Review Article - (2018) Volume 3, Issue 2
Ionization Dosimetry Principles for Conventional and Laser-Driven Clinical Particle Beams
2National Institute for Laser, Plasma and Radiation Physics, Atomistilor Str./409, Magurele, Ilfov, Romania
3University of Bucharest, Faculty of Physics, Atomistilor Street/407, Bucharest, Romania
4University of Bucharest, Faculty of Chemistry, Regina Elisabeta Street/4-12, 030018, Bucharest, Romania
5SC Bitsolutions SRL, Calea 13 Septembrie Street/201, Bucharest, Romania
6SC Medfam SCARLAT SRL, Bd. Ghencea/43 B, Bucharest, Romania
Received Date: Jul 18, 2018 / Accepted Date: Jul 24, 2018 / Published Date: Jul 30, 2018
Copyright: ©Scarlat Florea, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
In this paper after mentioning the clinical radiation fields of 20 keV-450 MeV/u, they are characterized by the number of particles and their energy. Particle energy is the quantity that determines radiation penetration at the depth at which the tumor is situated (Fig. 1). The number of particles (or beam intensity) is the second major quantity that assures the administration of the absorbed dose in the tumor. The first application shows the radiation levels planned for various radiation fields. Prior to interacting with the medium, the intensity (or energy fluence rate) allows the determination of energy density, energy, power and relativistic force. In the interaction process, it determines the absorbed dose, kerma and exposure. Non-ionizing radiations in the EM spectrum are used as negative energy waves to accelerate particles charged into special installations called particle accelerators. The particles extracted from the accelerator are the source of the corpuscular radiation for high-energy radiotherapy. Of these, light particle beams (electrons and photons) for radiotherapy are generated by betatron, linac, microtron, and synchrotron and heavy particle beams (protons and heavy ions) are generated by cyclotron, isochronous cyclotron, synchro-cyclotron and synchrotron. The ionization dosimetry method used is the ionization chamber for both indirectly ionizing radiation (photons and neutrons) and for directly ionizing radiation (electrons, protons and carbon ions). Because the necessary energies for hadrons therapy are relatively high, 50-250 MeV for protons and 100-450 MeV/u for carbon ions, the alternative to replace non-ionizing radiation with relativistic laser radiation for generating clinical corpuscular radiation through radiation pressure acceleration mechanism (RPA) is presented.
Introduction
Radiation (from fr. Radiation) is a physical phenomenon of emission and propagation from waves (wave radiation) or corpuscles (corpuscular radiation).
According the principle of relativity requiring the force field and the quantum principle requiring quanta of the field, i.e. force carrier particles, the photon is the quantum of the classical electromagnetic field and a matter particle is the quantum of some kind of matter field.
This paper deals with photons - the mediators of electromagnetic interaction, electrons which are fundamental particles and have no discernable substructure, and hadrons containing quarks, for example: baryons (protons = 2 up quark + 1 down quark and neutrons =1 up quark +2 down quark) and mesons (=1 quark + 1 antyquarks) [1].
The electromagnetic radiation spectrum can be divided into two areas: non-ionizing electromagnetic waves that start from radio frequency and end up with ultraviolet and indirectly ionizing radiation including photons (X rays, gamma and bremsstrahlung).
Leptons (electrons and positrons) and hadrons (protons, ions of other atoms) with charge are directly ionizing radiation. Charged particles in interaction with a target generate photons of type: - bremsstrahlung (B) when target is a solid material, synchrotron radiation (SR) when target is a magnetic field and - free electron laser radiation (FEL) when target is a periodic magnetic field
After presenting the physical data of the three types of radiation (non-ionizing, indirectly and directly ionizing), the radiation field is characterized and then the characteristics of the hadron accelerators using radio frequency (RF) waves with wavelengths of the order of meters for particle acceleration are mentioned [2].
The depth dose distribution of the proton and carbon ions absorbed has the most appropriate profile in setting the patient’s treatment plan due to the Bragg-Gray peak, which at the end of the range is about 4 times larger than the dose level to peak, Fig .1 [3]
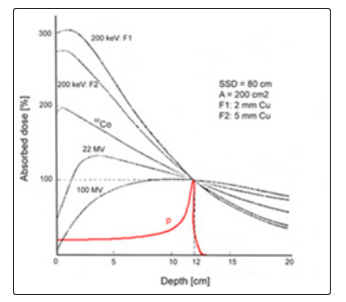
Figure 1. Depth dose in tissue for 60Co rays, X rays of 200 keV, photons of 22 MeV, 100 MeV and protons of 130 MeV
Also, some physical aspects of the interaction of particles with the environment related to ionization chamber dosimetry are presented [4-6].
Existence in the construction of high-power laser systems (10 PW) has reinforced the old idea of replacing the RF wave with relativistic laser wave to accelerate electrons and hadrons to the energies required by therapy: (50-250) MeV for protons and (100-450) MeV/u for carbon ions [7-9].
Dosimetric quantities for photons/electrons dosimetric are related to the energy fluence/particle fluence of the radiation field. It presents the calculation formalism for exposure, kerma and absorbed dose in the air and in the medium [10-13].
Determination of the absorbed dose is made in accordance with the charged particle equilibrium (CPE) principle in photon irradiation and the Bragg-Gray principle in irradiation with ionizing radiation [14-16].
The Spectrum of Electromagnetic Radiation
The electromagnetic spectrum (Fig. 2) is described with respect to a characteristic wavelength (λ) and associated energy, Ehw ≡ hω, where hw =1.055.10-34[J.s] is the smallest observable action value and ω= 2πc/λ), by relation:
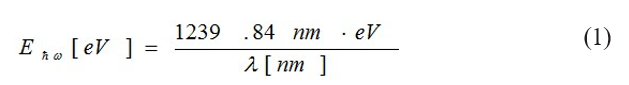
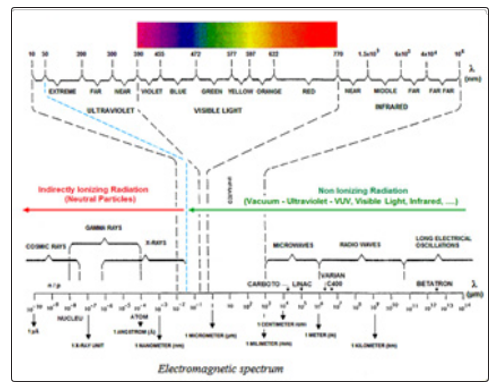
Figure 2: The spectrum of electromagnetic radiation [RCA Electro-Optics 1976]

In nature there is the principle that “Any body of nature with a temperature above 0 0K emits thermal radiation in space under electromagnetic waves”. The thermal energy emitted by the matter in thermodynamic equilibrium is related to the body temperature that emit the radiation through the relation Ehω = kT; where k = 1.38x10-23 J /K is Boltzman’s constant, which indicates the lowest value of entropy in nature (S ≥ k). Using the relation Ehω = kT, the following temperatures are obtained in degrees Kelvin – K, (T [K] = T [0C] + 273.15) for non-ionizing EM waves: radio wave - 1.44 x 10-2 ≥ K > 1.44 x 10-4, microwave
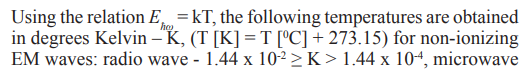
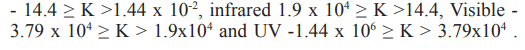
To date, MW, RW and LEO electromagnetic waves are used as negative energy waves in special devices called particle accelerators charged to obtain corpuscles radiation. Particles accelerated to the energy required for a particular application, are extracted from the accelerator, or are focuses on a metal target with large atomic number (Z) for generating braking radiation.
Among RF waves used in accelerators, we mention some values as a size order: 50 Hz for betatron (Kerst 1940), linear accelerator (3 GHz), microtron (3 GHz), cyclotron (80 MHz), syncrocycloton (150 MHz), isochronous cyclotron (75MHz) and synchrotron (100 MHz) [2].

Reference levels at frequency (electric field E strength [V/m],magnetic field H strength [A/m], B fied [μT] and equivalent plane wave density S[W/m2]) for general public exposure/occupational public exposure, are the following: 10 - 400 MHz (28/61, 0.073/0.16,0.092/0.2, 2/10) and for 2 -300 GHz (61/137, 0.16/0.36, 0.20/0.45, 10/50). These values are selected from the International Commission on Non-Ionizing Radiation Protection (ICNIRP [17].
The depth of penetration for tissues with high water content is 0.413 cm (8 GHz; 3.3x10-5 eV) and 0.343 cm (10 GHz; 4.13x10-5 eV) [18].
Indirectly Ionizing Radiation
A radiation that has sufficient energy to remove electrons from atoms or molecules as it passes through matter. Examples: photons, neutrons and other uncharged particle.
By introducing the photon energy Ehω = pc and the rest mass E0 =m c2 in the relation energy-impulse, E2 = E 2 + p2c2, the mass of the photon equals zero. In other words, when Ehω = pc a “particle” does not have a rest mass. Particles without mass do not have a rest frame of reference. They are moving at the speed of light in any frame of reference. Waves are pure energy.
The main characteristics of photon interaction with matter are: high energy transfer to direct ionizing radiation, the absorbed dose is given by direct ionizing radiation and the camera-detector response is given by the interaction of the photons with the air in the detector cavity and the fluence has exponential attenuation.
The depth dose distributions for photon energies of 6 MV and 20 MV (Mevatron KD) and 6 MV and 18 MV (Therac 20) are presented in Fig.3 [19].
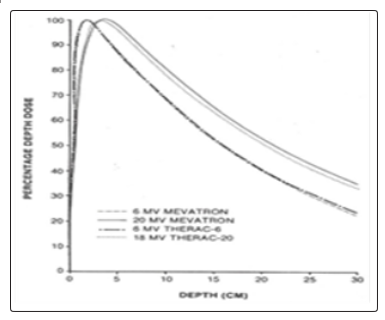
Figure 3: Central axis depth dose data: 10x10 cm2 field size, 100 cm SSD measured in water for the 6- and 20 -MV photon beams [19].
In this figure the general shape of the photon depth dose curve is presented. Absorbed dose measurements take place in a V volume of the ionization chamber centered at the depth xm at which the dose has the maximum value. Charged particle equilibrium (CPE) is said to exist at a point M, centred in a volume V, at depth xm, if each charged particle carrying out a certain energy from this volume is replaced by another identical particle which carrying the same energy into the volume.
Mathematically, CPE exists when the divergence of the charged particle beam energy fluence vector is equal to zero, divΨCPE = 0.In this case the dose D is equal to the collision kerma Kcool , D = Kcool for (g = 0).
In the case of indirectly ionizing radiation, the calculation relation for the radiation intensity is given by the expression
I [MeV/cm2 s] = 0.912x106 X / (μen/ρ)a (3)
where X is the exposure in air (a) in units of roentgen per minute [R/m] and (μen/ρ)a is the mass energy absorption coefficient expressed in units of [cm2/g] [14].

Also, the free electron laser (FEL) is added which, depending on the electron beam energy, E = γ.m0 c2, between 10 to 20 GeV, the undulator wavelength λu and the deflection parameter K. These generate SXR photons with wavelength λ0 given by

Directly ionizing radiation
Direct ionizing radiation consists of charged particles. The energy absorption per unit mass (dose) is mainly due to the phenomena of ionization and excitation. The response of the electron detector ionization chamber, which complies with the Bragg-Gray principle, is determined by the fluence of electrons released into the medium.
The main feature of directly ionizing radiation in interction with the medium is given by the range defined for leptons and hadrons, and especially the Bragg-Gray peak for hadrons (protons, antiprotons, carbon ions, alpha particles, etc.), Fig.4 [14].
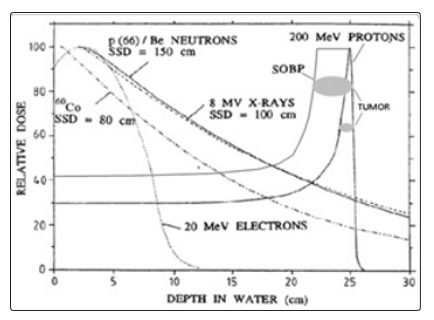
Figure 4: Physical characteristics of charged particle beams: 20 MeV- electron and 200 MeV proton
The important physical parameters of the charged particles are: the kinetic energy T, which determines their trajectory in the matter and the particle intensity I, which ensures obtaining the necessary dose for tumor sterilization or for radiography.
The electron beam provided by a linac, radiating a medium, in point of interest, absorbed dose rate is determined using the relationship Bragg-Gray,
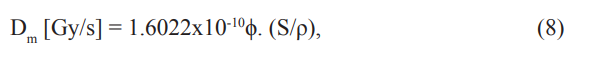
Where Φ is the electron fluence rate in traditionale units [e/cm2s] si S/ρ is the stopping power of energy in [MeVcm2/g].
Directly ionizing radiation has many areas of application, of which we mention: - electron therapy [4-25 MeV], - proton therapy [50-250 MeV], - heavy ions therapy [100-450 MeV/u], - meson therapy (supplying photons /electrons) 500-700 MeV, - electron beam angiography 500-1000 MeV, - electron beam computed tomography [MeV], - protons/ neutron production/therapy tens of MeV, sterilization, food preservation 2-10 MeV, - α emitters [4-9 MeV], - deuterons/radioisotope production 7-20 MeV, - protons/ radioisotope production 10-100 MeV [3].
To take a clinical treatment of a malignant tumor with a beam of photons of 10 MeV, (μen/ρ)a = 0.0150 cm2/g, Φ = 1 R/m, 9.7x10-6 W/cm2 per R/m, and energy density of U =3.23x10-10 J/m3. For administration of 10 Gy/weak, 2 Gy/day, with the absorbed dose of 1 Gy/m, is obtained a radiation pressure for hω = 10 MeV, in the standard input field A = 10x10 cm2, of 3.68 x10-8 [N/m2] < 4.51x 10-6 [N/m2] (solar pressure). The photon beam power propagating at the speed of light is P = 0.11 W < 13 W (solar power) and the force F acting on the A radiation field is F= 3.68 x 10-10 N < 4.51x10-8 N (solar force) [3].
Characterization of the ionizing radiation field
The radiation field is characterized by quantities based on the number of particles (N) and their energy, Ehω = hω for photons and Ehω = T - kinetic energy for particles, both expressed in SI in [J] or in traditional units [MeV].
Of the radiation fields used in dosimetry we mention two quantities. The first is the charged or uncharged particle fluence [m-2] = φ.t, which represents the number of particles per unit area. In the case of the Bragg-Gray chambers, this represents the fluence of the secondary electrons in the environment and multiplied by the stopping power determines the absorbed dose
The second quantity is the particle energy fluency, Ψ [J.m-2] = Φ.Φ = ψ.t, defined as the particle energy per unit area. This is the size is used for determination of the three dosimetric quantities (absorbed dose, kerma and exposure) in case of medium irradiation with a photon beam. The sizes of φ [m-2s-1] and ψ [J.m-2s-1] represent the particle fluence flow and the particle energy flow rate.
When charged or uncharged particle beams are polyenergetic, particle fluence and energy fluence are replaced with the particle fluence spectrum, ΦE = dΦ(E)/dE, and the energy fluence spectrum ΨE=dΨ(E)/dE = (dΦ(E)/dE)xE.
Clinical hadron accelerators
Heavy particle clinical beams are currently generated by two types of accelerators: classic cyclotron as synchrocyclotron and isochronous cyclotron and synchrotron. In 2012, of about 12,000 accelerators in medicine, only 39 work for proton and ion therapy [25].
Classical Cyclotron
The operating principle of classical cyclotron is shown in Figure 5 (Lawrence 1932) [26].
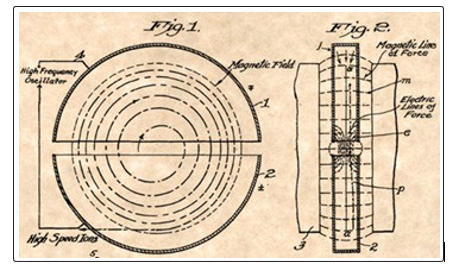
Figure 5: Front and profile view of patented cyclotron by E.O. Lawrence, US Patent Office [26].
The particles injected by a source located at the center of the magnet are accelerated on a spiral trace by the RF electrical voltage applied between two dees located inside a vacuum chamber, which in turn is located in the space between two magnetic poles providing the magnetic field with axial symmetry.

Synchrocyclotron

The synchrocyclotron works on the principle of phase stability (Veksler ’44, McMillan’45) which states that the ions would stay in phase with the RF, oscillating around a “synchronous phase” φs. Whereas the cyclotron can accelerate a stream of particles, the synchro-cyclotron can only accelerate “one bunch” of particles.
For proton therapy the Still Rivers Company conducted a Synchrocyclotron supraconductor (B = 9 T) of 250 MeV (Monarch 250) with a weight of 20 tons, Fig. 6 [28].
La Gatchina near St. Petersburg operates a 1000 MeV synchrocyclotron with standard magnets (B = 1.8 T) and weight of 10.000 tons (= weight of the Eiffel Tower).
Isochronous cyclotron is the second version of the classic cyclotron which maintains the constant frequency ω0 =qB↑/m0γ↑= const., and the increase of the magnetic field is synchronized with the mass increase.
This type of cyclotron works on the principle of alternating focusing (Christofilios1950). In the isochronous cyclotron, the magnetic field has a periodic spatial variation (azimuthal and radial, Kerst 1955) that provides the particle with a non-circular orbit.

Figure 7: Periodic spatial variation of magnetic field at Varian 250 MeV SC Compact Proton Cyclotron [29]
In Fig. 7 is seen the strong and weak alternating magnetic azimuth regions (Sectors of “hills” and “valleys”) in the isochronous cyclotron Varian SC 250 MeV [29].
We mention in this regard isochronous cyclotrons: IBA 235 proton therapy cyclotron (RF 106 MHz, NC coil: 1.7-2.2 T, 220 tons);VARIAN 250 MeV superconducting compact proton cyclotron (72.8 MHz, 2.4 – 3 T, < 90 tons) and IBA-JINIR C 400 carbon/proton therapy cyclotron (protons (260 MeV) and carbon ions (400MeV/u),75 MHz, B= 2.45-4.5 T, 660 tons [30].
Synchrotron
This change operating principle of cyclotron by replace the electrodes with smaller RF cavity (see Fig.8).
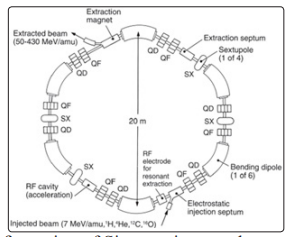
Figure 8: Configuration of Siemens ion synchrotron [31]
To obtain an equivalent circular orbit, particle injection into the synchrotron is done with a low energy accelerator. The synchrotron keeps the radius of the circle constant by increasing the strength of the magnetic field according to the relation R = p↑/0.3B↑ = const.
Mention for proton therapy and carbon therapy synchrotroanes of type: HIT, LLLMC, Siemens, Hitachi, Mitsubishi and Toshiba.
Currently, for radiotherapy, there are RD accelerators cyclinac (cyclotron + linac), fixed field alternating gradient accelerator - FFAG and dielectric wall accelerator - DWA using cyclotron technology with RF [2].
Replacing the RF (100 MHz, λ =300m) wave with laser wave (λ = 1μm =10-6 m) in the acceleration process and the magnetic field from the irradiation head (about 120-600 t) by the use of the channelling phenomenon in the bent crystals can reduce the dimensions of these accelerators and increase the multiplication factor of medical facilities to allow more patients to access therapy [32-35].
Laser-driven clinical hadron beams
Fig. 9 shows the second version of the laser accelerator in the article [9] when the target is located directly in the treatment room.
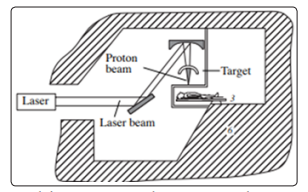
Figure 9: Laser driven proton therapy accelerator developed by Bulanov and Khoroshkov [9].
The laser beam (10 PW, 15 fs, 150 J, 1023 W/cm2) generated by Apollo Laser System, now under construction on Magurele Platform near Bucharest, can be used for technological research on the parameters of an clinic laser accelerator [36-37].
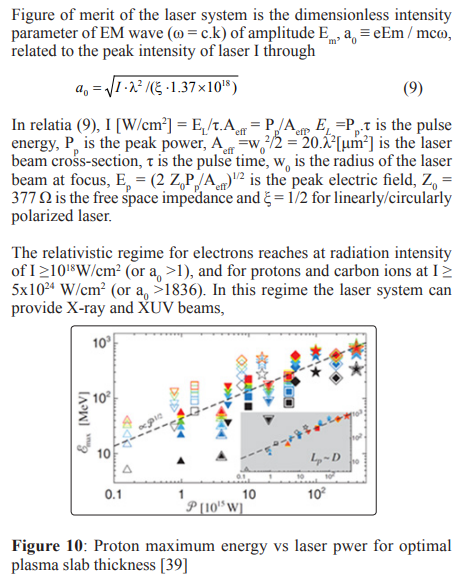
Figure 10 shows the maximum energy of the proton Φmax [MeV] according to the laser for ne =100 ncr, the laser pulse length of the order of the focal spot size, LP =D, for various ratio D/λ, Lp/λ and intensities I = 1020-1022 [W/cm2] [38-39].
The interpretation of the data in Figure 3 obtained in the RPA LS acceleration regime [30] indicates that the proton energy is proportional to the square root of the intensity for thin targets, ÃÃÃÃÂ????ÂÃÃÃÂ???ÂÃÃÂ??ÂÃÂ?ÂÂmax ≈ P1/2.
For use in hadron radiotherapy, schemas based on TNSA, RPA and other relativistic acceleration mechanisms are being researched and experimented (a0 > 1) [40-41].
Interaction of Photons with Matter
Major Processes
The main local processes through which photons of energy Φ=hω interact with the substance will be energy absorption and scattering [42-45].
The photoelectric effect (PE) takes place when the incident photon of energy hω interacts with an electron located on one of the orbits of the atom, with the energy of the bending B. The incident photon yields the energy B to extract the electron from the atom and the rest of the incident photon energy T = hω - B remains as the kinetic energy of the emerging electron. This process takes place when hω > B. The effect PE is characterized by the mass absorption coefficient, denoted by τ/ρ and expressed in [m2/kg] or in traditional units [cm2/g].
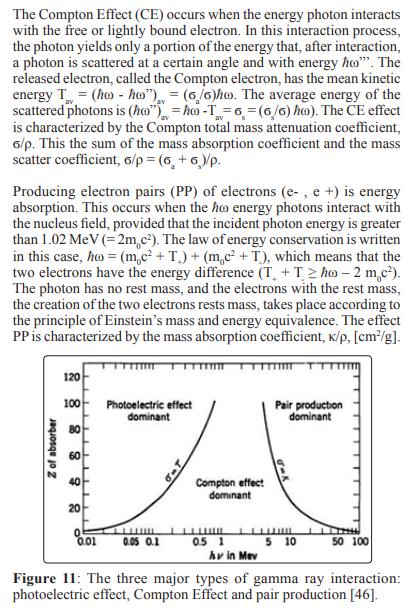
The three major effects produced by the interaction of the photon beam with medium are represented in Fig.11 [46]. The curves show the values of the atomic number Z and the energy of the photon â?ÂÃÃÂ??ÂÃÂ?ÂÂω for which the neighbouring effects are equal.Secondary photons and electrons can produce tertiary radiation (photon or electron δ) by ionisation or excitation outside the absorbed dose computation area.Photon interaction coefficients In the area where the three major processes occur, the energy transfer from the incident-primary photon to the secondary electrons (photoelectrons, T, electrons Compton, Tave, and electron pairs, (T- + T+) occurs. When the photoelectron is released, the atom remains positive with a certain electronic hole. Therefore, the total mass absorption coefficient is μa/ρ = τ/ρ + ÃÃÃÂ??ÂÃÂ?ÂÂÂa/ρ

Exponential Attenuation

Energy Transfer and Energy Absorption Coefficients

For the energies used in diagnostic radiology, g may be taken as zero. The value (1-g) slowly decreases with increasing photon energy
Because of their importance as gamma calibration sources for dosimetry, exact values of (1–g) have been determined for 137Cs photons (â?ÂÃÃÂ??ÂÃÂ?ÂÂω = 0.662 MeV) and 60Co photons (â?ÂÃÃÂ??ÂÃÂ?ÂÂω = 1.332 and 1.172 MeV) to be 0.9984 and 0.9968, respectively [44].
Exposure, Kerma and Absorbed Dose in Air

Kerma and Absorbed Dose in Medium


Absorbed Dose in Water via Kerma

Neutron Absorbed Dose to Muscle Tissue
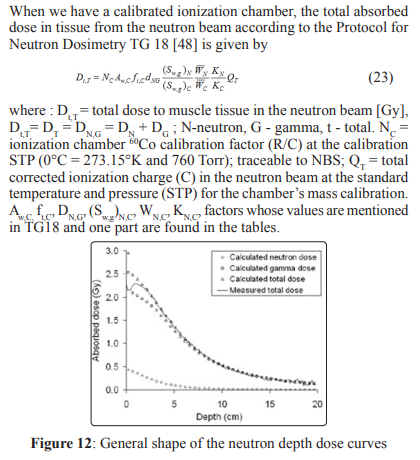
The general shape of the absorbed dose distribution generated by the neutrons (calculated and measured) for therapy is presented in Fig. 12 [49].
Interaction of electrons with matter
The major interacting processes of the secondary electrons resulting from EM radiation interactions with the substance, based on the Coulombian interaction with the orbital electrons of the atoms of the substance, are: excitation of the atom by the transfer of an orbit electron to a higher energy level and ionization of the atom by the separation of an orbital electron atom.
Both processes are considered inelastic processes, because by interaction some of the kinetic energy of the incident electron is consumed due to the binding energy of the target electron in the atom.
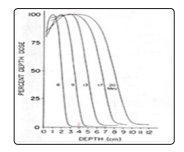
Figure 13: Central axis depth dose for large fields of Therac -20/ Saturne electrons [50].
The finite dose in water exposed to electron beams with the energies of 6, 9, 13, 17 and 20 MeV are shown in Fig. 13 [50].
Mass stopping power for electron and positron
If the indirect ionization radiation has been found to be the linear mass attenuation coefficient (μ/ρ)[cm2/g] for the direct ionization radiation, the mass stopping power(S/ρ) ≡ dT/ρdx [MeVcm2/g], in which T is the kinetic energy of the electron and dx is the length of the trajectory. The unit of measurement in SI is J.m2/kg or in traditionally units [MeV.cm2/g]. The total mass stopping power, (S/ρ) = (S/)col + (S/ρ)rad, consists of two components: mass collision stopping power (S/ρ)col resulting from electron-orbital electron interactions (atomic ionizations and atomic excitations) and mass radiation stopping power (S/ρ)rad resulting mainly from electron -nucleus interactions (bremsstrahlung production).
Stopping powers are rarely measured, rather they are calculated from theory. The calculation formula for the mass collision stopping power for electrons and positrons is taken from the ICRU Report No. 37 [51].

In Fig. 14 is shows the mass collision stopping power for electrons [52-53].
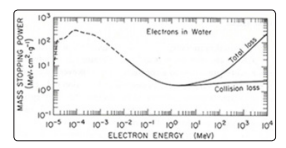
Figure 14: Stopping power of electrons in water as a functions of electron energy E [MeV] [52]
Restricted stopping power for charged particles
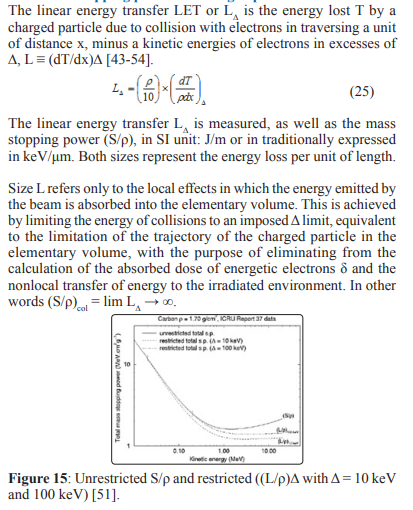
In Fig. 15 is presented the total mass stopping powers for carbon, based on data published in the ICRU Report 37. Vertical lines indicate the points at which restricted and unrestricted mass stopping powers begin to diverge as kinetic energy increases. As the threshold for maximum energy transfer in the restricted stopping power.
There is research linking LET to RBE. RBE is the ratio between the dose of a reference radiation with low LET and the dose of a considered radiation that causes an identical biological effect [54].
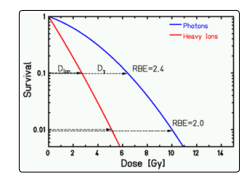
Figure 16: The dose response for cell survival.
The survival of a cell according to the administered dose (in the semi-logarithmic scale is presented in Fig. 16 (ICRU 2007). In this case, the RBE depends on: cell / tissue type, oxygen status, dose, dose rate and particle species. In this case Deff =Dphy.RBE.
Bragg-Gray Principle
The Bragg-Gray principle refers to the relationship between the dose absorbed in an irradiated medium and the dose absorbed in the ionization chamber cavity of sufficiently small size that does not disrupt the fluence of the secondary electrons to contribute to the dose absorbed in the ionisation chamber cavity [16].
The ionization chambers built on this principle have to meet two conditions: 1. The chamber cavity dimensions are small compared to the range of incidents charged particles and 2. The absorbed dose in the cavity is only from the charged particles that cross the cavity


Depth Dose for Hadrons
Protons and carbon ions are used in therapy due to the Bragg-Gray peak that occurs at the end range of these particles in the environment and due to the low-value constant dose of absorbed dose in adjacent tumor tissues that maintain a level until the BG peak appears.
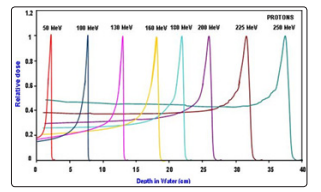
Figure 17: The distribution of the absorbed dose in water for protons with energy between 50 and 250 MeV.
In Fig. 17 shows the distribution of the absorbed dose in water for protons with energy between 50 and 250 MeV [55].
Mass Stopping Power for Heavy Charged Particles
The theory of the mass collision stopping power for heavy charged particles, electrons and positrons as a result of soft and hard collisions combines the Bethe theory for soft collisions with the stopping power as a result of energy transfers due to hard collisions. The result of this, for a heavy charged particle with mass M and velocity v, where the energy transfer due to hard collisions is limited to 2 mc2 β2 / (1-β2) with β=v/c, is [56].
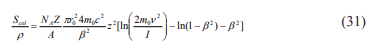
Where r0 is the classical electron radius (2.82 fm), z is the projectile charge in units of electron charge and I is the mean excitation potential of the medium.
Collision stopping power ratio for carbon ions with energy in the clinical domain of 100-450 MeV/u is presented in Fig. 18 [57].
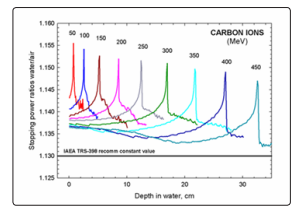
Figure 18: Stopping power ratio water/air for carbon ions
Absorbed Dose for Hadrons in Other Quality Q

Table 1: Specific factors for Q hadron beams and for the Qocalibration beam
|
kQ, Qo parameter |
Value for protons |
Values for carbon ions |
|
(sw,air) Qo |
1.133 |
1.133 |
|
(sw,air) Q |
Function of E |
Function of E |
|
(Wair/e) Qo |
33.97 eV |
33.97 eV |
|
(Wair/e) Q |
34.50 eV |
34.23 eV |
|
pQo |
1.009 |
1.009 |
|
pQ |
1.0 |
1.0 |
Equivalent Dose and Effective Dose Limits
In the field of radiobiology and radiotherapy, it is necessary to know the absorbed dose within a tissue or organ. In this respect, ICRP Publication 60 recommends the concept of tissue or organ absorbed dose DT, defined as the quotient of ET by mT, where ET is the total amount of energy deposited in the organ or tissue and mT is the mass of the organ or tissue, DT=ET/mT. The absorbed dose measurement unit is the Gray (1Gy = 1J/kg = 6.24 x 109 MeV/g =104 erg/g = 100 rad (radiation absorbed dose).

ICRP recommended anual dose limits for occupational personell/ general public, the effective dose E for whole body: 20 [mSv/1mSv] and the equivalent dose H for lens: 150/15 [mSv/mSv], skin 500/50 [mSv/mSv] and hands & feet 500 [mSv]. Note that such limits exclude any medical or natural background radiation doses.
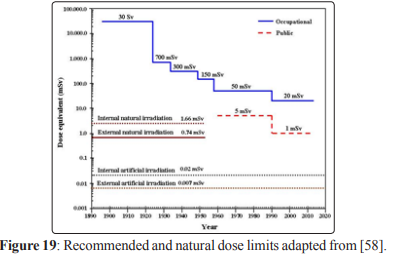
The dose limits recommended for exposure cases planned for equivalent dose for personnel working in the radiation domain and for members of the public is shows in Fig.19. Moreover, the levels of the dose equivalent for the internal and external natural irradiation as well as for the internal and external artificial irradiation are presented [58].
ICRP recommends for whole body E = 20 mSv for occupational personal and E = 1 mSv for general public, HT= 150 mSv for occupational and 15 mSv for general public, for skin HT = 500 mSv.
Conclusion
The principle of dosimetry with the ionization chamber for determining the absorbed dose applies to all kinds of radiation and materials, since the cavities of the electronic equilibrium ionization chambers respond to the electrons released inside the cavity, and the cavities of the Bragg-Gray chambers respond to electrons released outside the cavity (μen/ρ).
The principle of charged particle equilibrium (CPE) links the absorbed dose into the large cavity ionization chamber and the absorbed dose in the irradiated medium containing the ionization chamber. The energy fluence Ψ of the radiation field is a common factor in determining the three dosimetric sizes at the point where CPE exists, as follows: D = Ψ. (μen/ρ), K = Ψ. (μtr/ρ), Kcol = Ψ. (μen/ρ), and X = Ψ.(1/ΨR), where the multiplication is done with the inverse of the incident energy fluence, ΨR, of the 1 R radiation field.
The Bragg-Gray principle links the dose absorbed into the small-sized air cavity and thin walls and the absorbed dose in the irradiated medium containing the ionization chamber. For this use the secondary electrons fluence Φe generated in the irradiated medium. The absorbed dose is equal to the secondary electron fluency multiplied by the electron energy collision stopping power (S/ρ)col, D = Φe. (S/ρ)col.
The use of mechanisms to accelerate the hadrons using laser technology to the energies required for radiotherapy (50-250) MeV for protons and (100-450) MeV /u for carbon ions by means of the laser electric field instead of RF electric field will lead to the multiplication of hadron therapy in order to benefit as many patients as possible from these treatments.
References
- Bevalacqua J. J. (2007) Health Physics in the 21st Century, WILEY-VCH Verlag GmbH & Co. KGaA.
- Amaldi U, R. Bonomi, S. Braccinin, M. Crescenti, A. Degiovanni, et al. (2010) Accelerators for hadrontherapy: From Lawrence Cyclotrons to Linacs, Nucl. Instr. and Meth.in Phys. Res. A 620: 563-577.
- Scarlat F, Stancu E, Badita E, Scarisoreanu A, Vancea C, et al. (2017) Detection and Absorbed Dose Determination in Clinical Radiation Fields, Proc. of the 3rd Int. Conference on Sensors and Electronic Instrumentation Advances 113-118.
- Scarlat F, Scarisoreanu A, Badita E, Vancea C, Calina I, et al. (2015) Ionization Chamber Dosimetry for Conventional and Laser - Driven Clinical Hadron Beams, Journal of Biosciencis and Medicines 3: 8-17.
- Badita E, Vancea C, Calina I, Stroe D, Dumitrache M, et al. (2017) Long term stability of the performance of a clinical linear accelerator and z-score assessment for absorbed dose to water quantity, Romanian Reports in Physics vol. 69: 606.
- Stancu E, Vancea C, Valenta J, Zeman J, Badita E, et al. (2015) Assessment of absorbed dose to water in high energy photon beams using different cylindrical chambers, Romanian Reports in Physics 67: 693-699.
- Macchi A, Borghesi M, Passoni M (2013) Rev. Mod. Phys 85: 751.
- Robinson A.P.L, Gibbon P, Zepf M, Kar S, Evans R.G, et al. (2009) Relativistically correct hole boring and ion acceleration by circularly polarized laser pulses, Plasma Phys. Control. Fusion 51, 024004.
- Bulanov S.V, Khoroshkov V.S (2002) Feasibility of Using Laser Ion Accelerators in Proton Therapy, Plasma Physics Reports 28: 453-456.
- Stancu E, Badita E, Scarlat F, Scarisoreanu A, Scarlat F(2014) Evaluation of quality factor for clinical proton beams, Romanian Reports in Physics 66: 192-199.
- Stancu E, Vancea C, Valenta J, Zeman J, Badita E, et al. (2015) Absorbed dose to water measurements in high energy electron beams using different plane parallel chambers, Romanian Reports in Physics 67: 1152-1158.
- Scarisoreanu A, Scarlat F, Stancu E, Badita E, Dumitrascu M, et al. (2017) Absorbed dose to water and air kerma results for measurements carried out in an oncology radiotherapy laboratory, Romanian Reports in Physics 69: 605.
- Stancu E, Kapsch R.P, Badita E.S, Scarlat F, Scarisoreanu A.M (2018) The equivalence of the absorbed dose to water in high energy photon beams 12: 237-239.
- Scarlat F, Scarisoreanu A, Minea R, Badita E, Sima E, et al. (2013) Secondary Standard Dosimetry Laboratory at INFLPR, Optoelectronics and Advanced Materials - Rapid Communications 7: 618-624.
- Attix F. R, Roesch W.C, Tochilin E (1968) Radiation Dosimetry, Second Edition, Vol. I, Fundamentals (Academic Press, New York).
- Case K. R, Nelson W. R (1972) Concepts of Radiation Dosimetry, SLAC 153 UC 34, MISC.
- International Commission on Non-Ionizing Radiation Protection ICNIRP (1998), Publication - 1998, ICNIRP Guidelines for limiting Exposure to Time-Varying Electric, Magnetic and Electromagnetic Fields (up to 300GHz), Health Physics 74: 494-522.
- Johnson C. C, Guy A. W (1972) Non ionizing Electromagnetic Wave Effects in Biological Materials and Systems, Proc. IEEE 6: 692-718.
- Al-Ghazi M. S. A. L, Arjune B, Fiedler J. A, Sharma P. D (1988) Dosimetric aspects of the therapeutic photon beams from a dual- energy linear accelerator, Med. Phys 15: 250-257.
- Gibbon P (2011) Physics of High Laser Plasma Interactions, Varena Summer School on Laser Plasma Acceleration 6-132.
- Saldin E. L, Schneidmiller E.A, Yurkov M. W (2006) Coherence properties of the Radiation from X-ray free electron laser, DESY 288: 1179-1188.
- Sprangle P, Ting A, Esarey E, Fisher A (1992) Tunable short pulse x-rays from a compact laser synchrotron source, J. Appl. Phys 72: 5032-5038.
- Scarlat F, Oproiu C (1994) The 40 MeV Medical Betatron. Experience versus Predictions, Proceedings of the Fourth European Particle Accelerator Conference (EPAC-94), London, England 3: 2616- 2618.
- Scarlat F, Scarlat Fl (2002) A Potential X Ray Laser Synchrotron Source for Small Laboratories”, Proc of the 27 ARA Congress, (ARA-27), Oradea, Romania.
- Chernyaev A. P, Varzar S. M (2014) Particle Accelerators in Modern World, Physics of Atomic Nuclei 10: 1203-1215.
- Lawrence E.O (1932) Method and Apparatus for Acceleration for Ions, Fied Jan 20, 1932, US Patent Office, Serial No. 589033.
- Craddock M. K (2011) Cyclotrons: from Science to Human Health and Triumf, American Physical Society, April Meeting 2011, Anaheim, CA.
- Yves Jongen (2010) Review of cycloyrons for cancer therapy, FRM1CIO01, Proceedings of CYCLOTRONS 2010, Lanzhou,China 398- 403.
- Röcken H, Abdel-Bary M, Akcöltekin E, Budz P, Stephani T, et al. (2010) The VARIAN 250 MeV Superconducting Compact Proton Cyclotron: Medical Operation of the 2nd Machine, Production and Commissioning Statutus of Machines No. 3 to 7, Proceedings of Cyclotrons 2010, Lanzhou, ChinaTUM1CCO04: 283-285.
- Morozov N (2010) IBA-JINR 400 MeV/u Superconducting Cyclotron for Hadron Therapy, FRM1CIO03, Proceedings of CYCLOTRONS 2010, Lanzhou, China 398-403.
- Coutrakon G (2007) Accelerators for Heavy-charged-particle Radiation Therapy, Technology in Cancer Research & Treatment, Volume 6, Number 4 Supplement 6: 49-54.
- Scarlat F, Scarisoreanu A, Verga N, Scarlat Fl (2015) “Laser -Driven Hadron Therapy Project”, International Conference on Accelerators and Large Experimental Physics Control Systems, ICALEPCS 2015, Melbourne, Australia.
- Carrigan R.A. Jr (1982) On the possible applications of the steering of charged particles by bent single crystals including the possibility of separated charm particle beams 194: 205-208.
- Badita E, Vancea C, Stancu E, Scarlat F, Calina I, et al. (2016) Study on the development of a new single mode optic fiber radiation dosimeter for electron beams, Romanian Reports in Physics 68: 604-614.
- Badita E, Stancu E, Vancea C, Scarlat F, Calina I, et al. (2015) Influence of high energy ionizing radiation on single mode optical fiber proprieties, The third international conference on radiation and applications in various fields of research 41-45.
- Zamfir N.V (2012) Extreme Light Infrastructure - Nuclear Physics ELI-NP, Experimental Programme Workshop at ELI-NP, Bucharest.
- Verga N, Ursu I, Craciun L, Mirea D. A, Vasilescu R, et al. (2014) Eye proton therapy: proposed feasibility plan SF, Romanian Reports in Physics 66: 223-246.
- T. Esirkepov, M. Borghesi, S. V. Bulanov, G. Mourou, T. Tajima (2004) Highly Efficient Relativistic-Ion Generation in the Laser-Piston Regime, Phys. Rev. Lett 92: 175003.
- Esirkepov T, Yamagiva M, Tajima T (2006) Laser Ion Acceleration laws Scaling Seen Multiparametric Particle-in-cel Simulation, Phys. Rev. Lett 96: 105001.
- Scarlat F, Scarisoreanu A, Verga N, Scarlat Fl, Vancea C (2014) ”Towards Laser Driven Hadron Cancer Radiotherapy: Journal of Intense pulsed Lasers and Applications in Advances Physics 4: 55-64.
- Scarlat F, Verga N, Scarisoreanu A, Badita E, Dumitrascu M, et al. (2013) Journal of Intense Pulsed Lasers and Applications in Advanced Physics 3: 15-25.
- Fitzgerald J. J, Brownell G. L, Mahoney F. J. (1967) Mathematical Theory of Radiation Dosimetry (Gordon and Breach, New York).
- Dance D. R, Christofides S, Maidment A.D.A, McLean I. D,K. H. Ng (2014) Diagnostic Radiology Physics, A Handbook for Teachers and Students, IAEA Vienna.
- Martin J. E. (2006) Physics for Radiation Protection A Handbook, WILEY-VCH Verlag GmbH & Co. KGaA.
- Turner J. E. (2007) Atoms, Radiation, and Radiation Protection, WILEY-VCH Verlag GmbH & Co. KGaA.
- Evans R.D (1955) The Atomic Nucleus, McGraw-Hill Book Company, Inc., New York 2: 11s-12s.
- IAEA TRS 398 (2000) Absorbed dose determination in external beam radiotherapy. An international code of practice for dosimetry based on standards ao absorbed dose to water. Tehnical Report no 398.
- AAPM Report No 16 (1966) Protocol for heavy Charged particle Therapy beam dosimetry, A Report of Task group 20 Radiation Therapy Committee American Association of Physicists in Medicine, American Institute of Physics, New York 10017.
- https://www.google.ro/ search?q=depth+dose+in+water+for+neutrons.
- Sharma S. C, Wilson D. L (1985) Depth dose characteristics of elongated fields from electron beams from a 20 MeV accelerator, Med. Phys 12: 419-423.
- ICRU Report 37 (1984) Stopping Powers for Electrons and Positrons, International Commission on Radiation Units and Measurements, Bethesda, MD, USA.
- IAEA, TRS 188 (1979) Radiological safety Aspects of the operation of Electron Linear Accelerator, Vienna.
- ICRU Report 21 (1971) Radiation Dosimetry: Electrons with initial energies Between 1 and 50 MeV, International Commission on Radiation Units and Measurements, Washington D.C, USA.
- ICRP, 1991b. 1990 Recommendations of the International Commission on Radiological Protection. ICRP Publication 60, ann. ICRP 21 (1-3).
- The normalized (at peak) Bragg Curves for Various Proton Incident Energies in Water Phantom: A simulation with GEANT4 Monte Carlo Code, Abstract ID 8159, www.aapm. org/meetings/amos2/pdf.
- Berger M. J, S.M.Seltzer (1983) Stopping Powers and Ranges of Electrons and Positrons, NBSIR 82-2550-A, NBS - DC, U.S.A.
- Geithner O, Andreo P, Sobolevsky N, Hartman G, Jakel O (2006) Calculating of stopping power ratios for carbon ions dosimetry, Phys. Med. Biol 51: 2279-2292.
- Inkret W. C, Meinhold C. B, Tascher J. C (1995) A Brief History of Radiation Protection Standards, Los Alamos Science 23: 116-123.


