Review Article - (2024) Volume 2, Issue 2
Impact of Nanosilica on Anatomical, Morpho- Physiological and Yield Characters of Rice (Oryza Sativa l.) for Drought Tolerance – A Review
2Department of Crop Physiology, TNAU, Coimbatore, India
3Department of Nano Science and Technology, TNAU, Coimbatore, India
4Department of Agriculture, Vadupatti block, India
5Assistant Professor (SS&AC), KVK, Virudhunagar, India
6Professor (Crop Physiology), AC & RI, TNAU, India
7Department of Rice, TNAU, Coimbatore, India
Received Date: Aug 27, 2024 / Accepted Date: Sep 11, 2024 / Published Date: Sep 30, 2024
Copyright: ©©2024 N. Sritharan, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Surendar, K. K., Raja, R. K., Sritharan, N., Ravichandran, V., Kannan, M., et al. (2024). Impact of Nanosilica on Anatomical, Morpho- Physiological and Yield Characters of Rice (Oryza Sativa l.) For Drought Tolerance
Abstract
Rice (Oriza sativa L.) is considered as most staple food crops to world`s population. It is grown under irrigated ecosystem and some parts of India and Tamil Nadu cultivated under rain fed ecosystem also. Rice is also known as ‘Global grain’ because it is cultivated in more than 100 countries. Asian countries are the toppest in production and consumption of rice. Rice is one of the important staple food and most of the world population depends for its consumption and also income generation. Rice productions are severely affected by drought and its impacts yield. Agricultural scientists are mainly focus on drought stress, because climate change highly impact agricultural production through drought and it occurs frequently and unpredictable manner. Among the different growth stage of rice, reproductive stage is the sensitive stage to drought. Drought occurs during this reproductive period it will be directly reflected in yield traits.The major constraints in rice production is climate change (drought and flooding). Which generally affects the hydrological fluctuation levels and finally affecting agricultural mostly in developing countries. Drought is the major limiting factor for rice production. Therefore, enhancing rice yield with climate change and water limitation is the first approach for enhancing rice production to meet out the food demand in future, expected from the probable increasing in world population. Silicon is considered as non-essential nutrient because, silicon cannot fulfil the “essential criteria for a nutrient” Silicon deposition and cell wall modification change the turgor pressure of the guard cells, it results in a reduction in the stomatal conductance. In agriculture, silicon is considered as a quasi-essential element and beneficial effect on plant growth and development of the plant especially in abiotic stress conditions Nanotechnology has evolved into a rapidly growing field. Compared with normal material, nanomaterials have unique characters like high surface area, more reactive and a great surface volume ratio. Nanosilica had better results on mitigating the adverse effect of abiotic stress when compared with normal and bulk application of silicon fertilizers. Hence, with this background, the present review has been fremed to validate the aim of this study to understand and analyse the anatomical, morpho-physiological and yield changes under drought in rice varieties and also to study the amelioration response of drought stress by the foliar application of nanosilica.
Keywords
Nano Silica, Leaf Anatomy, Morpho-Physiology, Yield, Rice, Review
Introduction
Rice (Oryza sativa L.) is the most important food. Globally, four billion people consume it as stable food. Which was grown under various climatic conditions in the world. Economic part of the rice is grains, having a good source of nutrients and rich source of carbohydrate, proteins, fat and fibre. Based on the anatomical character rice classified as C3 tropical grass belonged to the Poaceae family. Rice brane are used for the production of oil and paddy straw is used for animal consumption and also used as a growth medium for mushroom productions. India ranks the second position in rice production in the world with an annual production of 118.8 million tonnes with a productivity of 2.72 tonnes per hectare. Worldwide demand for rice will reach up to 25 per cent, between the years 2010 to 2030 and undesirable environmental stresses also affects production. Among the stresses, drought impacts 50 per cent of rice cultivated lands in the world [1]. In India, drought is a major problem in rice production and Intergovernmental Panel for Climate Change (IPCC) has given the warning to India, climate change unpredictably leads to frequent drought occurrence. Severe drought are expected to have a 5 to 6 per cent impact on India’s Gross Domestic Product (GDP). In rice production, drought reduces grain yield due to adverse effects on plant vegetative, reproductive and maturity stages. It is a semi¬aquatic crop and it requires water throughout the life cycle and water requirement of rice is 1386 mm [2]. Drought is consider as the foremost confront in limiting the production and productivity of rice. In rice the drought drastically affects the morphological traits which leads to reduction in seed germination, plant height, number of productive tillers rhizosphere traits and leaf characters. It affects anatomical characters of reduction in leaf mesophyll thickness, stomatal length and breath. Trichome length and breadth were also affected due to drought. Drought stress also changes physiological and biochemical process and trait viz., photosynthetic efficiency, gas exchange measurement, stomatal diffusive resistance and conductance, stability of the membrane and chloroplast, water potential and changes in the molecular levels and in that way drought affects the yield in rice [3].
Rice crops are more sensitive to drought particularly in the critical stage (reproductive stage). The occurrence of 40% drought can cause yield losses up to 60 per cent. Carrijo et al. reported that soil dried below the 20Kpa resulted in 22.6 % yield loss in rice [4]. Venuprasad recorded that 64% of yield loss due to drought stress at the reproductive stage [5].
Silicon (Si) is a nontoxic element and second superabundant element available in the form of silicon dioxide and silicates in the earth crust. The late-stage of the 20th century and the early stage of the 21st century are considered as silicon age due to abundant usefulness in the various fields. In agriculture, silicon is considered as a quasi-essential element and beneficial effect on plant growth and development of the plant especially in abiotic stress conditions [6]. Silicon uptake by plants in the form of silicic acid (Si (OH) 4) [7]. Richmond reported that from a dry weight basis silicon deposition in plants ranges from 0.1 to 10% [8]. Based on the concentration of silicon accumulation, plants are classified as silicon accumulator, intermediate and non-accumulator [9]. Rice plants consider as silicon accumulators because shoot dry weight contains more than 10% of silicon, sometimes comparably higher than the essential elements [10]. In rice plants, silicon is mainly involved in the lodging resistance. Silicon is deposited in the leaf epidermis and form a double layer it helps to reduce the cuticular transpiration [11,12].
Silicon deposition and cell wall modification change the turgor pressure of the guard cells, it results in a reduction in the stomatal conductance [12]. Silicon can induce the potassium uptake in rice plants under drought [13]. Silicon can increase the nitrogen, potassium and phosphorus content in the paddy [14]. Potassium involved in the stomatal opening and closing and improve the grain quality and yield in rice plants due to application of silicon. Under drought conditions, silicon could increase the photochemical efficiency (Fv /Fm) and quantum efficiency [15].
Antioxidant enzymes are important for scavenging the reactive oxygen species under drought. One of the important antioxidant enzymes is catalase involves detoxifying the hydrogen peroxide (H2O2). Proline is produced in the cytoplasm and it acts as an imino acid and the accumulation of proline increase in the plants under drought conditions [16]. It is involved in protecting the plants from drought through stabilizing the protein structure, maintaining the enzyme activities and acting as an osmoprotectant and scavenger of reactive oxygen species in the cell by regulating the osmotic potential under drought [17,18]. The higher accumulation of proline as a best indicator of dehydration of leaf and sustainability under drought [19]. The proline, catalase, peroxidase and other antioxidantive enzymes are upregulated by the presence of silicon in rice under drought. Silicon influences the length of the trichrome characters and leaf surface structure of cucumber plants. Silicon can alleviate the drought by maintaining relative water content and it represents the higher water status of the tissues [20]. Moreover, a tentative role of silicon in the osmotic adjustment is to preserve the water content, proline and other osmolytes in the cell [21].
Nanotechnology has evolved into a rapidly growing field. Compared with normal material, nanomaterials have unique characters like high surface area, more reactive and a great surface volume ratio. Silicon dioxide is mostly presented in sand and quartz with a crystalline tetrahedral structure. Nanosilica is amorphous in nature and vary in size from 5 to 100 nm [22]. Nano fertilizer had high nutrient use efficiency [23]. Seed germination increased by exogenous application of nanosilica in plants [24]. Nanosilica had better results on mitigating the adverse effect of abiotic stress when compared with normal and bulk application of silicon fertilizers. The strengthening of organs involved in the transpiration process by the application of nanosilica astonishingly decreases the transpiration rate [25].
Drought
Drought is a prolonged period of dryness in the climatic pattern and it is classified into five different types viz., Meteorological drought, Hydrological drought, Agricultural drought, Ecological drought and Socio-economical drought. Agricultural scientists mainly focus on agricultural drought because it is directly affects the crop production. Agricultural drought defined as crop production affected by insufficient soil moisture at a particular crop growth period or stage. In India, 42 % of area considered as drought pretentions areas (Drought Early Warning System (DEWS, 2019).
Impacts of Drought on Plants
Drought is caused by lack (or) shortage of rainfall compared to long term normal rainfall pattern. Under drought conditions, plants are changes the morpho-physiological and biochemical characters [26]. Seed germination percentage, length and dry mater production of the root and shoot decorously reduced in drought conditions [27,28]. The impact of drought varies in different rice growth stage. Drought affects the plants differently at vegetative stage reduce plant height, but drought at reproductive stage caused more than 60 % of yield losses. Reduction of plant height due to reduction of internode elongation mainly due to moisture deficit condition in the root zone [29]. Moisture stress reduces the leaf size for minimizing the transpiration losses. Closing of stomata is the primary response for drought and the closing of stomata restrict the entry of CO2, which was ultimately reduce the photosynthetic rate [30].
Once root feels drought stress its immediately sends the signals to shoots for maintaining the water potential of the tissues and stomatal regulation [31]. Abscisic acid (ABA) an important stress hormone is produced in roots and then transported to shoots and act as a hormonal signal to regulate the stomatal activity [32]. Chlorophyll embedded with protein complex namely D 1 and D2 protein, these are highly sensitive to drought stress. Chlorophyll content and chloroplast stability are affected during drought [33,34]. Reddy et al. reported that drought stress induce the production of reactive oxygen species in the tissues[35].
The enzyme RuBisCO is made up of large and small subunits and Vu et al. observed that drought decreases the small subunit synthesis [36]. The level of sucrose and current photosynthetic rate determines photoassimilates transport from source to sinks in rice [37]. Drought limiting the carbohydrate assimilation into the sink organs. Invertase is the catalytic or hydrolytic enzyme of sucrose and its play as an important role in pollen abortion under drought. Kim et al. observed that invertase activity increased during drought and it enhances the carbohydrate limitation [38]. Improper phloem uploading due to the invertase activity leads seed abortion and unfilled grains.
Rice
Rice is an important cereal food crop grown all around the world with high genetic variability. Based on origin rice species are classified into two different types namely african rice (Oryza glaberrima steud.) and asian rice or common rice (Oryza sativa L.). Globally, 4 billion people depend on rice production and India poses second place in world rice production [39]. Rice production are severely affected by drought and its impacts yield. Agricultural scientists are mainly focus on drought stress, because climate change highly impact agricultural production through drought and it occurs frequently and unpredictable manner. Among the different growth stage of rice, reproductive stage is the sensitive stage to drought. Drought occurs during this reproductive period it will be directly reflected in yield traits.
Drought Affect the Critical Stage of Rice
In the moisture sensitive or critical stage, crop requires more water and this stage is more sensitive to the water stress. Drought occurs during the critical stage would reduce the yield progressively. Rice is semiaquatic crop and it require water for entire life.
According to O’ toole, 12 days before and 10 days after flowering is considered as a more sensitive period to the drought stress [40]. Variety have 110 days duration, this sensitive period generally occur between 68 days to 90 days. Critical periods of rice are active tillering stage and flowering or reproductive stage. Biglary et al. reported that drought at flowering stage causes maximum yield loss of 24% recorded [41]. Servestani et al. imposed the drought at three different stages (vegetative, flowering and grain filling) and observed the yield reduction of 21, 50, and 21 percent respectively [42]. It is indicated that flowering stages is the most critical or sensitive period of drought.
Silicon
Silicon is crystalline solid element present in the earth’s crust. It is second most abundant beneficial element (28.8%) [43]. Silicon rarely presented in purest form. The scientist Jöns Jakob Berzelius was a pioneer in silicon characterisation and purification. Silicon had more affinity to oxygen and forms silicate group. In periodic table Si placed in 14th group with atomic number of 14. Silica is considered as beneficial element and trace amount only available to the plant [44]. Based on the fraction, soil silica phase classified in three different phases viz., liquid, absorbed and solid phase. Among that liquid phase is biologically important because poly and mono silicic acid come under this phases. In rice plants, silicon is essential element for attaining the optimal growth and development. It also develop the pest and disease resistance and now a days many of the scientist are working or concentrating silicon fertilizer for developing drought tolerant crop (or) mitigating the crop to drought [45].
Silicon as a Beneficial Element
Silicon is considered as non-essential nutrient because, silicon cannot fulfil the “essential criteria for a nutrient” given by Arnon and Stout [46]. So, until 1994 silicon considered as non-essential element. After that, Epstein given the astonishing review on silicon in ‘The anomaly of silicon in plant biology’ change the prospects of silicon in the agricultural field [47]. Several studies reported that beneficial effects of silicon. ‘International Plant Nutrition Institute’ promote the silicon from non-essential to beneficial element (IPNI, 2015). Because small number of plant species requires silicon particularly in rice plant. McLarnon et al. reported that, silicon requirements increased during stress and silicon actively absorbed in some grass species [48].
Silicon Uptake by Plants
Plants absorbs the silicon in the form of monosilicic acid from the soil and availability of the silicon range varies from 0.1 to 0.6 mM [7,47]. In rice, lateral roots are play major role in mono-silicic acid absorption via xylem [49]. Three different mechanism are followed for absorption of silicon i.e. active absorption, passive absorption and rejective. In rice, special type of transporters involved in transport of silicon. Low silicon 1 (Lsi 1) and Lsi 2 are silicon transporter present in the rice, the transporter of Lsi 1 are involved in uptake of silicon and Lsi 2 is involved in efflux of silicon.
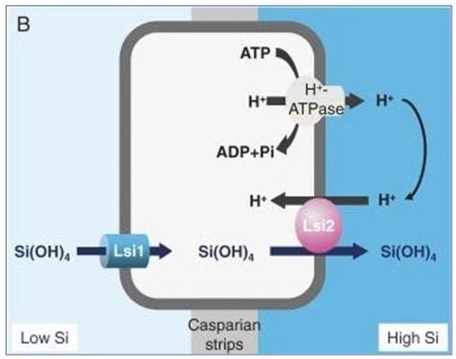
Figure 1. Transport of silicon via Lsi1 and Lsi2 transporters
Silicon Accumulation
Silicon deposition in the plants range between 0.1 to 10 % [8]. Plants are classified into three different types based on silicon accumulation in the plants. i.e. accumulator, intermediate and non-accumulators. Accumulators contains more than 1.5 % of silicon in the dry weight of plant. Intermediate contains 1.5-0.5% of silicon in dry weight basis. Non-accumulators having less than 0.5 % of silicon in the plant dry weight. Rice considered as a silicon accumulators it gives logging resistance to the plants through silicon bodies production in the epidermal cells. Same research reports also agreement with Savant reported that the silicon deposit in rice had more than 10 percent in the shoots [10].
Silicon Deposition on Leaf Epidermis in Rice
Silicon is absorbed by roots (mature region and root tips) and transported via xylem to shoots [50]. After translocation to the shoots silicon is deposited in different parts of the rice plant (hulls, blades and leaf sheath) particularly in the leaf. Silicon deposited beneath the cuticle layer (2.4µm) and from silicon cuticle double layers. According to Savant et al. the silicon involved in the function of increases the lodging resistance, reduce the water loss from cuticle and guard cell and also preventing the penetration of fungal hypha or hyphae [10].
In past two decades, numerous research reports shows that silicon alleviate the both biotic and abiotic stresses. Importantly abiotic stress like drought, salinity, UV radiation, high and low temperature, and heavy metal stresses. Moisture deficit conditions affects all developmental process (mentioned earlier). Application of silicon could improve the plant height, leaf area, plant dry mater production, relative water content and minimize the chlorophyll degradation by the way silicon ultimately increases the crop yield under drought stress [10].
Effect of Silicon on Plant Growth and Development
Plant Height
Plant height of the rice is based on length of the internode. Elongation of the internode is based on the activity of cells present in the internode. Internode cell expansion mainly depend on cell turgor pressure and it induce the loosening of the cell wall and elongation. Plant height are highly affected by drought. Okuda and Takahashi and Ma et al. reported that, applied silicon fertilizer on rice at different growth stages improved the plant height at all stages [51,52]. Application of sodium silicate at the rate of 7.14 mmol increases the plant height under drought in wheat [53]. In hydroponics100 ppm of silicon increases the plant height and logging resistance in rice. Under drought condition, basal application of 600 kg ha-1 of silicon fertilizer could improve the plant height, flag leaf length and productivity in rice. Ibrahim et al. opined that, application of 8.6 mg of potassium silicate at reproductive stage increases the plant height about 15.3 percent over the without potassium silicate treated plant in rice [54]. Pati et al. who found that, application of silicon through diatomaceous earth (silicon dioxide-SiO2- 63.70%) at the rate of 600 kg ha-1 might increases the plant height [14]. Ullah et al. reported that split dose of 300 kg Si ha-1at basal (25%) and panicle initiation (75%) stage had significant increases of plant height in rice under drought [55].
Leaf Area and Leaf Area Index Leaf area and leaf area index plays important role in photosynthesis, respiration and yield. Leaf surface acts in light trapping, production of photoassimilates and also act as a source organ during the vegetative stage positively correlated with productivity of the plants. Application of silicon improve the leaf area in rice [56]. Kang found that silicon increases the leaf area and leaf area index in rice [57]. Ahmed et al. suggested that, application of 200 mlL-1of potassium silicate improve the leaf area and leaf area index in sorghum plant under drought condition [58]. Seebold et al. revealed that applied calcium silicate fertilizer in rice crop, which improve the leaf surface area [56]. Silicon application (100 mg L−1 SiO2) improve the leaf surface area in cucumber plants. Application silicon in wheat under drought showed better leaf area index and leaf area [11].
Specific Leaf Weight
Specific leaf weight indirectly represent the thickness and density of the leaf [59]. It is directly influenced by environmental stimuli like intensity of the light and water status of the plant [60]. Light intensity and drought stress highly influence the specific leaf weight. Ahmed et al. reported that application of silicon improve the specify leaf weight in sorghum under drought condition [58]. Ibrahim et al. opined that, application of liquid potassium silicate on different growth period could improve the specific leaf weight in rice [54]. Under abiotic stress condition, foliar application of silicon increases the leaf thickness in pumpkin.
Leaf Angle
Silicon is an important element for inducing erectness of the leaves in rice and it also give mechanical strength to plant, which gives the logging resistance and good light interception [47,52,61]. Flag leaf photosynthesis are reported for maximum contribution for delivering photoasimilate to growing grains. If any abiotic stresses that affects flag leaf photosynthesis has a greater impact on rice grain yield and productivity [62]. Exogenous application of silicon increases the erectness of the leaf in rice wheat, kentucky bluegrass [11,52,63,64,65].
Effect of Silicon on Physiological and Biochemical Parameters
Gas Exchange Parameter
Yield of the crop is strongly correlated with the gas exchange parameter like photosynthetic rate, stomatal conductance and transpiration rate. Under drought condition, plants able to close their stomata for reducing the transpiration loss, which leads to reduce the exchange of CO2 and O2. RuBisCO enzyme activity is highly sensitive to water deficit conditions, which leads to decreasing of photosynthetic rate and stomatal conductance. Application of silicon improve the gas exchange parameter like photosynthetic rate, stomatal conductance and transpiration rate in rice [66]. According to Gong et al. observed that application of silicon increases the leaf net photosynthetic rate, stomatal conductance and transpiration rate under drought condition in rice [67]. Chen et al. also reported that, application of 1.5mM of silicon significantly improve the photosynthetic rate and stomatal conductance in wheat under drought condition [66].
Application of silicon (5g/pot) increases the transpiration rate in sorghum under drought condition [64]. But Matoh et al. and Yoshida et al reported that silicon involved in the formation of cuticle double layer, which reduce the transpiration rate in rice [68]. Maghsoudi et al. suggested that application of 6 mM of silicon increases the transpiration, stomatal conductance and photosynthetic rate in wheat [69]. Recent research reported that, amorphous silicon (SiO2) had maximum water holding capacity and make it to available to the plants. Application of 15 g silicon dioxide per kg of soil increases the transpiration rate, photosynthetic rate and stomatal conductance in rice under drought conditions [70]. Nwugo et al. stated that, silicon improve the transpiration, photosynthetic rate and stomatal conductance in rice under drought and heavy metal stress conditions [71].
Chlorophyll Fluorescence Parameters
Assessing the efficiency of photosystem II considered as an important for improving the crop productivity in physiological manner. Decreasing of Fv/Fm ratio in the plants indicates plant under stress. It is directly correlated with photosynthetic rate and yield characters. Photosystem is an essential tool for light absorption. Under drought condition application of silicon improve the efficiency of photosystem efficiency in plants.
Mahdieh et al. reported that application of 3mM of silicon increases the Fv/Fm ratio in rice under abiotic stress condition [72]. Under drought condition, rice plants show high response silicon application, 1.5mM of silicon on rice plants had significant increases of PS II photosynthetic efficiency (Fv/Fm ratio) [66]. Application of 0.5 mM of sodium metasilicate nonahydrate increases the PS II efficiency in rice under polyethylene glycol (PEG) drought conditions [73]. Nwugo et al. reported that silicon in the form of sodium silicate improve the photosystem efficiency in rice under low level cadmium stress and abiotic stress condition [74].
Chlorophyll Content and Chlorophyll Index (SPAD)
Photosynthetic pigments are important for in photosynthesis during the photochemical reaction and among different types of chlorophyll molecules chlorophyll a (chl a) and chlorophyll b (chl b) molecules are important for photosynthesis in plants [75]. Under drought, high production of reactive oxygen species (ROS) damage the thylakoid membrane and induce the leaf senescence by increased activity of chlorophyllase enzyme activities decrease the chlorophyll content in plants [76]. Swapna and Shylaraj reported that drought reduces the chlorophyll a and chlorophyll b content plants under drought condition [77]. Application of silicon improve the chlorophyll content in rice [66,73]. Biglary et al. reported that silicon improve the chlorophyll content in plants under oxidative stress [41]. Application of silicon in the form of sodium silicate helps to maintain the chlorophyll content under drought condition stated that 1 mM of silica increases the chlorophyll content in rice plant under mild drought stress.
Soluble Protein Content
Among the total leaf soluble proteins nearly 50 percent of soluble protein represent the RuBisCO. Water deficit condition make the RuBisCO enzyme very unstable because it prevent the generation of smaller sub units in rice [36]. Application of silicon shows significant increase of soluble protein content. Ding et al. reported that application of silicon at the rate of 1.0 mmol/L Si increases the soluble protein content in wheat [78]. Application of silicon on tomato increases the leaf soluble protein content in poly ethylene glycol (PEG) induced drought in tomato plants.
Gong et al. reported that soil application of silicon fertilizer improve the soluble protein content in wheat under drought [53]. Soluble protein content under abiotic stress condition increased by application of 3mM silicon in maize plants [79]. Al-aghabary et al. reported that silicon fertilizer improve the soluble protein contact in tomato under drought stress condition [80].
Effect of Silicon in Antioxidant Defense System
Drought stress dramatically increases the reactive oxygen species in almost all plant parts (i.e., chloroplast, mitochondria and apoplast etc.). It reduces the membrane integrity of the cell organelles. Plant has antioxidant defense mechanism which production both enzymatic and non-enzymatic antioxidants like proline, catalase (CAT), superoxide dismutase (SOD), ascorbate peroxidase (APX), ascorbate and glutathione etc.
Application of silicon in wheat plants under drought conditions could improve the plant growth and development through preventing the reactive oxygen species (ROS) by inducing the synthesis antioxidant enzymes like catalase (CAT), superoxide dismutase (SOD), ascorbate peroxidase (APX) [53]. Shi et al. also suggested that application of silicon at the rate of 0.5 mM improve the catalase and superoxide dismutase enzymes in tomato under PEG mediated drought stress [81,82]. However, Kim et al. observed that application silicon under salinity stress condition improve the catalase and peroxidase and superoxide dismutase content in rice [83].
Proline Content
Proline is an important component in protein synthesis like polyamines, glutamate and arginine. Proline synthesis is generally induced under stress condition. It acts as imino acid. Application of silicon improve the proline content in plants. Shen et al. reported that compared with stressed plants and plants with silicon applied plants, had a significant reduction in proline content [84]. Kumar et al. reported that under drought proline content significantly increased over the control plant [85]. Gunes et al. found that proline content significantly increased under drought condition then application of silicon shows decreased proline content in sunflower plants [86]. Mauad et al. suggested that silicon application decrease the proline content in upland rice under drought conditions [87].
Relative Water Content
Relative water content is another way of assessing the plant water content and cell metabolic activity, which was a standard method for asses drought tolerance [88]. Under drought condition relative water content significantly decreased. Maintaining the relative water content is important for noble growth and development. Zegaoui et al. reported that relative water content of the leaf reduced under drought condition [89]. Maghsoudi et al. found that silicon maintains higher relative water content and water status in leaf under drought in rice crop [69]. Mali et al. reported that silicon maintains the relative water content in wheat [90]. Biglary et al. suggested that silicon involve in the maintaining of relative water content and water status in rice leaf [41].
Chlorophyll Stability Index and Membrane Stability Index
Chlorophyll stability index and membrane stability index are important indicators for stress tolerance particularly drought tolerance. Maghsoudi et al. reported that cell membrane stability index and chlorophyll stability index increased by application of silicon in rice under drought condition [69]. Kafi et al. suggested that application of sodium silicate-nanoparticles (NaSiO3) increases the membrane stability in potato and sorghum plant under salinity stress [91]. Ashraf et al. observed in sunflower plant, under abiotic stress application of silicon improve the membrane stability and silicon involved in the maintenance of membrane integrity. Gong et al. opined that, application of silicon increases the chlorophyll stability index and minimise the electrolyte leakage [53]. Younis et al. found that application silicon nanoparticle improves the stability of chloroplast and membrane stability in wheat under drought and heat stress [92].
Yield and Yield Component
Yield components consist of number of productive tillers per m2, spikelets per panicle, spikelets filling percentage, and 1000 g weight [93]. Rice crop is highly water sensitive crop. Drought stress causes 40 to 60 percent yield loss in rice under reproductive stage. Chaiwong et al. reported that application of 100 mg SiO2 L-1 increases the grain yield about 23 to 44 percent in rice in hydroponic system [94]. Ming et al. suggested that application of silicon fertilizer at the rate of 600kg ha-1increases the grain yield and silicon positively correlated with yield [95]. Chu et al. found that application of silicon at tillering stage increases the yield characters of rice [96]. Cuong et al. also reported that application of 400 kg/hm2 along with recommended dose of fertilizers improve the yield characters like number of productive tillers, 1000 grain weight, seed setting percentage and straw yield in rice [97]. Application of 1.5mM potassium silicate could improved the grain yield, harvest index and grain quality parameter like grain length, breadth, carbohydrate, soluble protein content, secondary metabolic content like phenolic, flavonoid content are increased [98]. Bukhari et al. also suggested that foliar application of silicon at anthessis stage and fertigation method of application at tillering stage resulted better yield in rice under drought [99]. Under water deficit condition, application of silicon on rice showed 12 percent grain yield increment [85]. Nolla et al. reported that silicon alleviated that drought effect and increases the grain yield [100].
Leaf Surface Characters
Trichomes are unicellular unicellular (or) multi cellular branched hair like structure arises from the single (or) multi cell of protodermal cells [101]. Trichome contains essential oils, secondary metabolites and act as salt gland under salinity stress. Under drought condition trichomes are very use full trait for resistance against water deficient. Kennedy Trichomes involved in the production of secondary metabolites like terpenes for drought tolerance [102]. Guntwer et al. reported that density and length of the trichome are increased under drought conditions[103]. Rostkowska et al., who opined that, silicon deposited in the inside the trichomes and it helps to improve the length of the trichomes [104]. Takeda et al. reported that silicon deposited in the trichome increases the infra¬red light use efficiency in plant [105]. Wang et al. reported that size of the stomata reduced under drought [73]. Drought increases the density of the stomata (stomatal frequency) are increases, while stomata size was reduced which leads rapid stomatal conductance for increasing the photosynthetic rate Vandegeer et al. opined that, application of silicon helps to maintain the water content of the guard cell and improve the stomatal conductance to increases the photosynthetic rate in tall fescue [106]. Application of silicon deposited in the silico bodies, which increases the size [107]. Isa et al. reported that silicon may be deposited in the motor cells of leaf, it helps to maintain the water status of the leaf [108-112].
References
-
-
- Vibhute, S. D., Sarangi, A., & Singh, D. K. (2016). Development of crop water demand based water delivery schedule for a canal command. Journal of Agricultural Engineering, 53(2), 12-23.
- Pandey, V., & Shukla, A. (2015). Acclimation and tolerance strategies of rice under drought stress. Rice science, 22(4), 147-161.
- Carrijo, D. R., Lundy, M. E., & Linquist, B. A. (2017). Rice yields and water use under alternate wetting and drying irrigation: A meta-analysis. Field Crops Research, 203, 173-180.
- Venuprasad, R., Lafitte, H. R., & Atlin, G. N. (2007). Response to direct selection for grain yield under drought stress in rice. Crop Science, 47(1), 285-293.
- Epstein, E, and A Bloom. 2005. "Mineral nutrition of plants: principles and perspectives. Sinauer Associates." Inc. Sunderland, Mass.
- Ma, J. F., Tamai, K., Yamaji, N., Mitani, N., Konishi, S., Kat-suhara, M., ... & Yano, M. (2006). A silicon transporter in rice. Nature, 440(7084), 688-691.
- Richmond, K. E., & Sussman, M. (2003). Got silicon? The non-essential beneficial plant nutrient. Current opinion in plant biology, 6(3), 268-272.
- Mitani, N., & Ma, J. F. (2005). Uptake system of silicon in different plant species. Journal of experimental botany, 56(414), 1255-1261.
- Savant, N. K., Datnoff, L. E., & Snyder, G. H. (1997). Depletion of plantâ?available silicon in soils: A possible cause of declining rice yields. Communications in Soil Science and Plant Analysis, 28(13-14), 1245-1252.
- Gong, H. J., Chen, K. M., Chen, G. C., Wang, S. M., & Zhang,C. L. (2003). Effects of silicon on growth of wheat under drought. Journal of plant nutrition, 26(5), 1055-1063.
-
-
-
- Zhu, Y., & Gong, H. (2014). Beneficial effects of silicon on salt and drought tolerance in plants. Agronomy for sustainable development, 34, 455-472.
- Xu, C. X., Ma, Y. P., & Liu, Y. L. (2015). Effects of silicon (Si) on growth, quality and ionic homeostasis of aloe under salt stress. South African Journal of Botany, 98, 26-36.
- Pati, S., Pal, B., Badole, S., Hazra, G. C., & Mandal, B. (2016). Effect of silicon fertilization on growth, yield, and nutrient uptake of rice. Communications in soil science and plant analysis, 47(3), 284-290.
- Zhang, C., Xu, Y., Liu, J., Li, J., Xiang, J., Li, H., ... & Mirosh-nichenko, A. E. (2018). Lighting up silicon nanoparticles with Mie resonances. Nature Communications, 9(1), 2964.
- Szabados, L., & Savouré, A. (2010). Proline: a multifunction-al amino acid. Trends in plant science, 15(2), 89-97.
- Verbruggen, N., & Hermans, C. (2008). Proline accumulationin plants: a review. Amino acids, 35, 753-759.
- Ashraf, M. F. M. R., & Foolad, M. R. (2007). Roles of gly-cine betaine and proline in improving plant abiotic stress resistance. Environmental and experimental botany, 59(2), 206-216.
- Hanson, A. D., Nelsen, C. E., & Everson, E. H. (1977). Evaluation of Free Proline Accumulation as an Index of Drought Resistance Using Two Contrasting Barley Cultivars 1. Crop Science, 17(5), 720-726.
- Slayter, R. O. (1967). Plant-water relationships.
- Ahmed, M., Qadeer, U., Ahmed, Z. I., & Hassan, F. U. (2016). Improvement of wheat (Triticum aestivum) drought tolerance by seed priming with silicon. Archives of Agronomy and Soil Science, 62(3), 299-315.
- Imai, S., Yoshioka, Y., Morishita, Y., Yoshida, T., Uji, M., Na-gano, K., ... & Tsutsumi, Y. (2014). Size and surface modification of amorphous silica particles determine their effects on the activity of human CYP3A4 in vitro. Nanoscale Research Letters, 9, 1-7.
- Cui, H. X., Sun, C. J., Liu, Q., Jiang, J., & Gu, W. (2010,June). Applications of nanotechnology in agrochemical formulation, perspectives, challenges and strategies. In international conference on Nanoagri, Sao pedro, Brazil (pp. 28-33).
- Nair, R., Poulose, A. C., Nagaoka, Y., Yoshida, Y., Maeka-wa, T., & Kumar, D. S. (2011). Uptake of FITC labeled silica nanoparticles and quantum dots by rice seedlings: effects on seed germination and their potential as biolabels for plants. Journal of fluorescence, 21, 2057-2068.
- Aqaei, P., Weisany, W., Diyanat, M., Razmi, J., & Struik, P.C. (2020). Response of maize (Zea mays L.) to potassium nano-silica application under drought stress. Journal of Plant Nutrition, 43(9), 1205-1216.
- Zlatev, J., & McCune, L. (2014). Towards an integrated model of semiotic development. Cognitive development: Theories, stages, processes and challenges, 59-76.
- Kaya, M. D., Okçu, G., Atak, M., Çıkılı, Y., & Kolsarıcı, Ö. (2006). Seed treatments to overcome salt and drought stress during germination in sunflower (Helianthus annuus L.). European journal of agronomy, 24(4), 291-295.
- Zeid, I. M., & Shedeed, Z. A. (2006). Response of alfalfa to putrescine treatment under drought stress. Biologia plantarum, 50, 635-640.
- Bhatt, R. M., & Rao, N. S. (2005). Influence of pod load on response of okra to water stress. Indian journal of plant physiology, 10(1), 54.
- Lawlor, D. W., & Tezara, W. (2009). Causes of decreased photosynthetic rate and metabolic capacity in water-deficient leaf cells: a critical evaluation of mechanisms and integration of processes. Annals of botany, 103(4), 561-579.
- Tardieu, F. (2012). Any trait or trait-related allele can confer drought tolerance: just design the right drought scenario. Journal of experimental botany, 63(1), 25-31.
- Dodd, I. C. (2005). Root-to-shoot signalling: assessing the roles of ‘up’in the up and down world of long-distance signalling in planta. Plant and soil, 274, 251-270.
- Anjum, F., Yaseen, M., Rasool, E., Wahid, A., & Anjum, S. (2003). Water stress in barley (Hordeum vulgare L.) I. Effect on morpohological characters. seeds, 105, 266-271.
- Fu, J., & Huang, B. (2001). Involvement of antioxidants and lipid peroxidation in the adaptation of two cool-season grasses to localized drought stress. Environmental and Experimental Botany, 45(2), 105-114.
- Reddy, A. R., Chaitanya, K. V., & Vivekanandan, M. (2004). Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. Journal of plant physiology, 161(11), 1189-1202.
- Vu, J. C., Gesch, R. W., Allen Jr, L. H., Boote, K. J., & Bowes,G. (1999). C02 enrichment delays a rapid, drought-induced decrease in Rubisco small subunit transcript abundance. Journal of Plant Physiology, 155(1), 139-142.
- Komor, E. (2000). Source physiology and assimilate transport: the interaction of sucrose metabolism, starch storage and phloem export in source leaves and the effects on sugar status in phloem. Functional Plant Biology, 27(6), 497-505.
- Kim, J. Y., Mahé, A., Brangeon, J., & Prioul, J. L. (2000). A maize vacuolar invertase, IVR 2, is induced by water stress. Organ/tissue specificity and diurnal modulation of expression. Plant physiology, 124(1), 71-84.
- IRRI, 2019, Annual report. https:// www. goo-gle.com/url?sa=t&rct=j&q=&esrc=s&source= web&c-d=&ved=2ahUKEwjX24Rx870AhVJTGwGHZf8D-McQFnoECBcQAQ&url=http%3A%2F%2Fbooks.irri. org%2FAR2019_content.pdf&usg=AOvVaw313rsI47PUk-lKob8yKC_1p
- O’toole, J. C. Adaptation of rice environments. Drought Re-sistance in Crops with Emphasis on Rice, 195-213.
- Biglary, F., Haddad, R., Hosseini, R., & Sotudehniya, A. (2011). Roles of silicon in improving oxidative stress resistance by increase of chlorophyll content and relative water content of rice (Oryza sativa L.) genotypes. on Silicon in Agriculture, 9.
- Sarvestani, Z. T., Pirdashti, H., Sanavy, S. A., & Balouchi, H. (2008). Study of water stress effects in different growth stages on yield and yield components of different rice (Oryza sativa L.) cultivars. Pakistan journal of biological sciences: PJBS, 11(10), 1303-1309.
- McKeague, J. A., & Cline, M. G. (1963). Silica in soil solutions: I. The form and concentration of dissolved silica in aqueous extracts of some soils. Canadian Journal of Soil Science, 43(1), 70-82.
- Struyf, E., Mörth, C. M., Humborg, C., & Conley, D. J. (2010). An enormous amorphous silica stock in boreal wetlands. Journal of Geophysical Research: Biogeosciences, 115(G4).
- Tamai, K., & Ma, J. F. (2008). Reexamination of silicon effects on rice growth and production under field conditions using a low silicon mutant. Plant and Soil, 307, 21-27.
- Arnon, DI, and P Stout. 1939. "The essentiality of certain elements in minute quantity for plants with special reference to copper." Plant Physiology 14 (2):371.
- Epstein, E. (1994). The anomaly of silicon in plant biology. Proceedings of the National Academy of Sciences, 91(1), 11-17.
- McLarnon, E., McQueen-Mason, S., Lenk, I., & Hartley, S. E. (2017). Evidence for active uptake and deposition of Si-based defenses in tall fescue. Frontiers in Plant Science, 8, 1199.
- Ma, J. F., Miyake, Y., & Takahashi, E. (2001). Silicon as a beneficial element for crop plants. Studies in plant Science, 8, 17-39.
- Ma, J. F., & Takahashi, E. (2002). Soil, fertilizer, and plant silicon research in Japan. Elsevier.
- Okuda, A., & Takahashi, E. (1961). Studies on the physiological role of silicon in crop plants (Part 1); Discussion on the silicon deficient culture method. J. Sci. Soil Manure, Japan, 32, 475-480.
- Ma, J., Nishimura, K., & Takahashi, E. (1989). Effect of silicon on the growth of rice plant at different growth stages. Soil Science and Plant Nutrition, 35(3), 347-356.
- Gong, H., Zhu, X., Chen, K., Wang, S., & Zhang, C. (2005). Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant science, 169(2), 313-321.
- Ibrahim, M. A., Merwad, A. R. M., & Elnaka, E. A. (2018). Rice (Oryza Sativa L.) tolerance to drought can be improved by silicon application. Communications in soil science and plant analysis, 49(8), 945-957.
- Ullah, H., Luc, P. D., Gautam, A., & Datta, A. (2018). Growth, yield and silicon uptake of rice (Oryza sativa) as influenced by dose and timing of silicon application under water-deficit stress. Archives of Agronomy and Soil Science, 64(3), 318-330.
- Seebold, K. W., Kucharek, T. A., Datnoff, L. A., Correa-Victo-ria, F. J., & Marchetti, M. A. (2001). The influence of silicon on components of resistance to blast in susceptible, partially resistant, and resistant cultivars of rice. Phytopathology, 91(1), 63-69.
- Kang, Y. S. (1985). The influences of silicon on growth of rice plants.
- Ahmed, M., & Khurshid, Y. (2011). Does silicon and irrigation have impact on drought tolerance mechanism of sorghum?. Agricultural water management, 98(12), 1808-1812.
- Witkowski, E. T. F., & Lamont, B. B. (1991). Leaf specific mass confounds leaf density and thickness. Oecologia, 88, 486-493.
- Pearce, R. B., Carlson, G. E., Barnes, D. K., Hart, R. H., & Hanson, C. H. (1969). Specific leaf weight and photosynthesis in alfalfa 1. Crop Science, 9(4), 423-426.
- Lu, J. (1997). Effect of silicon fertilizer on wheat yield. Soil and fertilizer, 5, 42-43.
- Huang, G., Hu, H., Van de Meene, A., Zhang, J., Dong, L., Zheng, S., ... & Zhang, D. (2021). AUXIN RESPONSE FAC-TORS 6 and 17 control the flag leaf angle in rice by regulating secondary cell wall biosynthesis of lamina joints. The Plant Cell, 33(9), 3120-3133.
- Sattar, A., Cheema, M. A., Abbas, T., Sher, A., Ijaz, M., Wahid, M. A., & Hussain, M. (2017). Physiological response of late sown wheat to exogenous application of silicon. Cereal Research Communications, 45(2), 202-213.
- Hattori, T., Inanaga, S., Araki, H., An, P., Morita, S., Lux-ová, M., & Lux, A. (2005). Application of silicon enhanced drought tolerance in Sorghum bicolor. Physiologia Plantarum, 123(4), 459-466.
- Saud, S., Li, X., Chen, Y., Zhang, L., Fahad, S., Hussain, S.,... & Chen, Y. (2014). Silicon application increases drought tolerance of Kentucky bluegrass by improving plant water relations and morphophysiological functions. The Scientific World Journal, 2014(1), 368694.
- Chen, W., Yao, X., Cai, K., & Chen, J. (2011). Silicon alleviates drought stress of rice plants by improving plant water status, photosynthesis and mineral nutrient absorption. Biological trace element research, 142, 67-76.
- Gong, H., & Chen, K. (2012). The regulatory role of silicon on water relations, photosynthetic gas exchange, and carbox-ylation activities of wheat leaves in field drought conditions. Acta Physiologiae Plantarum, 34, 1589-1594.
- Yoshida, S., Navasero, S. A., & Ramirez, E. A. (1969). Effects of silica and nitrogen supply on some leaf characters of the rice plant. Plant and soil, 31, 48-56.
- Maghsoudi, K., Emam, Y., & Ashraf, M. (2016). Foliar application of silicon at different growth stages alters growth and yield of selected wheat cultivars. Journal of Plant Nutrition, 39(8), 1194-1203.
- Kuhla, J., Pausch, J., & Schaller, J. (2021). Effect on soil water availability, rather than silicon uptake by plants, explains the beneficial effect of silicon on rice during drought. Plant, Cell & Environment, 44(10), 3336-3346.
- Nwugo, C. C., & Huerta, A. J. (2008). Effects of silicon nutrition on cadmium uptake, growth and photosynthesis of rice plants exposed to low-level cadmium. Plant and soil, 311, 73-86.
- Mahdieh, M., Habibollahi, N., Amirjani, M. R., Abnosi, M. H., & Ghorbanpour, M. (2015). Exogenous silicon nutrition ameliorates salt-induced stress by improving growth and efficiency of PSII in Oryza sativa L. cultivars. Journal of soil science and plant nutrition, 15(4), 1050-1060.
- Wang, Y., Zhang, B., Jiang, D., & Chen, G. (2019). Silicon improves photosynthetic performance by optimizing thylakoid membrane protein components in rice under drought stress. Environmental and Experimental Botany, 158, 117-124.
- Nwugo, C. C., & Huerta, A. J. (2011). The effect of silicon on the leaf proteome of rice (Oryza sativa L.) plants under cadmium-stress. Journal of proteome research, 10(2), 518-528.
- Rabinowitch, E. I. (1965). The role of chlorophyll in photosynthesis. Scientific American, 213(1), 74-83.
- Farooq, M., Wahid, A., Kobayashi, N. S. M. A., Fujita, D. B. S. M. A., & Basra, S. M. (2009). Plant drought stress: effects, mechanisms and management. Sustainable agriculture, 153-188.
- Swapna, S., & Shylaraj, K. S. (2017). Screening for osmotic stress responses in rice varieties under drought condition. Rice science, 24(5), 253-263.
- DING, Y. F., LIANG, Y. C., ZHU, J., & Zhao-jun, L. (2007).Effects of silicon on plant growth, photosynthetic parameters and soluble sugar content in leaves of wheat under drought stress. Journal of Plant Nutrition and Fertilizers, 13(3), 471-478.
- Moussa, H. R. (2006). Influence of exogenous application of silicon on physiological response of salt-stressed maize (Zea mays L.). Int. J. Agric. Biol, 8(3), 293-297.
- Al-aghabary, K., Zhu, Z., & Shi, Q. (2005). Influence of silicon supply on chlorophyll content, chlorophyll fluorescence, and antioxidative enzyme activities in tomato plants under salt stress. Journal of plant nutrition, 27(12), 2101-2115.
- Shi, Y., Zhang, Y., Yao, H., Wu, J., Sun, H., & Gong, H. (2014). Silicon improves seed germination and alleviates oxidative stress of bud seedlings in tomato under water deficit stress. Plant Physiology and Biochemistry, 78, 27-36.
- Shi, Y., Zhang, Y., Han, W., Feng, R., Hu, Y., Guo, J., & Gong,H. (2016). Silicon enhances water stress tolerance by improving root hydraulic conductance in Solanum lycopersicum L. Frontiers in plant science, 7, 196.
- Kim, Y. H., Khan, A. L., Waqas, M., & Lee, I. J. (2017).Silicon regulates antioxidant activities of crop plants under abiotic-induced oxidative stress: a review. Frontiers in plant science, 8, 510.
- Shen, X., Zhou, Y., Duan, L., Li, Z., Eneji, A. E., & Li, J. (2010). Silicon effects on photosynthesis and antioxidant parameters of soybean seedlings under drought and ultraviolet-B radiation. Journal of plant physiology, 167(15), 1248-1252.
- Kumar, A., Nayak, A. K., Pani, D. R., & Das, B. S. (2019).Application of phosphorus, iron, and silicon reduces yield loss in rice exposed to water deficit stress. Agronomy Journal, 111(3), 1488-1497.
- Gunes, A, DJ Pilbeam, A Inal, and S Coban. 2008. "Influence of silicon on sunflower cultivars under drought stress, I: Growth, antioxidant mechanisms, and lipid peroxidation." Communications in Soil Science and Plant Analysis 39 (13-14):1885-1903.
- Mauad, M., Crusciol, C. A. C., Nascente, A. S., Grassi Filho, H., & Lima, G. P. P. (2016). Effects of silicon and drought stress on biochemical characteristics of leaves of upland rice cultivars. Revista Ciência Agronômica, 47(03), 532-539.
- Flower, D. J., & Ludlow, M. M. (1986). Contribution of osmotic adjustment to the dehydration tolerance of waterâ?stressed pigeon pea (Cajanus cajan (L.) millsp.) leaves. Plant, Cell & Environment, 9(1), 33-40.
- Zegaoui, Z., Planchais, S., Cabassa, C., Djebbar, R., Belba-chir, O. A., & Carol, P. (2017). Variation in relative water content, proline accumulation and stress gene expression in two cowpea landraces under drought. Journal of Plant Physiology 218, 26-34.
- Mali, M., & Aery, N. C. (2008). Influence of silicon on growth, relative water contents and uptake of silicon, calcium and potassium in wheat grown in nutrient solution. Journal of Plant Nutrition, 31(11), 1867-1876.
- Kafi, M., Nabati, J., Ahmadi-Lahijani, M. J., & Oskoueian,A. (2021). Silicon compounds and potassium sulfate improve salinity tolerance of potato plants through instigating the defense mechanisms, cell membrane stability, and accumulation of osmolytes. Communications in Soil Science and Plant Analysis, 52(8), 843-858.
- Younis, A. A., Khattab, H., & Emam, M. M. (2020). Impacts of silicon and silicon nanoparticles on leaf ultrastructure and TaPIP1 and TaNIP2 gene expressions in heat stressed wheat seedlings. Biologia plantarum, 64(1).
- YOSHIDA, K., NOMURA, F., & GOTOH, K. (1983). Signif-icance of Intra-plant Flowering Date in Soybean Seed Production: 2. Number of flowers, podding efficiency, nodal distribution of pods and yield components among different flowering dates. Japanese Journal of Crop Science, 52(4), 567-573.
- Chaiwong, N., Rerkasem, B., Pusadee, T., & Promâ?uâ?thai, C. (2021). Silicon application improves caryopsis development and yield in rice. Journal of the Science of Food and Agriculture, 101(1), 220-228.
- Ming, D. F., Pei, Z. F., Naeem, M. S., Gong, H. J., & Zhou, W.J. (2012). Silicon alleviates PEGâ?induced waterâ?deficit stress in upland rice seedlings by enhancing osmotic adjustment. Journal of Agronomy and Crop Science, 198(1), 14-26.
- Chu, M., Liu, M., Ding, Y., Wang, S., Liu, Z., Tang, S., ... & Li, G. (2018). Effect of nitrogen and silicon on rice submerged at tillering stage. Agronomy Journal, 110(1), 183-192.
- Cuong, T. X., Ullah, H., Datta, A., & Hanh, T. C. (2017). Effects of silicon-based fertilizer on growth, yield and nutrient uptake of rice in tropical zone of Vietnam. Rice Science, 24(5), 283-290.
- Emam, M. M., Khattab, H. E., Helal, N. M., & Deraz, A. E. (2014). Effect of selenium and silicon on yield quality of rice plant grown under drought stress. Australian Journal of Crop Science, 8(4), 596-605.
- Bukhari, M. A., Ashraf, M. Y., Ahmad, R., Waraich, E. A., & Hameed, M. (2015). Improving drought tolerance potential in wheat (Triticum aestivum L.) through exogenous silicon supply. Pak J Bot, 47(5), 1641-1648.
- Nolla, A., Faria, R. D., Korndoerfer, G. H., & Silva, T. D. (2012). Effect of silicon on drought tolerance of upland rice.
- Hülskamp, M., Miséra, S., & Jürgens, G. (1994). Genetic dis-section of trichome cell development in Arabidopsis. Cell, 76(3), 555-566.
- Kennedy, G. G. (2003). Tomato, pests, parasitoids, and predators: tritrophic interactions involving the genus Lycopersicon. Annual review of entomology, 48(1), 51-72.
- Guntzer, F., Keller, C., & Meunier, J. D. (2012). Benefits of plant silicon for crops: a review. Agronomy for sustainable development, 32, 201-213.
- Rostkowska, C., Mota, C. M., Oliveira, T. C., Santiago, F. M., Oliveira, L. A., Korndörfer, G. H., ... & Mineo, J. R. (2016). Si-accumulation in Artemisia annua glandular trichomes increases artemisinin concentration, but does not interfere in the impairment of Toxoplasma gondii growth. Frontiers in Plant Science, 7, 1430.
- Takeda, H., Ito, F., Yamanaka, S., Takiyama, N., & Yoshino,K. (2013). Roles of trichomes with silica particles on the surface of leaves in Aphananthe aspera. AIP Advances, 3(3).
- Vandegeer, R. K., Zhao, C., Cibilsâ?Stewart, X., Wuhrer, R., Hall, C. R., Hartley, S. E., ... & Johnson, S. N. (2021). Silicon deposition on guard cells increases stomatal sensitivity as mediated by K+ efflux and consequently reduces stomatal conductance. Physiologia Plantarum, 171(3), 358-370.
- Agarie, S., Uchida, H., Agata, W., Kubota, F., & Kaufman, P.B. (1998). Effects of silicon on transpiration and leaf conductance in rice plants (Oryza sativa L.). Plant Production Science, 1(2), 89-95.
- Isa, M., Bai, S., Yokoyama, T., Ma, J. F., Ishibashi, Y., Yuasa, T., & Iwaya-Inoue, M. (2010). Silicon enhances growth independent of silica deposition in a low-silica rice mutant, lsi1. Plant and soil, 331, 361-375.
- Ibrahim, H. I., Sallam, A. M., & Shaban, K. A. (2015). Impact of irrigation rates and potassium silicate fertilizer on seed production and quality of Fahl Egyptian clover and soil properties under saline conditions.
- Ning, X. A., Lin, M. Q., Shen, L. Z., Zhang, J. H., Wang, J. Y.,Wang, Y. J., ... & Liu, J. Y. (2014). Levels, composition profiles and risk assessment of polycyclic aromatic hydrocarbons (PAHs) in sludge from ten textile dyeing plants. Environmental research, 132, 112-118.
- Murphy, K. S. (1962). Modifications of the technique for determination of chlorophyll stability index in relation to studies of drought resistance in rice. Curr. Sci., 31, 470-471.
- Schuppler, U., He, P. H., John, P. C., & Munns, R. (1998). Effect of water stress on cell division and Cdc2-like cell cycle kinase activity in wheat leaves. Plant physiology, 117(2), 667-678.
-



