Case Report - (2025) Volume 10, Issue 3
Lip Lichen Planus Following COVID-19 Infection
2Community Dermatology, National Health Service, (HealthHarmonie) and The Midlands Medical Academy, United Kingdom
3Histopathology Department, Royal Wolverhampton NHS Trust, Wolverhampton, United Kingdom
4University of Wolverhampton, North Cumbria Integrated Care NHS Foundation Trust, United Kingdom
Received Date: Oct 28, 2025 / Accepted Date: Nov 27, 2025 / Published Date: Dec 08, 2025
Copyright: ©2025 Mohammed S Al Abadie, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Hamad, N., Sharara, Z., Maheshwari, M., Al Abadie, M. S. (2025). Lip Lichen Planus Following COVID-19 Infection. Int J Clin Expl Dermatol, 10(3), 01-04.
Abstract
Lichen planus is a dermatological disorder believed to be induced by various factors including viral infections, drug exposure, and contact with metals. The condition manifests with diverse patterns and can affect multiple body sites. Here, we describe a patient with histopathology proven lip lichen planus occurring after a coronavirus infection.
Keywords
Lichen Planus, Lichenoid Eruption, Wickham’s Striae, Pathology
Introduction
Lichen planus is a chronic inflammatory disorder involving the skin, mucous membranes, hair and nails, predominantly attributed to an autoimmune mechanism. Although its precise aetiology remains unclear, various triggers have been implicated. Factors identified are viral infections, certain medications such as antihypertensives, and exposure to metals like mercury, gold, copper. Psychological stress is also deemed a potential contributing factor [1]. The condition primarily involves individuals between the age of 30 and 60 years. The female to male ratio is approximately 1.5:1 [2].
Clinically, lichen planus presents with diverse morphologies, with the hallmark lesion being clusters of polygonal, purple papules that can be very pruritic. These papules often display a glistening surface characterized by Wickham’s striae [3].
Lesions may manifest in various distribution patterns, ranging from isolated to extensive, and may appear in distinct configurations such as annular, linear, or actinic arrangements. Commonly affected sites include the flexor aspects of the wrists, dorsal hands, lower back, shins, and ankles [2,3].
Mucosal involvement is frequent, particularly affecting the oral cavity, where six clinical subtypes of oral lichen planus have been described [4-6].
• Reticular (most common) - characterised by asymptomatic, white lines on both sides of the buccal mucosa
• Erosive and atrophic – often cause a burning sensation
• Papular, plaque-like and bullous forms are also recognised but less commonly observed
Biopsy and histopathology typically reveal degeneration of the basal layer of the epidermidis and a lymphocytic infiltrate along the dermo-epidermal junction. The epidermis shows irregular hyperplasia, producing a saw-tooth pattern accompanied by wedge-shaped areas of hypergranulosis [7].
Lichen planus (LP) has been associated with hepatitis C virus (HCV) infection, though prevalence varies widely by region, from 4% in Northern France to 62% in Japan [8].
Despite recent advances in understanding the immunopathogenesis of oral lichen planus (LP), the initial triggers of lesion formation and the essential pathogenic pathways are unknown [9].
Treatment
The treatment of oral and cutaneous lichen planus (LP) primarily focuses on alleviating symptoms and managing inflammation. Various therapeutic modalities are employed, with topical corticosteroids being the first-line treatment for both oral and cutaneous forms. Clobetasol propionate is commonly used, demonstrating significant improvement in lesions, Other options include topical tacrolimus for mucosal lesions and systemic corticosteroids for severe cases [10,11]. Additionally, alternative treatments such as retinoids, immunosuppressants, and photodynamic therapy have shown promise [12,13]. In refractory cases, oral alitretinoin has been effective in resolving symptoms [14].
Despite these treatment options, lichen planus can be persistent and resistant, particularly in mucosal areas, leading to a need for ongoing management and monitoring [11]. This highlights the complexity of LP and the necessity for individualized treatment plans
Case Report
A 60-year-old male with a history of coronavirus infection in November 2021 was referred to community dermatology services in July 2022 presenting with swelling, depigmentation, and thickening of the upper and lower lips. The patient reported accompanying symptoms of lip cracking, redness, and significant soreness, which had persisted since his COVID-19 illness. His medical history was otherwise unremarkable, with no ongoing medications or known allergies. He had undergone a two-week wait referral to maxillofacial surgery, where no signs of skin malignancy were identified. Notably, the patient was a smoker.
On clinical examination, lichenoid skin changes were observed on both the upper and lower lips, with pronounced thickening noted particularly on the lower lip.
Figure -1: Swelling, thickening and leukoplakia of the lips
Investigations revealed a normal complement C3 but a raised complement C4. A speckled staining pattern was seen when testing anti-nuclear antibodies, which may be seen with several connective tissue diseases, often associated with Ro and La in Sjögren’s syndrome.
Histopathology
The patient had a 3mm punch biopsy performed on the left lower lip which revealed marked hyperkeratosis with focal follicular plugging and almost wedge-shaped hypogranulosis Figure -2, Figure -3, and figure -4. There was evidence of basal cell damage with almost lichenoid inflammatory infiltrate at the epidermal dermal interface. Few apoptotic keratinocytes were noted in the basal epidermis. The dermis included showed no obvious peri-vascular or peri- adnexal inflammation. ABPAS stain showed some small rounded fungal debris admixed with keratin which appeared non-pathogenic while the dermis showed no obvious mucin. The appearances are of a lichenoid inflammation, and the features are suggestive of lichen planus. The patient was prescribed Elocon 0.1% ointment applied once a day on the lips with follow-up arranged.
Figure 2: Haematoxylin and Eosin staining (H&E) magnification 5.5x
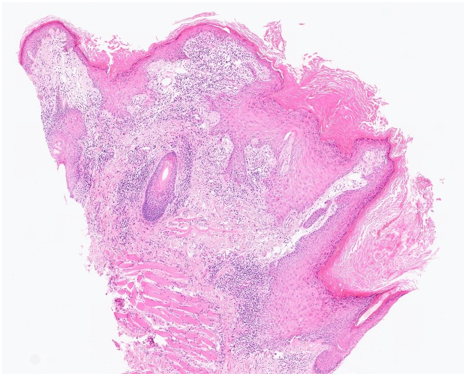
Figure 3: Haematoxylin and Eosin staining (H&E) magnification 10x
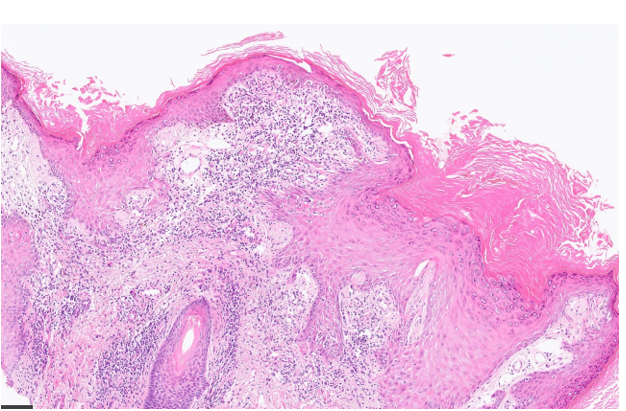
Figure 4: Haematoxylin and Eosin staining (H&E) magnification16x
Discussion
he aetiology of lichen planus (LP) remains poorly understood despite multiple known triggers. Current evidence supports LP as a T cell-mediated autoimmune disorder predominantly involving cytotoxic CD8+ T cells [15]. Potential triggers include viral infections such as COVID-19, certain medications, and exposure to metals, notably from dental restorations. The patient described above developed lip lesions following COVID-19 infection; although causality cannot be confirmed, similar associations have been frequently documented. An observational study demonstrated a statistically significant association between COVID-19 infection or vaccination and the onset of de novo LP, suggesting that COVID-19–related events may act as triggers [16]. A systematic review following PRISMA guidelines analyzed publications from December 12, 2019, to May 31, 2022, identifying 25 articles reporting 26 patients (mean age 55.3 years; 65.4% female) who developed LP after COVID-19 infection or vaccination.
Conclusion
The available evidence indicates a potential link between COVID-19 infection or vaccination and the onset of lichen planus, likely through immune-mediated mechanisms involving cytotoxic T cells. While causality remains to be definitively established, the consistent reporting of LP cases following COVID-19–related events highlights the need for heightened clinical awareness and further research to clarify these associations.
References
- British Association of Dermatologists (2016). Lichen planus [Internet]. London: British Association of Dermatologists; [cited 2025 Jul 27].
- Le, L. C., & Chosidow, O. (2012). Clinical practice. Lichen planus. N Engl J Med, 366(8), 723-732.
- Boyd, A. S, Neldner, K. H. (1991). Lichen planus. J Am Acad Dermatol, 25(4):593-619.
- Giannetti, L, Dello Diago, A. M, Spinas, E. (2018). Oral Lichen planus. J Biol Regul Homeost Agents, 32(2):391-395.
- Warnakulasuriya, S., Kovacevic, T., Madden, P., Coupland, V. H., & Sperandio, M., et al. (2011). Factors predicting malignant transformation in oral potentially malignant disorders among patients accrued over a 10â?year period in South East England. Journal of oral pathology & medicine, 40(9), 677-683.
- Ebrahimi, M., Lundqvist, L., Wahlin, Y. B., & Nylander, E. (2012). Mucosal lichen planus, a systemic disease requiring multidisciplinary care: a cross-sectional clinical review froma multidisciplinary perspective. Journal of lower genital tract disease, 16(4), 377-380.
- Sharquie, K. E., Noaimi, A. A., & Zeena, A. A. (2015). Upsurge of cases of lichen planus in Iraqi population in Baghdad City with frequency of hepatitis viruses. IOSR J Dent Med Sci, 14(12), 78-81.
- Lodi, G., Scully, C., Carrozzo, M., Griffiths, M., & Sugerman,P. B., et al. (2005). Current controversies in oral lichen planus: report of an international consensus meeting. Part 1. Viral infections and etiopathogenesis. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology, 100(1), 40-51.
- Rodrigues, R. R., Pinheiro, J. C., Silva, G. G., Barboza,C. A. G., & Leite, R. B. (2020). Oral lichen planus with cutaneous manifestations: case report with emphasis on dental diagnostic criteria. Jornal Brasileiro de Patologia e Medicina Laboratorial, 56, e2072020.
- Usatine, R. P., & Tinitigan, M. (2011). Diagnosis and treatment of lichen planus. American family physician, 84(1), 53-60.
- Aboushousha, A., Kamal, Y., & Ali, S. (2025). Supplementary zinc and vitamin D in management of symptomatic oral lichen planus: a three-arm randomized clinical trial. BMC Oral Health, 25(1), 872.
- Swain, N., Patel, S., Pathak, J., Shirke, K. J., & Patel, T., et al. (2017). Oral lichen planus–A brief review on treatment modalities. Journal of Contemporary Dentistry, 8(3), 137- 143.
- Kolios, A. G., Marques Maggio, E., Gubler, C., Cozzio, A., & Dummer, R., et al. (2013). Oral, esophageal and cutaneous lichen ruber planus controlled with alitretinoin: case report and review of the literature. Dermatology, 226(4), 302-310.
- Shiohara, T., Moriya, N., Mochizuki, T., & Nagashima, M. (1987). Lichenoid Tissue Reaction (LTR) Induced by Local Transfer of la-Reactive T-Cell Clones. II. LTR by Epidermal Invasion of Cytotoxic Lymphokine-Producing Autoreactive T Cells. Journal of investigative dermatology, 89(1).
- Arduino, P. G., Kubanov, A., Vlasova, A., Martynov, A., & Petti, S. (2025). Lichen Planus Following COVIDâ?19 Infection and Vaccination. Matched Case–Control Study. Australasian Journal of Dermatology.
- Nguyen, B., Perez, A. G., Elgart, G. W., & Elman, S. A. (2023). Lichen planus after COVIDâ?19 infection and vaccination: A systematic review. Journal of the European Academy of Dermatology & Venereology, 37(3).


