Research Article - (2018) Volume 3, Issue 1
Improvement of Bacterial Vaginosis and Vulvovaginal Candidiasis with a New Lactobacillus Strains Association: A Pilot Study
2Lower Genital Tract Disease Unit, V.Buzzi Hospital, University of Milan, Italy
Received Date: Jan 23, 2018 / Accepted Date: Jan 29, 2018 / Published Date: Feb 07, 2018
Copyright: ©Copyright: Ã?©2018 Filippo Murina. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Bacterial vaginosis and vulvo-vaginal candidacies are bacterial infections widely spread among women in childbearing age. These infections are associated with altered or disturbed vaginal microbiota, in a condition defined as vaginaldysbiosis. Restoration of the lactobacilli flora seems a promising strategy for the treatment of these pathologies.
The efficacy and safety of a new vaginal composition, containing three Lactobacillus strains selected for their activity against urogenital pathogens, was assessed in a pilot clinical trial for the treatment of bacterial vaginosis and vaginal candidacies. The study involved women with diagnosis of bacterial vaginosis (30) or vulvovaginal candidacies (24). Vaginal capsules, containing the three Lactobacillus strains, were administered following one application per die for ten consecutive days. Patients received gynecological examinations before and after treatment, with objective examination, assessment of clinical symptoms severity and vaginal wet mount microscopy.
The treatment showed a complete resolution in 60% of bacterial vaginosis cases and around 40% in vulvo-vaginal candidacies cases. Further analyzing the subgroup characterized by a recurrent vaginal candidacy, the success rate was significantly higher than in the acute cases, showing a stronger effect of the product against persistent and chronic condition. The product showed a resolution of the vaginal dysbiosis through vaginal wet mount examination and no adverse effect were reported.
In conclusion, the presented clinical data support the use of the Lactobacillus vaginal composition for the treatment of bacterial vaginosis and vulvo-vaginal candidacies.
Keywords
Bacterial vaginosis; vaginal candidacies; Lactobacillus; Dysbiosis
List Of Abbreviations
BV: bacterial vaginosis;
VVC: vulvovaginal candidacies;
RVVC: recurrent vulvovaginal candidacies.
Introduction
Vulvovaginal candidacies (VVC) is one of the most common women disease, an estimated 75% of women of all ages will have experienced at least one episode of VVC (particularly during childbearing age), and 40%–50% will have two or more episodes [1].
Bacterial vaginosis (BV) is the most common cause of vaginal symptoms among women ages 15-44 and its prevalence in the United States is estimated to be 21.2 million (29.2%) (2). Globally, more than 20% of the female population is affected by BV, with a lower rate in European countries, around 12% [3, 4].
BV is a condition that happens when an abnormal growth of anaerobic species (Mobiluncus, Gardnerella vaginalis, Prevotella, Mycoplasmahominis) in the vagina displaces the indigenous Lactobacillus flora [2].
This change the normal balance of bacteria in the vagina defined asdysbacteriosisor dysbiosis, causing vaginal pH alkalization coupled, in symptomatic cases, with malodorous discharge. Most women found to have BV reported no symptoms. BV is associated to preterm delivery, low birth weight and to a greater risk of developing pelvic inflammatory disease [5].
Recurrence rates in VVC and BV are one of the most critical aspects of these pathologies.
Around 15-20% of acute candidacies evolve into a recurrent form (RVVC), characterized by four or more yearly episodes [1]. Instead, more than 60% of women who experienced BV fall into recurrent or chronic disease [2]. Major responsible for recurrences is the formation of a pathogenic biofilm over the vaginal epithelium. Biofilms are microbial community systems that work as bacterial survival strategies in unfavorable environment; they are formed by an assemblage of surface-associated microbial cells that are enclosed in an extracellular polymeric substance matrix, self-produced by the bacterial community [6]. Biofilm acts as microbial reservoir and can provide a defensive barrier against anti-infective agents [7].
During childbearing age, in physiologic condition, more than 90% of vaginal microbiota comprises species belonging to Lactobacillus genus. When pathogens spread and overcome resident Lactobacilli, they may constitute a persistent biofilm [7]. Key factor in recurrent vaginal infections is the promotion of a Lactobacillus predominant microbiota, avoiding the formation of a persistent pathogenic biofilm. For this reason, seems fundamental reconsider the treatment for recurrent infections, aiming to recreate and maintain the healthful vaginal microbiota. The ability to create a favorable biofilm may be seen as a useful probiotic property, in promoting a durable colonization of vaginal mucosa. Aim of the study is evaluating the efficacy and tolerability of a medical device in vaginal capsules containing new strains of lactobacillus containing for vulvo-vaginal candidiasis and bacterial vaginosis treatment.
Methods
This observational and open-label study enrolled women of reproductive age and who presented with symptoms suggestive of BV and VVC. Pregnant women or women who intended to become pregnant during the study, or menopausal women were not allowed to participate. Patients with known allergy or hypersensitivity to product components were excluded. The use of antibiotic or antifungal agents or other products containing lactobacilli in the previous 4 weeks and women with any sexually transmitted disease or genital skin disorders were also exclusion criteria. The study was reviewed and approved by the local ethical committee and women were enrolled into the study after they provided written informed consent.
VVC was diagnosed by Sobel Score ≥ 2, a tool for measuring the severity of vulvo vaginal symptoms such as pruritus, burning, erythema and oedema with semi-quantitative scores of 0 (absent), 1 (mild), 2 (moderate) or 3 (severe) and by blastospores or hyphae on microscopic examination of a fresh sample [8].
BV was diagnosed if at least two of the following Damsels’ diagnostic criteria were present: whitish or greyish green homogeneous discharge on the vaginal walls, vaginal pH > 4.5, positive whiff test, and clue cells on fresh wet mount microscopy of vaginal fluid.
Verified the eligibility criteria and after exhaustive explanation of study protocol and outcomes, enrolled patients provide general and obstetric-gynecologic anamnesis data collection and undergo to an objective vulvo-vaginal examination confirming diagnosis of BV or VVC as above illustrated.
Subsequently, the patients received a new product formulated in vaginal capsules containing a total of 1,5*109 cfu/capsule of the following strains: Lactobacillus acidophilus PBS066 (or LA001) DSM 24936 +Lactobacillus plantarum PBS067 (or LP080) DSM24937 + Lactobacillus rhamnosus LRH020 DSM 25568; with the posology of one application per die for ten consecutive days. Patients were evaluated after two weeks from the end of the therapy, evaluating treatment compliance, clinical symptoms severity, possible adverse effects, objective vulvo-vaginal examination and vaginal wet mount microscopy. Primary endpoints comprise clinical symptoms severity improvement (burning sensation, itching, and dyspareunia) and objective evaluation (vaginal edema or erythema, pH and wet mount).Symptoms severity was ranked by a semi quantitatively scoring system, ranging from 0 (absent) to 4 (very intense). Similarly, vaginal discharge was recorded and classified as follow: 0 absence; 1 poor, liquid and transparent secretion; 2 evident, almost thick and whitish secretions; 3 abundant, very thick and white secretions.Vaginal wet mount microscopy outcomes were clustered into three distinct groups based on microbiota composition:
• Normal microbiota: strong and predominant presence of Lactobacillus species combined with the absence of anaerobic microorganisms, Gram-negative bacteria, fungal hyphae, spores or G.vaginalis.
• Partially disturbed microbiota: absence or reduced presence of Lactobacillus species with co-presence of anaerobic bacteria, gram-negative bacteria, hyphae, spores or G. vaginalis.
• Disrupted microbiota or dysbiosis: absence of Lactobacillus species in favor of strong presence of anaerobes, gram-negative bacteria, hyphae, spores and/or G. vaginalis.
An overall assessment of the treatment was considered as secondary endpoint, classifying patients such as: responders with negative wet mount examination or no vaginal microbiota alteration and without clinical features resolution; partial responders with positive wet mount examination (presence of bacteria or clue cells or hyphae) and complete or strongly symptoms remission and non-responders with unvaried or worsen clinical and microbiological features.
Results
54 patients were enrolled in the study, 30 of them were diagnosed with bacterial vaginosis and 24 with vulvo-vaginal candidiasis. A total of 53 subjects concluded the study; only one patient reported transitory burning at the end of the study. One drop-out occurred, motivated by low compliance of the therapy (poor dissolution of the vaginal capsule). Table 1 and Figure 1 summarize the results in terms of signs and symptoms before and after treatment. There was a statistically significant improvement in pruritus, burning, vulvovaginal oedema and erythema, dyspareunia and vaginal secretions in all diagnostic groups.
Table 1: symptoms and signs
|
Pathology |
Pruritus |
Burning |
Dyspareunia |
Erythema/Edemaand secretions |
||||
|
|
Basal |
Post-TP |
Basal |
Post-TP |
Basal |
Post-TP |
Basal |
Post-TP |
|
BV (n=30) |
1,9 |
0,9 |
2,6 |
1,8 |
3,1 |
1,1 |
3,6 |
1,3 |
|
VVC (n=23) |
3,4 |
2,1 |
3,1 |
2,2 |
2,9 |
0,9 |
3,4 |
2,3 |
Average score of symptoms and signs severity recorded prior (basal) and post therapy (post-TP)
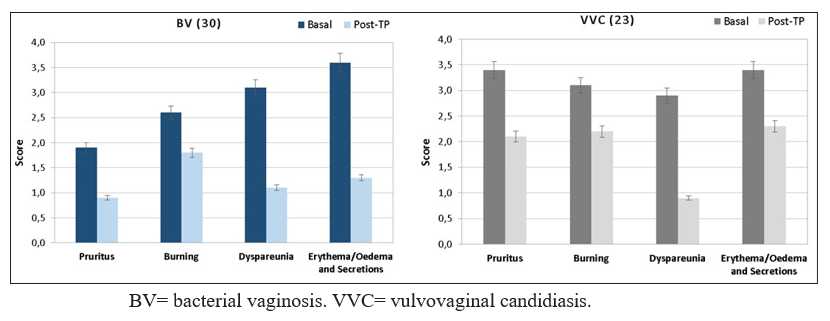
The parameters were assessed using a semi-quantitative scale from 0 to 3. Data are expressed as mean.
The final overall assessment of the treatment indicated resolution of the clinical features (responders and partially responders) in about 60% of cases with BV, about 40 % with VVC and about 83% with RVVC (Tab.2).At the end of the study, the microbiological evaluation improved in both the groups studied, re-establishing a healthy Lactobacilli-dominated vaginal microbiota (Fig.2).
|
Pathology |
Responders |
Partial Responders |
Non Responders |
|
Bacterial Vaginosis (n=32) |
16,8% |
43,5% |
39,7% |
|
Vulvo-vaginal Candidiasis (n=23) |
26,0% |
13,2% |
60,8% |
|
- Occasional or sporadic VVC (n=15) |
33,4% |
19,4% |
47,2% |
|
- Recurrent VVC (n=8) |
59,7% |
23,6% |
16,7% |
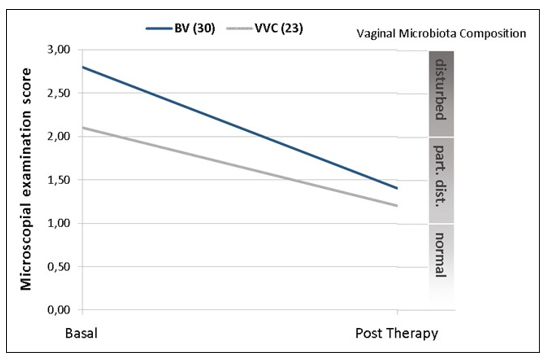
Figure 2: vaginal microbiota composition before and after therapy
Discussion
In terms of therapeutic efficacy, defined as clinical cure (resolution of signs and symptoms) and microbiological cure, women with BV had cure rates of about 60% with the new product containing peculiar lactobacillus strains. Patients with VVC reported a lower cure rate (about 40%).Interestingly, the subgroup characterized by a clinical history of recurrent candidiasis (RCCV), the more difficult to treat, were cured in higher percentage of cases (around 83%). Antibiotics and antimycotics generally work well against bacterial and mycotic infections but the potential side effects and progressive increase in bacterial and fungal resistance has stimulated the search for innovative therapeutic approaches. This is even more important for recurrent vaginal infections; in fact, epidemiological studies reveal that almost all women diagnosed with fluconazole-resistant C. albicans had experienced considerable previous exposure to Fluconazole [9]. that produce hydrogen peroxide and lactic acid, acting as a protective layer. This leads to an acidic pH, which inhibits the adhesion and growth of other bacteria, resulting in “good” protective bio film growth [10]. Biofilms are defined as microbial communities encased in a self-synthesized extracellular polymeric matrix, growing on abiotic or a biotic surface [6]. A “bad” biofilm is a strategy allowing some organisms to persist in harsh environments, and its physical removal is pivotal for eradication of the infection. Some studies have found that 90% of women with BV and 10% without BV have a complex polymicrobial biofilm, which can be demonstrated with electron microscopy of vaginal biopsies [11]. The tendency of Candida albicans to develop biofilms is also clinically relevant because biofilm-associated fungal cells are much more resistant to the conventional antifungal agents that act against their plank tonic counterparts and they resist host immune factors [12].
In our study, we used a therapy that included the use of particular probiotic strains that have proved probiotic properties, as B-group vitamins production, antioxidant power, immunomodulation and antimicrobial activity [13]. Particularly, the antimicrobial activity against the main pathogens responsible for VVC and BV was further investigated through two different experimental models. First, the probiotics bio film formation over the agar plate was able to inhibit the overgrowth of Candida albicans when co-cultured on the same plate. Second, the efficacy of the combination of these three strains in a single product was demonstrated in a reconstructed human vaginal epithelium model (RHVE Episkin). Two reconstructed tissues were infected by Gardnerella + vaginalis and by Atopobiumvaginae, and then treated with the probiotics blend. A 36% reduction in the pathogenic G. vaginalis bio film and a reduction by 40% against A.vaginae were observed after treatment.
As described previously, the cure rate was more relevant for BV and less significant for VVC patients. Nevertheless, differentiating between acute and recurrent vulvo-vaginal candidiasis, the percentage of responders rise significantly in the last, suggesting a tendency of the product to normalize the vaginal microbiota by replacing the pathogenic and persistent bio film with a healthier Lactobacillus microbiota. This observation aligns with the resulting analysis of vaginal fresh mount microscopy.
The role of the product in restoring and improving the composition of the vaginal microbiota is evident. Interesting is the standardization of vaginal flora at the microscopic examination, with an improvement of approximately 50% in both the groups studied and a complete resolution of the dysbiotic status.
Our study has certain limitations that must be acknowledged, such as the lack of randomization and the open-label nature of the trial. Therefore, a placebo effect cannot be ruled out. Moreover, the sample size for RVVC is small (eight patients).
In conclusion, the presented clinical data allow the newproduct in vaginal capsules containing peculiar strains of Lactobacillus to be recommended in the treatment of VVC and BV. The pharmacological and microbiological characteristics of the product suggest that this medical device may be a useful maintenance treatment if there are recurrent episodes.
References
- Sobel JD (2007) Vulvovaginal candidosis. Lancet. 369: 1961-1971.
- Nyirjesy P (2014) Management of Persistent Vaginitis. ObstetGynecol 124: 1135-1146.
- Kenyon C (2013) The global epidemiology of bacterial vaginosis: a systematic review. Am J Obstet Gynecol. 209: 505-523.
- Van de Wijgert J, Jespers V (2017) The global health impact of vaginal dysbiosis. Res Microbiol. S 168: 859-864.
- Gilbert GG Donder, Jana Zodzika, Dace Rezeberga (2014) Treatment of bacterial vaginosis: what we have and what we miss, Expert Opinion on Pharmacotherapy 15: 645-657.
- Ventolini G (2015) Vaginal Lactobacillus: bio film formation in vivo-clinical implications. Int J Women’s Health 7: 243-247.
- Verstraelen H, Swidsinski A (2013) The biofilm in bacterial vaginosis: implications for epidemiology, diagnosis and treatment. CurrOpin Infect Dis 26: 86-89.
- Sobel JD, Kapernick PS, Zervos M, Reed BD, Hooton T, et al. (2001) Treatment of complicated Candida vaginitis: comparison of single and sequential doses of fluconazole. Am J Obstet Gynecol 185: 363-369.
- Marchaim D, Lemanek L, Bheemreddy S, Kaye KS, Sobel JD (2012) Fluconazole-resistant Candida albicansvulvovaginitis. ObstetGynecol 120: 1407-1414.
- Terraf M, Juarez Tomas M, Nader-Macias M, Silva C (2012) Screening of bio film formation by beneficial vaginal lactobacilli and influence of culture media components. J Appl Microbiol 113: 1517-1529.
- Ventolini G (2015) Vaginal Lactobacillus: bio film formation in vivo-clinical implications. Int J Women’s Health 7: 243-247.
- Verstraelen H, Swidsinski A (2013) The bio film in bacterial vaginosis: implications for epidemiology, diagnosis and treatment. CurrOpin Infect Dis 26: 86-89.
- Chandra J, Kuhn DM, Mukherjee PK, Hoyer LL, McCormick T, et al. (2001) Bio film formation by the fungal pathogen Candida albicans: development, architecture, and drug resistance. J Bacteriol 183: 5385-5394.
- Presti I, D’Orazio G, Labra M, La Ferla B, Mezzasalma V, et al. (2015) Evaluation of the probiotic properties of new Lactobacillus and Bifidobacterium strains and their in vitro effect. Appl Microbiol Biotechnol 99: 5613-5626.


