Research Article - (2025) Volume 4, Issue 3
Impact of Neutrophils Lymphocytes Ratio on the Prognosis of Patients with Treated Metastatic Colorectal Cancer Undergoing Immunotherapy
Received Date: Jun 09, 2025 / Accepted Date: Jul 07, 2025 / Published Date: Jul 16, 2025
Copyright: ©©2025 Haichuan Su, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Wang, K., Deng, L., Yang, G., Su, H. (2025). Impact of Neutrophils Lymphocytes Ratio on the Prognosis of Patients with Treated Metastatic Colorectal Cancer Undergoing Immunotherapy. Biomed Sci Clin Res, 4(3), 01-11.
Abstract
Objective The aim of this study was to investigate the effect of factors such as neutrophils lymphocytes ratio(NLR) on the prognosis of patients with treated metastatic colorectal cancer who undergo immunotherapy, and the associated factors that influence the efficacy of immunotherapy.
Methods Colorectal cancer patients who attended the Department of Oncology of the Second Affiliated Hospital of Air Force Medical University from October 2019 to February 2023 were initially enrolled in this study, and 127 patients were finally included after the inclusion and exclusion criteria. Statistical methods were used to analyze the effects of whether or not to receive immune checkpoint inhibitors, the type of immune checkpoint inhibitors, and NLR on the evaluation of the efficacy of tumor treatment for patients with treated metastatic colorectal cancer. The effects of different factors such as gender, tumor site, KRAS status, mismatch repair protein(MMR), and NLR on the prognosis of treated metastatic colorectal cancer.
Results Of the 127 patients, 65 received immunotherapy and 62 did not. There was no statistically significant difference between the therapeutic efficacy of domestic and imported immune checkpoint inhibitors for metastatic colorectal cancer patients who had previously received more than one first-line treatment . Kaplan-Meier survival curves plotted by SPSS found that there was a statistical difference between NLR (P=0.007) and MMR (P=0.029) for progression free survival(PFS) in patients with treated metastatic colorectal cancer.Cox univariate survival analysis found that NLR (P=0.012) and MMR (P=0.046) were two factors affecting PFS in colorectal cancer patients. Cox multivariate analysis concluded that NLR<3 (P=0.024) was an independent risk factor affecting survival time of patients with treated metastatic colorectal cancer who received immune checkpoint inhibitors, with a statistically significant difference.
Conclusion For patients with treated metastatic colorectal cancer, NLR and MMR are important indicators of PFS, and NLR<3 is an independent risk factor for survival time of patients with treated metastatic colorectal cancer who receive immune checkpoint inhibitors, which has an important predictive value of prognosis.
Keywords
Neutrophils Lymphocytes Ratio, Immunotherapy, Colorectal Cancer, Prognosis
Background
Colorectal cancer, as a very common malignant tumor in the world, is always damaging people's lives and properties. According to the cancer data just released at the beginning of 2024, as of 2022, colorectal cancer has the second highest incidence rate among all malignant tumors in the whole population, with the fourth highest incidence rate among women and the second highest incidence rate among men [1]. In China, the 5-year survival rate of colorectal cancer patients is between 50% and 60%, which is slightly lower than that of the United States, Japan, and most European countries [2-4]. In addition, first-line treatment for metastatic colorectal cancer is effective, but benefit from back-line treatment remains limited [5]. At the same time, our country patients are mostly have good physical constitutions and have a higher likelihood of receiving back-line treatments, making more appropriate treatments especially important for them. Around 2020, the advent of immune checkpoint inhibitors, such as PD-L1 and CTLA-4 brought a new light to the treatment of malignant tumors [6-8]. However, in colorectal cancer patients, the efficacy of immunotherapy remains controversial due to issues such as its specific molecular mechanisms [9]. For colorectal cancer, there are currently limited biomarkers that can predict disease outcome [10]. It has been shown that microsatellite status, TMB, POLE/POLD1 mutation, and PD-L1 expression can assess the effectiveness of treatment with immune checkpoint inhibitors to varying degrees [11-13]. Currently, there is an increasing number of research that suggests that inflammatory markers such as the degree of lymphocyte and neutrophil infiltration can largely predict the outcome of malignant tumors, especially immunotherapy, and the evidence for this claim is inextricably linked to the doctrine of the tumor microenvironment [14-18]. For patients with early-stage colorectal cancer, perioperative NLR may have a certain relationship with their recurrence and prognosis [19]. However, NLR have not received a great deal of attention and there is no uniformity for colorectal cancer patients in advanced stages, especially those receiving back-line immunotherapy. In this paper, through a retrospective case study, we analyzed that NLR in patients with treated advanced colorectal cancer can serve as an important predictor of the efficacy of immunotherapy.
Materials and Methods
Clinical Date
A total of 474 patients with colorectal cancer attending the Department of Oncology of the Second Affiliated Hospital of Air Force Military Medical University from October 2019 to February 2023 were initially included in this study, and those with one of the following conditions were excluded by the exclusion criteria: (1) patients without a definitive pathological diagnosis; (2) patients receiving postoperative adjuvant chemotherapy or regular follow¬up; (3) patients without target lesions who cannot be evaluated for RESIST 1.1; (4) patients with advanced colorectal cancer receiving first-line treatment at first diagnosis; (5) patients with a combination of 2 or more malignancies; (6) end-stage patients receiving palliative symptomatic supportive care; (7) patients with unsuccessful follow-up or incomplete case data. Finally, 127 patients were included. Our study was approved by the Institutional Review Board of the Second Affiliated Hospital of Air Force Medical University. Patients who received immunotherapy were defined as the experimental group, patients who did not receive immunotherapy were defined as the control group. The selection of immunotherapy is strictly based on existing data from large randomized controlled trials. The primary endpoint was survival analysis, and the secondary endpoint was immunotherapy efficacy. The time from the start of immunotherapy to the occurrence of disease progression or death due to various causes in these patients was defined as PFS. Factors of stratified analysis included gender, MMR, tumor site, KRAS status, type of immune checkpoint inhibitor, whether combined with targeted therapy, whether liver metastatic cancer, whether lymph nodes metastases, and NLR level. NLR was defined as the ratio of the neutrophil count to the lymphocyte count of the peripheral blood, and NLR <3 was classified as the low NLR group, NLR ≥3 was classified as the high NLR group. MLH1, MSH2, MSH6 and PMS2 are the main proteins of MMR. When the expression of at least one of these proteins is missing, it is considered to be a mismatch repair gene defect (dMMR), and if all are positive, it is considered to be a mismatch repair gene intact (pMMR). Refer to Response Evaluation Criteria in Solid Tumors version 1.1(RECIST version 1.1), Complete Response (CR): Disappearance of all target lesions, any pathological lymph nodes (whether target or non-target) must have reduction in short axis to <10 mm; Partial Response (PR): At least a 30% decrease in the sum of diameters of target lesions, taking as reference the baseline sum diameters; Progressive Disease (PD): At least a 20% increase in the sum of diameters of target lesions, taking as reference the smallest sum on study (this includes the baseline sum if that is the smallest on study) [20]. In addition to the relative increase of 20%, the sum must also demonstrate an absolute increase of at least 5 mm. (Note: the appearance of one or more new lesions is also considered progression); Stable Disease (SD): Neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD, taking as reference the smallest sum diameters while on study.
Statistical Methods
SPSS version 25 was used for all statistical methods in this study. Use histograms and Q-Q plots to determine the normality of the data, and the evaluation of the group receiving immunotherapy and the group not receiving immunotherapy (including progressive disease, stable disease, and partial response), the evaluation of the group receiving domestic or imported immune checkpoint inhibitors, as well as the evaluation of the different NLR level groups were taken by the Chi-square test, and the Fisher's exact test was taken when the frequency number was less than 5, P<0.05 was considered statistically different. Kaplan-Meier survival analysis was used to plot the survival curves of different factor groups respectively; Cox univariate survival analysis was used to identify the important factors affecting the PFS of colorectal cancer patients among several factors respectively, and then Cox multivariate survival analysis was used to correct and identify the factors affecting the prognosis among the above factors, and the same was considered as statistically different with P<0.05.
Results
Patient Characteristics
127 patients were included, 87 males; 40 females; the age range was 41-81 years old, with a median age of 60 years; there were 65 patients who received immune checkpoint inhibitors(ICI) and 62 patients who did not receive immune checkpoint inhibitors; 84 cases of left colon cancer (including left transverse colon, descending colon, and sigmoid colon), and 43 cases of right colon cancer (including cecum, ascending colon, and right transverse colon); 9 cases of patients with dMMR, and 118 cases of patients with pMMR; 63 cases of patients with KRAS mutant type, and 64 cases of patients with KRAS wild type; 86 cases of patients who combined targeted therapy in the course of treatment, 41 cases of patients who did not combine targeted therapy, specific detailed data are shown in Table 1.
|
Characteristic |
Group A: with ICI (n=65) |
Group B: without ICI (n=62) |
|
Age(years), median(range) |
60 (46-78) |
58(41-81) |
|
Gender, n(%) |
|
|
|
Male |
46 (70.8) |
41 (66.1) |
|
Female |
19 (29.2) |
21 (33.9) |
|
Tumor location, n(%) |
|
|
|
Right colon |
24 (36.9) |
19 (30.6) |
|
Left colon |
41 (63.1) |
43 (69.4) |
|
MMR, n(%) |
|
|
|
dMMR |
9 (13.8) |
0 |
|
pMMR |
56 (86.2) |
62 (100) |
|
KRAS, n(%) |
|
|
|
KRAS mutant |
37 (56.9) |
26 (41.9) |
|
KRAS wild |
28 (43.1) |
36 (58.1) |
|
Combined with targeted therapy, n(%) |
|
|
|
Yes |
24 (36.9) |
62 (100) |
|
No |
41 (63.1) |
0 |
|
Liver metastases, n(%) |
|
|
|
Yes |
51 (78.5) |
45 (72.6) |
|
No |
14 (21.5) |
17 (27.4) |
|
Lymph nodes metastases, n(%) |
|
|
|
Yes |
48 (73.8) |
41 (66.1) |
|
No |
17 (26.2) |
21 (33.9) |
Table 1: Patient Characteristics
Abbreviations: ICI, Immune Checkpoint Inhibitors; MMR, Mismatch Repair; PMMR, Proficient Mismatch Repair; DMMR, Deficient Mismatch Repair
Comparison of Efficacy Between the Group Receiving ICI and the Group not Receiving ICI
After inclusion and exclusion criteria, 127 patients were enrolled in the study, including 65 patients who received immunotherapy and 62 patients who did not receive immunotherapy. Patients in both groups were evaluated retrospectively by imaging with enhanced CT at 2-month intervals. Refer to RECIST version 1.1, the results showed that of the 65 patients in the group receiving immunotherapy, 21 patients with PD, 40 patients with SD, 4 patients with PR, 4 patients met the criteria for objective response, and 44 patients met the criteria for disease control; and of the 62 patients in the group not receiving immunotherapy, 17 patients with PD, 43 patients with SD, 2 patients with PR, 2 patients met the criteria for objective response, and 45 patients met the criteria for disease control. The results of the different evaluations of the two groups of patients were statistically analysed separately, and the results using the Chi-square test and Fisher’s exact test were as follows: in the case of PD or not, there was no statistical difference between the two groups, P = 0.548; in the case of SD or not, there was no statistical difference between the two groups, P = 0.355; in the case of PR or not, there was no statistical difference between the two groups, P = 0.680; in the case of objective response or not, there is no statistical difference between the two groups, P = 0.680; in the case of disease control or not, there is no statistical difference between the two groups, P = 0.548, as shown in Table 2.
|
Variable |
Total (n = 127) |
Group A: with ICI (n=65) |
Group B: without ICI (n=62) |
X2 |
P |
|
Progressive Disease |
|
|
|
0.362 |
0.548 |
|
Yes |
38 (29.9 %) |
21 (32.3%) |
17 (27.42%) |
|
|
|
No |
89 (70.1 %) |
44 (67.7%) |
45 ( 72.58%) |
|
|
|
Stable Disease |
|
|
|
0.856 |
0.355 |
|
Yes |
83 (65.4 %) |
40 (61.5%) |
43 (69.35 %) |
|
|
|
No |
44 (34.6 %) |
25 (38.5%) |
19 (30.65 %) |
|
|
|
Partial Response |
|
|
|
0.129 |
0.680 |
|
Yes |
6 (4.7 %) |
4 (6.2%) |
2 (3.23 %) |
|
|
|
No |
121 (95.3 %) |
61 (93.8%) |
60 (96.77 %) |
|
|
|
objective response |
|
|
|
0.129 |
0.680 |
|
Yes |
6 (4.7%) |
4 (6.2%) |
2 (3.23 %) |
|
|
|
No |
121 (95.3%) |
61 (93.8%) |
60 (96.77 %) |
|
|
|
disease control |
|
|
|
0.362 |
0.548 |
|
Yes |
89 (70.1%) |
44 (67.7%) |
45 (72.58 %) |
|
|
|
No |
38 (29.9%) |
21 (32.3%) |
17 (27.42 %) |
|
|
Table 2: Comparison of Efficacy Between the Group with ICI and the Group Without ICI
Abbreviations: ICI, Immune Checkpoint Inhibitors
Comparison of the Efficacy Between the Domestic Immune Checkpoint Inhibitor Group and the Imported Immune Checkpoint Inhibitor Group
There were 65 patients receiving immunotherapy in this study, all of whom received PD-1 immune checkpoint inhibitors. Among them, there were 13 patients treated with imported immune checkpoint inhibitors, all of which were pembrolizumab, and 52 patients treated with domestic immune checkpoint inhibitors, which were sintilimab and tislelizumab. The results showed that among the 52 patients treated with domestic immune checkpoint inhibitors, 17 patients with PD, 32 patients with SD, 3 patients with PR, 3 patients met the criteria for objective response, and 35 patients met the criteria for disease control, while among the 13 patients treated with imported immune checkpoint inhibitors, 4 patients with PD, 8 patients with SD, 1 patient with PR, 1 patient met the criteria for objective response, and 9 patients met the criteria for disease control. The results of the different evaluations of the two groups of patients were statistically analysed separately, and the results using the Chi-square test and Fisher’s exact test were as follows: in the case of PD or not, there was no statistical difference between the two groups, P = 0.894; in the case of SD or not, there was no statistical difference between the two groups, P > 0.999; in the case of PR or not, there was no statistical difference between the two groups, P = 0.796; in the case of objective response or not, there is no statistical difference between the two groups, P = 0.796; in the case of disease control or not, there is no statistical difference between the two groups, P = 0.894, as shown in Table 3.
|
Variable |
Total (n = 65) |
Group A: domestic ICI (n=52) |
Group B: imported ICI (n=13) |
X2 |
P |
|
Progressive Disease |
|
|
|
0.018 |
0.894 |
|
Yes |
21 (32.3%) |
17 (32. 7 %) |
4 (30.8 %) |
|
|
|
No |
44 (67.7%) |
35 (67.3 %) |
9 (69.2 %) |
|
|
|
Stable Disease |
|
|
|
ï¼?0.001 |
ï¼?0.999 |
|
Yes |
40 (61.5%) |
32 (61.5%) |
8 (61.5 %) |
|
|
|
No |
25 (38.5%) |
20 (38.5%) |
5 (38.5 %) |
|
|
|
Partial Response |
|
|
|
0.067 |
0.796 |
|
Yes |
4 (6.2%) |
3 (5.8 %) |
1 (7.7 %) |
|
|
|
No |
61 (93.8%) |
49 (94.2 %) |
12 (92.3 %) |
|
|
|
objective response |
|
|
|
0.067 |
0.796 |
|
Yes |
4 (6.2%) |
3 (5.8 %) |
1 (7.7 %) |
|
|
|
No |
61 (93.8%) |
49 (94.2 %) |
12 (92.3 %) |
|
|
|
disease control |
|
|
|
0.018 |
0.894 |
|
Yes |
44 (67.7%) |
35 (67.3 %) |
9 (69.2 %) |
|
|
|
No |
21 (32.3%) |
17 (32. 7 %) |
4 (30.8 %) |
|
|
Table 3: Comparison of Efficacy Between the Domestic ICI Group and the Imported ICI Group
Abbreviations: ICI, Immune Checkpoint Inhibitors
Comparison of Efficacy Between High NLR and Low NLR Groups
A total of 127 treated metastatic advanced colorectal cancer patients were enrolled in this study, among which 65 patients received immune checkpoint inhibitor therapy. Peripheral blood of 65 patients was tested within one week before receiving immune checkpoint inhibitor therapy. The results showed that of the 24 patients in the high NLR group, 13 patients with PD, 10 patients with SD, 1 patient with PR, 1 patient met the criteria for objective response, and 11 patients met the criteria for disease control, while of the 41 patients in the low NLR group, 8 patients with PD, 30 patients with SD, 3 patients with PR, 3 patients met the criteria for objective response, and 33 patients met the criteria for disease control. The results of different evaluations of the two groups of patients were statistically analysed separately, and the results using the Chi-square test and Fisher’s exact test were as follows: in the case of PD or not, there was a statistical difference between the two groups, P = 0.004; in the case of SD or not, there was a statistical difference between the two groups, P = 0.012; in the case of PR or not, there was no statistical difference between the two groups, P = 0.610; in the case of objective response or not, there is no statistical difference between the two groups, P = 0.610; in the case of disease control or not, there is a statistical difference between the two groups, P = 0.004, as shown in Table 4.
|
Variable |
Total (n = 65) |
Group A: High NLR (n=24) |
Group B: Low NLR (n=41) |
X2 |
P |
|
Progressive Disease |
|
|
|
8.313 |
0.004 |
|
Yes |
21 (32.3 %) |
13 (54.2%) |
8 (19.5%) |
|
|
|
No |
44 (67.7 %) |
11 (45.8%) |
33 (80.5 %) |
|
|
|
Stable Disease |
|
|
|
6.348 |
0.012 |
|
Yes |
40 (61.5 %) |
10 ( 41.7%) |
30 (73.2%) |
|
|
|
No |
25 (38.5 %) |
14 (58.3%) |
11(26.8%) |
|
|
|
Partial Response |
|
|
|
0.260 |
0.610 |
|
Yes |
4 (6.2 %) |
1 (4.2%) |
3 (7.3%) |
|
|
|
No |
61 (93.8 %) |
23 (95.8 %) |
38 (92.7 %) |
|
|
|
objective response |
|
|
|
0.260 |
0.610 |
|
Yes |
4 (6.2 %) |
1 (4.2%) |
3 (7.3%) |
|
|
|
No |
61 (93.8 %) |
23 (95.8 %) |
38 (92.7 %) |
|
|
|
disease control |
|
|
|
8.313 |
0.004 |
|
Yes |
44 (67.7 %) |
11 (45.8 %) |
33(80.5 %) |
|
|
|
No |
21 (32.3 %) |
13 (54.2 %) |
8 (19.5 %) |
|
|
Table 4: Comparison of Efficacy Between High NLR and Low NLR Groups
Abbreviations: NLR, neutrophils lymphocytes ratio
Kaplan-Meier Survival Curves for Gender, Tumor Site, MMR, NLR and Other Factors
In this study, 65 patients who received immune checkpoint inhibitors were included for survival follow-up to determine PFS. According to different factors (including gender, MMR, tumor site, KRAS status, type of immune checkpoint inhibitor, whether combined with targeted therapy, whether liver metastatic cancer, whether lymph nodes metastases, and NLR level), they were divided into different groups, and the Kaplan-Meier survival curves of each group were plotted separately. The results showed that in the MMR grouping, there was a statistical difference between the PFS survival curves of the two, P = 0.029. In addition, in the NLR level grouping, there was a statistical difference between the PFS survival curves of the two, P = 0.007. Finally, there was no statistically significant difference in the PFS survival curves among the groups of gender, tumor site, KRAS status, type of immune checkpoint inhibitor, whether or not it was combined with targeted therapy, whether or not it was hepatic metastatic cancer and whether or not it was lymph nodes metastases, P > 0.05, as shown in Figure 1.
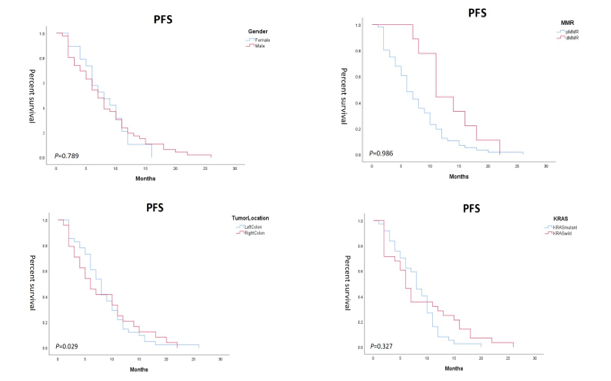
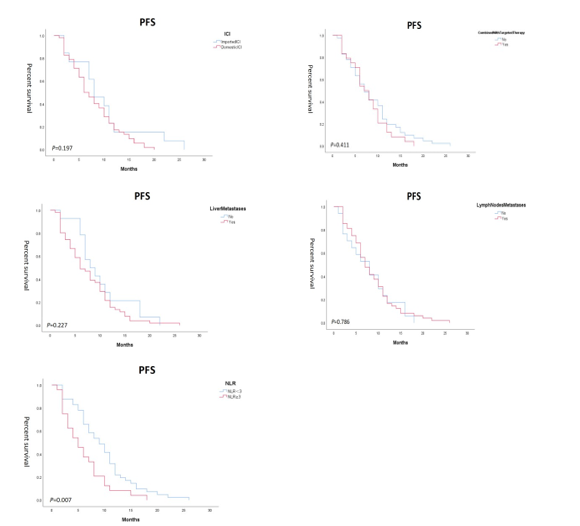
Figure 1: Kaplan-Meier Survival Curves for Several Factors
Cox Univariate Analyses and Cox Multivariate Analyses for Different Factors
A total of 65 out of 127 patients with treated metastatic advanced colorectal cancer who received immune checkpoint inhibitors were followed up. Age (≤60 vs. >60), gender (Female vs. Male), tumor site (Left colon vs. Right colon), MMR (pMMR vs. dMMR), KRAS status (KRAS mutant vs. KRAS wild), whether combined with targeted therapy (No vs. Yes ), whether liver metastatic cancer (No vs.Yes), whether lymph nodes metastases (No vs.Yes), type of immune checkpoint inhibitor (Domestic vs. Imported), and level of NLR (<3 vs.≥3) were used to perform Cox one-way survival analysis. The results showed that there was a statistically significant difference in PFS between the different groups of patients with MMR status and NLR level factors, P < 0.05, and no statistically significant difference in the other factors. The MMR and NLR level factors were then included in Cox multivariate survival analysis, and the results showed that there was a statistically significant difference in the PFS data between the patients in the high NLR group and the low NLR group, P = 0.024, as shown in Table 5.
|
|
Univariate analysis |
Multivariate analysis |
||||
|
|
HR |
95% CI |
P value |
HR |
95% CI |
P value |
|
Age(years) |
|
|
|
|
|
|
|
(≤60 vs.ï¼?60) |
1.006 |
0.610-1.659 |
0.981 |
|
|
|
|
Gender |
||||||
|
(Female vs.Male) |
0.933 |
0.541-1.609 |
0.802 |
|
|
|
|
Tumor location |
||||||
|
(Left colon vs.Right colon) |
1.004 |
0.602-1.676 |
0.987 |
|
|
|
|
MMR |
||||||
|
(pMMR vs.dMMR) |
0.483 |
0.237-0.988 |
0.046 |
0.447 |
0.196-1.019 |
0.055 |
|
KRAS |
||||||
|
(KRAS mutant vs.KRAS wild) |
0.783 |
0.463-1.323 |
0.360 |
|
|
|
|
Combined with targeted therapy |
||||||
|
(No vs.Yes) |
1.223 |
0.731-2.048 |
0.443 |
|
|
|
|
Liver metastases |
||||||
|
(No vs.Yes) |
1.412 |
0.774-2.576 |
0.261 |
|
|
|
|
Lymph nodes metastases |
||||||
|
(No vs.Yes) |
0.930 |
0.531-1.629 |
0.800 |
|
|
|
|
ICI |
|
|
|
|
|
|
|
(Domestic vs.Imported) |
1.496 |
0.774-2.890 |
0.231 |
|
|
|
|
NLR |
||||||
|
(ï¼?3 vs.≥3) |
1.944 |
1.155-3.272 |
0.012 |
1.918 |
1.088-3.382 |
0.024 |
Table 5: Cox Analyses for Different Factors
Abbreviations: CI, Confidence Interval; MMR, Mismatch Repair; ICI, Immune Checkpoint Inhibitors; PMMR, Proficient Mismatch Repair; DMMR, Deficient Mismatch Repair; NLR, Neutrophils Lymphocyte Ratio
Discussion
At present, there are numerous treatment modalities for colorectal cancer. Firstly, new form of chemotherapeutic drugs, the most representative of which is irinotecan hydrochloride liposome injection, PEPCOL and other relevant clinical research trials reveal that the traditional chemotherapeutic drug irinotecan, carried in liposomes, can play its efficient anti-tumor role while bringing fewer side effects to the patients [21,22]. Secondly, the newly discovered targeted treatment, KRAS target has been difficult to become a drug in the past. In recent years, through continuous scientific research and clinical medicine, it has been found that its corresponding targeted drugs combined with chemotherapy and immunotherapy can bring more benefits to colorectal cancer patients [23,24]. Moreover, even for antibody-drug conjugates(ADC), claudin 18.2, as a newly discovered site, has been highly expressed in gastric cancer as well as colorectal cancer, and the results of existing multi-center medical institution studies have shown that the relevant targeting drugs are highly expressed in gastric cancer as well as colorectal cancer, and existing multicentre medical institution studies have shown that related ADC are also effective in anti-tumor effects [25,26].
In our study, we found that for patients with treated advanced colorectal cancer, there was no statistically significant difference in patient outcomes between treatment with and without the selection of immune checkpoint inhibitors. It is well known that there is currently no uniform guideline or expert consensus on immunotherapy for patients with advanced colorectal cancer, except for patients with dMMR for whom an indication for treatment with immune checkpoint inhibitors [27]. So in this case, when the patient's physical status scores are good and they are able to tolerate the adverse effects caused by antineoplastic therapy, the choice of treatment may be varied and even chaotic. Targeted therapy, ADC, and immune checkpoint inhibitors may all be effective treatments for prolonging patient’s overall survival or progression free survival, however there is no doubt that the choice of immunotherapy will continue to be important, because of maintenance and consolidation therapy after disease stabilization [28].
This study also found no significant difference in the therapeutic effect of colorectal cancer patients in terms of the choice of domestic or imported immune checkpoint inhibitors. This conclusion is consistent with Si-Yang Maggie Liu's research [29]. Pembrolizumab injection is a PD-1 pathway inhibitor developed by Merck Sharp & Dohme, and is the immune checkpoint inhibitor with the widest range of indications, including adenocarcinoma of the lung, squamous carcinoma of the lung, esophageal carcinoma, gastric carcinoma, squamous cell carcinoma of the head and neck, triple-negative breast cancer, and hepatocellular carcinoma. However, due to export and tariff reasons, its price is relatively high in China, where the proportion of rural patients suffering from malignant tumors is more than half, which means that the majority of patients are unable to afford an imported immune checkpoint inhibitor like pembrolizumab injection due to financial reasons. In this study, of the 65 patients receiving back-line treatment with immune checkpoint inhibitors, only 13 patients were treated with imported immune checkpoint inhibitors, specifically pembrolizumab injection. The other 52 patients were treated with domestic immune checkpoint inhibitors, including sintilimab and tislelizumab. The prices of these domestic immune checkpoint inhibitors are relatively low, and some of them are also included in the reimbursement scope of China's national health insurance, which means that the final price may be one-tenth of that of the imported immune checkpoint inhibitors, which is the current status quo in the treatment of malignant tumors. Secondly, putting aside the economic factor, whether domestic or imported immune checkpoint inhibitors, their research, development and production processes are under appropriate national supervision, so the drugs themselves are absolutely qualified. In terms of the mechanism, all of them inhibit tumor immune escape and the related changes in the tumor microenvironment by blocking the PD-1 pathway, thus playing an anti-tumor therapeutic role. Therefore, fundamentally, the therapeutic effects of both domestic and imported immune checkpoint inhibitors on malignant tumors are the same, which was also confirmed in this study from the perspective of retrospective case analysis.
In addition, the most important point is that in this study, PFS survival curves were plotted by the Kaplan-Meier survival analysis method for different factor groups, including gender, MMR, tumor site, KRAS status, type of immune checkpoint inhibitor, whether or not combine with targeted therapy, whether or not with liver metastases, whether or not with lymph nodes metastases, and the level of NLR. These graphs demonstrate that gender, tumor site, KRAS status, type of immune checkpoint inhibitor, whether or not combination targeted therapy, whether or not with liver metastases, and whether or not with lymph nodes metastases were not statistically different for PFS in patients with treated advanced colorectal cancer, P > 0.05. However, in the factor of MMR, it can be clearly noticed from the graphs that there is a clear separation of the PFS survival curves of the dMMR and pMMR groups and that the PFS of the dMMR group is better than that of the pMMR, P = 0.029, which indicates a statistical difference. This result is also consistent with KEYNOTE177, which concluded that colorectal cancer patients with dMMR who treated with immune checkpoint inhibitors were able to demonstrate better efficacy. Therefore, for colorectal cancer patients, MMR is an important factor in the selection of immunotherapy. In addition, there was also a statistical difference in PFS between the high NLR and low NLR groups for patients with treated advanced colorectal cancer, with P = 0.007, suggesting that patients in the low NLR group undergoing immunotherapy can lead to longer PFS compared to the high NLR group. Ouyang H's research pointed out that low baseline NLR is significantly associated with a better prognosis in mCRC patients treated with immunotherapy. This is basically consistent with the conclusion of our study. Neutrophil infiltration in the tumor microenvironment is a complex biological process involving multiple functions and states [30-32]. Recent studies have revealed that neutrophils play an important role in tumor development and therapy [33,34]. In cancer, neutrophil infiltration exhibits a high degree of heterogeneity and can be classified into different subpopulations based on their specific status in the tumor microenvironment. For example, HLA-DR+CD74+ neutrophils were associated with better patient prognosis, suggesting that such neutrophils may have stronger anti-tumor activity [35]. Therefore, low NLR indicates that the degree of infiltration of inflammatory-related factors in the tumor microenvironment of this patient is well expressed, which is particularly important in terms of more lymphocytes and fewer neutrophils.
In Cox univariate analysis, it found that among several factors such as gender, MMR, tumor site, KRAS status, type of immune checkpoint inhibitor, whether or not combine with targeted therapy, whether or not with liver metastases, whether or not with lymph nodes metastases, and the level of NLR, the two factors of MMR and the level of NLR were correlated with the PFS of patients with advanced colorectal cancer who had been treated, P < 0.05. After Cox multivariate survival analysis, patients' NLR level was found to be an important predictor of PFS, with the low NLR group having a longer PFS than the high NLR group. Studies have shown that lymphocyte counts and neutrophil counts reflect the state of systemic inflammation, and that inflammation has a tumor-promoting effect, which contributes to tumor cell proliferation and survival as well as promotes angiogenesis and metastasis [36]. However, it should be noted that NLR is only an auxiliary indicator, and its interpretation and application should be combined with patient's specific situation and tumor type.
Of course, there are certain shortcomings in this study. Firstly, the small number in terms of sample size may lead to statistical bias in the final conclusion. Secondly, as this study only compared the efficacy of immunotherapy on patients with advanced colorectal cancer, but not the adverse effects of the treatment, the results may be relatively one-sided. However, the innovative idea of this study still has some research value, and more studies will be conducted to confirm this conclusion.
Conclusion
In summary, NLR can be an important biomarker for assessing the prognosis of patients with advanced tumors, especially NLR<3 is an independent risk factor for survival time of patients with treated metastatic colorectal cancer who receive immune checkpoint inhibitors. Its elevated level is usually associated with poorer prognosis, which helps doctors to develop more effective treatment strategies.
Data Sharing Statement
All data generated or analysed during this study are included in this manuscript.
Ethical Statement
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Institutional Review Board of the Second Affiliated Hospital of Air Force Medical University(TDLL-202411-06). Informed consent was waived due to the retrospective nature of this study and the anonymous processing of patient data.
Consent for Publication
Written informed consent for publication was obtained from all participants.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by the Key Research and Development Projects of Shaanxi Province to Haichuan Su (No.2022ZDLSF03-01) and Shaanxi Provincial Health Research Innovation Team Project (2024TD-04).
Disclosure
The authors declare that they have no conflict of interest.
References
- Bray, F., Laversanne, M., Sung, H., Ferlay, J., Siegel, R. L., Soerjomataram, I., & Jemal, A. (2024). Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians, 74(3), 229-263.
- Olukunle, O. F., Omoboyede, V., & Chukwuemeka, P. O. (2024). Network pharmacology and molecular docking-based identification of drug candidates and key targets of Allium sativum for colorectal cancer treatment. Journal of Biomolecular Structure and Dynamics, 42(9), 4442-4455.
- Ansa, B. E., Alema-Mensah, E., Sheats, J. Q., Mubasher, M., & Akintobi, T. H. (2023). Colorectal Cancer Knowledge and Screening Change in African Americans: Implementation Phase Results of the EPICS Cluster RCT. AJPM focus, 2(4), 100121.
- Allemani, C., Matsuda, T., Di Carlo, V., Harewood, R., Matz, M., NikšiÃÂ??, M., ... & Hood, M. (2018). Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. The Lancet, 391(10125), 1023-1075.
- Modest, D. P., Pant, S., & Sartore-Bianchi,A. (2019). Treatment sequencing in metastatic colorectal cancer. European Journal of Cancer, 109, 70-83.
- Xu, R., Wang, W., Zhu, B., Lin, X., Ma, D., Zhu, L., ... &Shen, L. (2020). Disease characteristics and treatment patterns of Chinese patients with metastatic colorectal cancer: a retrospective study using medical records from China. BMC cancer, 20, 1-10.
- Ren, W., Fang, Y., He, Y., Ren, Y., Wang, M., Xu, A., ... &Tao, Q. (2024). Efficacy and Safety of Programmed Death 1/ Programmed Death-Ligand 1 Plus Cytotoxic T-Lymphocyte–Associated Antigen 4 Inhibitors for Advanced or Metastatic Non–Small Cell Lung Cancer: A Meta-analysis Based on Randomized Controlled Trials. Therapeutic Drug Monitoring, 46(4), 422-433.
- Elez, E., Cubillo, A., Alfonso, P. G., Middleton, M. R., Chau, I., Alkuzweny, B., ... & Van Cutsem, E. (2024). Binimetinib in combination with nivolumab or nivolumab and ipilimumab in patients with previously treated microsatellite-stable metastatic colorectal cancer with RAS mutations in an open-label phase 1b/2 study. BMC cancer, 24(1), 446.
- Wang, Z. X., Peng, J., Liang, X., Cheng, Y., Deng, Y., Chen, K., ... & Xu, R. H. (2024). First-line serplulimab in metastatic colorectal cancer: Phase 2 results of a randomized, double-blind, phase 2/3 trial. Med, 5(9), 1150-1163.
- Zhong, W., Yu, Z., Zhan, J., Yu, T., Lin, Y., Xia, Z. S., ... &Chen, Q. K. (2015). Association of serum levels of CEA, CA199, CA125, CYFRA21-1 and CA72-4 and disease characteristics in colorectal cancer. Pathology & Oncology Research, 21, 83-95.
- Schrock, A. B., Ouyang, C., Sandhu, J., Sokol, E., Jin, D., Ross, J. S., ... & Fakih, M. (2019). Tumor mutational burden is predictive of response to immune checkpoint inhibitors in MSI-high metastatic colorectal cancer. Annals of oncology, 30(7), 1096-1103.
- Wang, F., Zhao, Q., Wang, Y. N., Jin, Y., He, M. M., Liu, Z.X., & Xu, R. H. (2019). Evaluation of POLE and POLD1 mutations as biomarkers for immunotherapy outcomes across multiple cancer types. JAMA oncology, 5(10), 1504-1506.
- Li, Y., Liang, L., Dai, W., Cai, G., Xu, Y., Li, X., ... & Cai, S.(2016). Prognostic impact of programed cell death-1 (PD-1) and PD-ligand 1 (PD-L1) expression in cancer cells and tumor infiltrating lymphocytes in colorectal cancer. Molecular cancer, 15, 1-15.
- Rozek, L. S., Schmit, S. L., Greenson, J. K., Tomsho, L. P., Rennert, H. S., Rennert, G., & Gruber, S. B. (2016). Tumor-infiltrating lymphocytes, Crohn’s-like lymphoid reaction, and survival from colorectal cancer. Journal of the National Cancer Institute, 108(8), djw027.
- Guren, M. G. (2019). The global challenge of colorectal cancer. The Lancet Gastroenterology & Hepatology, 4(12), 894-895.
- Kroemer, M., Turco, C., Spehner, L., Viot, J., Idirène, I., Bouard, A., ... & Borg, C. (2020). Investigation of the prognostic value of CD4 T cell subsets expanded from tumor-infiltrating lymphocytes of colorectal cancer liver metastases. Journal for immunotherapy of cancer, 8(2), e001478.
- Tamai, K., Okamura, S., Makino, S., Yamamura, N., Fukuchi, N., Ebisui, C., ... & Yano, M. (2022). C-reactive protein/ albumin ratio predicts survival after curative surgery in elderly patients with colorectal cancer. Updates in Surgery, 1-10.
- Petracci, E., Passardi, A., Biggeri, A., Valgiusti, M., Monti, M., Frassineti, G. L., ... & Scarpi, E. (2024). Baseline and Longitudinal Neutrophil-to-Lymphocyte Ratio as Prognostic Factor for Metastatic Colorectal Cancer: A Secondary Analysis of the ITACa Randomized Trial. JCO Precision Oncology, 8, e2300256.
- Ergen,S. A., Barlas, C., Yildirim, C., & Öksüz, D. Ç. (2022). Prognostic role of peripheral neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR) in patients with rectal cancer undergoing neoadjuvant chemoradiotherapy. Journal of Gastrointestinal Cancer, 1-10.
- Eisenhauer, E. A., Therasse, P., Bogaerts, J., Schwartz, L. H., Sargent, D., Ford, R., ... & Verweij, J. (2009). New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). European journal of cancer, 45(2), 228-247.
- Chibaudel, B., Maindrault-Gœbel, F., Bachet, J. B., Louvet, C., Khalil, A., Dupuis, O., ... & de Gramont, A. (2016). PEPCOL: a GERCOR randomized phase II study of nanoliposomal irinotecan PEP 02 (MM-398) or irinotecan with leucovorin/5-fluorouracil as second-line therapy in metastatic colorectal cancer. Cancer medicine, 5(4), 676-683.
- Kalra, A. V., Kim, J., Klinz, S. G., Paz, N., Cain, J., Drummond,D. C., ... & Fitzgerald, J. B. (2014). Preclinical activity of nanoliposomal irinotecan is governed by tumor deposition and intratumor prodrug conversion. Cancer research, 74(23), 7003-7013.
- Wang, J. X., Wu, H. L., Zhu, M., & Zhou, R. (2020). Role of anti-epidermal growth factor receptor therapy compared with anti-vascular endothelial growth factor therapy for metastatic colorectal cancer: an update meta-analysis of randomized clinical trials. Pathology & Oncology Research, 26, 159-166.
- Liang, X., & Shen, J. (2020). Impact of KRAS mutation status on outcomes of metastatic colorectal cancer treated with anti-angiogenic agents: a meta-analysis. Journal of Chemotherapy, 32(1), 41-48.
- Lordick, F., Thuss-Patience, P., Bitzer, M., Maurus, D., Sahin, U., & Türeci, Ö. (2023). Immunological effects and activity of multiple doses of zolbetuximab in combination with zoledronic acid and interleukin-2 in a phase 1 study in patients with advanced gastric and gastroesophageal junction cancer. Journal of cancer research and clinical oncology, 149(9), 5937-5950.
- Ricciuti, B., Lamberti, G., Andrini, E., Genova, C., De Giglio, A., Bianconi, V., ... & Pirro, M. (2021, February). Antibody–drug conjugates for lung cancer in the era of personalized oncology. In Seminars in cancer biology (Vol. 69, pp. 268-278). Academic Press.
- Hutchins, G., Southward, K., Handley, K., Magill, L., Beaumont, C., Stahlschmidt, J., ... & Quirke, P. (2011). Value of mismatch repair, KRAS, and BRAF mutations in predicting recurrence and benefits from chemotherapy in colorectal cancer. Journal of Clinical Oncology, 29(10), 1261-1270.
- Fan, H., Bai, L., & Bai, K. (2023). Longitudinal change of circulating tumor cell level and its relationship with immune checkpoint inhibitor-based treatment benefits in unresectable, metastatic colorectal cancer patients. Scandinavian Journal of clinical and laboratory inveStigation, 83(4), 227-233.
- Liu, S. Y. M., Huang, J., Deng, J. Y., Xu, C. R., Yan, H.H., Yang, M. Y., ... & Wu, Y. L. (2024). PD-L1 expression guidance on sintilimab versus pembrolizumab with or without platinum-doublet chemotherapy in untreated patients with advanced non-small cell lung cancer (CTONG1901): a phase 2, randomized, controlled trial. Science Bulletin, 69(4), 535-543.
- Andre, T., Amonkar, M., Norquist, J. M., Shiu, K. K., Kim,T. W., Jensen, B. V., ... & Le, D. T. (2021). Health-related quality of life in patients with microsatellite instability-high or mismatch repair deficient metastatic colorectal cancer treated with first-line pembrolizumab versus chemotherapy (KEYNOTE-177): an open-label, randomised, phase 3 trial. The Lancet Oncology, 22(5), 665-677.
- Ouyang, H., Xiao, B., Huang, Y., & Wang, Z. (2023). Baseline and early changes in the neutrophil–lymphocyte ratio (NLR) predict survival outcomes in advanced colorectal cancer patients treated with immunotherapy. International immunopharmacology, 123, 110703.
- Hedrick, C. C., & Malanchi, I. (2022). Neutrophils in cancer: heterogeneous and multifaceted. Nature Reviews Immunology, 22(3), 173-187.
- Shaul, M. E., & Fridlender, Z. G. (2019). Tumour-associated neutrophils in patients with cancer. Nature reviews Clinical oncology, 16(10), 601-620.
- Jaillon, S., Ponzetta, A., Di Mitri, D., Santoni, A., Bonecchi, R., & Mantovani, A. (2020). Neutrophil diversity and plasticity in tumour progression and therapy. Nature Reviews Cancer, 20(9), 485-503.
- Wu, Y., Ma, J., Yang, X., Nan, F., Zhang, T., Ji, S., ... &Gao, Q. (2024). Neutrophil profiling illuminates anti-tumor antigen-presenting potency. Cell, 187(6), 1422-1439.
- Mei, Z., Shi, L., Wang, B., Yang, J., Xiao, Z., Du, P., ... &Yang, W. (2017). Prognostic role of pretreatment blood neutrophil-to-lymphocyte ratio in advanced cancer survivors: a systematic review and meta-analysis of 66 cohort studies. Cancer treatment reviews, 58, 1-13.



