calculi in 140 renal units. Urolithiasis 42: 159-164.
- Tepeler A, Armagan A, Sancaktutar AA, Mesrur Selcuk Silay, Necmettin Penbegul, et al. (2013) The role of microperc in the treatment of symptomatic lower pole renal calculi. J Endourol 27: 13-18.
- Piskin Mesut, Selcuk Guven, Mehmet Kilinc, Mehmet Arslan, Emre Goger, et al. (2012) Preliminary, favorable experience with microperc in kidney and bladder stones. J Endourol 26: 1443-1447.



