Research Article - (2019) Volume 1, Issue 1
Lupic Nephropathy: Our experience at the Department of Nephrology
Received Date: Jul 08, 2019 / Accepted Date: Jul 20, 2019 / Published Date: Jul 30, 2019
Copyright: ©I Nouri, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Systemic lupus erythematosus (SLE) is the most common disease systemic autoimmune disorders that cause kidney damage. AT Conversely, kidney damage is the most common and the most severe visceral involvement of SLE. The most frequent renal involvement is glomerular and there are several types of glomerulonephritis (GN) Lupus now evaluated according to classification histological ISN / RPS (International Society of Nephrology/Renal Pathology Society) [1].
Introduction
Systemic lupus erythematosus (SLE) is the most common disease systemic autoimmune disorders that cause kidney damage. AT Conversely, kidney damage is the most common and the most severe visceral involvement of SLE. The most frequent renal involvement is glomerular and there are several types of glomerulonephritis (GN) Lupus now evaluated according to classification histological ISN / RPS (International Society of Nephrology/Renal Pathology Society) [1]. Other glomerular disorders such as a Nephrotic syndrome with minimal glomerular lesions are possible but rare. Vascular or interstitial lesions related to lupus may be associated with glomerular damage; they are rarely isolated. Finally, lupus nephropathy is sometimes mixed with renal diseases associated with lupus, the most common being renal antiphospholipid Syndrome.
Epidemiology
The global incidence and prevalence of SLE are between 1 and 5 per 100,000 and between 20 and 150 per 100,000, respectively. In France, they are estimated at 3.32 per 100,000 and 40.8 per 100,000. The frequency of the disease in women is 9 times higher than frequency encountered in men. Renal impairment is more common in subjects of African descent, Hispanic or Asian only in subjects of Caucasian origin [2]. African origin is probably a factor of severity. Reaching kidney is particularly common (50 to 80% of cases) and potentially more severe in pediatric cohorts. The male sex is a risk factor for early renal damage and greater severity; a low socio-economic level is a risk factor for lupus nephropathy and severity of impairment [3].
Diagnostic Approach: Clinical manifestations and indications kidney biopsy
Initialdiagnosis
Renal involvement of lupus occurs most often at the diagnosis of SLE or within 3 to 5 years. There does not seem to be any preferential association between renal impairment and other manifestations of lupus; also must we consider that all can accompany it [4]. Sometimes the damage renal is inaugural with litte or no clinical signs of lupus, the renal biopsy then being diagnosed with SLE. Triggers are those of SLE (sun exposure, stress, contraceptives including estrogen, pregnancy, etc.) and there is no factor specific to kidney damage [4,5]. The clinical manifestations have three characteristics:
• The important variability of the nephrological signs: simple silent proteinuria and/or microscopic hematuria associated renal failure, these manifestations may constitute chronic glomerulopathy syndrome, up to noisier manifestations such as glomerulonephritis syndrome quickly progressive, nephrotic syndrome or syndrome acute nephritic [6]. Aseptic leukocyturia accompanies sometimes glomerular signs [7-9].
• The clinical constant is proteinuria which must be take into account at a low flow rate (from 0.5 g / g creatinine urea to 0.3 g / g if it exists) concomitant hematuria), the other renal abnormalities being inconstant [10-12].
• There is no anatomic-clinical parallelism: it is not possible to predict the type of histological involvement and therefore the severity by the analysis semiological; so, for example, a proteinuria of 0.5 g / g can all as much to translate an Active Class III or IV GN as a Class III or IV chronic, that a class II or a class V [13, 14].
One can thus affirm the existence of a lupus GN as soon as the detection permanent proteinuria, but we cannot say how serious it is [15].
Immune abnormalities are associated with nephropathies the most severe lupus: consumption of complement by the way (C4 and C3 lowered) and the presence of anti-double DNA antibodies strand. Nevertheless, their predictive value is insufficient and only one renal biopsy can typify lupus GN and define the need treatment [16-18].
As a reminder, any patient with SLE should benefit from research renal anomaly, during diagnosis but also during follow-up. Currently, in the absence of contraindications, any patient with SLE and higher proteinuria 0.5 g per day must have a kidney biopsy because of the lack of correlation between clinical-biological presentation and lesions histological. Glomerular lesions are then graded according to the ISN-RPS classification from I to VI [19, 20].
• It is also important to quantify the activity or chronicity of the lesions because these parameters guide the clinician in indication as well as in the choice of treatments [21-23].
Histological features of the different classes of lupus nephritis (ISN / RPS 2003 classification)
Class Histological Features
The classification of LN is based on histological features, using the International Society of Nephrology (ISN) and Renal Pathology Society (RPS) criteria developed in 2003 (Table 1). Although the classification is mainly glomerulocentric, it includes features of tubulointerstitial disease, from which features of chronicity can be determined. The relationship between the histological class of LN and clinical course of the disease is well recognised. Patients with class II and class V (pure membranous LN) disease usually have a slow decline in renal function over long periods of observation. In contrast, patients with class III and class IV (or those with mixed class V) disease mostly have a more aggressive course of disease. Various studies have shown that the proliferative forms of LN (i.e. class III, class IV and mixed class V) occur more frequently than the other histological Morphologies [24-26].

Diagnosis
One of the major challenges with regard to SLE is its early recognition and diagnosis. Many physicians assume that SLE is a rare condition in Africa - it is therefore seldom considered as a differential diagnosis, except when patients present with classic features, such as malar rash and swollen painful joints of the hands and feet [27-29]. Hence, many patients tend to remain ill for prolonged periods before the diagnosis is made. In some instances, chronic damage to organs including the kidneys would have occurred [30, 31].
Role of Urinalysis and Urine Microscopy
LN is unlikely to present alone - it often manifests with other extra renal features such as joint pain, malar rash, oral ulcers and photosensitivity. The patient with LN is likely to present with nephritic syndrome (oliguria, minimal proteinuria, haematuria hypertension and azotaemia) or with features of nephrotic syndrome (anasarca, heavy proteinuria and hypoalbuminemia) [32, 33]. Urinalysis (dipstick and microscopic examination) presents the best opportunity for early identification of LN as a dipstick is likely to show the presence of blood and protein in the urine and urine microscopy enables identification of various urinary casts (red cells, granular, hyaline) [34-36]. LN must be strongly suspected in any SLE patient with high titres of double-stranded DNA (dsDNA) and a positive dipstick for blood. Urinalysis features have been shown to be correlated with the presence of proliferative class LN. Therefore, every SLE patient should have a urinalysis performed at every clinic visit [37, 38].
Role of Lupus Auto-Antibodies (Antinuclear Antibody and dsDNA) And Complements (C3/C4)
Auto-antibodies in SLE and complements (C3/C4) are known to be elevated or lowered, respectively, in patients with increased disease activity, especially in those with proliferative LN (class III, class IV and mixed class V). One study found proliferative LN to be significantly correlated with haematuria on dipstick (ρ<0.0001), proteinuria on dipstick (p=0.042), low complement C3 (p<0.0001), low complement C4 (p=0.009) and positive dsDNA (p=0.039) [39-41].
Role of Renal Biopsy
The definitive diagnosis of LN requires a kidney biopsy. Renal histology also enables classification of LN and assists in the prognosis. All guidelines recommend a renal biopsy when there is a suspicion of renal involvement, as clinical and laboratory parameters, although useful, cannot accurately predict the histological class. The indication for a renal biopsy in SLE patients includes persistent decline in renal function, proteinuria (>1.0 g/24 h) or proteinuria (>0.5 g/24 h) if associated with haematuria (5 red blood cells (RBCs)/ high power field) and active urinary sediment (granular casts, white blood cell (WBC) casts, RBC casts) [42,43]. It is recommended that the biopsy be examined by light microscopy, immunofluorescence (or immunohistochemistry) and where possible by electron microscopy. Quantification of activity and chronicity indices and description of vascular and interstitial lesions are also recommended. A repeat renal biopsy is indicated if there is evidence of worsening of the disease or disease refractory to treatment, evidence of relapse (to show transformation or progression in histological class or change in activity and chronicity cores) and to demonstrate other pathologies.
Fig. 1 shows the renal histology in a patient with class IV LN.
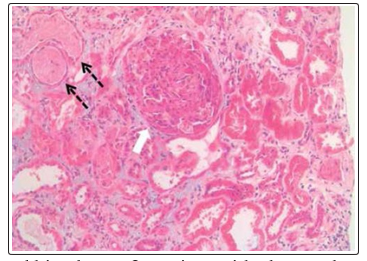
Figure 1: Renal histology of a patient with class IV lupus nephritis (white arrow shows a glomerulus with cellular crescent; black broken arrows show adjacent tubules with red cell casts)
Current Treatment
The treatment of lupus nephropathy is based on two main pillars: induction phase and the maintenance phase. The treatment of induction of NL is based on the combination of corticosteroids (CS) and cyclophosphamide (CYC) or mycophenolate mofetil (MMF). During the maintenance phase, treatment with azathioprine (AZA) or MMF can be used. The treatment of NL, however, remains imperfect. Indeed, the total re-emission rate is around 50%, generating a significant risk of evolution to the MRC, or even terminal renal failure [44,45].
The treatment of LN is dictated by the class of the disease and degree of activity and chronicity indices. All patients should receive adjuvant therapies as indicated and if tolerated. There should be a risk-benefit evaluation when deciding whether to use immunosuppression in patients with increased chronicity indices (i.e. glomerular sclerosis with tubulo-interstitial fibrosis) because of the increased risk of side-effects of treatment. The approach to treatment often involves two phases for patients with proliferative LN (class III, class IV and mixed classV) [46, 47].
Induction Therapy
Fig. 2 summarises the common approaches used for induction for the different classes of LN. Induction therapy is not used for patients with class I, II, V (pure class V with sub-nephrotic range proteinuria) and VI LN. The approach to induction involves the use of 3 consecutive pulses of intravenous methylprednisolone (500 - 750 mg daily) together with another immunosuppressive: cyclophosphamide (CYC) (daily oral or monthly intravenous pulse therapy) or mycophenolate mofetil (MMF). The patient should continue on oral prednisone (1 mg/kg/day) after completing the pulse treatment with methylprednisolone [48, 49]. CYC is often the agent of choice for many clinicians owing to easy accessibility and cost; however, the use of MMF is increasing. Various studies have assessed the efficacy and safety of CYC with MMF or placebo for induction therapy in patients with LN (reviewed in depth by Chan). The recommended duration of induction therapy is 6 months; during this time, the dose of oral corticosteroid should be weaned.
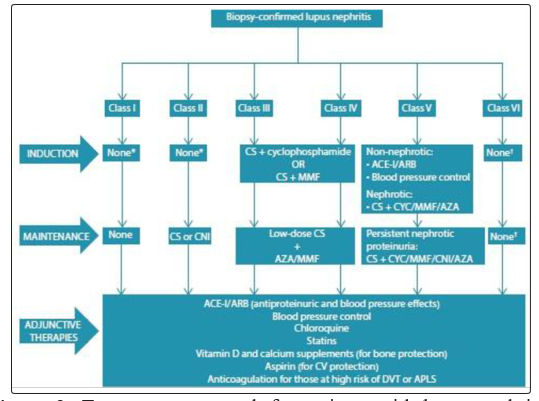
Figure 2: Treartment approach for patients with lupus nephritis (APLS = antiphospholipid syndrome; AZA = azathioprine; ACE-I=angiotensin converting enzyme inhibitor; ARB = angiotensin receptor blocker; CNI = calcineurin inhibitor; CS = corticosteroid; CYC = cyclophosphamide; DVT = deep-vein thrombosis; MMF = mycophenolate mofetil; CV = cardiovascular).
*Immunosuppre to be dictated by extrarenal manifestations. t Patients should be Prepared for renal replacement therapy (dialysis/ transplantation).
Maintenance Therapy
Treatment administered during the induction phase of therapy is de-escalated in the maintenance phase. The goal of the latter phase is to maintain the response (remission) gained during the induction phase and therefore to retard progression of chronic kidney disease. Immunosuppressive agents commonly used include MMF, azathioprine (AZA), and corticosteroids.
Calcineurin inhibitors may be used in special circumstances, such as in cases of intolerance to MMF or AZA or in patients with persistent heavy proteinuria (Fig.2). There is currently no consensus on the duration of maintenance therapy [50-52]. Nonetheless, the decision to withdraw maintenance immunosuppression should be guided by sustained complete clinical response over a period of at least 2 years. Withdrawal should be done gradually, starting with glucocorticoids before withdrawing immunosuppressive agents. In debilitating or life threatening complications of immunosuppression, therapy should be withdrawn [53, 54].
Adjunctive Therapies
Adjunctive therapies are usually started during the induction phase of treatment and although some need to be discontinued after completing this therapy, others will need to be continued during the maintenance phase [55-57]. Commonly recommended adjunctive therapies in LN include:
• Renin angiotensin aldosterone system (RAAS) inhibition for proteinuria and blood pressure treatment (Target<130/80 mmHg).
• Bone protection with calcium and vitamin D supplements.
• Chloroquine for all patients (unless contraindicated, e.g. visual disturbance).
• Treatment of hyperlipidaemia with statins (target low density lipoprotein <2.6 mmol/L).
• Low-dose acetylsalicylic acid in patients with antiphospholipid syndrome. • Anticoagulant to be considered in patients with nephrotic syndrome and albumin <20 g/L.
• Avoid vaccination with live or attenuated viruses during immune suppression.
• Tuberculosis prophylaxis with isoniazid (for those in highly endemic TB regions).
Treatment of Refractory Ln
Fewer than 50% of patients are able to achieve complete remission during the 6 months of induction therapy. It may take up to 2 years to reach remission in many patients [58, 59]. Switching to an alternative agent is recommended for patients who fail to improve within 3-4 months, or do not achieve a partial response after 6-12 months or a complete response after 2 years of treatment. Treatment options include switching from MMF to CYC or from CYC to MMF; rituximab may be given as add-on treatment or monotherapy. Other options include the use of calcineurin inhibitors, intravenous immunoglobulin and plasma exchange for patients with rapidly progressive glomerulonephritis [60, 61].
Treatment of Class Vi Ln
Immunosuppression for class VI patients must be dictated by extra renal manifestations of SLE. These patients should be prepared for renal replacement therapy (haemodialysis, peritoneal dialysis or transplantation). Treatment and Prevention of cardiovascular risk factors (e.g. blood pressure control, statins for dyslipidaemia) should be continued [62, 63].
Lupica Nephropathy
Our experience at the Department of Nephrology year 2015/2016/2017/2018:
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease; lupus nephritis is usually the most serious manifestation of SLE, burdened with significant morbidity and mortality. The pathophysiological development of the systemic form and involvement of lupus has been better and better understood over the years, and various so-called targeted therapeutic approaches have recently been developed.
Materials and Methods
*Type of study: Our study is retrospective it concerns 31 patients with lupic nephropathy in our department over a period of 4 years (2015-2018).
*Patients: they were selected from the hospitalization register of our department.
Results
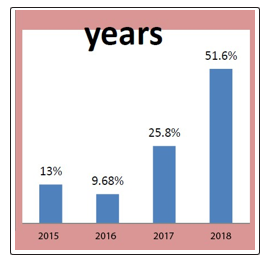
The incidence of lupus Nephropathy
|
EARS |
NUMBER |
FREQUENCY |
|
2015 |
4 |
13% |
|
2016 |
3 |
9.68% |
|
2017 |
8 |
25.8% |
|
2018 |
16 |
51.6% |
|
TOTAL |
31 |
100% |
|
female |
21 |
67.74% |
|
male |
8 |
25.8% |
|
child |
2 |
6.45% |
|
total |
31 |
100% |

|
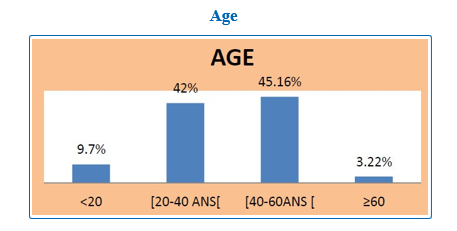
AGE |
Number |
Frequency |
|
<20 |
3 |
9.7% |
|
[20-40 ANS] |
13 |
42% |
|
[40-60 ANS] |
14 |
45.16% |
|
≥60 |
1 |
3.22% |
|
TOTAL |
31 |
100% |

*Family Antecedents(ATCP's)
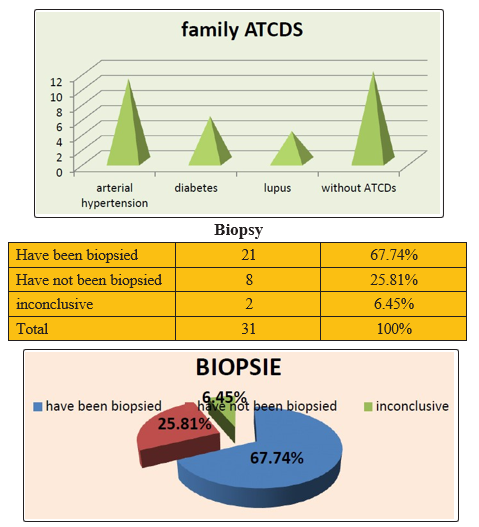
|
Biopsy |
Number |
|
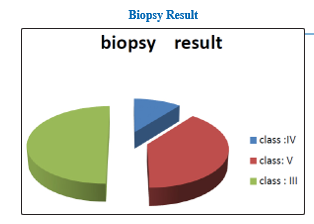
CLASS :IV |
3 |
|
CLASS :V |
11 |
|
CLASS :III |
7 |
Results
• The frequency of ludic nephropathy is increasing from 25.8% in 2017 to 51.6% in 2018, with a clear predominance of women at 67.74%.
• 38.7% of patients were known for lupus disease and 12.9% had familial SLE antecedents.
• Equal proportions between the age groups of 20-40 years and 40 and 60 years.
• On admission renal impairment was dominated by impure nephrotic syndrome and renal insufficiency found in more than 75% of cases
• Extra-renal signs were dominated by skin lesions and joint involvement with 62.2% of cases
• The association (anti-DNA antibodies Antinuclear antibodies (ANA)) was positive in 32% of cases, (anti-DNA antibodies+ ANA+ anti SM + anti SSA / SSB) in 25.81%.
• 23 of 31 patients were biopsied; Class V was dominant with more than 50%, elsewhere Class III and Class IV.
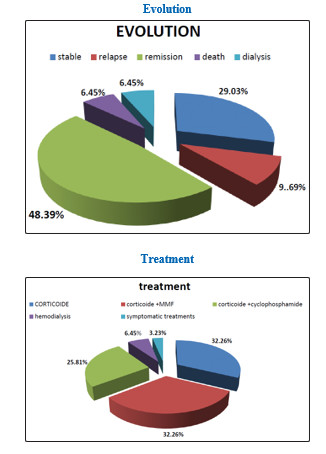
• 32.26% of patients received Corticoids + Cellcept (MMF), 25.81% Corticoids + Cyclophosphamides; 32.26% received Corticoids alone.
• The evolution was favourable in the majority of the cases with a percentage of remission of 48% and relapses in 9% of the cases; 6.45% required haemodialysis sessions.
Discussion
• The nature and severity of renal failure play an important role in the prognosis of SLE. in our series studied It is often a glomerular disease, but a tubulo-interstitial and vascular involvement are not negligible that can play an aggravating role and must also be evaluated.
• There is in our series a similarity in the distribution of histological classes with different series in which the non-proliferative forms predominates that’s why they received corticosteroids associated with MMF.
• 25% of the patients had repetition infections, which is why they received corticoids alone
• 9.69% of patients relapsed; there is no unanimous decision regarding the criteria used for renal recurrence, which is also based on observed changes in creatinine, proteinuria and urinary sediment levels compared to baseline values. Recurrences tend to be accompanied by a reduction in complement and an increase in anti-DNA antibody titers. In case of severe recurrence, deterioration of renal function may be present.
• Regarding treatment failure (lack of response or recurrence), it is often related to non-compliance, especially in young patients who have had difficulty coping with chronic illness.
Conclusion
Lupus nephritis remains the main determinant of mortality for patients with SLE. The assessment and management of lupus nephritis has seen major advances over the past 5 years. Whose classification for lupus nephritis has been updated to allow more accurate description of renal his to pathological specimens by the International Society of Nephrology and the Renal Pathology Society (2003). The future challenge remains the design of therapeutic regimens incorporating existing and newer therapies that will rapidly induce renal remission with minimum toxicity. Patients who have SLE should be followed in dedicated clinics, and early kidney involvement should be detected by very regular assessments of proteinuria.
References
- Lupic Nephritis: EMC 2018
- Golbus J, Mc Cune WJ (1994) Lupus nephritis Classification, prognosis, immunopathogenesis, and treatment. Rheum Dis Clin North Am 20: 213-42.
- Cameron JS (1999) Lupus nephritis. J Am Soc Nephrol 10:413-24.
- Houssiau FA, Vasconcelos C, D’Cruz D, Sebastiani GD, de Ramon Garrido E et al., (2010) The 10-year follow-up data of the Euro-Lupus Nephritis Trial comparing low-dose and high-dose intravenous cyclophosphamide. Ann Rheum Dis 69: 61.
- Rathi M, Goyal A, Jaryal A, Sharma A, Gupta PK et al., ( 2016) Comparison of low-dose intravenous cyclophosphamide with oral mycophenolate mofetil in the treatment of lupus nephritis. Kidney Int 89: 235-42.
- Ginzler EM, Dooley MA, Aranow C, Kim MY, Buyon J et al., (2005) Mycophenolate mofetil or intravenous cyclophosphamide for lupus nephritis. N Engl J Med 353: 2219-28.
- Tan EM, Cohen AS, Fries JF, Masi AT, McShane DJ et al., (1982) The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 25: 1271-7.
- Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40: 1725.
- Petri M, Orbai AM, Alarcón GS, Gordon C, Merrill JT et al., (2012) Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 64: 2677-86.
- Amezcua-Guerra LM, Higuera-Ortiz V, Arteaga-García U, Gallegos-Nava S, Hübbe- Tena C (2015) Performance of the 2012 Systemic Lupus International Collaborating Clinics and the 1997 American College of Rheumatology classification criteria for systemic lupus erythematosus in a real-life scenario. Arthritis Care Res (Hoboken) 67: 437-41.
- Lech M, Anders HJ (2013) The pathogenesis of lupus nephritis. J Am Soc Nephrol 24: 1357-66.
- Tojo T, Friou GJ, Spiegelberg HL (1970) Immunoglobulin G subclass of human antinuclear antibodies. Clin Exp Immunol 6: 145-51.
- Okamoto A, Fujio K, Tsuno NH, Takahashi K, Yamamoto K (2012) Kidney-infiltrating CD4+ T-cell clones promote nephritis in lupus-prone mice. Kidney Int 82: 969-79.
- Miyake K, Akahoshi M, Nakashima H (2011) Th subset balancein lupus nephritis. J Biomed Biotechnol 2011: 980286.
- Liu Y, Anders HJ (2014) Lupus nephritis: From pathogenesis to targets for biologic treatment. Nephron Clin Pract 128: 224-31.
- Mohan C, Putterman C (2015) Genetics and pathogenesis of systemic lupus erythematosus and lupus nephritis. Nat Rev Nephrol 11: 329-41.
- Saxena R, Mahajan T, Mohan C (2011) Lupus nephritis: Currentupdate. Arthritis Res Ther 13: 240.
- Ramos PS, Brown EE, Kimberly RP, Langefeld CD (2010) Genetic factors predisposing to systemic lupus erythematosus and lupus nephritis. Semin Nephrol 30: 164-76.
- Triantafyllopoulou A, Franzke CW, Seshan SV, Perino G, Kalliolias GD et al., (2010) Proliferative lesions and metalloproteinase activity in murine lupus nephritis mediated by type I interferons and macrophages. Proc Natl Acad Sci USA 107: 3012-7.
- Markowitz GS, D’Agati VD (2009) Classification of lupus nephritis. Curr Opin Nephrol Hypertens 18: 220-5.
- Ortega LM, Schultz DR, Lenz O, Pardo V, Contreras GN (2010) Review: Lupus nephritis: Pathologic features, epidemiology and a guide to therapeutic decisions. Lupus 19: 557-74.
- Weening JJ, D’Agati VD, Schwartz MM, Seshan SV, Alpers CE et al., (2004) The classification of glomerulonephritis in systemic lupus erythematosus revisited. J Am Soc Nephrol 15: 241-50.
- Hu W, Chen Y, Wang S, Chen H, Liu Z et al., (2016) Clinical-morphological features and outcomes of lupus podocytopathy. Clin J Am Soc Nephrol 11: 585-92.
- KDIGO Clinical Practice Guideline for Glomerulonephritis (2012). Agency for Healthcare Research and Quality (AHRQ) 2: 259-274.
- Hahn BH, McMahon MA, Wilkinson A, Wallace WD, Daikh DI et al., (2012) American College of Rheumatology guidelines for screening, treatment, and management of lupus nephritis. Arthritis Care Res (Hoboken) 64: 797-808.
- Bertsias GK, Tektonidou M, Amoura Z, Aringer M, Bajema I et al., (2012) Joint European League Against Rheumatism and European Renal Association-European Dialysis and Transplant Association (EULAR/ERA-EDTA) recommendations for the management of adult and paediatric lupus nephritis. Ann Rheum Dis 71: 1771-82.
- Chrysochou C, Randhawa H, Reeve R, Waldek S, Wood GN et al., (2008) Determinants of renal functional outcome in lupus nephritis: A single centre retrospective study. QJM 101: 313-6.
- Chen YE, Korbet SM, Katz RS, Schwartz MM, Lewis EJ (2008) Collaborative Study Group. Value of a complete or partial remission in severe lupus nephritis. Clin J Am Soc Nephrol 3: 46-53.
- Austin HA 3rd, Klippel JH, Balow JE, le Riche NG, Steinberg AD et al., (1986) Therapy of lupus nephritis. Controlled trial of prednisone and cytotoxic drugs. N Engl J Med 314: 614-9.
- Boumpas DT, Austin HA 3rd, Vaughn EM, Klippel JH, Steinberg AD et al., (1992) Controlled trial of pulse methylprednisolone versus two regimens of pulse cyclophosphamide in severe lupus nephritis. Lancet 340: 741-5.
- Gourley MF, Austin HA 3rd, Scott D, Yarboro CH, Vaughan EM et al., (1996) Methylprednisolone and cyclophosphamide, alone or in combination, in patients with lupus nephritis. A randomized, controlled trial. Ann Intern Med 125: 549-57.
- Grootscholten C, Ligtenberg G, Hagen EC, van den Wall Bake AW, de Glas-Vos JW et al., (2006) Azathioprine/ methylprednisolone versus cyclophosphamide in proliferative lupus nephritis. A randomized controlled trial. Kidney Int 70: 732-42.
- Nossent HC, Koldingsnes W (2000) Long-term efficacy of azathioprine treatment for proliferative lupus nephritis. Rheumatology (Oxford) 39: 969-74.
- Grootscholten C, Bajema IM, Florquin S, Steenbergen EJ, Peutz-Kootstra CJ et al., (2007) Treatment with cyclophosphamide delays the progression of chronic lesions more effectively than does treatment with azathioprine plus methylprednisolone in patients with proliferative lupus nephritis. Arthritis Rheum 56: 924-37.
- Arends S, Grootscholten C, Derksen RH, Berger SP, de Sévaux RG et al., (2012) Long-term follow-up of a randomised controlled trial of azathioprine/methylprednisolone versus cyclophosphamide in patients with proliferative lupus nephritis. Ann Rheum Dis 71: 966-73.
- Dooley MA, Cosio FG, Nachman PH, Falkenhain ME, Hogan SL et al., (1999) Mycophenolate mofetil therapy in lupusnephritis: Clinical observations. J Am Soc Nephrol 10: 833-9.
- Chan TM, Li FK, Tang CS, Wong RW, Fang GX et al., (2000) Efficacy of mycophenolate mofetil in patients with diffuse proliferative lupus nephritis. Hong Kong-Guangzhou Nephrology Study Group. N Engl J Med 343: 1156-62.
- Appel GB, Contreras G, Dooley MA, Ginzler EM, Isenberg D et al., (2009) Aspreva Lupus Management Study Group. Mycophenolate mofetil versus cyclophosphamide for induction treatment of lupus nephritis. J Am Soc Nephrol 20: 1103-12.
- Mok CC, Tong KH, To CH, Siu YP, Au TC (2005) Tacrolimus for induction therapy of diffuse proliferative lupus nephritis: An open-labeled pilot study. Kidney Int 68: 813-7.
- Tam LS, Li EK, Leung CB, Wong KC, Lai FM et al., (1998) Long-term treatment of lupus nephritis with cyclosporin A. QJM 91: 573-80.
- Austin HA 3rd, Illei GG, Braun MJ, Balow JE (2009) Randomized, controlled trial of prednisone, cyclophosphamide, and cyclosporine in lupus membranous nephropathy. J Am Soc Nephrol 20: 901-11.
- Faul C, Donnelly M, Merscher-Gomez S, Chang YH, Franz S et al., (2008) The actin cytoskeleton of kidney podocytes is a direct target of the antiproteinuric effect of cyclosporine A. Nat Med 14: 931-8.
- Gordon S, Denunzio T, Uy A (2013) Success using tacrolimus in patients with proliferative and membranous lupus nephritis and refractory proteinuria. Hawaii J Med Public Health 72: 18-23.
- Mok CC, Ying KY, Yim CW, Siu YP, Tong KH et al., (2016) Tacrolimus versus mycophenolate mofetil for induction therapy of lupus nephritis: A randomised controlled trial and long-term follow-up. Ann Rheum Dis 75: 30-6.
- Bao H, Liu ZH, Xie HL, Hu WX, Zhang HT et al., (2008) Successful treatment of class V+IV lupus nephritis with multitarget therapy. J Am Soc Nephrol 19: 2001-10.
- Liu Z, Zhang H, Liu Z, Xing C, Fu P et al., (2015) Multitarget therapy for induction treatment of lupus nephritis: A randomized trial. Ann Intern Med 162: 18-26.
- Tanaka H, Watanabe S, Aizawa-Yashiro T, Oki E, Kumagai N et al., (2012) Longterm tacrolimus-based immunosuppressive treatment for young patients with lupus nephritis: A prospective study in daily clinical practice. Nephron Clin Pract 121: c165-73.
- Yap DY, Ma MK, Mok MM, Kwan LP, Chan GC et al., (2014) Long-term data on tacrolimus treatment in lupus nephritis. Rheumatology (Oxford) 53: 2232-7.
- Contreras G, Pardo V, Leclercq B, Lenz O, Tozman E et al., (2004) Sequential therapies for proliferative lupus nephritis. N Engl J Med 350: 971-80.
- Houssiau FA, D’Cruz D, Sangle S, Remy P, Vasconcelos C et al., (2010) Azathioprine versus mycophenolate mofetil for long-term immunosuppression in lupus nephritis: Results from the MAINTAIN Nephritis Trial. Ann Rheum Dis 69: 2083-9.
- Dooley MA, Jayne D, Ginzler EM, Isenberg D, Olsen NJ et al., (2011) Mycophenolate versus azathioprine as maintenance therapy for lupus nephritis. N Engl J Med 365: 1886-95.
- Sahin GM, Sahin S, Kiziltas S, Masatlioglu S, Oguz F et al., (2008) Mycophenolate mofetil versus azathioprine in the maintenance therapy of lupus nephritis. Ren Fail 30: 865-9.
- Feng L, Deng J, Huo DM, Wu QY, Liao YH (2013) Mycophenolate mofetil versus azathioprine as maintenance therapy for lupus nephritis: A metaanalysis. Nephrology(Carlton) 18: 104-10.
- Maneiro JR, Lopez-Canoa N, Salgado E, Gomez-Reino JJ (2014) Maintenance therapy of lupus nephritis with mycophenolate or azathioprine: Systematic review and metaanalysis. Rheumatology (Oxford) 53: 834-8.
- Gatto M, Kiss E, Naparstek Y, Doria A (2014) In-/off-label use of biologic therapy in systemic lupus erythematosus. BMC Med 12: 30.
- Merrill JT, Neuwelt CM, Wallace DJ, Shanahan JC, Latinis KM et al., (2010) Efficacy and safety of rituximab in moderately-to-severely active systemic lupus erythematosus: The randomized, double-blind, phase II/III systemic lupus erythematosus evaluation of rituximab trial. Arthritis Rheum 62: 222-33.
- Reddy V, Jayne D, Close D, Isenberg D (2013) B-cell depletion in SLE: Clinical and trial experience with rituximab and ocrelizumab and implications for study design. Arthritis Res Ther 15: S2.
- Wallace DJ, Kalunian K, Petri MA, Strand V, Houssiau FA et al., (2014) Efficacy and safety of epratuzumab in patients with moderate/severe active systemic lupus erythematosus: Results from EMBLEM, a phase IIb, randomised, double-blind, placebo-controlled, multicentre study. Ann Rheum Dis 73: 183-90.
- Navarra, Hall S, Levy RA, Jimenez RE, et al., (2011) Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: A randomised, placebo controlled, phase 3 trial. Lancet 377: 721-31.
- Merrill JT, Burgos-Vargas R, Westhovens R, Chalmers A, D’Cruz D et al., (2010) The efficacy and safety of abatacept in patients with non-life-threatening manifestations of systemic lupus erythematosus: Results of a twelve-month, multicenter, exploratory, phase IIb, randomized, double-blind, placebo-controlled trial. Arthritis Rheum 62: 3077-87.
- Ginzler EM, Wax S, Rajeswaran A, Copt S, Hillson J et al., (2012) Atacicept in combination with MMF and corticosteroids in lupus nephritis: Results of a prematurely terminated trial. Arthritis Res Ther14: R33.
- Rovin BH, Furie R, Latinis K, Looney RJ, Fervenza FC et al., (2012) Efficacy and safety of rituximab in patients with active proliferative lupus nephritis: The Lupus Nephritis Assessment with Rituximab study. Arthritis Rheum 64: 1215-26.
- Weidenbusch M, Römmele C, Schröttle A, Anders HJ (2013) Beyond the LUNAR trial. Efficacy of rituximab in refractory lupus nephritis. Nephrol Dial Transplant.28: 106-11.



