Research Article - (2024) Volume 3, Issue 2
In Vitro Antimicrobial Activity Evaluation of Selected Medicinal Plants Against Multidrug-Resistant Isolates of Clinical Bovine Mastitis
Received Date: May 06, 2024 / Accepted Date: May 27, 2024 / Published Date: Jun 18, 2024
Copyright: ©©2024 Sultan Abda Neja, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Balcha, F. B., Sulayeman, M., Neja, S. A. (2024). In Vitro Antimicrobial Activity Evaluation of Selected Medicinal Plants Against Multidrug-Resistant Isolates of Clinical Bovine Mastitis. Archives Clin Med Microbiol, 3(2), 01-12.
Abstract
Mastitis is one of the challenges impairing the development of the dairy industry. Among mastitis-causing bacteria, Staphylococcus aureus (S. aureus) and Escherichia coli (E. coli) represent the main causative agents that threaten the farmers not only due to the high incidence rate and zoonotic potential but also due to reports of multi-drug resistance. The study was conducted to assess the antimicrobial activity of selected medicinal plants against multidrug-resistant bacterial isolates from clinical bovine mastitis. Milk samples from mastitis cases were aseptically collected and S. aureus and E .coli isolates were identified, the antibiotic sensitivity test pattern of the isolate were confirmed. Four medicinal plants were collected; extracted and their in vitro effectiveness on multidrug-resistant S. aureus and E. coli isolates of bovine mastitis was validated compared to the conventionally used antimicrobial drugs. Extracts from the four plants were effective against the two isolates. Extract of Crotona macrostachis at 100mg/mL minimum inhibitory concentration (MIC) showed the highest mean zone of inhibition (20.2 ± 1.48) on E. coli while Prunes Africana at 50mg/mL MIC showed 12.9 ± 0.74. The highest mean zone of inhibition 13.8±0.84 for Datura Stamonium at a MIC of 100mg/mL and lowest mean zone of inhibition 11.64 ± 0.86 for Crotona macrostachis at 100mg/mL MIC was observed on S. aureus. Moreover, compared to S. aureus, better efficacy was observed for all tested plant extracts against E. coli. This study indicated that traditional medicinal plant preparations might be considered as an alternative option for the treatment of resistant isolates of clinical bovine mastitis.
Keywords
Mastitis, Medicinal Plant, S. Aureus, E. Coli, Drug Resistance
Introduction
Mastitis is an infection of the udder that causes pathological changes in glandular tissues [1]. It is among the common and costly diseases that cause loss of milk yield, treatment cost for dairy farmers and culling of animals at unacceptable age. It is the major Cattle health problem and is considered as the most complex disease because of its multifactorial causation [2-4]. This disease is usually caused by microorganisms that enter through the teat end to the quarter and the majority of bacteria involved include Staphylococcus aureus, Streptococcus agalactiae, Corynebacterium bovis, Mycoplasma species, Streptococcus uberis, Serratia, Pseudomonas, Proteus species, environmental Streptococci, E. coli, Klebsiella species and Enterobacter Enterobacter species [5,6]. In addition to infection in animals with consequent loss of milk and milk products, many of these organisms are known to develop antimicrobial resistance potentially leading to therapeutic failure both in animals and humans [4].
S. aureus is the primary culprit among bacteria responsible for causing mastitis, posing a significant threat to farmers due to its high prevalence, potential for zoonotic transmission, and documented resistance to multiple drugs [7-10]. On the other hand, E. coli is a gram-negative bacterium that frequently causes subclinical mastitis, which can negatively impact dairy performance and pose public health risks. Infections with antibiotic-resistant bacteria have been known to be associated with frequent treatment failure and increased severity of the disease [11]. At a global level, there is an urgent need for the discovery of new drugs to counteract against the ever-increasing emergence and spread of AMR [12]. The spread of multidrug-resistant bacterial pathogens has driven the level of current investment on novel drug development, particularly in the fields of plant-derived natural-product and synthetic small molecules [12,13]. New agents displaying innovative chemistry and modes of action are desperately needed worldwide to tackle the public health menace posed by antimicrobial resistance.
Looking for affordable and alternative therapy in dairy mastitis, many Ethiopian ethnoveterinary practitioners traditionally use medicinal plants [14]. In fact, the use of medicinal plants in Ethiopia against human and animal infection goes a long back history and still 80% of people depend on traditional medicinal preparations [15]. Despite the presence of more than 6000 floral species of medicinal plants with an estimated 12% of which are endemic in Ethiopia, there is limited emphasis on the conservation and proper usage of the plants [16]. Studies in Ethiopia showed the presence of various medicinal plants that traditionally used for treatment of mastitis [17-20]. These studies recommended laboratory-based further investigation on extraction, pharmacological and biological activity tests as well as safety controls. The current increasing threat from drug-resistant bacteria also elicited the effort to search for novel solutions including natural products from medicinal plants [21,22].
In Ethiopia, there have been several studies conducted on the multidrug resistance profile of S. aureus (31.3-83%) and E. coli (26.1-72%) isolated from bovine mastitis and the recent report showed a very concerning increasing trend [23-28]. As a result, treatment failure in dairy mastitis remains to be the main challenge affecting the development of the dairy industry in Ethiopia. As part of global efforts against multi-drug resistant pathogens utilizing biochemical and molecular tools of drug discovery, investigating the antimicrobial activity of the most used locally available medicinal plants against antibiotic-resistant isolates from clinical mastitis is very important [29,30]. In this regard we used S. aureus and E. coli for the reason that these two test bacteria are the common bacterial isolate that has been reported to develop drug resistance [7-9,31,32]. The other reason is that it helps us to directly test the medicinal plant extract on gram-positive and gram-negative bacteria represented by S. aureus and E. coli respectively. Therefore, the objective of the present study was to assess the antimicrobial activity of selected medicinal plants against multidrug-resistant S. aureus and E. coli isolates of clinical bovine mastitis.
Materials and Methods
The study was conducted on purposely selected 76 lactating Cows from small-scale dairy farm at Shashemene District, Ethiopia. Following clinical examination and california mastitis test (CMT) screening test, 39 Cows were in sub-clinical mastitis while 9 Cows were in clinical mastitis. Milk samples from each quarter of 9 positive Cows of clinical bovine mastitis were aseptically collected and S. aureus and E. coli isolates were identified. Then the antibiotic sensitivity test pattern of the isolate was confirmed. Four medicinal plants were collected; extracted and their in vitro effectiveness on multidrug-resistant S. aureus and E. coli isolates of clinical bovine mastitis was validated in three experimental trials compared to the conventionally used antimicrobial drugs.
Isolation and Identification of Test Bacteria
Milk samples from 9 Cows of clinical mastitis were collected after the udder was washed with tap water and dried. Then the teat ends were swabbed with cotton soaked in 70% alcohol [33]. Approximately 5 mL of milk was collected into horizontally held sterile test tube after discarding the first 2-3 milking streams. The E. coli and S. aureus test bacteria were isolated and identified according to the standard protocols of cultural and biochemical tests [34,35].
Antimicrobial Sensitivity Test
Antimicrobial sensitivity test was conducted using the agar disc diffusion method. Briefly all S. aureus and E. coli isolates identified were inoculated into the nutrient broth and suspension was made in sterile saline. Turbidity of the bacterial suspension was adjusted by comparing with 0.05 McFarland turbidity standards and plated on Mueller- Hinton agar according to Clinical and Laboratory Standards Institute of USA and Kirby-Bauer Disk Diffusion Susceptibility Test Protocol on Mueller Hinton agar medium (HiMedia, India) [36,37]. The test isolates were confirmed to be multidrug-resistant when they are resistant to more than two antimicrobial classes according to the classification of Magiorakos et al., [38].
Selection and Collection Medicinal Plants
The plants selected for this study were those we identified through field surveys conducted in diverse regions of Ethiopia, where they are considered traditional medicinal plants [14,39,40]. Based on these surveys, parts of the plants often used by traditional healers to treat mastitis were considered. The aerial parts (leaves, stems bark) and roots of the medicinal plants (Table 1) were collected in fresh from public natural habitats in the Shashemene district and transported to the Hawassa University, Veterinary Microbiology Laboratory where the voucher specimens collected were reserved. The plant's parts were washed with tap water to remove unnecessary particles, chopped into pieces, dried under shade and grinded into powder forms using a large mortar and grinding machine.
Preparations of Extract
The material were sieved and the grounded powder was macerated in 80% methanol according to the methanol-based extraction of medicinal plants and slightly adopted by Tadeg et al., [41,42]. Briefly, 125-300 grams of each macerated herb leaves, bark or root were weighted and mixed by a flask shaker at maximum speed for 30 minutes and allowed to stand for 3 days at room temperature. Each extract was filtered by grade 3 size chromatographic filter paper. The filtrates were transferred to a round bottom flask and the solvent was evaporated by using rotary vapor while another solvent was evaporated at room temperature within 3 days to dry the extract. The residue left after filtration was macerated again in methanol solution for 24 hours, filtered and added to flask to evaporate the solvent. The procedure was repeated for the third time to have sufficient amounts of extracts. The crystals were collected and ready for use.
Determination of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
Determination of the MIC for bacterial isolates was carried out using tube dilution technique as described by Akinyemi et al. and the procedure adopted by Tadele et al., [43,44]. First, 0.8g of plant extract was mixed with 2ml Dimethyl Sulfoxide (DMSO) in the first test tube to prepare 400mg/mL solution. The remaining two-fold dilutions were made down to 12.5mg/ mL concentrations. The broth dilution method was used to determine MIC. Briefly 1mL of 24hrs culture of test organisms adjusted to 0.5 McFarland turbidity standard was incubated in serial dilution ranging from 12.5mg/mL to 400mg/mL of plant extracts at 37oc for 24 hrs. In the meantime, the vehicles used as a solvent (DMSO) to make different concentrations were used as a control. The concentration of the lowest dilution with no detectable bacterial growth was considered as MIC. MBC were determined by sub-culturing broth dilutions that inhibit growth of a bacterial organism observed by the absence of turbidity and confirmed by the absence of colony after inoculating each serial dilution above MIC on to the agar plates.
Preparation of Antimicrobial Discs from Extracts for in Vitro Experiment and Antimicrobial Sensitivity Test
Using agar diffusion methods with modification for plant extract testing according to previously adopted methods, a sterile Whatman filter paper discs of 6mm size were impregnated by the pre-determined MIC dilution of each test plant extract for 30minute [45-47]. The discs were allowed to dry at room temperature overnight. Each extract impregnated dried disc along with the selected antibiotics discs were applied at a space 15 mm away from the edge of the plate and 24 mm apart from the center where the DMSO-impregnated vehicle control disc was placed. The discs were gently pressed down to ensure complete contact with the Mueller- Hinton agar medium agar and the plates were inverted and incubated at 370C for 24hrs. The diameter of the zone of inhibition was measured in millimeters using a digital caliper and results were recorded and compared with standard values for each antibiotic disc [48]. Comparison of zone of inhibition of plant extracts at their pre-determined MIC with conventional antimicrobial discs were made against the test organisms.
Data Management and Analysis
The data obtained from laboratory results were analyzed by descriptive statistics. The MIC of the plant extracts was determined in milligram per milliliter (mg/mL). Data on the comparative analysis of zone of inhibition was measured in millimeters (mm) and presented as mean ± Standard deviation (SD) of biological replicates of the tested isolates.
Results
Antimicrobial Susceptibility of the Test Isolates
The antimicrobial sensitivity profile conducted following CLSI guidelines [48]. The test isolates were confirmed to be multidrug-resistant (Supplementary Table 1). The detail relative susceptibility pattern of each isolate against each antibiotic is indicated in Supplementary Table 2.
|
No |
Local name in Afan Oromo language |
Scientific name |
Parts of the plant used |
|
1 |
Asangera |
Datura Stamonium |
Leaf |
|
2 |
Bakenisa |
Crotona macrostachis |
Leaf |
|
3 |
Homi |
Prunes africana |
Bark |
|
4 |
Qomogno |
Brucie antidesentrica |
Root |
Table 1: List of Tested Medicinal Plants
In Vitro Antibiotic Activity Test of Herbal Preparations
Following determination of MIC each plant extract (Supplementary Fig. 1 and Table 3), the colony of both test isolates for all plant at each extract concentration were counted and MBC were determined (Table 2). Accordingly, no S. aureus colonies was observed for each Datura Stamoniums, Brucie antidesentrica, Crotona macrostachis at a concentration of 400mg/mL and 200mg/mL while colony count at 100mg/mL concentration were 300 colony-forming unit (CFU), 24CFU, 361CFU respectively. However, at the concentration of 12.5mg/ mL to 50mg/mL the colony of S. aureus that were too numerous to count (TNTC) were recorded for four tested plant extracts. But at a concentration of 50mg/mL, 500CFU and 358CFU colonies of S. aureus was counted for Prunes africana and Brucie antidesentrica plant extract respectively (Table 2).
|
Tested plants |
Bacterial Isolates |
Concentration (mg/ml) of tested plant extracts and number of colony (CFU) |
|||||
|
400 |
200 |
100 |
50 |
25 |
12.5 |
||
|
Datura stamonium |
S. aureus |
X |
X |
√ |
√ |
√ |
√ |
|
Colony count |
0 |
0 |
300 |
TNTC |
TNTC |
TNTC |
|
|
E. coli |
X |
√ |
√ |
√ |
|
√ |
|
|
Colony count |
0 |
280 |
TNTC |
TNTC |
TNTC |
TNTC |
|
|
S. aureus |
X |
X |
√ |
√ |
√ |
√ |
|
|
Crotona macrostachis |
Colony count |
0 |
0 |
361 |
TNTC |
TNTC |
TNTC |
|
E. coli |
X |
X |
√ |
√ |
√ |
√ |
|
|
Colony count |
0 |
0 |
268 |
TNTC |
TNTC |
TNTC |
|
|
S. aureus |
X |
X |
√ |
√ |
√ |
√ |
|
|
Prunes africanas |
Colony count |
0 |
3 |
5 |
500 |
TNTC |
TNTC |
|
E. coli |
X |
X |
√ |
√ |
√ |
√ |
|
|
Colony count |
0 |
2 |
260 |
TNTC |
TNTC |
TNTC |
|
|
S. aureus |
X |
X |
√ |
√ |
√ |
√ |
|
|
Brucie antidesentricas |
Colony count |
0 |
0 |
24 |
358 |
TNTC |
TNTC |
|
E. coli |
X |
X |
√ |
√ |
√ |
√ |
|
|
Colony count |
0 |
0 |
300 |
TNTC |
TNTC |
TNTC |
|
|
X, (no growth), √ (growth), Too numerous to count (TNTC) |
|||||||
Table 2: Effectiveness of Herbal Preparations for S. aureus and E. coli and Colony Count at Different Concentrations of Tested Plant Extracts
Prunes africana was also observed with zero to little colony count of S. aureus at 400mg/mL, 200mg/mL and 100mg/mL concentration. In other cases, zero to little E. coli colony was counted at a concentration of 400mg/mL and 200mg/mL for three plant extracts (Brucie antidesentrica, Crotona macrostachis and Prunes africana). Whereas 300CFU, 268CFU, and 260CFU E. coli colony was counted at a concentration of 100mg/mL for Brucie antidesentrica, Crotona macrostachis and Prunes africana respectively. But the highest concentration (400mg/ mL) with zero E. coli colony count was recorded for Datura stamoniums and 280CFU at 200mg/mL whereas TNTC were observed at 100mg/mL, 50mg/mL and 12.5mg/mL concentration (Table 2).
Determination of MIC and MBC of Medicinal Plants
The MIC values of plant extracts against tested bacteria have shown a range of 50mg/mL to 200mg/mL concentrations. Most of tested bacteria have shown MIC value of 100mg/mL, followed by 50mg/mL and 200mg/mL. The MIC value of Datura Stamoniums leaf extract ranged from 100mg/mL to 200mg/mL against S. aureus and E. coli. S. aureus have shown the lowest MIC value at 100mg/mL with 300CFU colony count while E. coli showed the highest MIC at 200mg/mL with 280CFU colony count against Datura Stamoniums leaf extract (Table 3).
Both E. coli and S. aureus have shown similar MIC of 100mg/ mL with colony count at this concentration 361 and 268CFU respectively for Crotona macrostachis leaf extract. At 50mg/mL MIC, Brucie antidesentricas root extract exhibited a colony count of 358 CFU for S. aureus, while at 100mg/mL MIC, the count was 300 CFU for E. coli. Methanolic extract of Prunes africanas stem bark has shown the MIC values as least as 50mg/mL with 500CFU of colony count for S. aureus and as high as 100mg/mL for E. coli with a colony count of 260CFU. Correspondingly, the MIC value against E. coli of Crotona macrostachis, Prunes africana, Brucie antidesentrica extract have shown the lowest MIC value of 100mg/mL while the highest MIC value of 200mg/ mL was recorded in the case of Datura stamoniums leaf extract. In contrast, the lowest MIC of 50mg/mL and the highest MIC value of 100mg/mL was recorded against S. aureus (Table 3).
|
Plants |
Bacterial species used for the test |
|||||||
|
E. coli |
S. aureus |
|||||||
|
MIC (mg/ml) |
Colony count |
MBC (mg/ml) |
Colony count |
MIC (mg/ml) |
Colony coun |
MBC (mg/ml) |
Colony count |
|
|
Datura Stamonium |
200 |
280 |
400 |
0 |
100 |
300 |
200 |
0 |
|
Crotona macrostachis |
100 |
268 |
200 |
0 |
100 |
361 |
200 |
0 |
|
Prunes africana |
100 |
260 |
100 |
2 |
50 |
500 |
100 |
5 |
|
Brucie antidesentrica |
100 |
300 |
100 |
0 |
50 |
368 |
200 |
0 |
Table 3: Determined MIC and MBC of each Plant Extract
In general, the MIC value recorded by each plant extract against E. coli was greater than that of S, aureus. Similarly, Prunes africanas plant extract showed the lowest MBC of 100mg/mL for S. aureus while all plant extract except Datura stamonium were recorded the lowest MBC against E. coli. The highest MBC value 200mg/mL has shown against S. aureus by Datura stamonium, Brucie antidesentrica and Crotona macrostachis extracts. Datura stamonium extract also showed MBC value of 400mg/mL against E. coli and lowest value of 200mg/mL against S. aureus respectively. The MBC and MIC values were found to be different both for S. aureus and E.coli methanol extract, because, according to this study those isolates with zero to the little colony to count were recorded as MBC while those tests with less than or equal to 500 CFU isolates were recorded as MIC (Table 3).
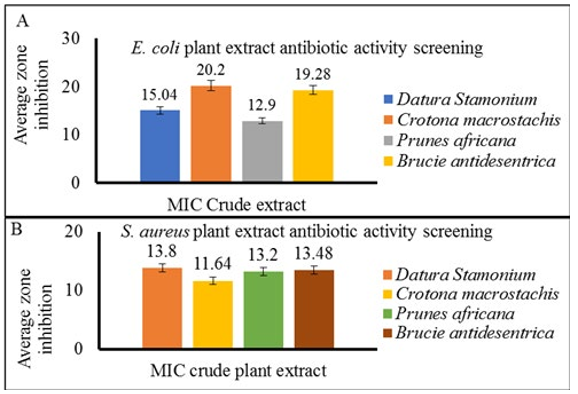
The effectiveness of each medicinal plant extract at each MIC concentration shown in Table 3 was evaluated against S. aureus and E. coli. At 100mg/mL MIC the mean zone of inhibition of Datura Stamonium on E. coli and at 200mg/mL MIC for S. aureus was 15.04mm and 13.8mm respectively. The mean zone of inhibition for Crotona macrostachis at 100mg/mL MIC for E. coli and S. aureus was 20.2mm and 11.64mm respectively (Supplementary Fig. 2A and B). Similarly, the mean zone of inhibition for Prunes africana at 50mg/mL MIC for E.coli was 12.9mm while at 100mg/mL for S. aureus was 13.2mm. Brucie antidesentrica was found to have a 19.29mm mean zone of inhibition at the lowest MIC value of 50mg/mL against S. aureus (Supplementary Fig. 2A and B). In this study, the highest mean zone of inhibition (20.2 ± 1.48) on E. coli was recorded by Crotona macrostachis at 200mg/mL MIC while the lowest mean (12.9 ± 0.74) was recorded by Prunes Africana at 100mg/mL concentration (Supplementary Fig. 2A and B). Concerning S. aureus there was a little difference between the mean zone inhibition at 50mg/mL and 100mg/mL concentration. So that the highest mean zone of inhibition 13.8 ± 0.84 for Datura Stamonium at a concentration of 100mg/mL and lowest mean zone of inhibition 11.64 ± 0.86 for Crotona macrostachis at 100mg/mL concentration was recorded (Supplementary Fig. 2A). In this study, the highest mean zone of inhibition at different MIC was also recorded for all tested plant extracts against E. coli as compared with S. aureus. During this study, the DMSO, which was the negative control did not show inhibitory activity.
Comparison of the MIC of Plant Extract with Conventional Antibiotics
Based on the comparative mean zone of inhibition, plant extracts at MHC showed better effectiveness compared to many antibiotics (Fig. 1). For instance, all four plant extracts were formed the largest average zone of inhibition against E. coli when compared to the other conventionally used antibiotics (Streptomycin, Ampicillin, Erythromycin, Amoxicillin, Penicillin and Oxytetracycline). However, chloramphenicol recorded the highest zone of inhibition, while the zone of inhibition for kanamycin and Oxytetracycline were recorded to be comparable with plant extract (Fig. 1A).
Figure 1: Antibiotics vs Plant extract MIC Comparison
Barograph of comparison of mean zone of inhibition (mm) of four plants crude extract MIC with conventional antimicrobial discs against both E. coli (Panel A) and S. aureus (Panel B) isolated from mastitic cow milk.
In the case of S. aureus, the average zones of inhibition formed by the plant extracts were higher than conventionally used antibiotics such as Ampicillin and Streptomycin while comparable antimicrobial activity with Kanamycin, penicillin, Cefoximine and Amoxicillin (Fig. 1B). On the other hand, chloramphenicol, oxytetracycline, Erythromycin, were recorded to have the higher average zone of inhibition as compared to plant extract. In general, the size of the diameter of inhibition zones exhibited by 100mg/mL concentration of Datura stamonium, 50mg/mL concentration of Brucie antidesentrica and 50mg/ mL Prunes africana plant extracts against S. aureus were found almost comparable to those of noble antibiotics discs (Fig. 1B and Fig. 2). While in the case of E. coli, 100mg/mL concentration of Brucie antidesentrica, Prunes africana, Crotona macrostachis and Datura stamonium at 200mg/mL concentrations were found almost comparable to those of antibiotics discs. The DMSO impregnated disc is used as vehicle control with no inhibition against any test organism which implies that the inhibition observed was exclusively by the extracts (Fig. 2).
Figure 2: Comparative Inhibitory Effect of Medicinal Plant
MIC of the four extracts was tasted compared to conventionally used Ampicillin, Kanamycin, Cefotaxime and Amoxicillin against clinical isolate of S. aures and E.coli.
Relative in Vitro Effectiveness of Plant Extract at MIC in Comparison with Antimicrobial Resistant Isolates
Based on the reading of zones of inhibition the standard classifications of the relative activity of antibiotics are often classified into susceptible, intermediate and resistant. The inhibition zone of < 13mm for Ampicillin and Amoxicillin, < 11mm for Oxy-tetracycline and Streptomycin against E. coli were considered as resistant. While inhibition zone of < 14mm for Oxy-tetracycline and < 28mm for Ampicillin and penicillin were taken as resistant for S. aureus [48]. Accordingly, we found E. coli were 100% resistant to Ampicillin and Amoxicillin while 85.7% and 42.9% were resistant to Oxy-tetracycline and streptomycin respectively. In the case of S. aureus 90%, 77.3% and 50% isolates were resistant to Ampicillin, penicillin G and oxytetracycline respectively (Supplementary Table 2).
The plant extract were effective compared to some conventional antimicrobial agents to which test organisms developed resistance according to clinical and laboratory standards institute (CLSI) standards [48]. For instance, the mean zone inhibition of 50mg/mL concentration Prunes Africanas, Brucie antidesentrica and 100mg/mL Datura Stamonium were higher than the mean zone inhibition of ampicillin-resistant S. aurues while 100mg/ mL Crotona macrostachis was recorded comparable mean zone inhibition as that of Ampicillin and Amoxacillin (Fig. 1B and Fig. 2). In contrast, the mean zone inhibition of 200mg/mL Datura Stamonium and 100mg/mL Brucie antidesentrica, Prunes Africana, Crotona macrostachis were higher than the mean inhibition zone of Streptomycin, Oxy-tetracycline, Ampicillin as well as Amoxicillin resistant E. coli (Fig. 1A and Fig. 2).
Discussion
In looking for affordable and alternative therapy in dairy mastitis, the present study investigated the in-vitro antimicrobial activity of four selected medicinal plants against S. aureus and E. coli clinical isolates of bovine mastitis. Our study showed that Brucea antidysenterica was effective at MIC of 100mg/mL on E. coli and 50mg/mL on S. aureus isolates and mean inhibition zone (mm) of 19.28 ± 1.302 and 13.48 ± 0.36 was also recorded at this MIC respectively. Compared to the standard cut-off value for the susceptibility range for the many antibiotics this plant can be considered as effective [48]. Although our test organisms are multidrug-resistant, the extracts have shown comparable inhibition to previous findings, which reported inhibition of S. aureus at concentration levels of 400mg/mL, 200mg/mL, 100mg/mL, 50mg/mL, and 25mg/mL. [49]. Additionally this finding was in line with the previous report of 8mm mean inhibition zone at 20% MIC against the S. aureus isolated from clinical bovine mastitis [16]. A relatively higher degree of wound healing percent of this plant extract at 50mg/mL- 100mg/ mL MIC was also reported [50].
The methanolic extracts of Crotona macrostachyus showed minimum inhibition zones at the concentration of 100mg/mL against both E. coli and S. aureus using broth dilution method and 20.2 ± 1.48 and 11.64 ± 0.86 mean zone inhibition (mm) at this concentration respectively. Compared to the standard cut-off value for the susceptibility range for the conventional antibiotics this plant can be considered effective [48]. This result agrees with the finding of Biruhalem et al., who reported the MIC of 12.5% against S. aureus and E. coli isolated from wound specimens at Aklilu Lemma Institute of Pathobiology, Ethiopia [51]. Sendeku et al., also reported efficacy of this plant extract with 1.5% MIC for both isolates and 8.33 ± 1.53 and 24.00 ± 1.73 mean zone of inhibition (mm) on both S. aureus and E. col isolated from patient specimens visiting Hospital respectively [52].
The methanolic extract of Datura Stamonium had recorded a higher MIC against E. coli. Accordingly, MIC up to 200mg/mL and 100mg/mL were recorded on E. coli and S. aureus isolates respectively while 15.04 ± 0.58 and 13.8 ± 0.84 mean zone inhibition (mm) at this MIC was also recorded using Muller Hinton agar diffusion method. The current study agrees with 20mg/mL MIC of methanol extract Datura Stamonium on E. coli procured from the microbial type culture collection [53]. However, a study conducted on S. aureus and E. coli clinical isolates of hospital patients at MIC of inhibition (mm) of 12.5mg/ mL and 6. 25mg/mL concentrations was 14.6 ± 2.3 and 9.2 ± 1.6 respectively [54]. Alemu et al., also reported at MIC 12.5mg/ mL the mean inhibition zone (mm) 13 and 15 on clinical isolates of pathogenic bacteria of S. aureus and E. coli, obtained from Hospital [55]. The current finding is nearly in agreement with this finding concerning the concentration levels and the mean zone inhibition at a concentration of MIC against S. aureus.
The extract of Prunes africanas was also tested for its effect as an antimicrobial against E. coli and S. aureus isolates of mastitic cow milks. During this study 50mg/mL and 100mg/mL MIC of Prunes africanas against S. aurues and E. coli respectively. Likewise, 12.9 ± 0.7416198 and 13.2 ±1.643168 visible mean zone of inhibition (mm) at this MIC was recorded on E. coli and S. aureus respectively. The variation of efficacy among the above-mentioned phytopreparations could be attributed to the way of plant preparation, the season of collection, stage of the plant, place of collection, way of extract drying, means of extraction, the solvent used, preservation or storage of the extract till evaporation and other unnoticed factors.
The result from comparison of MIC of plant extract with conventional antimicrobial discs showed that the efficacy of all tested plant extract against S. aureus at their respective MIC were comparable except with Chloramphenicol. The least mean inhibitory zone recorded by Prunes Africana against E. coli might be due to the capacity of this plant extract penetrating through the media [56]. The mean inhibition zone obtained by Oxytetracycline was almost comparable to that obtained by Prunes Africana and Datura Stamonium against E. coli while Crotona macrostachis and Brucea antidysenterica was greater than the drugs. The mean inhibition zone of Erythromycin, Ampicillin, Amoxicillin was less than that of all tested plant extracts, however, chloramphenicol and cefotaxime were higher than that of all tested plant extract against E. coli. Similarly, the mean inhibition zone obtained by Kanamycin was almost comparable to that obtained by Datura Stamonium while less than that of Crotona macrostachis and Brucea antidysenterica and a little higher than that of Prunes Africana against E. coli as reported by previous study [57].
In case of S. aurues the mean zone inhibition of Datura Stamonium, Brucea antidysenterica and Prunes Africana was comparable with that of Amoxicillin, Streptomycin and Cetaximine. Almost nearly equivalent mean zone of inhibition were noted by Crotona macrostachis, to that of Ampicillin resistant isolates of S. aureus, however a bit lower than the mean inhibition zone of Brucea antidysenterica, Datura Stamonium and Prunes Africana. The mean inhibition zone obtained by Penicillin G against resistant isolates of S. aureus was also higher than that obtained by four tested plant extract. Although there is no established standard formula to judge the level of the zone of inhibition to say resistant, intermediate, and susceptible for plant extract, the mean inhibition zone of Penicillin G against resistant isolates of S. aureus in this study was below the established standard of resistance formulated by CLSI [48].
Interestingly, all tested plant extracts showed a greater inhibitory zone than Ampicillin, Amoxaccillin and Streptomycin resistant isolates of E. coli. The mean inhibition zone obtained by Oxytetracycline against both E. coli and S. aureus was smaller than that obtained by Brucea antidysenterica, Datura Stamonium and Prunes Africana and still it was less than the result obtained by Crotona macrostachis. The comparison among these test materials suggests that the herbal preparations do have a capacity to inhibit the growth of test organisms and thus can be used to treat an emerging antibiotic-resistant pathogens that has implications for not only veterinary patients, but also humans [58].
The overall finding showed that medicinal plant extract has the highest inhibition zone against E. coli organism at the highest MIC (100-200mg/mL) as compared to S. aureus in which the lowest inhibition zone was seen at the lowest MIC (50-100mg/ mL). The observed difference in antibacterial activities of extracts between E. coli and S. aureus clinical isolate may be attributed to the difference in the outer membrane of both isolates. Gram-negative (E. coli) bacteria possess a high permeability barrier for numerous antibiotic molecules similarly for these extracts. Their periplasmic space also contains enzymes, which are capable of breaking down foreign molecules and appears to be less susceptible to plant extracts than gram-positive bacteria [59]. Despite the limitation of infrastructures and facilities to conduct HPLC-based purify the active ingredient from the plant and genetic based identification of drug resistant gene, this study delineated the possibility that phytopreparations could play an important role in the treatment of resistant isolates of mastitis causal agents and it could be the alternative solution to prevent the problem of ever-emerging resistant isolates in any disease situation.
Conclusion
The study validated the effectiveness and future use of the medicinal plants for treatment of antibiotic-resistant strains of clinical bovine mastitis. The four medicinal plant extract showed promising in vitro efficacy at its MIC compared to conventional antimicrobial. The study implicates that bioactive ingredient in those plants can be used as alternative option to treat diseases caused by the two tested bacteria in animals and humans. Hence further studies on the purification of active ingredients, in vivo validation and development into new medicaments is needed [60].
Declarations
Conflict of Interest
The authors declare that they have no competing interests.
Acknowledgments
The authors extend gratitude to Hawassa University Faculty of Veterinary Medicine for the provision of logistics and laboratory reagent needed for this research. The authors thank Mizan Regional Veterinary Laboratory for sharing some drug-sensitivity discs used in this study. The authors were also thankful to the dairy farmers in Shashemene town and other study participants. A preprint has previously been published.
Funding
This work was financially supported by the Faculty of Veterinary Medicine of Hawassa University and the West Guji zone Kercha woreda livestock and fishery production office, Ethiopia to allow the authors to carry out the study. The funding bodies played no role in the design of the study and collection, analysis, interpretation of data, and in writing the manuscript.
Availability of data and Materials
The data supporting the findings are presented in the manuscript. The corresponding author can also be reached for any data inquiry.
Consent for Publication
Not applicable.
Ethics Approval and Consent
The study was approved by the research review committee of the faculty of veterinary medicine, Hawassa University. Informed consent was obtained for milk sample collection to keep the confidentiality of dairy farms at the time of sample collection. The four-plant specimens were collected from public natural habitats in the Shashemene district after permission was obtained from the local governing body according to the legislation and regulation (recently as Regulation no. 1112/2019). All methods were carried out in accordance with relevant guidelines and regulations.
SAN: Conceptualizing the study, data analysis, writing, and edit the manuscript. FBB: Data curation and laboratory analysis. MS: Supervision and reviewing the manuscript.
References
- Radostits, O. M., Gay, C. C., Hinchcliff, K. W., & Constable,P. D. (2007). Veterinary Medicine (10th edn.): A Text Book of Diseases of Cow, Buffalo, Horse, Sheep, Goat and Pig.
- Neja, S. A., & Gari, Y. (2020). Study on the Major Cattle Health and Production Constraints in and Around Haramaya Town, Ethiopia. Journal of Biology, Agriculture and Healthcare, 10, 1-9.
- Biffa, D., Debela, E., & Beyene, F. (2005). Prevalence and risk factors of mastitis in lactating dairy cows in Southern Ethiopia. International Journal of Applied Research in Veterinary Medicine, 3(3), 189-198.
- Moges, N., Asfaw, Y., Belihu, K., & Tadesse, A. (2011). Aantimicrobial susceptibility of mastitis pathogens from smallholder dairy herds in and around Gondar, Ethiopia. Journal of Animal and Veterinary Advances, 10(12), 1616-1622.
- Erskine R. (2001). Mastitis Control in Dairy Herds. Herd Health: Food Animal Production Medicine, third edition, edited by OM Radstits. WB Saunders, Philadelphia, Pennsylvania.
- Quinn, P. J. (1994). Clinical veterinary microbiology.
- Deyno, S., Fekadu, S., & Astatkie, A. (2017). Resistance of Staphylococcus aureus to antimicrobial agents in Ethiopia: a meta-analysis. Antimicrobial Resistance & Infection Control, 6, 1-15.
- Kalayu, A. A., Woldetsadik, D. A., Woldeamanuel, Y., Wang, S. H., Gebreyes, W. A., & Teferi, T. (2020). Burden and antimicrobial resistance of S. aureus in dairy farms in Mekelle, Northern Ethiopia. BMC veterinary research, 16, 1-8.
- Tibebu, L., Belete, Y., Tigabu, E., & Tsegaye, W. (2021). Prevalence of Staphylococcus aureus, methicillin-resistant Staphylococcus aureus and potential risk factors in selected dairy farms at the interface of animal and human in Bishoftu, Ethiopia. Veterinary Medicine: Research and Reports, 241-251.
- Balcha, F. B. SulayemanM and Neja SA*.(2022). Prevalence and Antimicrobial Susceptibility of Staphylococcus Aureus And Escherchia Coli Isolates of Bovine Mastitis And Associated Risk Actors in Shashemene Town, Ethiopia. J Vet Heal Sci, 3 (4), 361, 372.
- Finch, R., & Hunter, P. A. (2006). Antibiotic resistance— action to promote new technologies: report of an EU Intergovernmental Conference held in Birmingham, UK, 12–13 December 2005. Journal of Antimicrobial Chemotherapy, 58(suppl_1), i3-i22.
- Miethke, M., Pieroni, M., Weber, T., Brönstrup, M., Hammann, P., Halby, L., ... & Müller, R. (2021). Towards the sustainable discovery and development of new antibiotics. Nature Reviews Chemistry, 5(10), 726-749.
- Atanasov, A. G., Zotchev, S. B., Dirsch, V. M., & Supuran,C. T. (2021). Natural products in drug discovery: advances and opportunities. Nature reviews Drug discovery, 20(3),200-216.
- Tadesse, B., Mulugeta, G., Fikadu, G., Sultan, A., & Nekemte, E. (2014). Survey on ethno-veterinary medicinal plants in selected Woredas of east Wollega zone, western Ethiopia. Journal of Biology, Agriculture and Healthcare, 4(17), 97-105.
- Fassil, H. (2005). Beyond Plants Professionals & Parchments: The role of home-based medicinal plant use and traditional health knowledge in primary health care in Ethiopia. Ethnobotany Research and Applications, 3, 037-050.
- Mengistu, A. K. (2004). The Effect of Herbal Preparations on Staphylococcus aureus and Streptococcus agalactiae isolated from clinical bovine mastitis (Unpublished MSC thesis Faculty of veterinary medicine, AAU).
- Haile, A. A. (2021). Important medicinal plants in Ethiopia: a review in years 2015–2020. Herbs and spices-new processing technologies.
- Kalayou, S., Haileselassie, M., Gebre-Egziabher, G., Tiku'e, T., Sahle, S., Taddele, H., & Ghezu, M. (2012). In–vitro antimicrobial activity screening of some ethnoveterinary medicinal plants traditionally used against mastitis, wound and gastrointestinal tract complication in Tigray Region, Ethiopia. Asian Pacific Journal of Tropical Biomedicine, 2(7), 516-522.
- Faye, G., Birhanu, T., & Belete, T. (2021). Survey and antimicrobial activity study of ethnomedicinal plants in selected districts of North Shewa Zone, Oromia, Ethiopia. Infection and Drug Resistance, 5511-5520.
- Neja, S. A., & Bogale, E. (2022). Ethnoveterinary Survey on Medicinal Plants in Aleta-Chuko District of Sidama Reginal State, Ethiopia. J Vet Heal Sci, 3 (2), 125, 136.
- Theuretzbacher, U. (2012). Accelerating resistance, inadequate antibacterial drug pipelines and international responses. International journal of antimicrobial agents, 39(4), 295-299.
- Verpoorte, R., Choi, Y. H., & Kim, H. K. (2005). Ethnopharmacology and systems biology: a perfect holistic match. Journal of ethnopharmacology, 100(1-2), 53-56.
- Asmelash, T., Mesfin, N., Addisu, D., Aklilu, F., Biruk, T., & Tesfaye, S. (2016). Isolation, identification and drug resistance patterns of methicillin resistant Staphylococcus aureus from mastitic cows milk from selected dairy farms in and around Kombolcha, Ethiopia. Journal of Veterinary Medicine and Animal Health, 8(1), 1-10.
- Bekele, T., Zewde, G., Tefera, G., Feleke, A., & Zerom, K. (2014). Escherichia coli O157: H7 in raw meat in Addis Ababa, Ethiopia: prevalence at an abattoir and retailers and antimicrobial susceptibility. International Journal of Food Contamination, 1, 1-8.
- Shecho, M., Thomas, N., Kemal, J., & Muktar, Y. (2017). Cloacael carriage and multidrug resistance Escherichia coli O157: H7 from poultry farms, eastern Ethiopia. Journal of veterinary medicine, 2017.
- Elemo, K. K., Sisay, T., Shiferaw, A., & Fato, M. A. (2017). Prevalence, risk factors and multidrug resistance profile of Staphylococcus aureus isolated from bovine mastitis in selected dairy farms in and around Asella town, Arsi Zone,South Eastern Ethiopia. African Journal of MicrobiologyResearch, 11(45), 1632-1642.
- Ababu, A., Endashaw, D., & Fesseha, H. (2020). Isolation and antimicrobial susceptibility profile of Escherichia coli O157: H7 from raw milk of dairy cattle in Holeta district, Central Ethiopia. International Journal of Microbiology, 2020, 1-8.
- Gemeda, B. A., Assefa, A., Jaleta, M. B., Amenu, K., & Wieland, B. (2021). Antimicrobial resistance in Ethiopia: A systematic review and meta-analysis of prevalence in foods, food handlers, animals, and the environment. One Health, 13, 100286.
- Vivas, R., Barbosa, A. A. T., Dolabela, S. S., & Jain, S. (2019). Multidrug-resistant bacteria and alternative methods to control them: an overview. Microbial Drug Resistance, 25(6), 890-908.
- Balcha, F. B., & Neja, S. A. (2023). CRISPR-Cas9 mediated phage therapy as an alternative to antibiotics. Animal Diseases, 3(1), 4.
- Messele, Y. E., Abdi, R. D., Tegegne, D. T., Bora, S. K.,Babura, M. D., Emeru, B. A., & Werid, G. M. (2019). Analysis of milk-derived isolates of E. coli indicating drug resistance in central Ethiopia. Tropical animal health and production, 51, 661-667.
- Balemi, A., Gumi, B., Amenu, K., Girma, S., Gebru, M. U., Tekle, M., ... & Kerro Dego, O. (2021). Prevalence of mastitis and antibiotic resistance of bacterial isolates from CMT positive milk samples obtained from dairy cows, camels, and goats in two pastoral districts in Southern Ethiopia. Animals, 11(6), 1530.
- Committee NMCR. (1990). Microbiological procedures for the diagnosis of bovine udder infection. National mastitis Council Incorporated.
- International Organization for Standardization. (1999). Water Quality-Enumeration of Culturable Micro-organisms-Colony Count by Inoculation in a Nutrient Agar Culture Medium. International Organization for Standardization.
- Quinn, P. J., Markey, B. K., Carter, M. E., Donnelly, W. J. C., & Leonard, F. C. (2002). Veterinary microbiology and microbial disease. Blackwell science.
- Humphries, R. M., Ambler, J., Mitchell, S. L., Castanheira,M., Dingle, T., Hindler, J. A., ... & Sei, K. (2018). CLSImethods development and standardization working group best practices for evaluation of antimicrobial susceptibility tests. Journal of clinical microbiology, 56(4), 10-1128.
- Hudzicki, J. (2009). Kirby-Bauer disk diffusion susceptibility test protocol. American society for microbiology, 15(1), 1-23.
- Magiorakos, A. P., Srinivasan, A., Carey, R. B., Carmeli, Y., Falagas, M. E., Giske, C. G., ... & Monnet, D. L. (2012). Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clinical microbiology and infection, 18(3), 268-281.
- Kebede, A., Ayalew, S., Mesfin, A., & Mulualem, G. (2017). An ethnoveterinary study of medicinal plants used for the management of livestock ailments in selected kebeles of Dire Dawa Administration, Eastern Ethiopia. Journal of Plant Sciences, 5(1), 34-42.
- Wubetu, M., Abula, T., & Dejenu, G. (2017). Ethnopharmacologic survey of medicinal plants used to treat human diseases by traditional medical practitioners in Dega Damot district, Amhara, Northwestern Ethiopia. BMC research notes, 10, 1-13.
- Pandey,A., & Tripathi, S. (2014). Concept of standardization, extraction and pre phytochemical screening strategies for herbal drug. Journal of Pharmacognosy and phytochemistry, 2(5), 115-119.
- Tadeg, H., Mohammed, E., Asres, K., & Gebre-Mariam, T. (2005). Antimicrobial activities of some selected traditional Ethiopian medicinal plants used in the treatment of skin disorders. Journal of ethnopharmacology, 100(1-2), 168-175.
- Akinyemi, K. O., Oladapo, O., Okwara, C. E., Ibe, C. C., & Fasure, K. A. (2005). Screening of crude extracts of six medicinal plants used in South-West Nigerian unorthodox medicine for anti-methicillin resistant Staphylococcus aureus activity. BMC complementary and alternative medicine, 5, 1-7.
- Tadele Tolosa, T. T., Henok Wagaye, H. W., & Fekadu Regassa, F. R. (2010). A study on in-vitro antimicrobial effects of some selected plants on Staphylococcus aureus isolated from bovine clinical mastitis.
- Clinical and Laboratory Standards Institute. (2015). Performance Standards for Antimicrobial Disk Susceptibility Tests for Bacteria Isolated from Animals: CLSI Supplement VET01S; Replaces VET01-S2. Clinical and Laboratory Standards Institute.
- Jain, P., Nimbrana, S., & Kalia, G. (2010). Antimicrobial activity and phytochemical analysis of Eucalyptus tereticornis bark and leaf methanolic extracts. Int J Pharm Sci Rev Res, 4(2), 126-128.
- Hammud, K. K., Neema, R. R., Ali, S. G., & Hamza, I. S. (2015). Direct Solid Disc as a Novel antibacterial testing method. Int. J. Adv. Pharm. Biol. Chem, 4, 844-851.
- CLSI. (2018). Performance Standards for Antimicrobial Susceptibility Testing: Clinical and Laboratory Standards Institute (CLSI). CLSI Suppliment M100. Wayne PA. 28th ed, 38:296.
- Belay, T. (2003). Screening of 14 traditional medicinal plants for antimicrobial properties. DVM thesis. Addis Ababa University Ethiopia. (37).
- Mekonnen, H., Workineh, S., Bayleyegn, M., Moges, A., & Tadele, K. (2005). Antimicrobial susceptibility profiles ofmastitis isolates from cows in three major Ethiopian dairies.
- Taye, B., Giday, M., Animut, A., & Seid, J. (2011). Antibacterial activities of selected medicinal plants in traditional treatment of human wounds in Ethiopia. Asian Pacific Journal of Tropical Biomedicine, 1(5), 370-375.
- Sendeku, W., Alefew, B., Mengiste, D., Seifu, K., Girma, S., Wondimu, E., ... & Berhane, N. (2015). Antibacterial activity of Croton macrostachyus against some selected pathogenic bacteria. Biotechnoloy International, 8(1), 11-20.
- Sharma, R. A., Sharma, P. A. L. L. A. V. I., & Yadav, A.N. K. I. T. A. (2013). Antimicrobial screening of sequential extracts of Datura stramonium L. International Journal of Pharmacy and Pharmaceutical Sciences, 5(2), 401-4.
- Solomon Baynesagne, S. B., Nega Berhane, N. B., Wagaw Sendeku, W. S., & Ai LianZhong, A. L. (2017). Antibacterial activity of Datura stramonium against standard and clinical isolate pathogenic microorganisms.
- Alemu, T., Kinfe, G., Mequanent, D. Antibacterial Activities Of The Leaf Extract Of Datura Stramonium Linn. International Journal Medicine Microbiol 202, 8( 11):1-8.
- Olajuyigbe, O. O., & Afolayan, A. J. (2011). In vitro antibacterial activities of the methanol extract of Ziziphus mucronata Willd. subsp. mucronata Willd. J. Med. Plants Res, 5(37913795), 18.
- Deresa, D. A., Abdissa, Z., Gurmessa, G. T., & Abdissa, N. (2022). Chemical constituents of the stem bark of Prunus africana and Evaluation of their Antibacterial Activity. Journal of the Turkish Chemical Society Section A: Chemistry, 9(2), 395-414.
- Hoffman, S. J., Caleo, G. M., Daulaire, N., Elbe, S., Matsoso, P., Mossialos, E., ... & Røttingen, J. A. (2015). Strategies for achieving global collective action on antimicrobial resistance. Bulletin of the World Health Organization, 93, 867-876.
- Duffy, C. F., & Power, R. F. (2001). Antioxidant and antimicrobial properties of some Chinese plant extracts. International journal of antimicrobial agents, 17(6), 527-530.
- Balcha, F. B., Sulayeman, M., & Neja, S. A. (2023). In vitro antimicrobial activity evaluation of selected medicinal plants against multidrug-resistant isolate of Clinical Bovine Mastitis.
Supplementary Data
Supplementary Table 1: Multidrug Resistance Patterns of E. coli and S. Aureus Isolates
|
|
Number of drugs |
Antimicrobial’s name |
Number resistant isolates |
|
E. coli |
Six drugs |
AMP, AMC, OT, P, E S, |
3 |
|
AMP, AMC, CTX, P, E, S, |
2 |
||
|
AMP, K, AMC, CTX, P, E |
1 |
||
|
Seven drugs |
AMP, K, AMC, CTX, P, E S, |
1 |
|
|
Total |
|
7 |
|
|
S. aureus |
Two drugs |
E, P |
1 |
|
AMP, P |
1 |
||
|
AMP, OT |
1 |
||
|
Three drugs |
E, P, OT |
1 |
|
|
AMP, CTX, S |
1 |
||
|
AMP, P, AMC |
1 |
||
|
AMP, P, CTX |
3 |
||
|
Four drugs |
AMP, E, P, S |
1 |
|
|
AMP, P, OT, S |
3 |
||
|
AMP, E, S, AMC |
1 |
||
|
Five drugs |
AMP, E, K, P, S |
1 |
|
|
AMP, E, P, OT, S |
1 |
||
|
AMP, CTX, E, P, S |
1 |
||
|
AMP, E, P, OT, AMC |
2 |
||
|
Six drugs |
AMP, P, OT, CTX, S, AMC |
2 |
|
|
Seven drugs |
AMP, CTX, E, OT, P, S, AMC |
1 |
|
|
Total |
|
22 |
|
|
Ampicillin (AMP), Amoxicillin (AMC), Cefotaxime (CTX), Erythromycin (E), Oxytetracycline (OT), Penicillin(P), Kanamycin(K) and Streptomycin(S) |
|||
Supplementary Table 2: Antimicrobial Susceptibility Pattern of E. coli and S. Aureus Isolated from Dairy Mastitis.
|
|
Antimicrobial Agents |
Susceptible â?? (%) |
Intermediate â?? (%) |
Resistant â?? (%) |
|
E. coli |
Streptomycin (S10 |
- |
1(14.3) |
6(85.7) |
|
Erythromycin (E15 |
- |
- |
7(100) |
|
|
Ampicillin (AMP10 |
- |
- |
7(100) |
|
|
Kanamycin (K30 |
- |
5(71.4) |
2(28.6) |
|
|
Chloramphenicol (C30 |
7(100) |
- |
- |
|
|
Amoxicillin (AMC30 |
- |
- |
7(100) |
|
|
Penicillin-G (P10 |
- |
- |
7(100) |
|
|
Oxytetracycline (OT30 |
4(57.1) |
- |
3(42.9) |
|
|
Cefotaxime (CTX30 |
1(14.3) |
2(28.6) |
4(57.1) |
|
|
S. aureus |
Streptomycin (S10 |
7(31.8) |
4(18.2) |
11(50) |
|
Erythromycin (E15 |
8(36.4) |
2(9.1) |
12(54.5) |
|
|
Ampicillin (AMP10 |
2(9.1) |
- |
20(90) |
|
|
Kanamycin (K30 |
14(63.6) |
7(31.8) |
1(4.6) |
|
|
Chloramphenicol(C30 |
22(100) |
- |
- |
|
|
Amoxicillin (AMC30 |
13(59.1) |
2(9.1) |
7(31.8) |
|
|
Penicillin G (P10 |
5(22.7) |
- |
17(77.3) |
|
|
Ox tetracycline (OT30 |
11(50) |
- |
11(50) |
|
|
|
Cefotaxime (CTX30 |
6(27.3) |
7(31.8) |
9(40.9) |
Supplementary Figure. 1: Serial Dilution of Plant Extract. Plant Extracts Were Serially Diluted for Estimation of MIC to S. Aureus (Upper Panel) and to E. Coli Test Isolates (Lower Panel). An Equal Volume of DMSO was Used as Vehicle Control
Supplementary Figure. 2: Average Zone Inhibition of Plant Extract. The Zone of Inhibition of Plant Extract was Determined at MIC of Crude Extract Against and E. coli (A) and S. aureus (B). Average one of Inhibition was Measured in Millimeters (mm) and Presented as Mean ± Standard Deviation (SD) of Biological Replicates of the Tested Isolates



