Review Article - (2024) Volume 2, Issue 1
Increase in Clostridioides Difficile Infection Resulting from Over Usage of Antibiotics During Covid 19 Pandemic
2Clinical Pharmacist, NMC Specialty Hospital, Abu Dhabi, UAE
Received Date: Feb 03, 2024 / Accepted Date: Mar 02, 2024 / Published Date: Mar 25, 2024
Copyright: ©©2024 Sreehari Karuna Karan Pillai, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Pillai, S. K., Shuriqy, N. (2024). Increase in Clostridioides Difficile Infection Resulting from Over Usage of Antibiotics During Covid 19 Pandemic. COVID Res OA, 2(1), 01-07.
Abstract
Healthcare-associated infections (HAIs) due to Clostridioides difficile infections (CDIs) are a significant public health problem globally. The emergence of the novel coronavirus disease (COVID-19) caused by Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) in 2019 has exacerbated the situation. Elderly and chronically ill individuals are particularly vulnerable to COVID-19, and gastrointestinal (GI) symptoms are increasingly recognized as essential symptoms of COVID-19. Bacterial infections in COVID-19 patients are prevalent, and the rates of Clostridioides difficile infection (CDI) are high and associated with antibiotic use. The study aims to investigate the correlation between CDI and community antibiotic usage patterns during COVID-19 in 2021 compared to the previous year to identify the impact on overall CDI infection rates. The study design is a non-interventional retrospective study evaluating antibiotic usage patterns in CDI patients during the pandemic, and the data will be analysed based on the number of patients and test positivity rates. The preliminary findings of the study reveal a 27% increase in the number of symptomatic CDI infections in 2021 as compared to pre-COVID years.
Keywords
Covid-19, C. Difficile, CDI, Antibiotic Associated CDI, Gastrointestinal Symptoms,HaisIntroduction
Healthcare-associated infections (HAIs) due to Clostridioides difficile infections (CDIs) [1]is one of the leading reason of hospitalization that affects public health globally[2].The 2019 novel coronavirus (2019-nCoV), also known as the novel coronavirus disease (COVID-19), which is caused by the Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2), first appeared in Wuhan (China) in early December 2019[3]. It has since spread quickly throughout the world, resulting in a major pandemic[4]. The most vulnerable people to COVID-19 infections are the elderly and those with underlying illnesses and chronic disorders. When compared to young and middle- aged adults, the elderly are particularly seriously affected by COVID-19, both in terms of the disease's prevalence and mortality[4-5]. Most of the time, patients present with severe respiratory symptoms like fever, sore throat, cough, fatigue, dyspnea, sputum production, myalgia, fatigue, and headaches. However, it has been reported that about 19% of COVID-19 patients exhibit gastrointestinal (GI) symptoms[6], including 2-10% diarrhoea, 1-3.6% loss of appetite, nausea, vomiting, and abdominal pain, which are now universally recognised as crucial COVID-19 symptoms[7].
Although few studies have been published thus far, the rates of bacterial infection in COVID-19 patients are high and are likely underreported because it is difficult to diagnose bacterial infections in the midst of a health emergency[8]. Atypical COVID-19 clinical presentations in the elderly can make it challenging to distinguish COVID-19 from bacterial infections, and there may be an overuse of antibiotics patients with chronic underlying conditions[9].Clostridium difficile infection (CDI) in COVID-19 patients had received less attention from the scientific community than other bacterial infections, despite the fact that CDI is frequently linked to the use of broad-spectrum antibiotics, hospital overcrowding, gut microbioma changes, patient ageing, and frailty[10-11].
This study aims to investigate the correlation between C. Difficile infection with community usage of antibiotics for respiratory and other infections in 2021 as compared to the year 2020, to identify if antibiotic usage patterns in COVID-19 had an impact on overall C. Difficile infection rates.
Study design
This study is a Non-Interventional Retrospective Study evaluating Antibiotic usage patterns in patients with Clostridium difficile infection during COVID 19 pandemic to identify the impact on overall Clostridium difficile infection rates and will be conducted NMC Specialty Hospital Abu Dhabi, United Arab Emirates.
Patients: Patients were eligible for analysis if they were infected with CDI and been to NMC Specialty hospital. We are committed to protecting patient privacy and complying with the Good Clinical Practice (ICH-GCP E6(R2) and with the ethical principles stated in the Declaration of Helsinki 1964, as revised in Fortaleza, 2013.
Study Objectives
Primary: To investigate the correlation between Clostridium difficile infection with community usage of antibiotics for respiratory and other infections in 2021 as compared to the year 2020, to identify if antibiotic usage patterns in COVID-19 had an impact on overall Clostridium difficile infection rates. Secondary: Follow up on the impact of the corrective measures taken in 2021.
Analysis
Year 2019 is taken for comparison prior to the onset of COVID 19. Year 2020 saw an overall reduction in non-COVID hospitalisation. By 2021, the hospital visits had returned to normal, although the community continued to face waves of COVID-19 infections [Figure 1]. Vaccination program was going on in the community and new COVID 19 case volumes gradually returned to baseline by October 2021.
Diagnosis of Clostridium difficile is made by testing for the presence of C. Difficile antigen A or antigen B in those patients who present with diarrhoea, using immunochromatographic test. In the outpatient’s service, investigations are carried out in those with no apparent reason for the diarrhoea and common aetiologies have been excluded. This is different from the practice in the inpatient’s service, where a new onset diarrhoea is promptly screened for C Difficile antigen in the interest of safe infection control practices.
Figure 1: Covid-19 Cases in UAE 2019-2023 Courtesy Worldometers.Info
From July 2021, a spike in the total number of C difficile turning positive was observed among patients visiting out- patients department as well as admitted patients. This remained consistently above the average incidence in similar months in the preceding years [Figure 2]. These patients included those presenting to the outpatient’s services with symptoms of diarrhoea, abdominal pain and fever. During the same period, a spike was also noted in the number of admitted patient’s developing C Difficile diarrhoea.
Figure 2: Percentage of C. difficile antigen tests that were positive from 2019 to 2022
As compared to the pre-COVID year, there was an increase in symptomatic C difficile infections in 2021[Table 1].
|
Year |
Total tests |
Positive |
Percentage |
|
2019 |
813 |
51 |
6% |
|
2020 |
589 |
28 |
5% |
|
2021 |
620 |
65 |
10% |
|
2022 |
720 |
11 |
2% |
Table 1: Percentage Positivity of C Difficile Test
A similar spike for noted in the infection rates among the in-patients, in the same period between July to December 2021[Table 2].
|
Year |
C Difficile infection in IP |
|
2019 |
0 |
|
2020 |
1 |
|
2021 |
4 |
|
2022 |
0 |
Table 2: C. Difficile Among Admitted Patients
On analysing the demographics, no significant sex difference was observed among patients. Highest occurrence was among the younger age group between 18 to 39 years [Table 3].
|
Demographics |
|
2021 |
|
Sex |
|
|
|
Male |
38 |
58% |
|
Female |
27 |
42% |
|
Total |
65 |
|
|
Age |
|
|
|
<18 |
3 |
5% |
|
18 to 39 |
32 |
49% |
|
40 to 59 |
21 |
32% |
|
>60 |
9 |
14% |
|
Total |
65 |
|
Table 3: Demographic Distribution of C Difficile Antigen Positive Patients in 2021
Among those infected, 48.3 % had prior antibiotic prescription from the hospital or by doctors with referral links to the hospital.
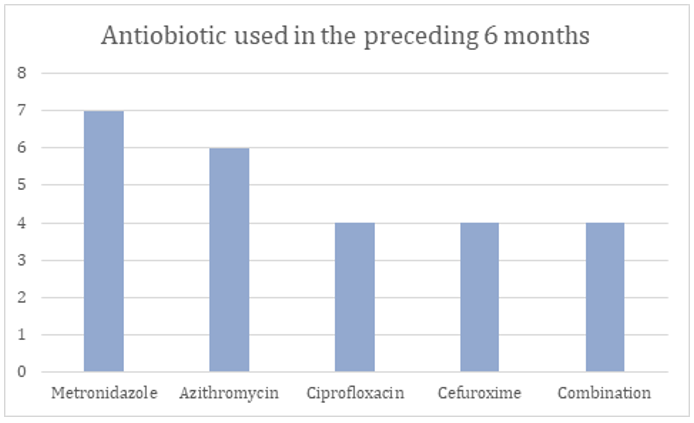
Figure 3: Antibiotic Usage Identified in the 6 Months Preceding C Difficile Infection
|
Antibiotic |
Number of patients |
% |
Indication |
|
Metronidazole |
7 |
25% |
Diarrhoea |
|
Azithromycin |
6 |
21% |
Respiratory infection |
|
Ciprofloxacin |
4 |
14% |
Urinary infection |
|
Cefuroxime |
4 |
14% |
Respiratory/Urinary infection |
|
Combination antibiotics |
4 |
14% |
Sepsis |
Table 4: Indications for Prior Antibiotic Use.
Out of the 65 positive cases, prior antibiotic usage in the preceding 6 months was noted in 28 (48%) out of the 65 patients. The commonest antibiotic used was Metronidazole, immediately followed by Azithromycin [Figure 3]. The reasons for prior antibiotic usage were as per [Table 4].
Discussion
There are significant concerns about a potential rise in Clostridioides difficile infections (CDIs) as a result of the widespread use of broad-spectrum antibiotics during the historic COVID-19 pandemic[12]. Clostridioides difficile is a multi- resistant pathogen that is known as the most common cause of diarrhoea in healthcare settings. Due to its association with antibiotic use, high morbidity, and mortality rates, it is considered one of the most important public health threats[13-14]. C. difficile causes over 500,000 infections and 29,000 deaths annually in the United States (US)[15], whereas in Europe, it causes 8382 annual deaths and 152,905 instances of C. difficile infection (CDI)[16].The elderly, immune-compromised individuals, hospitalised patients, or those receiving antibiotic treatment, which is the primary risk factor for this infection, are the target populations for CDI[17].
Our retrospective study conducted at NMC Specialty Hospital in Abu Dhabi, revealed a 27% increase in the number of symptomatic CDI infections in 2021 compared to 2019, and the highest occurrence was among the younger age group between 18 to 39 years. The analysis revealed a spike in the total number of CDI positive patients, both among those visiting outpatient departments and among admitted patients, from July to December 2021.This is consistent with the previously published data as some single-center studies also reported an increase in HA-CDI cases during the COVID-19 pandemic[18]. Boccolini et al[19] reported that CDI rates in an intensive care unit (ICU) in an Italian hospital climbed from 0.0% to 3.2% between March-April 2019 and March-April 2020,whereas Lewandowski et al[18] have noticed an increase in CDI rates in a Warsaw hospital from 2.6% in the pre-pandemic era to 10.9% during the COVID pandemic.
Despite a straightforward WHO recommendation against the use of broad-spectrum empirical antibiotics in COVID-19 patients, there had been excessive use of antimicrobial drugs in these patients, particularly in those with asymptomatic or mild- to-moderate disease[21]. The majority of pulmonary bacterial super-infections appear to be nosocomial and were linked to the use of empirical antibiotic therapy[22]. Several studies have presented data on antibiotic use in patients with COVID-19. It has been estimated that about 72% of COVID-19 patients were treated with broad-spectrum antibiotics, mostly respiratory quinolones, to prevent bacterial co-infections and super- infections[22]. About 75% of long term care residents receive at least one course of antibiotics during 6 or more months of permanence[23]. In addition to being more vulnerable to CDI, these patients may also be more likely to develop new, aggressive strains of C. Difficile due to antibiotic misuse, which will lead to negative consequences[24].
Our study also revealed that 48.3% of infected patients had prior antibiotic prescriptions from the hospital or referral links. Azithromycin usage for respiratory infections was noted as the second-highest association in patients who had developed C. Difficile infections following prior antibiotic usage. This is in contrast to another study which revealed that azithromycin has been demonstrated to not have any effect on CDI development in patients with COVID-19 compared with other antibiotics, probably due to a lower risk of CDI associated with this antibiotic[24]. Antibiotics affects the biomass, composition and function of the gut microbiota and consequently reduce colonization resistance capacity against opportunistic pathogens such as C. Difficile, and subsequently develop CDI Furthermore, the promiscuous use of antibiotics alters the overall diversity and makeup of the gut microbiota and throws off the delicate balance of microbes. Antibiotic-induced disruption of the gut microbiota is a risk factor for CDI[25-26].
It is crucial to keep in mind that the COVID-19 pandemic could make CDI diagnosis more difficult. In fact, patients with COVID-19 may not have a clinical suspicion for CDI because SARS-CoV-2 commonly induces gastrointestinal symptoms such nausea, vomiting, and diarrhoea[27-28]. Moreover, due to concerns about the presence of SARS-CoV-2 in feces[29], stool tests may have been discouraged during the pandemic to avoid nurse contact with faeces during collection, which led to an underreporting of CDI. Regarding this, delays in CDI diagnosis have been noted while COVID-19 patients were hospitalised[30-31]. A recent study conducted in nine hospitals in Massachusetts reports that about 40% of patients with COVID-19 have experienced diarrhea after admission and that two of these patients, critically ill, died prior to receiving a correct therapy for CDI[31].
The study's strength lies in its non-interventional retrospective design, which makes it less prone to biases. The study also adhered to ethical principles stated in the Declaration of Helsinki and Good Clinical Practice (ICH-GCP E6(R2)).However, this study has some limitations. Firstly, it was a single-center study which may limit the generalizability of the findings to other populations or settings and the lack of standards for the treatment of CDI in COVID-19. This highlights the importance of judicious use of antibiotics for potential secondary bacterial infection in patients with COVID-19. Further studies are needed to better assess the impact of infection control procedures reinforcement during the COVID-19 pandemic on CDI incidence and to clarify the interplay between COVID-19 and CDI.
Conclusion
The study highlights the potential correlation between community antibiotic usage for respiratory and other infections during the COVID-19 pandemic and the incidence of C. difficile infection. It emphasises the need for judicious use of antibiotics, improved surveillance, and the identification of patients at risk to prevent C. difficile infection. The findings suggest the need for more judicious antibiotic prescribing practises and greater awareness among physicians and the public about the potential risks associated with rampant antibiotic usage. Such measures can help reduce the incidence of CDI and other antibiotic- resistant infections, particularly during a pandemic. It will also guide antibiotic stewardship programmes to mitigate the risk of C. difficile infections in the future. Moreover, further research is needed to explore the long-term impact of COVID-19 on antibiotic usage patterns and C. difficile infection rates.
Declaration
Ethics Approval and Consent to Participate
The study protocol received approval from the Regional Research Ethics Committee on NMC Specialty Hospital in Abu Dhabi, UAE.
The study adhered to ethical principles stated in the Declaration of Helsinki and Good Clinical Practice (ICH-GCP E6(R2)).
An approval for waiver of informed consent was obtained from the Regional Research Ethics Committee on NMC Specialty Hospital in Abu Dhabi, UAE. This was granted on the basis of non-interventional retrospective nature of the study.
Consent for Publication
Not applicable.
Availability of Data and Materials
The datasets generated and analyzed during the study are not publicly available due to it containing patient-related information. However, they can be made available by the corresponding author upon reasonable request after anonymization.
Competing Interests
The authors declare no competing interests.
Funding
No funding was received for this study.
Authors' Contributions
Sreehari Karuna Karan Pillai conceived the study, designed the study protocol, and drafted the manuscript. Naseem Shuriqy collected and analyzed the data and created the figures. Both authors critically reviewed and edited the manuscript, and approved the final version for submission. Sreehari Karuna Karan Pillai and Naseem Shuriqy are responsible for the overall content and integrity of the study.
Acknowledgements
The authors would like to acknowledge the support provided by the infection control team members and research administration in collecting and clearing the data through scientific, ethics, and regulatory review processes.
References
- Lawson, P. A., Citron, D. M., Tyrrell, K. L., & Finegold,S. M. (2016). Reclassification of clostridium difficile as clostridioides difficile (Hall and O’Toole 1935) Prévot 1938. Anaerobe, 40, 95-99.
- Finn, E., Andersson, F. L., & Madin-Warburton, M. (2021). Burden of Clostridioides difficile infection (CDI)-a systematic review of the epidemiology of primary and recurrent CDI. BMC infectious diseases, 21(1), 456.
- Zhu, N., Zhang, D., Wang, W., Li, X., Yang, B., Song, J.,... & Tan, W. (2020). A novel coronavirus from patients with pneumonia in China, 2019. New England journal of medicine, 382(8), 727-733.
- Leung, C. (2020). Clinical features of deaths in the novel coronavirus epidemic in China. Reviews in medical virology, 30(3), e2103.
- Zhang, J. J., Dong, X., Cao, Y. Y., Yuan, Y. D., Yang, Y. B., Yan, Y. Q., ... & Gao, Y. D. (2020). Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy, 75(7), 1730-1741.
- Spigaglia, P. (2020). COVID-19 and Clostridioides difficile infection (CDI): Possible implications for elderly patients. Anaerobe, 64, 102233.
- Ramachandran, P., Onukogu, I., Ghanta, S., Gajendran, M., Perisetti, A., Goyal, H., & Aggarwal, A. (2020). Gastrointestinal symptoms and outcomes in hospitalized coronavirus disease 2019 patients. Digestive Diseases, 38(5), 373-379.
- Hendaus, M. A., & Jomha, F. A. (2021). Covid-19 induced superimposed bacterial infection. Journal of Biomolecular Structure and Dynamics, 39(11), 4185-4191.
- Frisina Doetter, L., Preuß, B., & Rothgang, H. (2021). Taking stock of COVID-19 policy measures to protect Europe’s elderly living in long-term care facilities. Global Social Policy, 21(3), 529-549.
- Páramo-Zunzunegui, J., Ortega-Fernández, I., Calvo- Espino, P., Diego-Hernández, C., Ariza-Ibarra, I., Otazu- Canals, L., ... & Menchero-Sánchez, A. (2020). Severe Clostridium difficile colitis as potential late complication associated with COVID-19. The Annals of The Royal College of Surgeons of England, 102(7), e176-e179.
- Sandhu, A., Tillotson, G., Polistico, J., Salimnia, H., Cranis, M., Moshos, J., ... & Chopra, T. (2020). Clostridioides difficile in COVID-19 patients, Detroit, Michigan, USA, March–April 2020. Emerging Infectious Diseases, 26(9),2272.
- Bartlett, J. G. (2002). Antibiotic-associated diarrhea. New England journal of medicine, 346(5), 334-339.
- Cox, M. J., Loman, N., Bogaert, D., & O'Grady, J. (2020). Co-infections: potentially lethal and unexplored in COVID-19. The Lancet Microbe, 1(1), e11.
- Huttner, B. D., Catho, G., Pano-Pardo, J. R., Pulcini, C., & Schouten, J. (2020). COVID-19: don't neglect antimicrobial stewardship principles! Clinical Microbiology and Infection, 26(7), 808-810.
- Sa'ed, H. Z. (2022). Global research on Clostridium difficile- associated diarrhoea: A visualized study. World Journal of Gastroenterology, 28(28), 3720.
- Wiuff, C., Banks, A. L., Fitzpatrick, F., & Cottom, L. (2018). The need for European surveillance of CDI. Updates on Clostridium dificile in Europe: Advances in Microbiology, Infectious Diseases and Public Health Volume 8, 13-25.
- Keller, J. M., & Surawicz, C. M. (2014). Clostridium difficile infection in the elderly. Clinics in geriatric medicine, 30(1), 79-93.
- Lewandowski, K., Rosolowski, M., Kaniewska, M., Kucha, P., Meler, A., Wierzba, W., & Rydzewska, G. (2021). Clostridioides difficile infection in coronavirus disease 2019 (COVID-19): an underestimated problem. Pol Arch Intern Med, 131(2), 121-127
- Baccolini, V., Migliara, G., Isonne, C., Dorelli, B., Barone,L. C., Giannini, D., ... & Villari, P. (2021). The impact of the COVID-19 pandemic on healthcare-associated infections in intensive care unit patients: a retrospective cohort study. Antimicrobial Resistance & Infection Control, 10(1), 87.
- Granata, G., Bartoloni, A., Codeluppi, M., Contadini, I., Cristini, F., Fantoni, M., ... & CloVid Study Group. (2020). The burden of Clostridioides difficile infection during the COVID-19 pandemic: a retrospective case-control study in Italian hospitals (CloVid). Journal of clinical medicine, 9(12), 3855.
- Getahun, H., Smith, I., Trivedi, K., Paulin, S., & Balkhy,H. H. (2020). Tackling antimicrobial resistance in the COVID-19 pandemic. Bulletin of the World Health Organization, 98(7), 442.
- Buehler, P. K., Zinkernagel, A. S., Hofmaenner, D. A., Garcia, P. D. W., Acevedo, C. T., Gómez-Mejia, A., ... & Brugger, S. D. (2021). Bacterial pulmonary superinfections are associated with longer duration of ventilation in critically ill COVID-19 patients. Cell Reports Medicine, 2(4).
- Jump, R. L., Crnich, C. J., Mody, L., Bradley, S. F., Nicolle,L. E., & Yoshikawa, T. T. (2018). Infectious diseases in older adults of long-term care facilities: update on approach to diagnosis and management. Journal of the American Geriatrics Society, 66(4), 789-803.
- Brown, K. A., Langford, B., Schwartz, K. L., Diong, C., Garber, G., & Daneman, N. (2021). Antibiotic prescribing choices and their comparative C. difficile infection risks: a longitudinal case-cohort study. Clinical Infectious Diseases, 72(5), 836-844.
- Ferreira, E. D. O., Penna, B., & Yates, E. A. (2020). Should we be worried about clostridioides difficile during the SARS-CoV2 pandemic?. Frontiers in Microbiology, 11, 581343.
- Reeves, A. E., Theriot, C. M., Bergin, I. L., Huffnagle, G. B., Schloss, P. D., & Young, V. B. (2011). The interplay between microbiome dynamics and pathogen dynamics in a murine model of Clostridium difficile infection. Gut microbes, 2(3), 145-158.
- Khanna, S., & Kraft, C. S. (2021). The interplay of SARS- CoV-2 and Clostridioides difficile infection. Future Microbiology, 16(6), 439-443.
- Luo, Y., Grinspan, L. T., Fu, Y., Adams-Sommer, V., Willey,D. K., Patel, G., & Grinspan, A. M. (2021). Hospital-onset Clostridioides difficile infections during the COVID-19 pandemic. Infection Control & Hospital Epidemiology, 42(9), 1165-1166.
- Chen, Y., Chen, L., Deng, Q., Zhang, G., Wu, K., Ni, L., ...& Cheng, Z. (2020). The presence of SARS-CoV-2 RNA in the feces of COVID-19 patients. Journal of medical virology, 92(7), 833-840.
- Laszkowska, M., Kim, J., Faye,A. S., Joelson,A. M., Ingram, M., Truong, H., ... & Freedberg, D. E. (2021). Prevalence of Clostridioides difficile and other gastrointestinal pathogens in patients with COVID-19. Digestive diseases and sciences, 66, 4398-4405.
- Allegretti, J. R., Nije, C., McClure, E., Redd, W. D., Wong,D., Zhou, J. C., ... & Chan, W. W. (2021). Prevalence and impact of Clostridioides difficile infection among hospitalized patients with coranavirus disease 201 9. JGH Open, 5(5), 622-625.



