Research Article - (2022) Volume 7, Issue 1
Marital Status Independently Predicts Survival in Patients with Small Cell Lung Cancer: A SEER Database Analysis
#Equally contribution
Received Date: Feb 09, 2022 / Accepted Date: Feb 14, 2022 / Published Date: Feb 20, 2022
Copyright: ©Pei Luo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Pei Luo, Keyu Zhao, Liping Yang, Xin Yao, Chao Pan, Aijing Bao, Can Huang, Jun Guo. (2022). Marital status independently predicts survival in patients with small cell lung cancer: A SEER database analysis. Int J Women's Health Care, 7(1), 19-27.
Abstract
Purpose: This study will investigate the relationship between marital status and prognosis in small cell lung cancer patients.
Methods: Patients of SCLC was selected from the SEER database (1973-2013) and the patient sinformation. Kaplan-Meier analysis, log-rank test and Cox regression model were used for studying patient prognosis.
Result: 27069 SCLC patients eligible for inclusion were screened from the SEER database. Kaplan-meier test showed that the median OS values were 8, 7, 6 months in married, single and SDW patients, respectively.
Conclusion: This study shows that marital status is an independent prognostic factor for overall survival in SCLC patients. Married patients with small cell lung cancer have better prognosis than those who were divorced/separated, widowed and single.
Keywords
Marital status, Small cell lung cancer, SEER, Overall survival
Introduction
Lung cancer is the most common malignant tumor in the world and the number one cause of cancer death [1]. Among them, small cell lung cancer (SCLC) accounts for 15% to 20% of the total number of newly diagnosed lung cancers in the world each year [2]. Since SCLC is the least differentiated but most malignant type in lung cancer, it has a short doubling time, high blood vessel density, and genomic instability Compared to non-small cell lung cancer, the characteristics of stable and extremely distant metasta-sis have shown little improvement in SCLC detection, treatment and survival in the past 30 years [3,4]. If no measures are taken for treatment, the median survival time is generally only 2 to 4 months after comprehensive treatment, the median survival time for patients with limited period can reach 15-20 months [5,6]. The 2-year survival rate is 20% -40%, and the median survival period for patients with extensive stage can reach 7-13 months, about 5% reaches 2 years [7]. Facing the grim situation, we are urged to con¬tinuously increase our research on SCLC.
As an important factor affecting human physical and mental health and quality of life, the quality of marriage has received increasing attention [8,9]. Some studies have shown that harmonious mar¬riage can prolong the life of cancer patients [10]. About 750,000 patients with different types of tumors were selected. These tumors include colon cancer, prostate cancer, etc., it was found that before the tumor appeared to metastasize, tumor patients with a harmoni¬ous and happy marriage had a 20% reduction in the probability of dying from cancer [11]. A harmonious marriage could improve the survival rate of tumor patients. After analyzing relevant data from 127,753 patients with colon cancer, the researchers found that married patients diagnosed at earlier period had mortality rate 14% lower than that of unmarried, widowed or divorced patients [12]. The exact mechanism of how marriage plays a positive role in co-lon cancer diagnosis and treatment is not fully understood [13]. The spouse can play a key positive role in the disease, and this effect is conducive to the improvement of survival rate [14,15]. However, there are no reports about the relationship between mar¬ital status and prognosis in patients with small cell lung cancer. In this study, we performed a comprehensive population-based anal¬ysis using the SEER database to clarify the prognostic significance of marital status for SCLC patient survival.
Data and Methods
Study Population and Data Collection
Patients diagnosed with small cell lung cancer from 1973 to 2013 in the American Surveillance, Epidemiology, and End Results (SEER) database were selected. The data was analyzed using the SEER database (version 8.3.2, updated April 14, 2016).
The Inclusion Criteria
• Patient was histologically confirmed with small cell lung can-cer (ICD-O-3 histology code: 8041, 8042, 8043, and 8044)
• Patient was over 18 years old
• Full survival follow-up time
The Exclusion Criteria
• Patients with non-primary SCLC
• Missing marital status data
• Patient baseline information was incomplete, such as sex, race and age
• Tumor information (histology grade) was not recorded
• Patient treatment information (radiotherapy and surgery) was unclear
Methods
The case-control studies were used to divide patients into married group, single group, and SDW groups (divorced, separated, wid-owed) based on their marital status, the outcome index was over-all survival (OS). The variables included in this study were sex, marital status, age, race, tumor classification, surgical status, and radiotherapy status. Marital status was divided into three groups: married group, single group and SDW group (divorced, separated, and widowed). Race was divided into white group, black group and other races group. Age is divided into various age groups. The tumors histological grades were divided into I group, II group, III group and IV group. According to whether surgery is divided into two groups: surgery and non-operation; according to whether ra¬diotherapy is divided into two groups: radiotherapy and non-ra-diotherapy.
Statistical Methods
The data was extracted by Perl 5.26.2 software, data were collated with Excel 2013 software, statistical analysis was performed with SPSS 23.0 and R 3.35 software, chi-square test was used for pa¬tient baseline characteristics test, Kaplan-Meier and Log rank test were used for single factor survival efficiency analysis and surviv¬al curve was drawn, Cox multivariate analysis study The effect of each variable on the survival prognosis of lung cancer patients, P <0.05 was statistically significant.
Results
Baseline Characteristics
A total of 27069 eligible SCLC patients aged from 18 to 98 were included during the study period (from 1973 to 2013) in the SEER database, with an average age of (67.71 ± 10.32) years old. Among them, 14451 (53.39%) were married, 3167(11.70%) were never married, 9451 (34.91%) were in SDW group, which included 4955 (52.43%) widowed, 3706 (39.21%) divorced, and 790 (8.36%) sep¬arated. Age, race, histological grade, radiotherapy and surgery were different in marital composition (P<0. 01), as shown in Table 1.
Table 1: basic characteristics of patients with small cell lung cancer (n=27069)
|
Item |
n |
Marital status |
P value |
||
|
married |
single |
SDW |
|||
|
Total number (%) |
27069(100) |
14451(53.39) |
3167(11.70) |
9451(34.91) |
|
|
Sex |
|||||
|
Male (%) |
13439(49.65) |
8846(65.82) |
1696(12.62) |
2897(21.56) |
<0.001 |
|
Female (%) |
13630(50.35) |
5605(41.12) |
1471(10.79) |
6554(48.09) |
|
|
Race |
|||||
|
Black (%) |
2119(7.83) |
715(33.74) |
575(27.14) |
829(39.12) |
<0.001 |
|
White (%) |
23945(88.46) |
13089(54.66) |
2489(10.39) |
8367(34.95) |
|
|
Other (%) |
1005(3.71) |
647(64.38) |
103(10.25) |
255(25.37) |
|
|
Age/years |
|||||
|
65(%) |
109(0.40) |
51(46.79) |
17(15.60) |
41(37.61) |
<0.001 |
|
≥65 (%) |
1174(4.34) |
586(49.91) |
270(23.00) |
318(27.09) |
|
|
Histological grade |
|||||
|
I (%) |
92(0.34) |
56(60.87) |
12(13.04) |
24(26.09) |
0.024 |
|
II (%) |
282(1.04) |
146(51.77) |
48(17.02) |
88(31.21) |
|
|
III (%) |
5932(21.92) |
3133(52.82) |
771(13.00) |
2028(34.18) |
|
|
IV (%) |
20763(76.70) |
11116(53.54) |
2336(11.25) |
7311(35.21) |
|
|
Radiotherapy |
|||||
|
Yes (%) |
12622(46.63) |
7164(56.76) |
1427(11.31) |
4031(31.93) |
<0.001 |
|
No (%) |
14447(53.37) |
7287(50.44) |
1740(12.04) |
5420(37.52) |
|
|
Surgery |
|||||
|
Yes (%) |
779(2.88) |
475(60.98) |
59(7.57) |
245(31.45) |
<0.001 |
|
No (%) |
26290(97.12) |
13976(53.16) |
3108(11.82) |
9206(35.02) |
|
Relationship between Patient Characteristics and Sur-vival Prognosis
Kaplan-meier analysis showed that age, race, marital status, his-tological grade, radiotherapy and surgery were all related to the survival prognosis of patients. The prognosis of patients with bad marriage is significantly worse than that of married patients. There were statistically significant differences in prognosis between patients of different genders. There was significant difference in prognosis among different races (P<0.01). The prognosis of pa-tients <65 years old was significantly better than that of patients ≥65 years old. The prognosis of the patients after radiotherapy was significantly better than that of the patients without radiotherapy; the difference of prognosis in different histological stages was sta¬tistically significant (P<0.01). The prognosis of patients undergo¬ing surgery was significantly better than that of patients without surgery, as shown in Figure A1-7.
Figure A1: Kaplan-Meier Survival Curves of Overall Survival by Married Status
Figure A2: Kaplan-Meier Survival Curves of Overall Survival by Sex
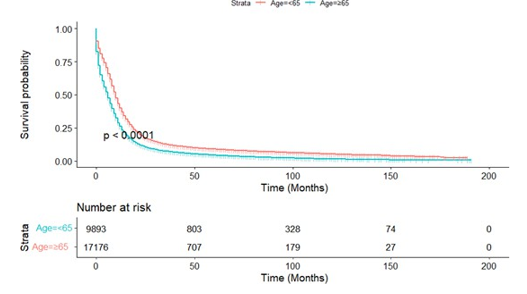
Figure A3: Kaplan-Meier Survival Curves of Overall Survival by Surgery or Age
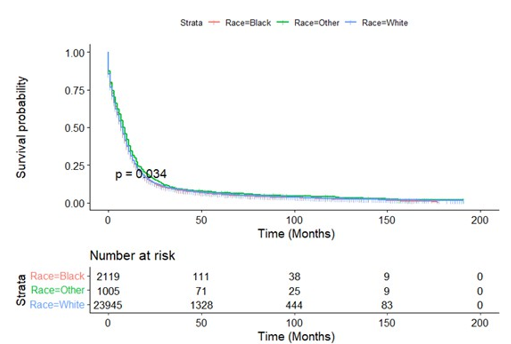
Figure A4: Kaplan-Meier Survival Curves of Overall Survival by Race
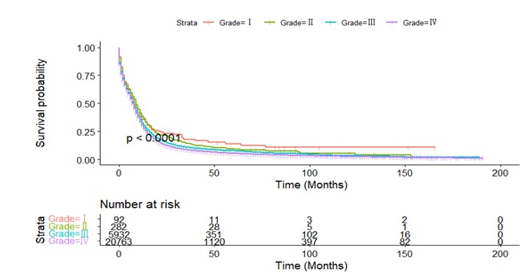
Figure A5: Kaplan-Meier Survival Curves of Overall Survival by Grade
Figure A6: Kaplan-Meier Survival Curves Of Overall Survival by Radiotherapy or Not
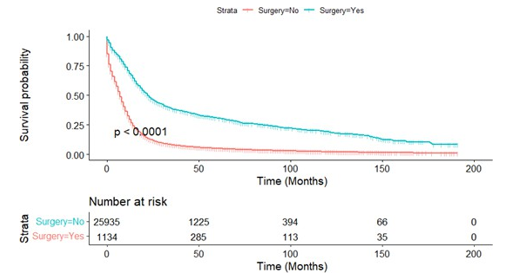
Figure A7: Kaplan-Meier Survival Curves Of Overall Survival by Surgery or Not
In order to remove the influence of confounding factors, the above analysis factors with P<0.05 were included in the COX regression model for multivariate analysis. The results showed: age, race, marital status, histological grade, radiotherapy or not and surgery or not were independent factors of survival prognosis in SCLC, as shown in Table 2 and multivariate Cox forest map (Figure B).
Figure B: Multivariate Cox Forest Map
Table 2: Univariate and multivariable Cox regression model of overall survival of patients with small cell lung cancer
|
|
Median OS |
Univariate |
|
Multivariable |
|
|
Case |
(month) |
Log rank |
P value |
HR (95%CI) |
P value |
|
Marital status |
|
113.33 |
<0.001 |
|
|
|
Married |
8 |
|
|
Ref |
|
|
Single |
7 |
|
|
1.15(1.10-1.20) |
<0.001 |
|
SDW |
6 |
|
|
1.10(1.07-1.13) |
<0.001 |
|
Sex |
8 |
126.42 |
<0.001 |
|
|
|
Male |
7 |
|
|
Ref |
|
|
Female |
8 |
|
|
0.85(0.83-0.87) |
<0.001 |
|
Race |
|
6.79 |
0.034 |
|
|
|
Black |
7 |
|
|
Ref |
|
|
White |
7 |
|
|
0.99(0.95-1.03) |
0.728 |
|
Other |
8 |
|
|
0.89(0.82-0.96) |
0.002 |
|
Age |
|
1449.18 |
<0.001 |
|
|
|
1-39 |
12 |
|
|
Ref |
|
|
40-49 |
11 |
|
|
1.11(0.89-1.37) |
0.337 |
|
50-59 |
10 |
|
|
1.17(0.95-1.44) |
0.131 |
|
60-69 |
9 |
|
|
1.35(1.10-1.66) |
0.004 |
|
70-79 |
6 |
|
|
1.62(1.32-1.99) |
<0.001 |
|
80+ |
3 |
|
|
2.06(1.67-2.53) |
<0.001 |
|
Grade |
|
41.90 |
<0.001 |
|
|
|
I |
8 |
|
|
Ref |
|
|
II |
9 |
|
|
1.02(0.80-1.32) |
0.860 |
|
III |
8 |
|
|
1.22(0.98-1.53) |
0.078 |
|
IV |
7 |
|
|
1.30(1.04-1.63) |
0.019 |
|
Radiotherapy |
|
2469.72 |
<0.001 |
|
|
|
No |
4 |
|
|
Ref |
|
|
Yes |
11 |
|
|
0.56(0.55-0.58) |
<0.001 |
|
Surgery |
|
480.97 |
<0.001 |
|
|
|
No |
7 |
|
|
Ref |
|
|
Yes |
21 |
|
|
0.38(0.35-0.41) |
<0.001 |
Subgroup Analysis Based On Survival Rate
In order to exclude the influence of all other possible factors on the ultimate survival, we did a stratified analysis to determine the independent prognostic effect of marital status on small cell lung cancer patients. Kaplan-Meier survival curves stratified by sex, age (cut-off value of 65 years), radiotherapy and surgery were shown in Figures A8-11, respectively. Subgroup analysis stratified by sex, age and radiotherapy displayed that married group had better survival compared with patients in SDW and single groups (Figures A8-10). Figure A11 depicted that married people had lon¬ger survival compared with those in SDW and single groups when individually analyze data from patients not receiving surgery.
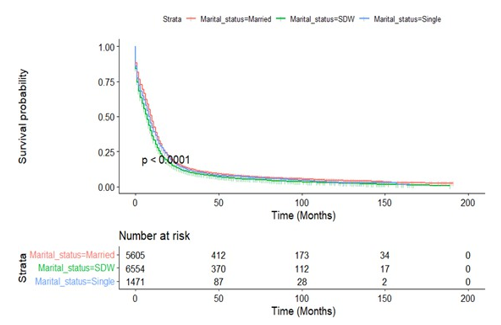
Figure A8.1: Kaplan-Meier Survival Curves of Overall Survival by Female
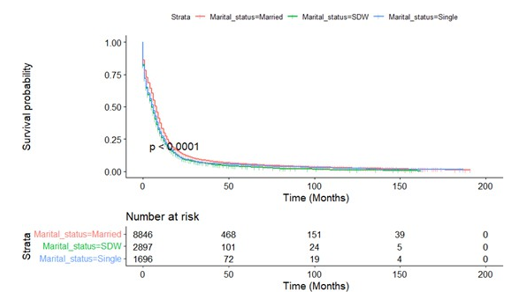
Figure A8.2: Kaplan-Meier Survival Curves of Overall Survival by Male
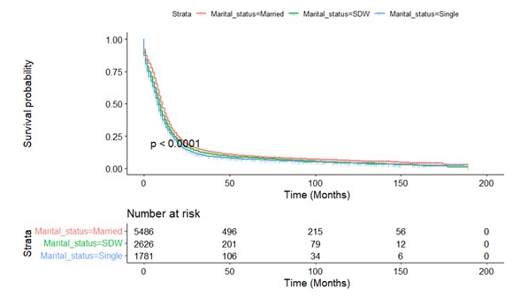
Figure A9.1: Kaplan-Meier Survival Curves Of Overall Survival by Age<65
Figure A9.2: Kaplan-Meier Survival Curves of Overall Survival by Age≥65
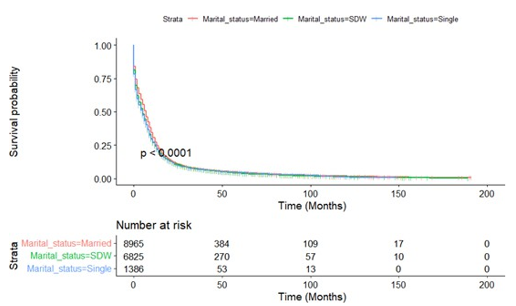
Figure A10.1: Kaplan-Meier Survival Curves of Overall Survival by Radiotherapy
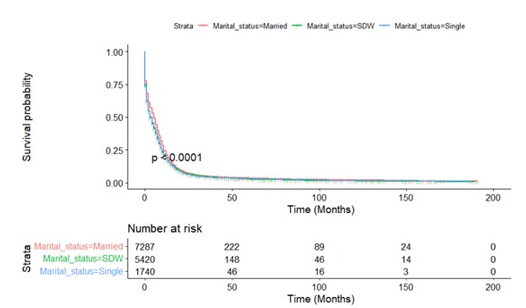
Figure A10.2: Kaplan-Meier Survival Curves of Overall Survival by Without Radiotherapy
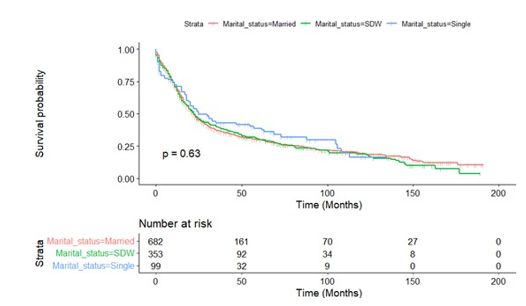
Figure A11.1: Kaplan-Meier Survival Curves of Overall Survival by Surgery
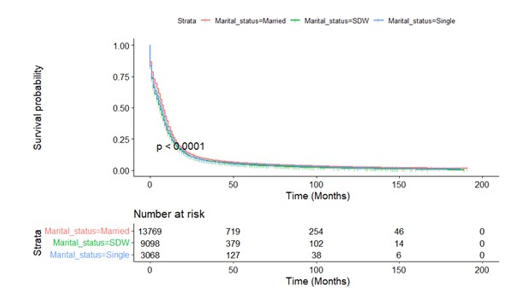
Fig.A11.1: Kaplan-Meier Survival Curves of Overall Survival by Without Surgery
Discussion
The results of this study showed that marital status is an indepen-dent prognostic factor for patients with small cell lung cancer. SCLC patients in Single group and SDW group had poor progno-sis and a higher risk of death. Therefore, the inharmonious marital state is one of the risk factors for poor prognosis of small cell lung cancer. Without the companion, it may easily lead to the change of patients’ mood, affect their quality of life, change their role func¬tion, and cause serious adverse effects on their psychological state Studies have found that mental state can affect the progress of tu¬mor, aggravate the clinical symptoms of patients, and then affect the clinical prognosis [16-18]. Positive emotions have a buffering effect on stress, can alleviate the psychological burden, stimulate patients to take positive behaviors, and significantly improve the psychological state and prognosis of cancer patients [19]. Howev¬er, negative emotions may induce changes in the respiratory sys¬tem and cardiovascular system, which may lead to poor prognosis [20].
The researchers conducted a large epidemiological survey and analyzed data from 1,260,898 patients with various types of tu- mors, such as prostate cancer, breast cancer, lymphoma, colorec¬tal cancer, pancreatic cancer, ovarian cancer, esophageal cancer, lung cancer, head and neck tumors, etc [21,22]. The results showed that after correction for all confounding factors, married cancer patients had a lower risk of death from cancer. Researchers have analyzed that cancer patients in happy families were more likely to receive health checkups recommended by their family members, so that they can be diagnosed and treated early in the disease [23]. At the same time, after being diagnosed, these patients are more willing to strictly follow the treatment plan and conduct the follow up supervision regularly. The results show social support is im¬portant for cancer patients [24].
Cancer has an impact on both physiology and psychology of pa¬tients. For example, the clinical symptoms of the disease will bring physical pain to patients, and the fear of disease recurrence and death will make patients suffer from psychological loneliness and depression [25]. Social support is considered that individuals re¬ceive support from the outside world, including material and spir¬itual guarantees, which directly promote the physical and mental health of individuals Study showed that social support, can inhibit the progress of cancer [26]. Compared with the control group, the survival rate of the group participating in social support was sig¬nificantly improved. Marriage is a form of social support. Social support in marriage mainly comes from the spouse. The patient’s spouse can provide effective emotional comfort and social en-vironment support in time to reduce the psychological stress of the patient [27]. Therefore, single or SDW patients with SCLC should be provided with active psychological counseling. The doctors should answer patient questions patiently, communicate in a timely manner and give psychological intervention to reduce the negative emotions of patients, to improve their quality of life and prolong their survival time. It is also suggested for the medi¬cal staff to give patients more care that is humane and emotional support while treating tumors; this will reduce their psychological pain, improve their internal control, and then effectively improve their psychological state [28].
The SEER database provides us the opportunity to perform large, population-based studies. However, there are several limitations in our study. An obvious limitation is that this is a retrospective study that has a greater probability of bias than prospective stud¬ies. In addition to demographic and clinical factors, there is a rel¬ative lack of control variables. For example, the database cannot provide important confounding variables such as smoking history, patient complications, and other treatment options. Secondly, the marital status recorded in the SEER database is only the marital status at the time of diagnosis. As for whether the patient’s subse¬quent marital status has changed, no follow-up visits and records have been performed, so the impact of this change on the study cannot be estimated. Thirdly, the SEER database does not incor¬porate data on the life quality of married patients, such as marital satisfaction or financial status.
Despite of these potential limitations, this is the first study investi-gating the effect of marital status on prognosis of SCLC by big data. It suggests that doctors should pay more attention to unmarried or SDW patients. This will improve the mental health problems of such patients caused by the lack of companion, thus achieve better clinical treatment measures and improve the long-term prognosis.
Abbreviations
SEER: Surveillance Epidemiology and results
SCLC: Small Cell Lung Cancer
OS: Overall Survival
SDW: Divorced, Separated, Widowed
HR: Hazard ratio
Ethics Approval
All procedures in studies involving human participants were per-formed in accordance with the ethical standards of the institutional review board of the seventh affiliated hospital of zunyi medical university Ethics Committee basing on the 1964 Helsinki declara¬tion and its later amendments.
Acknowledgements
The authors thank the reviewers for their helpful comments on this article.
Availability of Data and Materials
The dataset generated and analyzed during the current study is available in the Surveillance Epidemiology and End Results (SEER) Database repository [https://seer.cancer.gov/].
Authors Contribution
PL provided contribution to (i) data analysis, interpretation, (iii) manuscript drafting, and critical revising. PL and KY-Z contribut-ed to (i) experimental design, (ii) data analysis and interpretation and (iii) critical manuscript revising for important intellectual con-tent. LP-Y and CP provided contribution to (i) data analysis and (ii) critical manuscript revising for important intellectual content. XY, AJ-B and CH contributed to the modification of (iv) images and (v) table contents. JG contributed to (i) data analysis and in-terpretation and (ii) critical manuscript revising for important in-tellectual content. All authors have approved the final version and submission of this manuscript.
References
- Siegel R. L., Miller K. D. and Jemal A. (2020) Cancer statistics. CA Cancer J Clin.2020, 70(1): 7-30.
- Rudin C. M., Brambilla E., Faivre-Finn C., Julien Sage. (2021) Small-cell lung cancer. Nat Rev Dis Primers. 7(1): 3.
- Wang, Y., Zou, S., Zhao, Z., Liu, P., Ke, C., & Xu, S. (2020).New insights into small-cell lung cancer development and therapy. Cell Biology International, 44(8), 1564-1576.
- Islami, F., Torre, L. A., & Jemal, A. (2015). Global trends of lung cancer mortality and smoking prevalence. Translational lung cancer research, 4(4), 327.
- Peng, A., Li, G., Xiong, M., Xie, S., & Wang, C. (2019). Role of surgery in patients with early stage small-cell lung cancer. Cancer Management and Research, 11, 7089.
- Yang, Y., Yuan, G., Zhan, C., Huang, Y., Zhao, M., Yang, X., & Wang, Q. (2019). Benefits of surgery in the multimodality treatment of stage IIB-IIIC small cell lung cancer. Journal of Cancer, 10(22), 5404.
- Zhao, X., Kallakury, B., Chahine, J. J., Hartmann, D., Zhang, Y., Chen, Y., & Giaccone, G. (2019). Surgical resection of SCLC: prognostic factors and the tumor microenvironment. Journal of Thoracic Oncology, 14(5), 914-923.
- Hofman, A., Zajdel, N., Klekowski, J., & Chabowski, M. (2021). Improving Social Support to Increase QoL in Lung Cancer Patients. Cancer Management and Research, 13, 2319.
- Lan, T., Lu, Y., Luo, H., He, J., He, J., Hu, Z., & Xu, H.(2020). Effects of marital status on prognosis in women with infiltrating ductal carcinoma of the breast: a real-world 1: 1 propensity-matched study. Medical science monitor: international medical journal of experimental and clinical research, 26, e923630-1.
- Aizer, A. A., Chen, M. H., McCarthy, E. P., Mendu, M. L., Koo, S., Wilhite, T. J., & Nguyen, P. L. (2013). Marital status and survival in patients with cancer. Journal of clinical oncology, 31(31), 3869.
- Hanske, J., Meyer, C. P., Sammon, J. D., Choueiri, T. K., Me-non, M., Lipsitz, S. R., & Trinh, Q. D. (2016). The influence of marital status on the use of breast, cervical, and colorectal cancer screening. Preventive medicine, 89, 140-145.
- Li, Q., Gan, L., Liang, L., Li, X., & Cai, S. (2015). The in-fluence of marital status on stage at diagnosis and survival of patients with colorectal cancer. Oncotarget, 6(9), 7339.
- Feng, L., Yang, Y. J., Du, J., Yu, Y. J., & Diao, J. D. (2020).Marital status and survival of patients with colorectal signet ring cell carcinoma: a population-based study. Scientific Reports, 10(1), 1-10.
- Rendall, M. S., Weden, M. M., Favreault, M. M., & Waldron,H. (2011). The protective effect of marriage for survival: a review and update. Demography, 48(2), 481-506.
- Liu, Y. L., Wang, D. W., Yang, Z. C., Ma, R., Li, Z., Suo, W.,... & Li, Z. W. (2019). Marital status is an independent prognostic factor in inflammatory breast cancer patients: an analysis of the surveillance, epidemiology, and end results database. Breast cancer research and treatment, 178(2), 379-388.
- Tatangelo, G., McCabe, M., Campbell, S., & Szoeke, C. (2017). Gender, marital status and longevity. Maturitas, 100, 64-69.
- Wu, Y., Ai, Z., & Xu, G. (2017). Marital status and survival in patients with non-small cell lung cancer: an analysis of 70006 patients in the SEER database. Oncotarget, 8(61), 103518.
- Applebaum, A. J., Stein, E. M., Lordâ?Bessen, J., Pessin, H., Rosenfeld, B., & Breitbart, W. (2014). Optimism, social support, and mental health outcomes in patients with advanced cancer. Psychoâ?oncology, 23(3), 299-306.
- Dunstan, D. A., & Scott, N. (2020). Norms for Zung’s self-rat-ing anxiety scale. BMC psychiatry, 20(1), 1-8.
- Hu, T., Xiao, J., Peng, J., Kuang, X., & He, B. (2018). Relationship between resilience, social support as well as anxiety/ depression of lung cancer patients: a cross-sectional observation study. Journal of cancer research and therapeutics, 14(1), 72.
- Fitzmaurice, C., Abate, D., Abbasi, N., Abbastabar, H., Abd-Allah, F., Abdel-Rahman, O., & Derakhshani, A. (2019). Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-ad-justed life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the global burden of disease study. JAMA oncology, 5(12), 1749-1768.
- Henley, S. J., Thomas, C. C., Lewis, D. R., Ward, E. M., Islami, F., Wu, M., & Richardson, L. C. (2020). Annual report to the nation on the status of cancer, part II: Progress toward Healthy People 2020 objectives for 4 common cancers. Cancer, 126(10), 2250-2266.
- Chen, Z. H., Yang, K. B., Zhang, Y. Z., Wu, C. F., Wen, D. W.,Lv, J. W., & Lin, L. (2021). Assessment of modifiable factors for the association of marital status with cancer-specific sur-vival. JAMA network open, 4(5), e2111813-e2111813.
- Osborne, C., Ostir, G. V., Du, X., Peek, M. K., & Goodwin,J. S. (2005). The influence of marital status on the stage at diagnosis, treatment, and survival of older women with breast cancer. Breast cancer research and treatment, 93(1), 41-47.
- Luszczynska, A., Boehmer, S., Knoll, N., Schulz, U., & Schwarzer, R. (2007). Emotional support for men and women with cancer: do patients receive what their partners provide?. International Journal of Behavioral Medicine, 14(3), 156-163.
- Aydın Sayılan, A., & Demir DoÄ?an, M. (2020). Illness perception, perceived social support and quality of life in patients with diagnosis of cancer. European journal of cancer care, 29(4), e13252.
- Gao, Z., Ren, F., Song, H., Wang, Y., Wang, Y., Gao, Z., & He,X. (2018). Marital status and survival of patients with chon-drosarcoma: a population-based analysis. Medical science monitor: international medical journal of experimental and clinical research, 24, 6638.
- Yan, X. R., Chen, X., & Zhang, P. (2019). Prevalence and risk factors of depression in patients with lung cancer: protocol for a systematic review and meta-analysis. BMJ open, 9(8), e028994.


