Research Article - (2020) Volume 2, Issue 1
In-vitro Antibacterial activity of Rhus javanica Against Multidrug-Resistant Uropathogens
2Department of Pharmacy, Janamaitri Foundation Institute of, Health Sciences (JFIHS), Hattiban, Lalitpur, Nepal
Received Date: Nov 21, 2019 / Accepted Date: Dec 09, 2019 / Published Date: Jan 08, 2020
Copyright: ©Bhuvan Saud. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background and objective: Antibiotic resistance is emerging as a major public health concern. From the ancient time in history, different plants and herbs have been known to have medicinal value. Rhus javanica has been found to show antibacterial activity against pathogenic bacteria. Thus, present study is designed to investigate the prevalence of urinary tract infection causing bacterial pathogens, its antibiogram and effect of Rhus javanica in standard in-vitro condition against multidrug-resistant.
Materials and Methods: Cross-sectional study was conducted in urinary tract infection suspected patients visiting a clinic in Kalimati, Kathmandu, from August to October 2019. A total of 133 midstream urine samples were collected and cultured in MacConkey agar and Blood agar media and isolates were identified by standard biochemical tests. Antibiotic susceptibility testing was performed according to Clinical and Laboratory Standard Institute (CLSI) guideline. Leaves extract of Rhus javanica was obtained by maceration using 50.0% methanol andsusceptibility testing was performed by using cork borer method in Mueller-Hinton agar.
Results: Out of total 133 samples, 35.3%showed significant bacterial growth (i.e. 105 cfu/ml) on agar plate. The most predominant organism was Escherichia coli 17.3%, followed by Enterococcus faecalis 6.8%, Klebsiella pneumoniae 3.8%, Pseudomonas aeruginosa 3.0%, Proteus vulgaris 2.3%, and Staphylococcus aureus 2.3%. Out of 47 isolates, 53.2% were Multi Drug Resistant (MDR). Various concentrations of the crude extract used (25, 50, 75 and 100 mg/ml) showed susceptibility to all the bacterial pathogens isolated, with the diameter of zone of inhibition ranging from 12 to 28 mm.
Conclusion: Methanolic extract of Rhus javanica showed antibacterial activity against multidrug-resistant isolates of bacterial uropathogens in standard in-vitro condition.
Keywords
Rhus javanica, Escherichia coli, antibacterial activity; multidrug-resistant
Introduction
Antimicrobials are medicines used to prevent and treat infections in human, animals and plants. Haphazard use or misuse of antimicrobials in daily life leads to evolutionof alteration in microbial response and become antibiotic resistant. Globally, Antimicrobials resistance is rising rampantly which leads to increased mortality and morbidity [1]. In developed country like United State, 2 million peoples are infected and 23,000 peoples die due to antibiotic-resistant bacteria every year [2]. In Nepal the mortality rate is high due to diarrheal diseases, respiratory tract infection, blood stream infection, urinary tract infection, tuberculosis and sexually transmitted diseases [3]. The evidence of multidrug resistant pathogens is reported from the various studies conducted in different part of Nepal [4, 5]. The major culprits include pathogens like extended-spectrum β-lactamase-producing Escherichia coli and Klebsiella pneumonia, carbapenem-resistant Enterobacteriaceae, Methicillin-Resistant Staphylococcus aureus (MRSA), Vancomycin Resistant Enterococcus (VRE), Pseudomonas aeruginosa and Acinetobacter baumannii.
A total of 75% of the population of developed and developing countries have been using plant extracts for medicinal purpose and it is believed that plants provide variety and best source of drug [6]. In Nepal, about 70-80% population from mountain and hilly regions is dependent on traditional medicine for primary health care [7]. Globally, various studies revealed that natural herbs are effective against human pathogenic bacteria in both in-vitro and in-vivo conditions [8, 9]. Rhus javanica, a member of Anacardiaceae is traditionally used to treat dysentery and diarrhea [7]. In phytochemical screening it is evident that, Rhus javanica contains phenolics, glycosides, flavonoids, terpenoids and organic acids. The antibacterial activity might be due to the presence of phenolic compound [10]. However, there is limited evidence regarding the effect of Rhus javanicaon Multi Drug Resistant (MDR) bacterial isolates from Urinary Tract Infection (UTI) patients. In this study, we aimed to investigate the prevalence of UTI causing bacterial pathogens and its Antibiotic Sensitivity Testing (AST) pattern, and evaluation of antibacterial effect of Rhus javanica against MDR bacterial isolates in standard in-vitrocondition.
Materials and Methods
Cross-sectional study was designed from August to October 2019 urine samples were collected from 133 UTI suspected participants visiting clinic in Kathmandu. Further processing was done in the Department of Medical Laboratory Technology, JF Institute of Health Sciences, Hattiban, Lalitipur. Samples were inoculated into MacConkey Agar (Hi-media,India) and Blood Agar(Hi-media, India) by standard loop inoculation method (1μl volume) and incubated in controlled in-vitro condition. Standard biochemical tests were performed for further identification.
Antibiotic susceptibility testing for the pathogenic organism isolated in culture was done by Kirby-Bauer Disk Diffusion method as per the recommendation of Clinical and Laboratory Standard Institute (CLSI) using Mueller Hinton agar (MHA) (Hi-media, India). McFarland turbidity standard 0.5 was used and Hi-media, India antibiotic disk were used for AST, which can be enlisted as follows: Amoxycillin (10 μg) Nalidixic acid (30 μg), Nitrofurantoin (300 μg), Norfloxacin (10 μg), Ofloxacin (5 μg), Amikacin (30 μg), Cefixime (30 μg), Ampicillin (10 μg) and Ceftriaxone (30 μg) [11].
Rhus javanica,selected in this study was collected from Swoyambhu area of Kathmandu valley. The identification of the specimen was confirmed by Departmentof Plant Resources, Godawari, Nepal and voucher specimen was deposited at the Herbarium of the Department. Dried leaves, exactly, 600 g was soaked in 3000 mL of 50.0% methanol for 72 hour at room temperature. The extracted solution was filtered and evaporated under reduced pressure to yield methanol extract of 42.4 g (7.1%). After the extract was thoroughly dried to facilitate complete removal of the solvent, the dry extract was dissolved in Dimethyl sulfoxide (DMSO) to give the desired stock solution. Antibacterial testing was performed in MHA media by cork borer method in different concentrations of extract (i.e. 25, 50, 75 and 100 mg/ml. and 0.1% DMSO as control) prepared in 0.1 % DMSO. All experiments were performed in duplicate. Data were entered and analyzed by using the Statistical Package for Social Sciences (SPSS) version 20.
Results
Out of total 133 urine samples, 35.33% (47/133) showed bacterial growth and 64.67% (86/133) did not show any bacterial growth. Among them, female to male ratio was 2:1. Among the total positive cases of 47, 65.95% (31/47) of the females were reported with urinary tract infection whereas 34.05% (16/47) of the males were reported with urine infection. Highest number of positive cases were observed from the age of 60-70 years (27.7%; 13/47), followed by age group 10-20 and 40-50 years (17.0%; 8/47). The least number ofpositive cases was obtained from the age group of less than 10 years (0.0%; 0/47). During the study period, 6 different species of bacteria were isolated.Escherichia coli 23 (17.3%) was the predominant organism among the total organism followed by Enterococcus faecalis 9(6.8%), Klebsiella pneumoniae 5(3.8%), Pseudomonas aeruginosa 4(3%), Proteus vulgaris 3(2.3%), and Staphylococcus aureus 3(2.3%) as shown in figure 1.
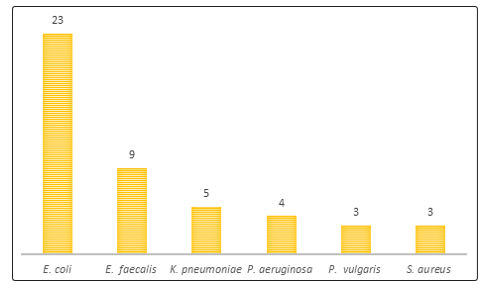
Figure 1: Bacterial isolates from UTI suspected patients urine samples
Among the antibiotics used against the isolated organism, the most effective antibiotic was Nitrofuration (70.2%) followed by Levofloxacin (68.0%) and Amikacin (53.2%). Most of the organisms were resistance to Amoxycillin(95.7%) followed by Ampicillin(85.1%), Nalidixic acid (80.9%) shown in figure 2.
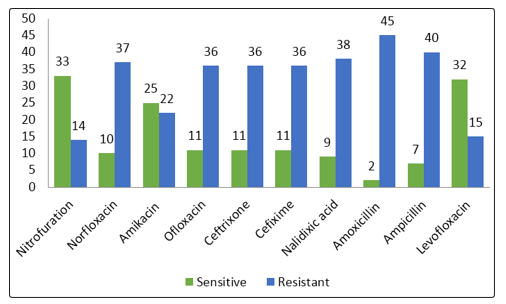
Figure 2: Antibiotic susceptibility pattern of bacterial isolates
Most of the bacteria isolated (53.2%)(n=25) were found to be resistant to antibiotics and were considered MDR and the remaining 46.8% (n=22)were non-MDR. It was found that 17(36.1%) isolates were sensitive to all antibiotic and (5.5%) (n=4) were resistant to 1 drug. Among the total isolates, 25 were MDR strains, where MDR strains in individual bacteria was 66.6% (n=2) in Proteus vulgaris and Staphylococcus aureus, 55.5% (n=5) in Enterococcus faecalis, 52.2% (n=12) in Escherichia coli, 50.0% (n=2) in Pseudomonas aeruginosa and 40% (n=2) Klebsiella pneumoniae, as shown in Table 1
Table 1: Resistance pattern and distribution of MDR bacterial isolates
|
Organisms |
Total Isolates |
Resistance |
Total MDR (%) |
|||
|
0 |
1 |
2 |
>3 drugs |
|
||
|
Escherichia coli |
23 |
9 |
2 |
3 |
9 |
52.2 |
|
Pseudomonas aeruginosa |
4 |
1 |
1 |
1 |
1 |
50.0 |
|
Klebsiella pneumoniae |
5 |
3 |
0 |
1 |
1 |
40.0 |
|
Staphylococcus aureus |
3 |
0 |
0 |
2 |
0 |
66.6 |
|
Enterococcus faecalis |
9 |
3 |
1 |
3 |
2 |
55.5 |
|
Proteus vulgaris |
3 |
1 |
0 |
1 |
1 |
66.6 |
Methanolic extract of Rhus javanica showed antibacterial effect against all MDR bacterial isolates of Escherichia coli, Enterococcus faecalis, Klebsiella pneumoniae, Pseudomonas aeruginos, Proteus vulgaris and Staphylococcus aureu. Zone of Inhibition (ZOI) was seen to increase with increasing concentration of extract. Diameter of inhibition was found to range from12 mm.to 28 mm, as shown in table 2.
Table 2: Effect of methanolic extract of Rhus javanica against MDR bacterial isolates
|
Organisms |
Total Isolates |
ZOI(mm) |
|||
|
Concentration (mg/ml) |
|||||
|
25 |
50 |
75 |
100 |
||
|
Escherichia coli |
12 |
17-20 |
18-22 |
21-28 |
22-28 |
|
Pseudomonas aeruginosa |
2 |
13-18 |
14-20 |
16-19 |
21-26 |
|
Klebsiella pneumoniae |
2 |
12-15 |
16-19 |
18-23 |
22-27 |
|
Staphylococcus aureus |
2 |
14-16 |
15-22 |
17-22 |
21-24 |
|
Enterococcus faecalis |
5 |
13-15 |
13-20 |
16-20 |
17-21 |
|
Proteus vulgaris |
2 |
15-18 |
16-17 |
16-20 |
18-25 |
Discussion
Development of antimicrobial resistant in bacteria, virus and fungi is become major issue for human, animal and plants. Both developed and developing countries are equally affected by this burden. In present study, the prevalence of bacterial infection among study subjects is found to be 35.3%, in which Escherichia coli is the predominant organism and followed by Enterococcus faecalis and Klebsiella pneumoniae. AST showed that, out of total isolates, 53.2% were MDR.Similar finding was reported by Shakya et al.,where 52.3% of the total isolates were MDR, in which the main pathogens are Escherichia coli 79.7%, Providencia species 5.1%,and Klebsiella pneumoniae 3.4% [4]. A study from eastern Nepal also showed that, 43.98 % of urine samples showed significant growth and pathogens were resistant to amoxycillin, co-trimoxazole, flouroquinolones and third-generation cephalosporins [12]. In addition, Chaudhary et al. reported that 39.2% of the total isolates were MDR main pathogens are Staphylococcus aureus and Escherichia coli in various surgery sites [13].
In the present study, we examined the antimicrobial activity of Rhus javanica extract against MDR urinary tract bacterial pathogens. Our study showed that, 25 to 100 mg/mL of the methanol extract inhibitedgrowth of MDR isolates of Escherichia coli, Enterococcus faecalis, Klebsiella pneumoniae, Pseudomonas aeruginos, Proteus vulgaris and Staphylococcus aureus in standard in-vitro condition. The diameter of zone of inhibition is high with increased concentration of extract where the diameter of inhibition was found range from12 to 28 mm in MHA. A study conducted in MRSA, revealed that extract of Rhus javanica inhibited the bacterial growth by significantly decreasing the expression of mecA, sea, agrA and sarA gene [10]. Another study showed that, extract showed inhibitory effect against shigellosis causing pathogens,Shigella flexneri and Shigella dysenteriae, like other bacterial pathogens [14].
Rhus javanica has been used to treat dysentery and diarrhea in different Asian countries including Korea, China, and Japan [15]. Several studies showed that, Rhus javanica extract was effective against animal husbandry disease-related bacteria like Listeria monocytogenes and Staphylococcus epidermidis, fish pathogens like Vibrio ichthyoenteri and Streptococcus iniae [16, 17]. Leaves of Rhus javanica primarily contains gallic methy lester, syringic acid, protocatechuic acid, and 1, 2, 3, 4, 6-penta-O-galloyl--D-glucose [18]. You et.al., suggested that phenolics may have been responsible for the antibacterial activity and Leaves of Rhus javanica contains phenolics, glycosides, flavonoids, steroids and organic acids [10]. Various factors like lack of knowledge and awareness regarding rational use of antibiotics in human and animals, lack of sophisticated laboratory for diagnosis and practice of medicine that usually overlooks standard microbiology protocol, are responsible for emergence of antibiotic resistance in Nepal [3, 19]. Our study revealed that crude extract of Rhus javanica shows antimicrobial activity against all MDR uropathogens in standard in-vitro condition.
Conclusion
Crude extract of Rhus javanica shows antibacterial activity in all the MDR isolates. Further study is needed for purification and isolation of the active compound responsible for such activity in vitro.
Authors Contribution
BS, GP &TS- Study design and laboratory test. BS, GP & MG - data analysis and prepare manuscript, MG, RKS &NB- plant collection and processing and GD & VS- critical comment on manuscript.
References
- World Health Organization (WHO). Antibiotic resistance. 2018. https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance
- Centers for Disease Control and Prevention (CDC). Antibiotic / Antimicrobial Resistance (AR / AMR);2019. https://www.cdc. gov/drugresistance/about.html
- Adhikari S, Saud B, Paudel G, Bajracharya D (2019) Emergence of Antimicrobial Drug Resistant Bacteria in Nepal: A Current Scenario. Proteomics Bioinformatics Current Res 1: 31-33.
- Shakya P, Shrestha D, Maharjan E, Sharma VK, Paudyal R (2017) ESBL production among E. coli and Klebsiella spp. causing urinary tract infection: A hospital based study. Open Microbiol J 11: 23-30.
- Ansari S, Nepal HP, Gautam R, Sony Shrestha, Puja Neopane, et al. (2014) Childhood septicemia in Nepal: Documenting the bacterial etiology and its susceptibility to antibiotics. Int J Microbiol 2014: 1-6.
- Saud B, Malla R, Shrestha K (2019) A Review on the Effect of Plant Extract on Mesenchymal Stem Cell Proliferation and Differentiation. Stem Cells International 2019.
- Kunwar RM (2003) Ethnobotanical notes on flora of Khaptad National Park (KNP), far-western Nepal. Himalayan Journal of Sciences 1: 25-30.
- Shan B, Cai YZ, Brooks JD, Corke H (2007) The in vitro antibacterial activity of dietary spice and medicinal herb extracts. International Journal of food microbiology 117: 112-119.
- Choi JG, Kang OH, Lee YS, Hee-Sung Chae, You-Chang Oh, et al. (2011) In vitro and in vivo antibacterial activity of Punica granatum peel ethanol extract against Salmonella. Evidence-Based Complementary and Alternative Medicine 2011.
- You YO, Choi NY, Kang SY, Kim KJ (2013) Antibacterial activity of Rhus javanica against methicillin-resistant Staphylococcus aureus. Evidence-based complementary and alternative medicine 2013.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing 27th ed. CLSI supplement 2017: M100: P. A. Wayne, https://clsi.org/ media/1469/m100s27_sample.pdf .
- Chaudhari BK, Singh GK, Parajuli KP, Shrestha K (2016) Incidence and susceptibility of uropathogens isolated among the patients at tertiary care hospital in Eastern Nepal. Journal of Nobel Medical College 5: 51-55.
- Chaudhary R, Thapa SK, Rana JC, Shah PK (2017) Surgical site infections and antimicrobial resistance pattern. J Nepal Health Res Counc 15: 120-123.
- Yang SK, Kim HJ, Oh SW (2014) Antimicrobial Activities of Ethanol Extracts of R hus javanica and T erminalia chebula Against S higella Species. Journal of Food Safety 34: 95-102.
- Im Seon Lee, Oh SR, Ahn KS, Lee HK (2001) Semialactone, isofouquierone peroxide and fouquierone, three new dammarane triterpenes from Rhus javanica. Chemical and Pharmaceutical Bulletin 49: 1024-1026.
- Korean Society of Food Science and Nutrition 32: 1214-1220.
- Kim KH, Kim AR, Cho EJ, Seong-Je Joo (2014) Antibacterial activity of Rhus javanica against the Fish Pathogens Vibrio ichthyoenteri and Streptococcus iniae. Korean Journal of Fisheries and Aquatic Sciences 47: 18-22.
- Cha B, Lee S, Rhim T, Lee K (2000) Constituents of antioxidative activity and free radical scavenging effect from Galla Rhois (Rhus javanica Linne). Korean Journal of Pharmacognosy 31: 185-189.
- Saud B, Paudel G, Khichaju S, Bajracharya D, Dhungana G et al. (2019) Multidrug-Resistant Bacteria from Raw Meat of Buffalo and Chicken, Nepal. Veterinary medicine international 2019.
- Choi I (2003) Antimicrobial activity of Rhus javanica extracts against animal husbandry disease-related bacteria. Journal of the



