Research Article - (2022) Volume 7, Issue 1
Long Covid Syndrome: A Multi Organ Disorder
2Neuro-Cardiology Research Corporation, LLC, Wilmington, DE, USA
3Pennsylvania Hospital of the University of Pennsylvania Health System, Philadelphia, PA, USA
4CTO and Sr. Medical Director, Physio PS, Inc., Atlanta, GA, USA
Received Date: Mar 04, 2022 / Accepted Date: Mar 14, 2022 / Published Date: Mar 22, 2022
Copyright: ©Joe Colombo. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Nicholas L DePace, Joe Colombo (2022) Long-Covid Syndrome: A Multi-Organ Disorder. Cardio Open, 7(1): 204-215.
Abstract
Long-COVID Syndrome, a multi-organ disorder, that persists beyond 12 weeks post-acute SARS-CoV-2 Infection COVID-19. Here we provide a definition for this syndrome and discuss the organs that are involved. The involvement of multiple organs is due to that fact that angiotensin converting enzyme-2 receptors (the entry points for the virus), inflammation and oxidative stress (the immediate effects of the virus) effect all systems of the body. We suggest that this is then perpetuated by a resulting autonomic dysfunction. Since the autonomic dysfunction also effects all systems of the body, the initial infection is compounded and perpetuated by the resulting autonomic dysfunction underlying the Long-COVID Syndrome. We discuss the symptoms and suggest therapies that target the underlying autonomic dysfunction to relieve the symptoms, rather than merely treating symptoms. In addition to treating the autonomic dysfunction, the therapy also treats chronic inflammation and oxidative stress. To fully document the autonomic dysfunction, a full assessment of the autonomic nervous system is recommended, including Cardio-Respiratory Monitoring. Specific measurements of Parasympathetic and Sympathetic activity, both at rest and in response to challenges, connects all symptoms of Long-COVID to the documented autonomic dysfunction(s)
Introduction
Long-COVID, Multi-Organ Disorder, Autonomic Dysfunction, Inflammation, Oxidative Stress, Symptoms, Treat- ment, Quality of Life
Introduction
SARS-CoV-2 Infection COVID-19 is a major pandemic that is worldwide and causing significant mortality and morbidity. About 80% have mild to moderate disease. However, among the 20% with severe disease, 5% develop a critical illness. There is a subset of patients, however, who will have lingering, persistent or prolonged symptoms for weeks or month afterwards, which we termed “Post-Acute Sequelae of SARS-CoV-2 infection” syndrome or simply the “Post-Acute COVID-19 Syndrome” or "Long COVID-19" or just "Long COVID" or "Long Haul- ers COVID-19" or simply “Long Haulers” or “Post-Covid Syn- drome.” This has extended the significant worldwide morbidity from this pandemic. It is estimated that about 10% of patients who tested positive for SARS-COVID-19 will remain ill beyond three weeks and a smaller proportion for months. This is a sub- set that constitutes the Long-COVID syndrome. Globally, there are estimated over 200 million confirmed cases of COVID-19. Although the majority of infected individuals recover, we still do not know the exact percentage that will continue to experience symptoms or complications after the acute phase of the illness is over.
This prolonged phase with morbidity and ongoing symptoms creates significant burden to the patient and burden to the health- care system and is not completely understood. To complicate matters, not only do the long-term effects of those infected by the virus remain largely unknown, there are reports highlighting that sustained transmission and emergent variants continue to cause challenges to healthcare providers throughout the world, and therefore we do not know when the pandemic will cease. While it is estimated that 10% will develop a chronic syndrome, or symptoms that are persistent, this statistic may actually in- crease. Since this is a new illness, we do not know the cause or characteristics of the long-term sequelae of someone who has re- covered from acute COVID, not just the quality of life, including mental health, but the employment and productivity issues be- come paramount when the acute phase of COVID, the subacute, and the chronic phases occur. In our experience, approximately 20% of people will exhibit symptoms for more than five weeks and 10% will have symptoms for more than 12 weeks.
History
COVID-19 was reported in Wuhan, China in December 2019. It is caused by a small novel coronavirus. The acute phase of COVID-19 infected patients has been well described and may a have varying number of symptoms and intensity. The majority of patients have fever, sore throat, cough, shortness of breath, and chest pain. Although, multiorgan involvement may become extensive. COVID symptoms may be identified in six clusters [1]. These include:
1. Flu-like with no fever, which consist of headache, loss of smell or taste, cough, muscle pains, sore throat, chest pains.
2. Flu-like with fever, which consists of headache, loss of smell or taste, cough, sore throat, hoarseness, fever, loss of appetite.
3. Gastrointestinal, which consists of headache, loss of smell or taste, loss of appetite, diarrhea, sore throat, chest pain, but no cough.
4. Severe level one, fatigue with headache, loss of smell or taste, cough, fever, hoarseness, chest pain.
5. Severe level two, which consists of confusion with head- ache, loss of smell, loss of appetite, cough and fever, hoarse- ness, sore throat, chest pain, fatigue, and muscle pain.
6. Severe level three, which is abdominal and respiratory dysfunction with headache, loss of smell or taste, loss of appetite, cough, fever, hoarseness, chest pain, fatigue, sore throat, confusion, muscle pain, diarrhea, shortness of breath and abdominal pain. The recovery from COVID-19 usually occurs at seven to ten days after the onset of symptoms in mild disease, but could take up to six weeks in severe or critical illness. It is for this reason that mild cases are usually quarantined for between 7-10 days, and severe illnesses are for a more extended period of time. However, it is believed that even when one is ill for 3-6 weeks, they are probably not actively contagious. Some studies have shown that active coughing is indicative of continuing conta- giousness. This has not been clarified. Studies have shown that household cases support the highest incidences of contagious- ness and that rational for masks appears to be most beneficial with close contacts for prevention.
The most common feature of acute illness is interstitial pneu- monia, which may in some cases be complicated by the serious acute respiratory distress syndrome where individuals require high doses of oxygen. This has a high mortality particularly in elderly people who have comorbidities. The cough is usually dry. Laboratory abnormalities may be present and include low lymphocyte counts, elevated inflammatory markers, such as Sed Rate, C-reactive protein, Ferritin, Interleukin 1 and 6, and Tu- mor Necrosis Factor abnormalities, and others, which will be discussed later. Coagulation system abnormalities may occur (to be discussed later). Clots may form in the acute phase as well as in the subacute phase, especially if there is a history of clots.
Defining Long-Covid
What exactly is Long-COVID Syndrome? There are many defi- nitions that have been offered. Basically, there are individuals who do not completely recover over a period of weeks, usually 2-3 weeks. Since COVID-19 is a novel disease, there is still no consensus of the definition of Long-COVID-19 symptoms. In a study by Salamanna and coworkers, 20% of the reports of long- term COVID symptoms involved abnormal lung function, 24% involved neurological complaints and olfactory dysfunction, and 55% on specific widespread symptoms, mainly chronic fatigue and pain [2]. The World Health Organization (WHO) developed a clinical case definition of Long-COVID-19 by Delphi method- ology that included 12 domains. However, the understanding of this definition has been going through changes as new evidence emerges and we are gaining a better understanding of the conse- quences of COVID-19 and its mutations. Usually, three or more months past the acute COVID-19 infection, symptoms that last for at least two months and cannot be explained by alternate di- agnoses, may fit this definition. These symptoms include fatigue, shortness of breath, cognitive dysfunction, and symptoms that affect the functional capacity of individuals with daily living. Symptoms may fluctuate, flare up or relapse over time.
Long-COVID or Post-COVID-19 is an umbrella term that refers to symptoms persisting past the initial phase. In addition, Long- COVID-19 may adversely affect multiple organ systems, which include the kidneys, lungs, pancreas and heart. Unfortunately, the lack of a standardized definition for Long-COVID Syndrome presents obstacles for researchers in studying the condition with controlled studies and arriving at a precise diagnosis, algorithms and treatment algorithms. In addition, many patients with Long Covid Syndrome require rehospitalization especially those with comorbidities, such as cardiovascular disease, Diabetes Mellitus, obesity, cancer and kidney disease. There is data to suggest that the evolution of Long-COVID Syndrome is driven by chemicals produced in the body from inflammation called cytokines. Re- search is ongoing regarding pathogenesis that will be discussed later in this communication. Post-infection olfactory dysfunc- tion occurs in over 60% of those with acute SARS-CoV-2 in- fection, even in asymptomatic infection, and this represents an important frequent symptom of Long-COVID-Syndrome. Also, loss of taste has been seen. Many patients are not diagnosed with Long-COVID-Syndrome, when in reality their taste or smelling remains impaired as the only symptom.
Meta-analysis has shown the prevalence of Long-COVID man- ifestations. Fatigue and muscle weakness are by far the major symptoms followed by dyspnea and then pain and discomfort. Then there is anxiety and depression and impaired concentra- tion. Insomnia and sleep disorders present with lower frequency. Interestingly, Alopecia has been described as occurring. Chronic cough may persist and arthralgias and myalgias may be pres- ent. Chest pain, cognitive impairment, dizziness and headache are also symptoms but less common than the ones above. Per- sistent sore throat, palpitations, lack of smell, diarrhea, vomit- ing, fever, blurry vision, lack of taste, nasal congestion, anorex- ia, nausea, ringing in the ears and rash may be present but at a much lower prevalence. Interestingly, autonomic clusters in the Long-COVID-Syndrome (by COMPASS 31, which is a ques- tionnaire for autonomic symptoms) have identified two clusters. Impaired visual activity and blurry vision were more frequently registered during the acute phase in patients, while depression, chills, weakness, diarrhea, musculoskeletal, palpitations, tachy- cardia, dryness, cognitive dysfunction, headache, dizziness and tinnitus were more frequently observed in the Long-COVID second cluster. Levels of antibodies to the SARS virus were not different between two clusters. This has given us some insight into one of the main mechanisms of Long COVID Syndrome, mainly autonomic dysfunction, which is an imbalance between the Parasympathetic and Sympathetic nervous systems. It is be- lieved that a chronic inflammatory process, or autoimmune, or even a hormonal imbalance as a consequence of alterations in the hypothalamus-pituitary- adrenal axis may also be operative in conjunction with this acquired autonomic dysfunction with constitutional symptoms as described above.
The term Long-COVID syndrome was first used by Perego and social media to describe symptoms weeks or months after the initial SARS-CoV2 infection [3]. The term "long haulers" was used by Watson and by Yong [3-5]. There may be persistence of one or more symptoms of acute COVID or appearance of new symptoms and they may be relapsing and remitting; therefore, showing the variable presentation of Long-COVID-Syndrome. Interestingly, the majority of people with Long-COVID-Syn- drome are PCR negative including any microbiological recov- ery. It is interesting how many people continue to see infectious disease doctors and seek out other alternative infection diagno- ses when serology is continuously negative. Long-COVID-Syn- drome or Long-COVID has been defined as the time lag between the microbiologic recovery and clinical recovery [6].
In fact, the majority of those with Long Covid Syndromes have biochemical and radiological recovery. It has been postulated that there are two stages of Long-COVID-Syndrome: 1) symptoms that extend beyond three weeks, but less than 12 weeks, which is more of a subacute phase; and 2) chronic COVID symptoms that extend beyond 12 months. An interesting diagram and timeline have been postulated, which shows that Short-COVID will generally last less than three weeks from onset of symptoms [7].
Post-acute COVID, or subacute COVID will last from onset of symptoms approximately up to 10-12 weeks and chronic COVID will last from onset of symptoms beyond 12 weeks. See Figure. It would make sense to group the Post-Acute or Subacute COVID, which lasts from up to 10-12 weeks, and then chronic COVID, which lasts more than 12 weeks as Long COVID Syn- dromes. In this review, however, we are more concerned with those symptoms that last more than 12 weeks, the true long- or prolonged-COVID Syndrome Sometimes individuals are fairly asymptomatic, and by the time they develop Long-COVID symptoms, we do not know when the initial infection occurred nor are we certain. They did not undergo any test to confirm SARS-CoV2. When these individu- als develop multiple symptoms consistent with a Long-COVID Syndrome, we oftentimes consider them as probable, or possible Long COVID Syndrome, since we cannot confirm that they in- deed did have COVID exposure since they were asymptomatic. With individuals who were symptomatic but not tested it is more likely that this is a Long-COVID-Syndrome.
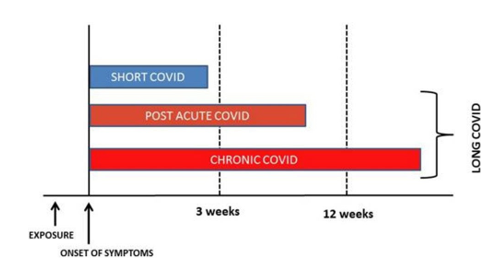
Figure : Classification of long COVID [7].
The problem is not only in those who have persistent symptoms who have never tested positive for COVID, but similarly in individuals who had upper respiratory tract infections and had a negative COVID test and then developed long prolonged symptoms. The question must be asked, “Did they have a false-negative test performed too early or too late in the disease course?” Antibodies are unreli- able as up to 1/5 patients do not seroconvert, and antibody levels decrease over time and by three months oftentimes are not mea- surable. Not only is the morbidly important with Long-COVID- Syndrome, but the economic cost to society, as 1/3 people in one survey did not return to their job for up to three weeks after being COVID-positive. Further, this is more common in older people. Not returning to work is often associated with obesity and psychiatric disease. In addition, given the similarity between Long-COVID Syndrome and Autonomic Dysfunction, and the ability of psychosocial stress to trigger symptoms of Autonomic Dysfunction further blurs the distinction between Long-COVID and Autonomic Dysfunction.
Of all the risk factors for individuals that develop the Long Covid Syndrome, it is noted that the risk of COVID is twice as common in women as it is in men. Increasing age is also a risk factor for development of Long-Covid Syndrome. Studies show that individuals who develop Long Covid Syndrome are four years older than those who did not [8]. Patients with five or more symptoms in the acute phase of illness, have a higher likelihood of developing a Long Covid Syndrome. We have em- pirically found that obesity and increased body mass index is an extremely important risk factor as is female gender. Symptoms that various studies report to be predictors of Long-COVID- Syndrome include diarrhea, anosmia, dyspnea, pleurisy, skin sensitivity, and A blood type [9]. One study showed that a lower SARS-CoV2 IgG titer at the beginning of the observation pe- riod was associated with a higher frequency of Long-COVID- Syndrome [10]. Also, the severity of acute COVID-19 suggests that the recovering critically ill patient commonly experiences long-lasting mental health issues that include depression, anx- iety, post-traumatic stress, memory disorders, attention deficit disorders, and ongoing brain fog. This is controversial as others have shown that acute disease was not associated with Long- COVID-Syndrome. All of this negativity speaks to a heteroge- nous presentation of Long-COVID-Syndrome with risk factors including its time-line.
Even if the virus is cleared, there were high neutralized anti- body titers that suggest that the immune system could continue to be overactive and induce this syndrome. Therefore, the ques- tion of autoimmunity is raised. This is especially true since viral shedding has shown that persistent fragments of viral genes and body secretions may cause hyperimmune responses, and this may explain some of the persistent symptoms in Long-COVID- Syndrome. Although, lingering Autonomic Dysfunction also presents with auto-immune-like symptoms. Meta-analysis of prolonged COVID-19 looking at age, gender, comorbidities, ethnicity and severity of acute disease confirm that female gen- der and Long-COVID risk are associated. In addition, increas- ing age, minority/ethnicity was also associated with Long Covid Syndrome. However, some of these results have been inconsis- tent [11]. Studies have shown that Long-Covid Syndrome effects with previously hospitalized and non-hospitalized patients are similar. Perhaps, the best-known study from the Office for Na- tional Statistics (ONS) study including controlled participants reports that the most common symptoms persisting for 12 or more weeks include fatigue (8.3%), headaches (7.2%), cough (7%, myalgias (5.6%), [12].
A lack of case-controlled studies presents a direct attribution of symptoms that are solely related to COVID-19; however, larg- er perspective studies with match-controlled groups are needed. Similarly, many of the studies and statistics on Long Covid Syn-drome are based on Long-COVID cohorts produced in Western Europe on patients recently discharged from the hospital. As a result, there was not enough information on Long-COVID-19 patients in low-to middle income countries and people who were not hospitalized. Also, there are no large studies that focus on children, specifically. This is despite the fact that young children are affected and may be affected by Long-COVID.
Multiple Organ Systems Involved
The Long-COVID-Syndrome is a complex condition with pro- longed heterogenous symptoms that are related to multiple organ involvement. Long-COVID-Syndrome involve hyperinflamma- tory and hypercoagulable states that affect all organ systems. It reflects a maladaptation of the Angiotensin Converting Enzyme 2 (ACE 2) pathway. ACE-receptors are present in virtually every organ system. Therefore, it is understandable that many organ systems are susceptible to involvement from direct viral infec- tion and inflammation. As previously mentioned, depending on duration of symptoms, Long-COVID Syndrome may be divided into two stages post-acute COVID, or symptoms extending be- yond three weeks but less than 12 weeks, and chronic or Long- COVID where symptoms extend beyond the 12 weeks [13].
Multiple organ sequelae and abnormalities of COVID-19 that may go beyond the acute phase of infection are increasingly be- ing noted as data are being collected. Assessment of key clinical, serological, and imaging features are important to understand the natural history of this disease as it progresses beyond the acute viral infection phase. Workup consisting of pulmonary function tests, chest X-rays, six-minute walk tests, pulmonary embolism workups (when needed), echocardiograms (even se- rially), and (at times) high resolution CT scans (to assess for fi- brosis) should also be considered. These will be discussed under the Pulmonary section. Hematological assessment may lead to extending thromboprophylaxis against clots and high-risk sur- vivors. A neuropsychiatric screening for anxiety, posttraumatic stress, sleep disorders, depression, cognitive impairment, mem- ory abnormalities and other factors associated with brain fog is important. A neuropsychiatric screening should include a full au- tonomic dysfunction test, especially in patients with orthostatic intolerance symptoms and chronic fatigue syndromes. Amnesia, sore throat, hoarseness, runny nose, hair loss, hearing loss and tremors have been reported at low incidences in Long-COVID Syndrome [14]. If there are renal function abnormalities, Ne- phrology follow-up and creatinine clearance determination with urinalysis evaluation may be needed. These may be performed in person or on virtual clinical visits. Next is a discussion of some of the various organ systems and how they are affected.
Pulmonary System
The pulmonary system is the most commonly involved. Chronic complications, such as chronic cough, fibrotic lung changes also known as Long-COVID fibrosis or post-ARDS fibrosis, bron- chiectasis and pulmonary vascular disease may occur [15]. Even if a person is asymptomatic, they may have CT scan abnormal- ities that are seen many months after infection has resolved. If COVID-19 leads to pulmonary fibrosis it may result in shortness of breath and the need for supplemental oxygen. In a post-acute COVID-19 Chinese study approximately 50% of 349 patients who underwent high resolution computer tomography of the chest at six months had at least one abnormality pattern [16]. These changes involved the appearance of ground-glass opaci- ties. There are also long-term risks of pulmonary embolisms and chronic pulmonary hypertension, but at this point it is unknown and further research is being conducted. It is thought that pa- tients with a greater severity of acute COVID, especially those on high-flow nasal cannula and noninvasive or invasive ventila- tion are at the highest risk for long-term severe pulmonary com- plications that include even pulmonary function test abnormali- ties such as persistent diffusion impairment. Pulmonary vascular micro-clotting or micro thrombosis has been observed in 20% to 30% of patients with COVID-19 [17,18].
Persistent lung symptoms are to be expected in many patients with Long-COVID Syndrome because the lungs appear to be one of the main organs affected by the virus. This may result in
(1) diffuse damage to the alveoli, the lining of the small sacs in the lungs,
(2) desquamation of alveolar epithelial type 2 cells,
(3) pulmonary fibrosis,
(4) Hyaline membranes,
(5) scattered interstitial inflammation, and
(6) monocytes and macrophages [19].
Up to six months after hospitalization, pulmonary function abnormalities or structural changes may occur. In severe COVID-19 patients, up to 35% of patients at six months may show fibrotic-like changes after infection [20]. Abnormal airway function may occur up to 11 months in severe COVID-19 in- fections [21]. Persistent diffusion lung carbon monoxide impair- ment also described the noncritical COVID patients who present with shortness of breath may occur up to four months after infec- tion [22]. Other abnormalities include abnormalities in total lung capacity, poor expiratory Vol at 1 second (FEV1), forced vital capacity (FVC), FEV1 to FVC ratio, and small airway function abnormalities [23]. Mild cases usually have persistent chronic cough, which may be due to fibrosis, bronchiectasis and pulmo- nary vascular disease. Individuals also have oxygen desaturation during exertion compared to individuals who have no symptoms of shortness of breath post-COVID [24].
Cardiac Involvement
Common cardiac problems may occur with labile heart rate and blood pressure response to activity. Myocarditis and pericarditis may occur chronically. In the acute stages, myocardial infarc- tion, cardiac failure, life-threatening arrhythmias and sudden cardiac death have been described. The incidents of arrhythmias in Long-COVID-Syndrome is unknown, but many individu- als have palpitations and studies using ambulatory monitoring need to be further conducted. Sequelae from acute COVID may occur, such as coronary artery aneurysm, aortic aneurysm, ath- erosclerosis, venous and arterial thrombotic disease including life-threatening pulmonary embolism [25]. These structural ab- normalities may manifest itself in Long-COVID Syndrome long after recovery of acute illness and predispose to arrhythmias, breathlessness, and acute coronary events, such as heart attacks and chest pain syndromes.
Myocardial injury is the most common abnormality detected with acute COVID infection. It is usually detected even when patients are asymptomatic with no cardiac symptoms with el- evated cardiac troponin levels, which may be evident in a high percentage of patients with COVID-19 [26]. Further research is ongoing as to whether this myocardial injury pattern, even when subclinical, may lead to increased arrhythmias and heart failure in the long-term. Echocardiographic studies have shown abnor- malities with COVID, including right ventricular dysfunction 26.3%, left ventricular dysfunction 18.4%, diastolic dysfunction 13.2% and pericardial perfusion 7.2%. To what extent this is re- versible in patients who go on to Long-COVID-Syndrome is not known [27]. In addition, sleep abnormalities and difficulties that reduce quality of life have been noted in Long-COVID-19 Syn- drome patients. These may also adversely affect cardiac func- tion, provoke arrhythmias, elevate blood pressure and exacer- bate or cause hypertensive states.
Chest pain and palpitations are status post-acute phase of COVID. In a Chinese study, palpitations were reported in 9% and chest pain in 5% of patients six months after follow-up [16]. To track heart inflammation, one of the most effective and sensi- tive tests is cardiac magnetic resonance imaging (MRI). Inflam- mation rates may be as high as 60% more than two months after a diagnosis of COVID, although this is a very difficult test to obtain in many centers that do not have it readily available [28]. Long-COVID Syndrome patients may be present with chest pain in 17% of patients, palpitations in 20% of patients, and dyspnea on exertion 30% of patients [27]. The question of myocarditis is always raised especially in children, but adults are also known to have myocarditis and this may be smoldering. One study showed that in healthy collage athletes the mild symptoms were asymptomatic up to 15%, and they have evidence of MRI find- ings consistent with myocarditis on a screening study [29]. More importantly, many of the chest pains and palpitations, which ap- pear to be cardiology in etiology, are actually due to autonomic dysfunction, including the postural orthostatic tachycardia state. Therefore, the importance of not only doing cardiac imaging, ambulatory monitoring, stress testing, six-minute walk test, echocardiography and other noninvasive cardiac workup, but also autonomic testing, such as cardio-respiratory monitoring, HRV interval testing, beat-to-beat blood pressure with tilt testing and sudomotor testing may be useful in diagnosing autonomic nervous dysfunction. Arrhythmias are noted Long-COVID but attention to the use of anti-arrhythmic drugs, Amiodarone for example, must be used carefully in patients who have fibrotic pulmonary changes after COVID-19 [30].
Neurological
Encephalitis, seizures, and other conditions including prolonged brain fog may occur for several months after acute COVID in- fection [31]. Three months after follow-up MRI, abnormalities may be demonstrated [32]. Neuropsychiatric sequelae are often common and reported with many post-viral symptoms, such as chronic tiredness, myalgias, depressive symptoms, non-restor- ative sleep [33]. Migraine headaches, often refractory to treat- ment, and late-onset headaches have been presumed to be due to high cytokine levels [34]. In a follow up study of 100 patients, 38% had ongoing headaches after six weeks [35]. Loss of taste and smell may also persist for up to six months and longer on follow-up of patients. Brain fog, cognitive impairment, con- centration, memory difficulties, receptive language, executive function abnormalities may also persist over a long period of time. This may be related to autonomic dysfunction and other factors [36,37]. Psychiatric manifestations are also common in COVID-19 survivors of more than one month. In an Italian study, approximately 15% have at least some evidence of de- pression and post-traumatic stress, anxiety, insomnia and obses- sive-compulsive behavior [38]. Some studies have shown up to 30-40% of patients with COVID-19 have depression and anxiety for prolonged periods of time [39].
After ischemic or hemorrhagic strokes, hypoxic and endemic damage, may lead to reversible encephalopathy syndrome and acute disseminated myelitis. These and other neurological de- fects may require extensive rehabilitation and their sequelae may linger. Also, acute critical illness, myopathy and neuropathies resulting during acute COVID-19 may leave residual symptoms weeks to months later [40]. Again, Long-COVID often involves brain fog. This may involve mechanisms of cardiac decondition- ing, post-traumatic stress or dysautonomia. Long-term cognitive defects may be seen occurring in up to 20% - 40% of patients [41-43]. The association between Long-COVID-Syndrome and brain fog may be the result of the autonomic dysfunction, spe- cifically Sympathetic Withdraw, and the associated decreased cerebral perfusion [44,45].
Autonomic dysfunction has been noted to be significant. Patients with orthostatic tachycardia and inappropriate sinus tachycardia may benefit from heart rate management including beta-block- ers and other autonomic therapies [personal observations] [46]. Muscle wasting and fragility are often seen prolonged. This is because COVID-19 when it is severe may cause catabolic mus- cle weakness and feeding difficulties [47]. Neuropsychiatric manifestations of COVID-19 have been documented in a British study including stroke and altered mental status. Multiple psy- chiatric symptoms emanating from encephalopathy and enceph- alitis and primary psychiatric diagnoses were noted in young adults [48]. Prolonged effects on patients who have had prone ventilation and COVID may cause focal and multifocal periph- eral nerve damage [49]. Symptoms consistent with orthostatic hypotension syndrome and painful small fiber neuropathy were reported in as short as three weeks and as long as three months [50-53].
The most frequent neurological long-term symptoms in patients were myalgias, arthralgias, sleeping troubles and headaches [54]. Autonomic dysfunction findings resulting from analysis of Compass-31 Questionnaires demonstrates significant symptom- atology [55]. Based on autonomic symptoms by Compass-31, two clusters were obtained with differential characteristics. Clus- ter 2 exhibited high scores of Compass-31. This accounted for 31% of all patients included. Median Compass-31 score of 22 suggesting that one third of patients with PCS may yield higher scores when compared with the general population [56]. Patients with higher scores exhibited more clinical manifestations and depression. These clusters have therapeutic implications and cli- nicians should be aware of particular manifestations during the follow-up and all psychosocial intervention may reduce the bur- den of Long-COVID Syndrome [57]. Orthostatic intolerance has been described from a series of patients that had either resting or postural hypotension or tachycardia [50]. Orthostatic intolerance symptoms presented from orthostatic hypotension, vasovagal syncope, and postural orthostatic tachycardia syndrome that all occur and last for a prolonged period of time after COVID viral infection. The Symptoms are often associated with palpitations, breathlessness and chest pain, which are common symptoms seen in Long-COVID patients. It is postulated that high cate- cholamine levels may lead to paradoxical vasal dilatation and increased activation of the Vagus nerve that may result even in syncope and also sympathetic activity withdraw [58,59].
Hypovolemia may also exacerbate or worsen these symptoms. The relationship between COVID-19 infection and how it affects the autonomic nervous system is not completely understood, but it is believed to be due to inflammatory cytokine release and a cytokine storm, which results from the initial sympathetic acti- vation produced by a pro-inflammatory cytokine release [60]. However, vagal stimulation (i.e., in coordination with the im- mune response to COVID-19) may also result in anti-inflamma-tory responses [61]. The COVID-19 relationship to autonomic dysfunction may be by the viral infection itself. Immuno-medi- ated neurological syndromes have been described [62]. It is well known that some forms of orthostatic hypotension and POTS are associated with autoantibodies perhaps caused by other viruses in the past, and this may also be the case with COVID; for exam- ple, antibodies against Alpha and Beta and muscarinic receptors.
Treatment of these autonomic nervous systems disorders in- volves exercise with both aerobic and resistant elements in a graded fashion that oftentimes begin with recumbent exercises, “low and slow” [63], Fluid and electrolyte repletion is required. Avoiding exacerbating factors, such as prolonged sitting and warm environments is recommended. Some counter maneuvers and isometric exercises, compression garments especially up to the waist, or abdominal binders are recommended. Pharmaco- logical treatment that may involve many different regimens may be prescribed, such as volume expanders (e.g., Fludrocortisone or Desmopressin) may be used along with vaso activation (e.g., Midodrine or Mestinon). If there are prominent hyperadrener- gic symptoms, Propranolol, Clonidine, Methyldopa or other be- ta-blockers may be considered, especially for a postural ortho- static tachycardia response.
Again, the questionnaire that addresses autonomic symptoms, such as the Composite Autonomic Symptom Scale-31 (Com- pass-31), may be used to validate dysautonomia symptoms in post COVID patients. In a study of 180 participants (70% males), orthostatic hypotension was found in 13.8% of the sub- jects [64]. The mean Compass-31 score was 17.6 with the most affected domains being orthostatic intolerance, sudomotor, gas- trointestinal, and pupillomotor dysfunction. Higher Compass-31 scores were found in those with neurological symptoms due to more severe orthostatic intolerance symptoms. Interestingly, gastrointestinal, urinary and pupillomotor domains were more represented than non-neurological symptom groups. Therefore, this study concluded that it is important to monitor autonomic nervous system symptoms as a complication of COVID-19 dis- ease that may persist in a post-acute period. Other prominent au- tonomic dysfunctions in these post-COVID individuals included secretomotor and sweating abnormalities in about half the study population and thermoregulatory alterations.
Another feature of COVID-19 involves the urinary tract and bladder incontinence. Urinary symptoms may occur with in- flammation and demyelination of the pudendal nerve. This has been reported in other viral infections [65,66]. Up to a half of Long-COVID Syndrome patients may have urinary dysfunction [64]. It is also suggested that sexual impairment such as erectile dysfunction is possible after COVID infections which may af- fect both endothelial and autonomic components. However, the psychiatric component due to stress and anxiety cannot be sep- arated [67,68]. Compass-31 scoring highlights gastrointestinal symptoms after COVID. These may occur along with a combi- nation of other symptoms, including abnormal appetite, nausea and diarrhea. This is because in the GI tract there is a signifi- cant amount of angiotensin coenzyme ACE-2 receptors present. [69]. Vision disturbances may also occur in Long-COVID syn- dromes including sore eyes and light sensitivity [70]. In acute conjunctivitis, an abnormal pupil response has been noted [71]. Light sensitivity is often a common complaint. Nearly 50% of study patients complained of ocular abnormalities [64]. There- fore, multiple organ systems may manifest symptoms in Long- COVID syndrome which may be attributed to the autonomic dysfunction, and this need not necessarily just be orthostatic intolerance symptoms [72]. As noted, GI, ocular, urological, su- domotor, and endocrine organs may be affected.
Dysautonomia, including anosmia, hypogeusia, headaches and hypoxia has been shown to be components of the Long-COVID syndrome [73]. The dysautonomia is hypothesized to occur sec- ondary to COVID-19 infection. The demonstrated dysregulation of heart rate variability (HRV) testing is reflected in abnormal Nociception Level (NOL) index; an index that refers to abnor- mal parasympathetic-sympathetic balance. The NOL algorithm with a multiparameter, non-linear combination of (1) heart rate, (2) HRV, (3) amplitude of the finger, photoplethysmography, skin conduction level, and (4) time derivatives were obtained using a noninvasive finger probe [73]. Autonomic dysfunction has been reported in up to 63% of patients presenting with spe- cific symptoms having survived a COVID-19 infection between March 2020 and January 2021. Diagnoses were revealed by recording and evaluating beat-to-beat blood pressure and heart rate during head-up tilt [74]. As long as six to eight months after recovery from COVID-19, patients still suffer residual autonom- ic symptoms and 60% are unable to return to work and only 50% have completely recovered [75].
Chronic fatigue syndrome, also known as post-infective syn- drome, has been commonly recognized in the Long-COVID Syndrome. A study of 20 fatigued and 20 non-fatigued Long- COVID patients with a mean age 44.5 (90% female) were fol- lowed up for 166 days. A scale known as a Chalder Fatigue Scale was used. They underwent an Ewing autonomic function test battery, including deep breathing, active standing, Valsalva and cold-pressure testing, with continuous electrocardiogram, blood pressure monitoring, and cerebral oxygenation. A 24-hour am- bulatory blood pressure monitoring was conducted. Findings included no differences between the fatigued and non-fatigued patients in autonomic testing. There was an that increase in anx- iety, strongly associated with Chronic Fatigue Syndrome. This, however, is in contradistinction to other studies which have shown autonomic dysfunction with Long-COVID syndrome and chronic fatigue [76].
Post infectious Myalgic Encephalomyelitis, which is also syn- onymous with Chronic Fatigue Syndrome, has been recognized along with sleep disturbances, neurocognitive changes, ortho- static intolerance and post-exertional malaise [77,78]. Fatigue at three weeks post symptoms may occur in 13-33% of patients. There are many factors responsible which include sleep distur- bances, autonomic dysfunction with sympathetic predominance, endocrine disturbances, abnormalities of the hypothalamic-pi- tuitary-adrenal axis, reactive mood disorders and depression and anxiety. Findings therefore concluded that chronic fatigue post Long-COVID Syndrome is multifactorial. More testing is required to see the association of autonomic disturbances with the emergence of a chronic fatigue syndrome. This has been demonstrated with other viral infections. In fact, in the cohort at our autonomic clinic we have found significant disturbanc- es in Cardiorespiratory and heart rate variability testing in pa- tients with chronic fatigue with Long-COVID-Syndrome with abnormal autonomic responses, including sympathetic withdraw (associated with orthostatic dysfunction) and vagal excess with postural change (associated with pre-syncope symptoms). Both autonomic dysfunctions are associated with poor cerebral and possibly coronary perfusion. These symptoms present regardless of whether they have drops in blood pressure, postural rise in heart rate or none of the above changes.
It has been widely reported that autonomic dysfunction and var- ious viral infections including HIV, Herpes virus, enterovirus 71, flavivirus, Human-T lymphotropic virus, etc. may result in loss of heart rate variability, hypersalivation, photophobia, and dyspnea. Epstein-Barr virus may lead to dysautonomia through alterations of cerebral autoregulation by high nitric oxide release with subsequent autonomic nervous system dysfunction [79]. It has been postulated that the coronavirus shares a common fea- ture of neuroinvasion [80].
SARS-CoV-2 may also reach the autonomic nervous system
1. by employing the retrograde axonal transport by the olfac- tory nerve [81].
2. via ACE 2 receptors and brain stem pathways [82].
3. via systemic blood circulation [83].
4. by immune injury [84], and
5. neuronal pathways [85].
The virus may also indirectly invade the autonomic nervous sys- tem by the enteric nervous system (ENS) and its sympathetic afferent neurons by infecting the GI tract [86,87]. In a study of 50 patients showed that 26% of patients had sweat dysfunction as measured by SUDOSCAN test with motor, sensory and auto- nomic dysfunction [88]. Abnormalities of heart rate variability is more common in COVID-19 patients who develop fatigue, Long-COVID-19 is found to be associated with persistent symp- toms, such as hypoxia and fatigue due to development of dysau- tonomia [89-91]. It is initially believed that SARS-COVID-19 causes sympathetic nervous system activation with catechol- amine excess of activation in a sympathetic storm which acti- vates the renin angiotensin system. Simultaneously, there is inhibition of the parasympathetic nervous system mediated an- ti-inflammatory effect, that leads to a decrease in neuro-vagal anti-inflammatory response and enhances the cytokine storm. This all leads to cardiopulmonary complications and COVID- 19-induced dysautonomia [87]. The Parasympathetic responses reported above are found at rest. There is an abnormal Parasym- pathetic response to stress that may also occur, exaggerating the dynamic Sympathetic response to stress, thereby amplifying and prolonging the Sympathetically-mediated inflammatory, hista- minergic, pain, and anxiety responses in a post-traumatic-like fashion [92].
Gastrointestinal Symptoms Associated with Long- Covid Syndrome
COVID-19 may cause intestinal dysfunction due to changes in intestinal microbes and an increase in inflammatory cytokine. Multiple organ systems may evolve acutely and may even pro- duce septic shock. Increasing diarrhea may be an early mani- festation that may continue in the chronic phase. In the chronic phase the Gastrointestinal (GI) sequelae may include persistent anorexia, vomiting, abdominal pain along with diarrhea. It ap- pears gastrointestinal (GI) symptoms are accompanied by in- flammation or intestinal damage. Coronavirus has also been found in the stool [93]. Acute hepatocellular injury was also identifying with elevated liver enzymes. Fecal oral transmis- sion of COVID-19 may be suspected because of the presence of COVID-19 RNA in stool samples of COVID-19-positive pa- tients [94]. GI upset may also be due to gastric and intestinal motility dysfunction due to COVID-induced Parasympathetic dysfunction. For example, diarrhea and abdominal pain may be due to excessive Parasympathetic activity causing overly-rapid GI motility.
At least 76% of patients had one of the GI sequelae symptoms 90-days after discharge of COVID-19, persisting to six months after disease onset [95]. Only 13% of patients recorded GI symptoms on admission and 49% during hospitalization. After discharge, GI symptoms were present in 44% of patients. The most common GI sequelae were loss of appetite at 24%, nausea 18%, acid reflux 18%, and diarrhea 15%. Less common symp- toms were abdominal distention 14%, belching 10%, vomiting 9%, abdominal pain 7% and bloody stools 2%. Patients who had GI symptoms going beyond 90 days more often had shortness of breath and myalgia as coexisting presenting symptoms in the acute phase. Interestingly, patients with GI sequelae were less frequently severely ill than those without GI sequelae 17% ver- sus 37%. These patients with GI sequelae appeared to have a lower frequency of supplemental needs in the long-term also. Also, patients with GI sequelae at 90 days were treated more often with proton-pump inhibitor and corticosteroids and were less frequently treated with enteral nutrition. This suggests that there are different mechanisms in Long-COVID Syndrome for GI abnormalities versus pulmonary or cardiac. GI symptoms six-months after coronavirus showed that 29% of 220 patients reported GI symptoms six months after COVID-19; 9.6% had diarrhea, 11% had constipation, 9.4% abdominal pain, 7.1% nausea and vomiting and 16% with heartburn [96].
A GI symptom was the most bothersome recurrent symptoms in 83 patients, 11%. Many symptoms resembled Irritable Bowel Syndrome. There were 39% who met criteria for the Rome IV Criteria for Irritable Bowel Syndrome, and women were much more likely to report this than men. These symptoms were not trivial as almost 10% of patients reported this as their most sig- nificant symptom [96]. Studies are currently evaluating the long- term consequences of COVID-19 on the GI system, including postinfectious, Irritable Bowel Syndrome, and Dyspepsia.
Hematological Sequelae of Long-Covid Syndrome
Long-COVID venous thromboembolism has been estimated to be less than 5%. A single center report of 163 patient in the United States without post-discharge thrombotic prophylaxis suggested a 2.5% cumulative incidence of thrombosis at 30 days following discharge. This included segmental pulmonary embo- lism, intracardiac thrombus, thrombosed AV fistulas and isch- emic stroke [97]. Late onset hematological complications with Long-COVID-19 has become an emerging medical problem for the hematologists. These include coagulopathy disorders, immune-thrombotic states, and hemorrhagic events. Late onset thrombocytopenia related to immune system dysregulation has also been reported as a rare complication of COVID-19 [98].
Kidney Disease
Patients that are extremely frail and have chronic comorbidities are at an increased risk for kidney disease and progression of kidney failure after infection of SARS-CoV-2. The kidney func- tion gradually declines over time even in the absences of the virus infection [99]. Initially, approximately 28% of patients hospitalized with COVID are diagnosed with acute kidney inju- ry and up to 9% have shown a received kidney replacement ther- apy. A study of 26 autopsy patients who died from COVID-19 were reported in China. Clusters of coronavirus-like particles were noted in epithelial cells and diffuse proximal tubal injury was demonstrated [100]. Others found that older-aged diabetes, cardiovascular disease, black race, hypertension, and need for ventilation and vasopressor medications are at risk for acute kid- ney injury in COVID-19 [101].
Olfactory and Gustatory Abnormalities
Recovery of the Olfactory and Gustatory system may last more than one month after the onset of smell and taste loss [102,103]. Loss of olfactory receptor neurons may be the mechanism in some of these cases. Long-lasting effects on taste and smell are uncommon but have been noted in isolated cases.
Pathophysiology of Long Covid-19
The mechanism behind the causation of Long-COVID Syndrome may be multifactorial. As mentioned earlier, immune response, antibody generations, direct effects of the virus, complications of the critical illness, psychosocial factors and post-intensive care syndrome, post-traumatic stress and oxidative stress may be operative mechanism. Deconditioning may also be a factor [7]. The mechanism of heart failure as another organ system in- volved pro-inflammatory cytokines with interleukin 1 and inter- leukin 6 tumor necrosis factors, and these may cause prolonged effects [104]. Redox imbalance linking COVID-19 and chronic fatigue syndromes and systemic inflammation and neuroinflam- mation have also been postulated [105]. Oxidative phosphoryla- tion may be operative in a hyperinflammatory state with altered cardiorespiratory function. It is thought that viral infections cause a shift in Mitochondrial energy system contribution from ATP synthesis to innate immune signaling occurs in order to eradicate pathogens, promote inflammation, and eventually re- store tissue homeostasis [106]. An increased rate of glycolysis and down-regulation of oxidative phosphorylation is seen [107]. Oxidative stress has been implicated in many acquired myocar- dial disorders and may lead to significant autonomic dysfunction [108].
Treatment in Long-Covid Syndrome
For the most part, supportive therapy for Long-COVID symp- toms is a keystone and there is treatment for autonomic dys- function that may be demonstrated objectively in a laboratory. As mentioned earlier, volume expanders and vasodilators in ad- dition to fluids, electrolytes, compression garments, and various exercise techniques have been prescribed for orthostatic intol- erance symptoms. Omega-3 fatty acid and dietary supplemen- tation have been investigated [109]. It is believed that Omega-3 may help resolve inflammatory imbalance. L-arginine has also been proposed as a treatment modality [110]. Included in the L-arginine pathway is the production of Nitric Oxide. Nitric Ox- ide maintains or improves the health and function of endothelial cells and benefits the immune system, especially in chronic fa- tigue states. Various antioxidants and zinc have also been used empirically, but there are no controlled studies to confirm their utility. All of these therapies also effect proper autonomic func- tion to help relieve Long-COVID symptoms [111]. Vaccination has been suggested as possibly a factor that may ease symptoms of Long-COVID. In one large survey, 57% of responders report- ed an overall improvement in their symptoms following vacci- nation and around 19% reported an overall deterioration [112]. Mental health conditions may be treated with various psycho- logical aides, such as cognitive behavioral therapy as well as antidepressants, including tricyclics. Treatment of liver function abnormalities Irritable Bowel Syndrome, dyspepsia and other GI symptoms are very challenging. Renal dysfunction should be followed serially, nephrotoxins avoided and proper hydration maintained. However, from a large study of fully vaccinated people 2.6% were found to contract the virus (labeled as “Break- through” patients) [113]. Among 1497 fully vaccinated health care workers for whom RT-PCR data were available, 39 SARS- CoV-2 breakthrough infections were documented. Neutralizing antibody titers in case patients during the peri-infection period were lower than those in matched uninfected controls (case-to- control ratio, 0.361; 95% confidence interval, 0.165 to 0.787). Higher peri-infection neutralizing antibody titers were associat- ed with lower infectivity (higher Ct values). Most breakthrough cases were mild or asymptomatic, although 19% had persistent symptoms (>6 weeks). The B.1.1.7 (alpha) variant was found in 85% of samples tested. A total of 74% of case patients had a high viral load (Ct value, <30) at some point during their infec- tion; however, of these patients, only 17 (59%) had a positive result on concurrent Ag-RDT. No secondary infections were documented. Among fully vaccinated health care workers, the occurrence of breakthrough infections with SARS-CoV-2 was correlated with neutralizing antibody titers during the peri-infec- tion period. Most breakthrough infections were mild or asymp- tomatic, although persistent symptoms did occur.
LONG-COVID
Cardiopulmonary testing for unexplained dyspnea post COVID-19 was reported [114]. Patients with symptoms con- sistent with chronic fatigue had an abnormal pattern of oxygen uptake on cardiopulmonary testing consistent with what is seen with chronic fatigue syndrome. Circulatory impairment, abnor- mal ventilatory pattern and chronic fatigue syndrome may be common in patients with post-acute sequelae of post-concussive syndrome, and this accounts for the mechanism of dyspnea in many patients who do not have pulmonary disease from Long- COVID. The symptoms of Long-COVID Syndrome may all be associated with autonomic dysfunction as measured with Car- dio-Respiratory testing [111] and relieved with appropriate auto- nomic therapy based on the Cardio-Respiratory test [108].
Conflicts of Interest
Only Dr. Colombo has any conflicts of interest, specifically as inventor of P&S Monitoring technology and officer of Physio PS, Inc.
References
- Sudre, C. H., Lee, K. A., Ni Lochlainn, M., Varsavsky, T., Murray, B., & et al. (2021). Symptom clusters in COVID-19: A potential clinical prediction tool from the COVID Symp- tom Study app. Science advances, 7(12), eabd4177.
- Salamanna, F., Veronesi, F., Martini, L., Landini, M. P., & Fini, M. (2021). Post-COVID-19 syndrome: the persistent symptoms at the post-viral stage of the disease. A systemat- ic review of the current data. Frontiers in medicine, 8, 392.
- ht tps:/ / twit t er .com / el isape rego78/ st a -tus/1263172084055838721, Long Covid.
- https://www.nbcnews.com/nightly-news-netcast/video/ nightly-news-full-broadcast-july-31st-89377349826. COVID-19 'long-haulers' report nearly 100 symptoms for more than 100 days.
- https://www.theatlantic.com/health/archive/2021/11/ health-care-workers-long-covid-are-being-dis- missed/620801 Even Health-Care Workers with Long COVID Are Being Dismissed by Ed Yong.
- Garg, P., Arora, U., Kumar, A., & Wig, N. (2020). The" post-COVID" syndrome: How deep is the damage. Journal of medical virology, 93(2), 673-674.
- Raveendran, A. V., Jayadevan, R., & Sashidharan, S. (2021). Long COVID: an overview. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 15(3), 869-875
- Nabavi, N. (2020). Long Covid: How to define it and how to manage it. BMJ, 7(370), m3489.
- Cirulli, E. T., Barrett, K. M. S., Riffle, S., Bolze, A., Neveux, I., & et al. (2020). Long-term COVID-19 symptoms in a large unselected population. Medrxiv.
- Augustin, M., Schommers, P., Stecher, M., Dewald, F., Gieselmann, L., & et al. (2021). Post-COVID syndrome in non-hospitalised patients with COVID-19: a longitudinal prospective cohort study. The Lancet Regional Health-Eu- rope, 6, 100122.
- Michelen, M., Manoharan, L., Elkheir, N., Cheng, V., Da- gens, A., & et al. (2021). Characterizing long COVID: a liv- ing systematic review. BMJ global health, 6(9), e005427.
- https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/bulletins/preva- lenceofongoingsymptomsfollowingcoronaviruscovid19in- fectionintheuk/1july2021. Prevalence of Ongoing Symp- toms Following Coronavirus (COVID-19) Infection in the UK: 1 July 2021.
- Greenhalgh, T., Knight, M., Buxton, M., & Husain, L. (2020). Management of post-acute covid-19 in primary care. BMJ, 370, m3026.
- Aiyegbusi, O. L., Hughes, S. E., Turner, G., Rivera, S. C., McMullan, C., & et al. (2021). Symptoms, complications and management of long COVID: a review. Journal of the Royal Society of Medicine, 114(9), 428-442.
- Fraser, E. (2020). Long term respiratory complications of covid-19. BMJ, 370, m3001.
- Huang, C., Huang, L., Wang, Y., Li, X., Ren, L., & et al. (2021). 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. The Lancet, 397(10270), 220-232.
- Cui, S., Chen, S., Li, X., Liu, S., & Wang, F. (2020). Prev-alence of venous thromboembolism in patients with severe novel coronavirus pneumonia. Journal of Thrombosis and Haemostasis, 18(6), 1421-1424.
- Klok, F. A., Kruip, M. J. H. A., Van der Meer, N. J. M.,Arbous, M. S., Gommers, D. A. M. P. J., & et al. (2020). Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thrombosis research, 191, 145- 147.
- Hall, J., Myall, K., Lam, J. L., Mason, T., Mukherjee, B., & et al. (2021). Identifying patients at risk of post-discharge complications related to COVID-19 infection. Thorax, 76(4), 408-411.
- Han, X., Fan, Y., Alwalid, O., Li, N., Jia, X., & et al. (2021). Six-month follow-up chest CT findings after severe COVID-19 pneumonia. Radiology, 299(1), E177-E186.
- Zhu, M., Chen, D., Zhu, Y., Xiong, X., Ding, Y., & et al. (2021). Long-term sero-positivity for IgG, sequelae of re- spiratory symptoms, and abundance of malformed sperms in a patient recovered from severe COVID-19. European Journal of Clinical Microbiology & Infectious Diseases, 40(7), 1559-1567.
- Bellan, M., Soddu, D., Balbo, P. E., Baricich, A., Zeppegno, P., & et al. (2021). Respiratory and psychophysical sequelae among patients with COVID-19 four months after hospital discharge. JAMA network open, 4(1), e2036142-e2036142.
- Mo, X., Jian, W., Su, Z., Chen, M., Peng, H., & et al. (2020).Abnormal pulmonary function in COVID-19 patients at time of hospital discharge. European Respiratory Journal, 55(6), 2001217.
- Cortes-Telles, A., López-Romero, S., Figueroa-Hurtado, E., Pou-Aguilar, Y. N., Wong, A. W., & et al. (2021). Pulmo- nary function and functional capacity in COVID-19 survi- vors with persistent dyspnoea. Respiratory Physiology & Neurobiology, 288, 103644.
- Becker, R. C. (2020). Toward understanding the 2019 Coro- navirus and its impact on the heart. Journal of Thrombosis and Thrombolysis, 50(1), 33-42.
- Sandoval, Y., Januzzi Jr, J. L., & Jaffe, A. S. (2020). Cardiac troponin for assessment of myocardial injury in COVID-19: JACC review topic of the week. Journal of the American college of cardiology, 76(10), 1244-1258.
- Giustino, G., Croft, L. B., Stefanini, G. G., Bragato, R., Sil- biger, J. J., & et al. (2020). Characterization of myocardial injury in patients with COVID-19. Journal of the American College of Cardiology, 76(18), 2043-2055.
- Puntmann, V. O., Carerj, M. L., Wieters, I., Fahim, M., Ar- endt, C., & et al. (2020). Outcomes of cardiovascular mag- netic resonance imaging in patients recently recovered from coronavirus disease 2019 (COVID-19). JAMA cardiology, 5(11), 1265-1273.
- Rajpal, S., Tong, M. S., Borchers, J., Zareba, K. M., Obar- ski, T. P., & et al. (2021). Cardiovascular magnetic reso- nance findings in competitive athletes recovering from COVID-19 infection. JAMA cardiology, 6(1), 116-118.
- Kociol, R. D., Cooper, L. T., Fang, J. C., Moslehi, J. J., Pang, P. S., & et al. (2020). Recognition and initial man- agement of fulminant myocarditis: a scientific statement from the American Heart Association. Circulation, 141(6), e69-e92.
- Zubair, A. S., McAlpine, L. S., Gardin, T., Farhadian, S., Kuruvilla, D. E., & et al. (2020). Neuropathogenesis and neurologic manifestations of the coronaviruses in the age of coronavirus disease 2019: a review. JAMA neurology, 77(8), 1018-1027.
- Lu, Y., Li, X., Geng, D., Mei, N., Wu, P. Y., & et al. (2020).Cerebral micro-structural changes in COVID-19 patients– an MRI-based 3-month follow-up study. EClinicalMedi- cine, 25, 100484.
- Fauci. (2020) International AIDS Conference: YouTube 2020 and Nordvigas: Potential Neurological Manifestations of COVID-19: Neurology Clinical Practice 2020.
- Belvis, R. (2020). Headaches during COVIDâ?19: My clini- cal case and review of the literature. Headache: The Journal of Head and Face Pain, 60(7), 1422-1426.
- Pozi-Rosich: Virtual Annual Scientific Meetings 2020.
- Henek A. (2020) Alzheimer's Research Therapeutics Vol 1269, 2020.
- Kaseda, E. T., & Levine, A. J. (2020). Post-traumatic stress disorder: A differential diagnostic consideration for COVID-19 survivors. The Clinical Neuropsychologist, 34(7-8), 1498-1514.
- Mazza, M. G., De Lorenzo, R., Conte, C., Poletti, S., Vai, B., & et al. (2020). Anxiety and depression in COVID-19 survivors: Role of inflammatory and clinical predictors. Brain, behavior, and immunity, 89, 594-600.
- Nalbandian, A., Sehgal, K., Gupta, A., Madhavan, M. V., McGroder, C., & et al. (2021). Post-acute COVID-19 syn- drome. Nature medicine, 27(4), 601-615.
- Tankisi, H., Tankisi, A., Harbo, T., Markvardsen, L. K., An- dersen, H., & et al. (2020). Critical illness myopathy as a consequence of Covid-19 infection. Clinical Neurophysiol- ogy, 131(8), 1931-1932.
- Novac E (2021) Neurologicals, 21, 100-276.
- Shaw, B. H., Stiles, L. E., Bourne, K., Green, E. A., Shi- bao, C. A., & et al. (2019). The face of postural tachycardia syndrome–insights from a large crossâ?sectional online com- munityâ?based survey. Journal of internal medicine, 286(4), 438-448.
- Sakusic, A., & Rabinstein, A. A. (2018). Cognitive out- comes after critical illness. Current opinion in critical care, 24(5), 410-414.
- Colombo, J., Arora, R. R., DePace, N. L., & Vinik, A. I. (2014). Clinical Autonomic Dysfunction: Measurement. In- dications, Therapies, and Outcomes.
- Stefano et.al. 2021
- Raj, S. R., Black, B. K., Biaggioni, I., Paranjape, S. Y., Ramirez, M., & et al. (2009). Propranolol decreases tachy- cardia and improves symptoms in the postural tachycardia syndrome: less is more. Circulation, 120(9), 725-734.
- Hosey, M. M., & Needham, D. M. (2020). Survivorship af- ter COVID-19 ICU stay. Nature reviews Disease primers, 6(1), 1-2.
- Varatharaj, A., Thomas, N., Ellul, M. A., Davies, N. W., Pol- lak, T. A., & et al. (2020). Neurological and neuropsychiat- ric complications of COVID-19 in 153 patients: a UK-wide surveillance study. The Lancet Psychiatry, 7(10), 875-882.
- Malik G.R. (2020) Medrxiv 20, 2020.
- Dani, M., Dirksen, A., Taraborrelli, P., Torocastro, M., Pa- nagopoulos, D., & et al. (2021). Autonomic dysfunction in ‘long COVID’: rationale, physiology and management strategies. Clinical Medicine, 21(1), e63.
- Hellmuth, J., Barnett, T. A., Asken, B. M., Kelly, J. D., Torres, L., & et al. (2021). Persistent COVID-19-associat- ed neurocognitive symptoms in non-hospitalized patients. Journal of neurovirology, 27(1), 191-195.
- Novak E (2020) Neurological Science.
- Rahimi, M, M. (2021) British Medical Journal and Case Re- port. 14e, 240178.
- Salamanna, F., Veronesi, F., Martini, L., Landini, M. P., & Fini, M. (2021). Post-COVID-19 syndrome: the persistent symptoms at the post-viral stage of the disease. A systemat- ic review of the current data. Frontiers in medicine, 8, 392.
- Sletten, D. M., Suarez, G. A., Low, P. A., Mandrekar, J., & Singer, W. (2012, December). COMPASS 31: a refined and abbreviated Composite Autonomic Symptom Score. In Mayo Clinic Proceedings. 87(12), 1196-1201.
- Rodríguez, Y., Rojas, M., Ramírez-Santana, C., Acos- ta-Ampudia, Y., Monsalve, D. M., & et al. (2018). Auto- nomic symptoms following Zika virus infection. Clinical Autonomic Research, 28(2), 211-214.
- Anaya, J. M., Rojas, M., Salinas, M. L., Rodríguez, Y., Roa, G., & et al. (2021). Post-COVID syndrome. A case series and comprehensive review. Autoimmunity reviews, 20(11), 102947.
- Freeman, R., Abuzinadah, A. R., Gibbons, C., Jones, P., Mi- glis, M. G., & et al. (2018). Orthostatic hypotension: JACC state-of-the-art review. Journal of the American College of Cardiology, 72(11), 1294-1309.
- Jardine, D. L., Wieling, W., Brignole, M., Lenders, J. W., Sutton, R., & et al. (2018). The pathophysiology of the va- sovagal response. Heart Rhythm, 15(6), 921-929.
- Konig, M. F., Powell, M., Staedtke, V., Bai, R. Y., Thomas,D. L., & et al. (2020). Preventing cytokine storm syndrome in COVID-19 using α-1 adrenergic receptor antagonists. The Journal of clinical investigation, 130(7), 3345-3347.
- Fudim, M., Qadri, Y. J., Ghadimi, K., MacLeod, D. B., Mo- linger, J., & et al. (2020). Implications for neuromodulation therapy to control inflammation and related organ dysfunc- tion in COVID-19. Journal of cardiovascular translational research, 13(6), 894-899.
- Guilmot, A., Maldonado Slootjes, S., Sellimi, A., Bron- chain, M., Hanseeuw, B., & et al. (2021). Immune-mediated neurological syndromes in SARS-CoV-2-infected patients. Journal of neurology, 268(3), 751-757.
- Fu, Q., VanGundy, T. B., Shibata, S., Auchus, R. J., Wil- liams, G. H., & et al. (2011). Exercise training versus pro- pranolol in the treatment of the postural orthostatic tachy- cardia syndrome. Hypertension, 58(2), 167-175.
- Buoite Stella, A., Furlanis, G., Frezza, N. A., Valentinotti, R., Ajcevic, M., & et al. (2022). Autonomic dysfunction in post-COVID patients with and witfhout neurological symp- toms: A prospective multidomain observational study. Jour- nal of Neurology, 269(2), 587-596.
- McMillan, M. T., Pan, X. Q., Smith, A. L., Newman, D. K., Weiss, S. R., & et al. (2014). Coronavirus-induced de- myelination of neural pathways triggers neurogenic blad- der overactivity in a mouse model of multiple sclerosis. American Journal of Physiology-Renal Physiology, 307(5), F612-F622.
- Pourfridoni, M., Pajokh, M., & Seyedi, F. (2021). Bladder and bowel incontinence in COVIDâ?19. Journal of Medical Virology, 93(5), 2609-2610.
- Sansone, A., Mollaioli, D., Ciocca, G., Limoncin, E., Col- onnello, E., & et al. (2021). Addressing male sexual and reproductive health in the wake of COVID-19 outbreak. Journal of endocrinological investigation, 44(2), 223-231.
- Sansone, A., Mollaioli, D., Ciocca, G., Colonnello, E., Li- moncin, E., & et al. (2021). Mask up to keep it up: prelim- inary evidence of the association between erectile dysfunc- tion and COVID-19. Andrology, 9(4), 1053-1059.
- Xiao, F., Tang, M., Zheng, X., Liu, Y., Li, X., & et al. (2020). Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology, 158(6), 1831-1833.
- Domínguez–Varela, I. A., Rodríguez–Gutiérrez, L. A., Mo- rales-Mancillas, N. R., Barrera–Sánchez, M., Macías-Ro- dríguez, Y., & et al. (2021). COVID-19 and the eye: a re- view. Infectious Diseases, 53(6), 399-403.
- Karahan, M., DemirtaÅ?, A. A., Hazar, L., Erdem, S., Ava, S., & et al. (2021). Autonomic dysfunction detection by an automatic pupillometer as a non-invasive test in patients re- covered from COVID-19. Graefe's Archive for Clinical and Experimental Ophthalmology, 259(9), 2821-2826.
- Goodman, B. P., Khoury, J. A., Blair, J. E., & Grill, M. F.(2021). COVID-19 dysautonomia. Frontiers in neurology, 12, 543.
- Barizien, N., Le Guen, M., Russel, S., Touche, P., Huang, F., & et al. (2021). Clinical characterization of dysautonomia in long COVID-19 patients. Scientific reports, 11(1), 1-7.
- Shouman, K., Vanichkachorn, G., Cheshire, W. P., Suarez,M. D., Shelly, S., & et al. (2021). Autonomic dysfunction following COVID-19 infection: an early experience. Clini- cal Autonomic Research, 31(3), 385-394.
- Blitshteyn, S., & Whitelaw, S. (2021). Postural orthostatic tachycardia syndrome (POTS) and other autonomic disor- ders after COVID-19 infection: a case series of 20 patients. Immunologic research, 69(2), 205-211.
- Townsend, L., Dowds, J., O’Brien, K., Sheill, G., Dyer, A. H., & et al. (2021). Persistent poor health after COVID-19 is not associated with respiratory complications or initial disease severity. Annals of the American Thoracic Society, 18(6), 997-1003.
- Poenaru, S., Abdallah, S. J., Corrales-Medina, V., & Cow- an, J. (2021). COVID-19 and post-infectious myalgic en- cephalomyelitis/chronic fatigue syndrome: a narrative review. Therapeutic Advances in Infectious Disease, 8, 20499361211009385.
- Sandler, C. X., Wyller, V. B., Moss-Morris, R., Buchwald, D., Crawley, E., & et al. (2021). Long COVID and post-in- fective fatigue syndrome: a review. In Open forum infec- tious diseases, 8(10), ofab440.
- Steinberg (2012)
- Khatoon, F., Prasad, K., & Kumar, V. (2020). Neurologi- cal manifestations of COVID-19: available evidences and a new paradigm. Journal of neurovirology, 26(5), 619-630.
- Li, W., Li, M., & Ou, G. (2020). COVIDâ?19, cilia, andsmell. The FEBS Journal, 287(17), 3672-3676.
- Kanwar, D., Baig, A. M., & Wasay, M. (2020). Neurologi- cal manifestations of COVID-19. JPMA. The Journal of the Pakistan Medical Association, 70(5 (Suppl 3)), S101.
- Tetlow, S., Segietâ?Swiecicka, A., O’Sullivan, R., O’Hallo- ran, S., Kalb, K., & et al. (2021). ACE inhibitors, angioten- sin receptor blockers and endothelial injury in COVIDâ?19. Journal of internal medicine, 289(5), 688-699.
- Wu, J., Liang, B., Chen, C., Wang, H., Fang, Y., & et al. (2021). SARS-CoV-2 infection induces sustained humor- al immune responses in convalescent patients following symptomatic COVID-19. Nature communications, 12(1), 1-9.
- McGovern & Kang. (2011).
- Toljan, K. (2020). Letter to the editor regarding the view- point “evidence of the COVID-19 virus targeting the CNS: tissue distribution, host–virus interaction, and proposed neurotropic mechanism”. ACS chemical Neuroscience, 11(8), 1192-1194.
- Al-Kuraishy, H. M., Al-Gareeb, A. I., Qusti, S., Alsham- mari, E. M., Gyebi, G. A., & et al. (2021). Covid-19-in- duced dysautonomia: a menace of sympathetic storm. ASN neuro, 13, 17590914211057635.
- Hinduja, A., Moutairou, A., & Calvet, J. H. (2021). Sudo- motor dysfunction in patients recovered from COVID-19. Neurophysiologie Clinique, 51(2), 193-196.
- Barizien, N., Le Guen, M., Russel, S., Touche, P., Huang, F.,& et al. (2021). Clinical characterization of dysautonomia in long COVID-19 patients. Scientific reports, 11(1), 1-7.
- Becker, R. C. (2021). Autonomic dysfunction in SARS- COV-2 infection acute and long-term implications COVID-19 editor’s page series. Journal of thrombosis and thrombolysis, 52(3), 692-707.
- Crook, H., Raza, S., Nowell, J., Young, M., & Edison, P. (2021). Long covid-mechanisms, risk factors, and manage- ment. BMJ, 374.
- Tobias, H., Vinitsky, A., Bulgarelli, R. J., Ghosh-Dastidar, S., & Colombo, J. (2010). Autonomic nervous system mon- itoring of patients with excess parasympathetic responses to sympathetic challenges-clinical observations. US Neurol, 5(2), 62-66.
- Villapol, S. (2020). Gastrointestinal symptoms associated with COVID-19: impact on the gut microbiome. Transla- tional Research, 226, 57-69.
- Groff, A., Kavanaugh, M., Ramgobin, D., McClafferty, B., Aggarwal, C. S., & et al. (2021). Gastrointestinal manifes- tations of COVID-19: a review of what we know. Ochsner Journal, 21(2), 177-180.
- Weng, J., Li, Y., Li, J., Shen, L., Zhu, L., & et al. (2021).Gastrointestinal sequelae 90 days after discharge for COVID-19. The Lancet Gastroenterology & Hepatology, 6(5), 344346.
- Blackett, J. W., Li, J., Jodorkovsky, D., & Freedberg, D.E. (2022). Prevalence and risk factors for gastrointestinal symptoms after recovery from COVIDâ?19. Neurogastroen- terology & Motility, 34(3), e14251.
- Patell, R., Bogue, T., Koshy, A., Bindal, P., Merrill, M., & et al. (2020). Postdischarge thrombosis and hemorrhage in patients with COVID-19. Blood, 136(11), 1342-1346.
- Korompoki, E., Gavriatopoulou, M., Fotiou, D., Ntanasisâ? Stathopoulos, I., Dimopoulos, M. A., & et al. (2022). Lateâ? onset hematological complications post COVIDâ?19: An emerging medical problem for the hematologist. American Journal of Hematology, 97(1), 119-128.
- Silver, S. A., Beaubien-Souligny, W., Shah, P. S., Harel, S., Blum, D., & et al. (2021). The prevalence of acute kidney injury in patients hospitalized with COVID-19 infection: a systematic review and meta-analysis. Kidney Medicine, 3(1), 83-98.
- Su, H., Yang, M., Wan, C., Yi, L. X., Tang, F., & et al. (2020). Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney inter- national, 98(1), 219-227.
- Hirsch, J. S., Ng, J. H., Ross, D. W., Sharma, P., Shah, H. H., Barnett, R. L., ... & Abate, M. (2020). Northwell COVID-19 research consortium; northwell nephrology COVID-19 re- search consortium. Acute kidney injury in patients hospital- ized with COVID-19. Kidney Int, 98(1), 209-218.
- Addison, A. B., Wong, B., Ahmed, T., Macchi, A., Kon- stantinidis, I., & et al. (2021). Clinical Olfactory Working Group consensus statement on the treatment of postinfec- tious olfactory dysfunction. Journal of allergy and clinical immunology, 147(5), 1704-1719.
- Le Bon, S. D., Pisarski, N., Verbeke, J., Prunier, L., Cave- lier, G., & et al. (2021). Psychophysical evaluation of che- mosensory functions 5 weeks after olfactory loss due to COVID-19: a prospective cohort study on 72 patients. Eu- ropean Archives of Oto-Rhino-Laryngology, 278(1), 101-108.
- Adeghate, E. A., Eid, N., & Singh, J. (2021). Mechanisms of COVID-19-induced heart failure: a short review. Heart Failure Reviews, 26(2), 363-369.
- Paul, B. D., Lemle, M. D., Komaroff, A. L., & Snyder, S.H. (2021). Redox imbalance links COVID-19 and myalgic encephalomyelitis/chronic fatigue syndrome. Proceedings of the National Academy of Sciences, 118(34).
- Sander, L. E., & Garaude, J. (2018). The mitochondrial respiratory chain: A metabolic rheostat of innate immune cell-mediated antibacterial responses. Mitochondrion, 41, 28-36.
- Burtscher, J., Cappellano, G., Omori, A., Koshiba, T., & Millet, G. P. (2020). Mitochondria: in the cross fire of SARS-CoV-2 and immunity. IScience, 23(10), 101631.
- DePace, N. L., & Colombo, J. (2019). Autonomic and mito- chondrial dysfunction in clinical diseases: diagnostic, pre- vention, and therapy.
- Weill, P., Plissonneau, C., Legrand, P., Rioux, V., & Thi-bault, R. (2020). May omega-3 fatty acid dietary supple- mentation help reduce severe complications in Covid-19 patients? Biochimie, 179, 275-280.
- Adebayo, A., Varzideh, F., Wilson, S., Gambardella, J., Ea- cobacci, M., & et al. (2021). l-Arginine and COVID-19: An Update. Nutrients, 13(11), 3951.
- Colombo, J., Weintraub, M, I., Munoz, R., Verma, A., Ahmed, G., & et al. (2022). Long-COVID and the Auto- nomic Nervous System: The journey from Dysautonomia to Therapeutic Neuro-Modulation, Analysis of 152 Patient Retrospectives. Submitted, 2022.
- Medical News Today
- Bergwerk, M., Gonen, T., Lustig, Y., Amit, S., Lipsitch, M., & et al. (2021). Covid-19 breakthrough infections in vacci- nated health care workers. New England Journal of Medi- cine, 385(16), 1474-1484.
- Mancini, D. M., Brunjes, D. L., Lala, A., Trivieri, M. G., Contreras, J. P., & et al. (2021). Use of Cardiopulmonary Stress Testing for Patients with Unexplained Dyspnea Post– Coronavirus Disease. Heart Failure, 9(12), 927-937.




