Research Article - (2022) Volume 6, Issue 1
Immune Hemolytic Anemia in Patients with Lymphoid Malignancy in Tanzania
Received Date: May 31, 2022 / Accepted Date: Jun 15, 2022 / Published Date: Jun 23, 2022
Copyright: ©Copyright: Ã?©2022 Gilbert Uwizeyimana, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Citation: Gilbert Uwizeyimana, Mwashungi Ally, Mbonea Yonazi, Clara Chamba, Ahlam Nasser, et al. (2022). Immune Hemolytic Anemia in Patients with Lymphoid Malignancy in Tanzania. Stem Cell Res Int 6(1), 46-53.
Abstract
Background: Hematological malignancies of the lymphoid lineage such as multiple myeloma, lymphomas and lymphoid leukemia are a burden to the world especially in Tanzania; anaemia is one of the presenting symptoms of these conditions and is found in almost 100% of the patients at diagnosis. Of particular importance Immune Hemolytic Anemia (IHA) which has a poor response to treatment and subsequently poor prognosis when it is associated with lymphoid malignancies. The aim of this study is to determine the magnitude of immune hemolytic anemia in lymphoid malignancies in Tanzania
Methods: This was a cross sectional study that recruited 86 patients with lymphoid malignancies attending both Ocean Road Cancer Institute (ORCI) and Muhimbili National Hospital (MNH). The clinical features were captured using a well-structured questionnaire. Laboratory results for Full Blood Count (FBC), reticulocytes count, total, indirect and direct bilirubin and Lactate Dehydrogenase (LDH) were retrieved from the laboratory reporting system. Additionally, a blood sample for the Direct Antiglobulin Test (DAT) was taken from all participants. This test was performed at the Central Pathology Laboratory (CPL) of MNH. Data analysis was performed using Statistical Package for the Social Sciences (SPSS 20.0) and Microsoft excel 2010. Data analysis was performed using South Texas Art Therapy Association (STATA 15.0).
Results: IHA, indicated by a positive DAT, was found in 10% of patients with lymphoid malignancies. Analysis of DAT among patients never exposed to chemotherapy showed a prevalence of 22%. Among 9 IHA cases, 2 were CLL (Chronic Lymphoid Leukaemia), 2 were MM (Multiple Myeloma), 3 was NHL (Non-Hodgkin Lymphoma), 1 was HL (Hodgkin Lymphoma) and 1 SLL (Small Lymphocytic Lymphoma). All 9 cases had decompensated hemolytic anemia.
Conclusion: IHA is a real complication of chronic lymphoid malignancies and DAT should be included in tests done during the course of management.
Keywords
Immune Hemolytic Anemia (IHA), Autoimmune Hemolytic Anemia (AIHA), Lymphoid Malignancies, Tanzania
Background
Cancer is a common health threat all over the world; According to the WHO, cancer is one of the leading causes of morbidity and mortality worldwide; In 2018, there were approximately 9.6 mil- lions deaths with about one in every six people dying from cancer related causes; In 2014, there were approximately 21,000 reported deaths from cancer in Tanzania and 35,000 new diagnosis across all ages [1].
Immune phenomena are a well-known complication of hematolog- ical malignancies, and especially of those involving the lymphoid system. Indeed, three immune hematologic conditions associated with hematological malignancy are immune hemolytic anemia, id- iopathic thrombocytopenic purpura and pure red cell aplasia: of these, immune hemolytic anemia is the most common, and it is more common in chronic hematological malignancy [2]. Immune hemolytic anemia can be classified as (i) auto-immune hemolytic anemia (AIHA) that is the most commonly associated with he- matological malignancies; (ii) allo-immune hemolytic anemia as a consequence of blood transfusion that is common practice among hematological malignancy [3]; (iii) drug induced hemolytic ane- mia. IHA is also classified as primary, or idiopathic, when thereis no associated condition; or secondary when an underlying disorder can be identified: the latter is estimated to be about one-half of all cases of AIHA. Hematological malignancy is thought to be the number one cause of the secondary type [4]. However, patients with ‘primary’ AIHA may be seen to develop subsequently NHL, multiple myeloma, CLL or other lymphoid malignancies [5].
The Direct Antiglobulin Test (DAT) in the diagnostic test for IHA, but it is not routinely carried out in most hospitals in Tanzania [6]. Anemia is one of the commonest symptoms of hematological malignancy and very frequently causes the patient to seek medical attention even before the cancer is diagnosed. Proper management of anaemia depends on the cause of anaemia and can significantly improve the quality of life [6, 7].
The aim of this study is to determine the magnitude of IHA in B cell lineage malignancies in Tanzania.
Methods
This was a descriptive cross-sectional hospital-based study on pa- tients with hematological malignancies attended in the two major cancer referral hospitals (Muhimbili National Hospital and Ocean Road Cancer Institute) in Tanzania from November 2019 to March 2020. These hospitals serve the whole country, with patients being transferred from regional hospital or through direct consultations. Approximately 96 patients with lymphoma and 45 patients with other chronic hematological malignancies are attended in both hospitals per year.
The study population consisted of patients with chronic lymphoid malignancies attended at MNH and ORCI from November 2019 to March, 2020. The minimum sample size required was calculated using one group proportion formula. The prevalence rate comput- ed in this formula was 5.6% which was from a previous study done in Nigeria [7]. Therefore, minimum projected sample size was 86. All patients with chronic lymphoid malignancies (Multiple my- eloma, lymphomas and chronic lymphocytic leukaemia) aged 18 and above were eligible to participate in the study. Patients with acute lymphoblastic leukaemia and those known to have IHA were excluded from the study.
A well-structured questionnaire was used to capture the infor- mation on age, sex, reason of admission/consultation, history of transfusion, symptoms of anemia, and examination findings such as paleness, jaundice, splenomegaly, lymphadenopathies, hepato- megaly and treatment were assessed. Diagnosis of a lymphoid malignancy was confirmed from peripheral blood smear, bone marrow, serum electrophoresis and lymphnode biopsy results documented in the patients’ records. Results for Full Blood Count (FBC), reticulocytes count, total, indirect and direct bilirubin and Lactate Dehydrogenase (LDH) were retrieved from the patients’ records. DAT was performed at the CPL in MNH
We received ethical approval from the MUHAS institutional re- view board and permission to conduct the study from the hospital administration of ORCI and MNH.
The total of 94 patients with lymphoid malignancies consulted MNH and ORCI during our study period. Seven patients were excluded from the study because they had no hemolytic markers results in the hospital reporting system and one excluded because of failure to consult for participating in the study. We therefore enrolled 86 participants.
Results
Demographic and Baseline Characteristics The majority (80%) of the participants were enrolled from MNH (69/86); 58% (50/86) were male, and the mean age of participants was 56 years (56 ± 16). With regards to diagnosis, 42% (36/86) had lymphoma, 34% (29/86) had multiple myeloma, and 24% had CLL. (Table 1).
Table 1: Demographic and Baseline Characteristics of Patients with Lymphoid Malignancy
|
Characteristic |
Lymphoid malignancies: N=86 |
Percentage |
|
Age, Mean ± SD |
56 ± 16 |
|
|
Gender |
|
|
|
Female |
36 |
42 |
|
Male, |
50 |
58 |
|
Hospital where treated |
|
|
|
ORCI |
17 |
18 |
|
MNH, |
69 |
80 |
|
Diagnosis |
|
|
|
CLL |
21 |
24 |
|
Multiple myeloma |
29 |
34 |
|
Lymphomas |
36 |
42 |
|
PS+BMB: peripheral smear plus bone marrow biopsy; N or n: number of participants; SPE: serum protein electrophoresis, MNH: Muhimbili National Hospitals; ORCI: Ocean Road Cancer Institute |
||
Clinical Presentation of Participants
The most common symptom was fatigue related to anemia that was manifested followed by other symptoms of anemia such as palpitations, dizziness, headache. The history of transfusion with blood products and time from last transfusion was assessed, 62% (53/86) of the patients had a history of blood transfusion at some point in their life time, with most common blood product being packed red cells (52/53). For these participants with a transfu- sion history, 53% (28/53) reported to have received the transfusion more than three months prior the time we collected blood sample for DAT. 64% (56/86) of patients had been on steroids (55 was on chemotherapy containing steroids and one on steroids alone). (Table 2)
Table 2: Clinical Information of Participants
|
Characteristic |
Lymphoid malignancies: N=86 |
Percentage |
|
Symptoms |
|
|
|
Fatigue score/10, Median (IQR) |
4 (1-5) |
|
|
Palpitations |
19 |
22 |
|
Dizziness |
24 |
28 |
|
Headache |
32 |
37 |
|
Signs |
|
|
|
|
|
|
|
Pallor Moderate |
15 |
17 |
|
Mild |
20 |
23 |
|
No pallor |
24 |
28 |
|
Severe |
27 |
31 |
|
With hepatomegaly |
6 |
7 |
|
With jaundice |
12 |
14 |
|
With Splenomegaly |
29 |
34 |
|
With Lymphadenopathy |
35 |
41 |
|
Transfusion history in life |
53 |
62 |
|
Blood product |
|
|
|
PRBCSs +Platelets |
1 |
1 |
|
PRBCs |
52 |
61 |
|
Number of units of transfusion, Median (IQR) |
4 (0-6) |
|
|
0-72 hours |
4 |
5 |
|
3-120 days |
21 |
24 |
|
>120 days |
28 |
33 |
|
On Steroids |
56 |
65 |
|
Time on Chemotherapy in months, Median (IQR) |
3 (0-7) |
|
|
FBC: Full Blood Count, PRBCS: Packed red blood cells, IQR: Interquartile range FBC was done to assess the level of anemia. The median hemoglobin level was 8.8 (6.2-10.5) in the lymphoid malignancies. |
||
Prevalence of IHA in lymphoid malignancies
Nine patients were found to have positive DAT making the preva- lence of IHA among lymphoid malignancies 10% (9/86).
Frequency of IHA in individual sub-types of lymphoid malignancies
Frequency of IHA was highest in lymphoma, where it was 14% (5/36). Multiple myeloma had 7% (2/29) and CLL had 9% (2/21). (Table 3)
Table 3: Frequency of IHA in Different Sub-Types of Lymphoid Malignancies
|
Type of lymphoid malignancies |
IHA Positive N=9 |
IHA Negative N=77 (%) |
Total N=86 |
|
Lymphomas, n (%) |
5 (14) |
31(86) |
36 |
|
NHL, n (%) |
3 (16) |
16 (84) |
19 |
|
HL, n (%) |
1 (14) |
6 (86) |
7 |
|
SLL, n (%) |
1 (50) |
1 (50) |
2 |
|
MCL, n (%) |
0 (0) |
5 (100) |
5 |
|
Splenic lymphoma, n (%) |
0 (0) |
2 (100) |
2 |
|
Cutaneous Lymphoma, n (%) |
0 (0) |
1 (100) |
1 |
|
Multiple myeloma, n (%) |
2 (7) |
27 (93) |
29 |
|
CLL, n (%) |
2 (9) |
19 (91) |
21 |
|
CLL: chronic lymphocytic leukemia, NHL: Non-Hodgkin lymphoma, HL: Hodgkin lymphoma, MCL: mantle cell lymphoma, SLL: small lymphocytic lymphoma |
|||
Compensated versus de-compensated hemolytic anemia (N=9)
Among 9 participants who were found to have immune hemolytic anemia, all had decompensated anemia; the lowest Hemoglobin was 5.1 g/dl and the highest one was 9.8 g/dl.
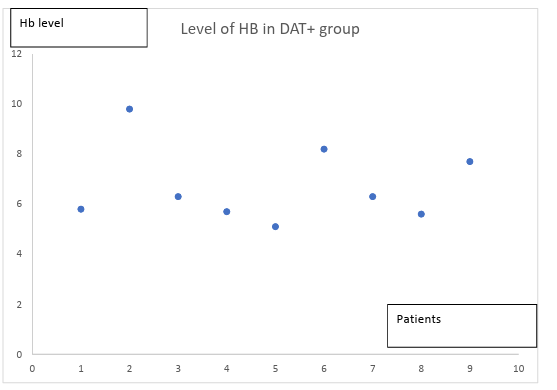
Figure a: Severity of Anemia in Patients with IHA
Treatment History and Transfusion History in Patients with IHA
As summarized in table 4, among 9 participants with IHA, two patients were on steroids (one patient was on steroid alone and one was on chemotherapy containing steroid). The odd ratio of being on steroids versus not on steroids was found to be 8.2 with 95% confidence interval of 1.6-42.5. Therefore, the risk of developing IHA while not on steroids was 8.2 times more compared to if on steroids.
Table 4: IHA in Relation To Chemotherapy and Blood Transfusion
|
Parameter |
IHA Positive N=9 |
IHA Negative N=77 |
p-value |
Odds ratio (95% CI) |
|
Types of malignancy not on steroids, n (%) |
7 (23) |
23 (77) |
|
|
|
Multiple myeloma |
2 (20) |
8 (80) |
|
|
|
CLL |
1 (20) |
4 (80) |
|
|
|
Lymphomas |
4 (27) |
11 (73) |
|
|
|
Steroids |
|
|
0.008 |
|
|
On steroids, n (%) |
2 (4) |
54 (96) |
|
|
|
Not on steroids Not on steroids Vs on steroids |
7 (23) |
23 (77) |
|
8.2 (1.6-42.5) |
|
Transfusion history in life |
|
|
0.295 |
|
|
Has been transfused, n (%) |
4 (8) |
49 (92) |
|
|
|
Never transfused, Trans- fused Vs Never transfused |
5 (15) |
28 (85) |
|
0.46 (0.11-1.84) |
|
Time from the last trans- fusion Never transfused |
5 (15) |
28 (85) |
0.295 |
|
|
>120 days, n (%) |
1 (4) |
27 (96) |
|
|
|
3-120 days, n (%) |
1(25) |
3(75) |
|
|
|
0-3 days, n (%) |
2 (10) |
18 (90) |
|
|
|
OR: odds ratio |
||||
IHA and Hemolytic Markers
Apart from hemoglobin and reticulocyte count, the hemolytic markers were significantly higher in patients with IHA compared to those without IHA. (Table 5)
Table 5: Laboratory Findings in Patients with Lymphoid Malignancies with and Without IHA
|
Parameter |
IHA positive, N= 9 |
IHA negative, N= 77 |
p-value |
|
Hemoglobin: n= 86, (Mean±SD) |
6.85±1.48 |
8±2.57 |
0.054 |
|
Reticulocytes: n=86 Median (IQR) |
80700 (45200-99000) |
41600 (23800-55800) |
0.0814 |
|
Total Bilirubin Median (IQR) |
34.9 (30.9-62.5) |
10.5 (6.8-15.6) |
<0.0001 |
|
Direct Bilirubin Median (IQR) |
10.9 (8-15.4) |
4.3 (3-7.5) |
0.0017 |
|
Indirect Bilirubin Median (IQR) |
24 (15.5-54.1) |
5.5(3.9-8.1) |
<0.00001 |
|
LDH Median (IQR) |
516 (324-867) |
293 (222-432) |
0.0062 |
Discussion
In this cross-sectional study, we have established the prevalence of immune hemolytic anemia among our patients with lymphoid malignancies: specifically CLL, MM and lymphomas.
Prevalence of Immune Hemolytic Anemia among Lym- phoid Malignancy
The prevalence of immune hemolytic anemia among lymphoid malignancies was 10%. Almost the same percentage was reported in the study done in India by Ghosh et al where the prevalence was 10.6% [28]. Thus, patients with lymphoid malignancy must be regarded as being at a higher risk of developing IHA, as from a re- view article on causes of anemia in cancer by Balana Louis Gaspar et al [27]. A study done in south Nigeria by Kaladada et al showed a prevalence of AIHA of about 6% (2/35) among B cell neoplasm; this lower might be explained by the fact that most of their partic- ipants were already on chemotherapy, and all those with positive DAT were not on chemotherapy [7]. In our study, we observed 4 out of 9 patients with positive DAT had never been transfused in life and one had been transfused in more than 120 days ago; That means the prevalence of AIHA in our study can be estimated to be 6% (5/86) that is almost the same in the study of south Nigeria [7].
Frequency of IHA among Different Types of B cell Neo- plasms
The prevalence of IHA among CLL/SLL, Multiple myeloma, NHL and HL was 13% (3/23), 7% (2/29), 13.8% (4/29) and 14% (1/7) respectively. Almost the same prevalence among CLL/SLL was reported in the study done in Egypt by Basma et al that showed the prevalence of AIHA in CLL to be 11.9% [10]. Some studies reported prognostic implication of lymphoid malignancies when presented with IHA [11-13].
We have low prevalence of IHA among multiple myeloma com- pared to other few published studies. The study done by Rajesh et al in India showed a slightly higher prevalence of 10.6% that is different from ours that is 7%; this difference can be explained by the fact that most of our participants were already on chemother- apy containing steroids that may have reduced the frequency of IHA [14].
So far, there are very few studies done showing prevalence of IHA in lymphomas. A study done by Ji Cheng in China showed a preva- lence of AIHA among NHL of 0.91%, which is significantly lower than the present study that showed a prevalence of 14% (1/7). Sim- ilarly, the present study reports prevalence of 14% for HL but other studies reported that IHA is rare among HL. However, our sample size for lymphomas was small and also our patients present at late stage of the disease which is a risk factor of developing AIHA in hematological malignancy as explained by Ji Cheng et al [15]. The same as others wherever these conditions develop AIHA, the prognosis is poor but our study did not assess the survival rate as it was cross sectional study [16, 17].
Association between IHA and Treatment Offered To Pa- tients with Chronic Lymphoid Malignancies.
The prevalence of IHA among participants who had never been exposed to chemotherapy or steroids was 23% (7/31). Among 9 participants with positive DAT, 78% (7/9) had never been on ste- roids whereas 22% (2/9) had been on the latter: this difference was statistically significant with P value of 0.012 and this showed that steroids may be protective to develop IHA. The observed odds ra- tio was 8.2 that means the risk of developing IHA while not on steroids is 8.2 times more if you are on steroids. This can be ex- plained by the fact that first line and most effective treatment of IHA is steroids and almost all chemotherapy regimen for lymphoid malignancies include steroids, so there is much chance that if the participant had IHA but was on steroids, the disease will clear out and hence negative DAT [18].
4 out 9 positive DAT meaning 44% (4/9) had been transfused in less than 120 days that is the life span of red cells; this means that they may have allo-immune hemolytic anemia or AIHA.
Status of Different Hemolytic Markers
DAT is the gold standard for the diagnosis of AIHA; but other pa- rameters help to assess the severity of hemolysis. These includes reticulocytes count, indirect bilirubin and LDH [19-22]; they may suggest AIHA even in the rare cases (estimated 5%) when DAT is negative [23]. They are also very important in non-immune hemo- lytic anemia where they will raise whereas DAT is negative.
In our study the median absolute reticulocytes count among IHA and non IHA was 80700/L and 41600/L: a considerable difference, though not statistically significant. The highest reticulocyte count that was reported among IHA patients was 557480/L was and low- est was 1380/L. Three out of nine participants had reticulocytes count below the low limit of reference range and two had the one higher than the upper limit of reference range, four remaining had the one within the limits of normal range that is 50,000-100,000/L. Therefore, reticulocytopenia with IHA was found in 33.3% (3/9). This findings is similar in other studies where cases of AIHA with reticulocytopenia have been reported like Lockard et al reported four cases of AIHA and reticulocytopenia with erythroid hyper- plasia in the bone marrow [24]; the same findings were reported by William et al who assessed 55 cases of AIHA and he found 14 (25.5%) of them had reticulocytopenia and they had poor progno- sis compared to those with normal or higher reticulocytes count [25].
In our study, we have found that total, conjugated, unconjugated bilirubins were higher in IHA group compared to non IHA and the difference was statistically significant. These findings may suggest that total and indirect bilirubin can be a good marker of IHA for treatment in case DAT is not available, however, more studies are required to confirm this concept. Jorge et al reported a case of high indirect bilirubin in hemolysis [26].
LDH was high in both IHA and non IHA groups but higher in IHA with a median of 516 and 293 respectively and a P value of 0.0062. LDH usually rises in case of malignancies and it has been used for long time as treatment response monitoring for lymphomas, the lower value observed in the non IHA group should be due to the fact that most of them were on chemotherapy for long period of time [27]. LDH has been used for long time as a marker of hemo- lysis especially intravascular hemolysis and our study can show how it is even relevant in extravascular hemolysis [28].
Conclusion
To our knowledge, this is the first study in Tanzania, indeed in East Africa, exploring the association between immune hemolyt- ic anemia and any type of haematological malignancy. We had a good sample size and in order to get a sufficiently large number of participants, thus increasing the power of our study, we needed a multi-center data collection (MNH and ORCI), that is also strength of our study. IHA specifically AIHA among lymphoid malignan- cies is a big concern, it has both clinical and prognostic impli- cation. Its main presentation after symptoms of anemia, there is jaundice with possible splenomegaly. Indirect bilirubin and LDH raises; and DAT will be positive.
Our study has found that the prevalence of IHA among chronic lymphoid malignancy including those on and off chemotherapy is 10%. This means that among 10 patients with chronic lymphoid malignancy who attend MNH and ORCI, one may have IHA. This is a big number to show that the magnitude of IHA among chron- ic lymphoid malignancy is not neglectable. Because of the short time for data collection, we couldn’t determine the prognosis for those with and without IHA; despite that most literatures we cited showed that those with IHA have poor prognosis compared with those without IHA.
List of Abbreviations
Ethics Approval and Consent to Participate
It is attached. We have gotten ethics approval from the Muhimbili University of Health and Allied Sciences (MUHAS)
Consent for Publication
N/A. Our manuscript does not contain any individual person’s data in any form (including any individual details, images or videos)
Availability of Data and Materials
Dataset will be available
Competing Interests
We declare no conflict of interests.
Funding
This study was conducted as part of the corresponding author’s completion of master’s in medicine of hematology and blood transfusion at Muhimbili University of Health and Allied Sciences (MUHAS). He was funded by Government of Rwanda in all his studies including this research.
Authors’ Contributions
The research protocol was prepared by the first author in close col- laboration with the last author and second author. The first author collected, analyzed data under supervision of the last and second authors. Manuscript was prepared by the first author and corrected by all authors mentioned.
Acknowledgements
We wish to acknowledge the support of Muhimbili National Hos- pital/department of hematology and blood transfusion, Ocean Road Cancer Institute (ORCI) and the cooperativeness of the study participants.
References
- Kohi, T. W., von Essen, L., Masika, G. M., Gottvall, M., & Dol, J. (2019). Cancer-related concerns and needs among young adults and children on cancer treatment in Tanzania: a qualitative study. BMC cancer, 19(1), 1-9.
- Mauro, F. R., Foa, R., Cerretti, R., Giannarelli, D., Coluzzi, S., Mandelli, F., & Girelli, G. (2000). Autoimmune hemolytic anemia in chronic lymphocytic leukemia: clinical, therapeu- tic, and prognostic features. Blood, The Journal of the Ameri- can Society of Hematology, 95(9), 2786-2792.
- Ddungu, H., Krantz, E. M., Kajja, I., Naluzze, S., Nabbanja, H., Nalubwama, F., ... & Kiwanuka, N. (2020). transfusion challenges in patients with Hematological Malignancies in Sub-Saharan Africa: A prospective observational Study from the Uganda cancer institute. Scientific reports, 10(1), 1-9.
- Lauda-Maillen, M., Catroux, M., Roy-Peaud, F., Souch- aud-Debouverie, O., El Masmouhi, B., & Roblot, P. (2017). Diagnostic et prise en charge de l’anémie hémolytique au- to-immune à l’exclusion des formes secondaires à une cause néoplasique. Adéquation de la prise en charge au PNDS oc- tobre 2009. La Revue de Médecine Interne, 38(10), 648-655.
- Hill, Q. A., Stamps, R., Massey, E., Grainger, J. D., Pro- van, D., & Hill, A. (2017). British Society for Haematology Guidelines. Guidelines on the management of drug-induced immune and secondary autoimmune, haemolytic anaemia. Br J Haematol, 177(2), 208-220.
- Liebman HA, Weitz IC. Liebman, H. A., & Weitz, I. C. (2017). Autoimmune hemolytic anemia. Medical Clinics, 101(2), 351-359.
- Korubo, K. I., Nwauche, C. A., & Ejele, O. A. (2013). Auto- immune Hemolytic Anemia In Cancer Patients At a Tertiary Center In Southern Nigeria. Blood, 122(21), 4658.
- Ghosh, J., Singh, R. K., Saxena, R., Gupta, R., Vivekanan- dan, S., Sreenivas, V., ... & Kumar, L. (2013). Prevalence and aetiology of anaemia in lymphoid malignancies. Natl Med J India, 26(2), 79-81.
- Gaspar, B. L., Sharma, P., & Das, R. (2015). Anemia in malig- nancies: pathogenetic and diagnostic considerations. Hema- tology, 20(1), 18-25.
- Atef, B., Azmy, E., Aladle, D., & Mabed, M. (2019). The prevalence and prognostic significance of autoimmune cyto- penias in a cohort of Egyptian patients with chronic lympho- cytic leukemia. Hematology/oncology and stem cell therapy, 12(2), 97-104.
- Shvidel, L., Tadmor, T., Braester, A., Bairey, O., Rahimi-Lev- ene, N., Herishanu, Y., ... & Polliack, A. (2013). Pathogene- sis, prevalence, and prognostic significance of cytopenias in chronic lymphocytic leukemia (CLL): a retrospective com- parative study of 213 patients from a national CLL database of 1,518 cases. Annals of hematology, 92(5), 661-667.
- Kyasa, M. J., Parrish, R. S., Schichman, S. A., & Zent, C. S. (2003). Autoimmune cytopenia does not predict poor progno- sis in chronic lymphocytic leukemia/small lymphocytic lym- phoma. American journal of hematology, 74(1), 1-8.
- Rogers, K. A., & Woyach, J. A. (2016, April). Secondary autoimmune cytopenias in chronic lymphocytic leukemia. In Seminars in Oncology (Vol. 43, No. 2, pp. 300-310). WB Saunders.
- Kashyap, R., Singh, A., & Kumar, P. (2016). Prevalence of au- toimmune hemolytic anemia in multiple myeloma: a prospec- tive study. Asiaâ?Pacific Journal of Clinical Oncology, 12(2), e319-e322.
- Uwizeyimana, G., Ally, M., Yonazi, M., Chamba, C., Nasser, A., & Luzzatto, L. (2022). Immune Hemolytic Anemia in Pa- tients with Lymphoid Malignancy in Tanzania.
- Sallah, S., Sigounas, G., Vos, P., Wan, J. Y., & Nguyen, N.P. (2000). Autoimmune hemolytic anemia in patients with non-Hodgkin's lymphoma: characteristics and significance. Annals of Oncology, 11(12), 1571-1577.
- Doni, E., Carli, G., Di Rocco, A., Sassone, M., Gandolfi, S., Patti, C., ... & Visco, C. (2017). Autoimmune haemolytic anaemia in mantle cell lymphoma: an insidious complication associated with leukemic disease. Hematological oncology, 35(1), 135-137.
- Zanella, A., & Barcellini, W. (2014). Treatment of autoim- mune hemolytic anemias. Haematologica, 99(10), 1547.
- Müller, A., Zimmermann, R., & Krause, S. W. (2011). Hemo- lytic anemias in adults. Deutsche Medizinische Wochenschrift (1946), 136(45), 2308-2312.
- Barcellini, W., & Fattizzo, B. (2015). Clinical applications of hemolytic markers in the differential diagnosis and manage- ment of hemolytic anemia. Disease markers, 2015.
- Packman, C. H. (2015). The clinical pictures of autoimmune hemolytic anemia. Transfusion Medicine and Hemotherapy, 42(5), 317-324.
- Dhaliwal, G., Cornett, P. A., & Tierney Jr, L. M. (2004). He- molytic anemia. American family physician, 69(11), 2599- 2606.
- Jaime-Pérez, J. C., Aguilar-Calderón, P. E., Salazar-Cavazos, L., & Gómez-Almaguer, D. (2018). Evans syndrome: clini- cal perspectives, biological insights and treatment modalities. Journal of blood medicine, 9, 171.
- Packman, C. H. (2015). The clinical pictures of autoimmune hemolytic anemia. Transfusion Medicine and Hemotherapy, 42(5), 317-324.
- Crosby, W. H., & Rappaport, H. (1956). Reticulocytopenia in autoimmune hemolytic anemia. Blood, 11(10), 929-936.
- Maldonado, J. E., Kyle, R. A., & Schoenfield, L. J. (1974). Increased serum conjugated bilirubin in hemolytic anemia. Postgraduate Medicine, 55(2), 183-190.
- Long, G., Tang, W., Fu, X., Liu, D., Zhang, L., Hu, G., ... & Sun, W. (2019). Pre-treatment serum lactate dehydrogenase predicts distant metastasis and poor survival in nasopharyn- geal carcinoma. Journal of Cancer, 10(16), 3657.
- Kato, G. J., McGowan, V., Machado, R. F., Little, J. A., Tay-lor, J., Morris, C. R., ... & Gladwin, M. T. (2006). Lactate dehydrogenase as a biomarker of hemolysis-associated nitric oxide resistance, priapism, leg ulceration, pulmonary hyper- tension, and death in patients with sickle cell disease. Blood, 107(6), 2279-2285.


