Research Article - (2023) Volume 6, Issue 1
Heavy Metals Speciation, Mobility, and Bioavailability in Constructed Wetland Sediment Treating Polluted River Water: Implication for Environmental Safety
Received Date: Oct 23, 2023 / Accepted Date: Nov 11, 2023 / Published Date: Nov 20, 2023
Copyright: ©Â©2023 Deshu Mamo. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Mekuria, M.D.,( 2023) Heavy Metals Speciation, Mobility, and Bioavailability in Constructed Wetland Sediment Treating Polluted River water: Implication for Environmental Safety. Toxi App Pharma Insights, 6(1), 104-120.
Abstract
The presence of toxic heavy metals in sediments beyond allowable limits are often posing a serious threat to local environment. Constructed wetland sediments contamined with heavy metals can affect sediment-dewlling biota such as benthic organisms, wetland plants, aquatic life including amphibian and reptiles. The objective of this study was to investigate heavy metals, mobility and bioavailability, level of environmental risks, and it implication for environmental safety. The study was conducted using the horizontal subsurface flow (HSSF) constructed wetland (CW) sediments designed for treating polluted Little Akaki River water. The sediment samples were collected from the CW and heavy metals speciation determined following the European Community Bureau References (BCR) sequential extraction procedures. The results showed that the highest percentage of metal fractions: Zn = 71.67%, Cr =82.20%, Cd = 76.47% and Pb = 74.96% were bound to residual fraction(R), implying that majority of the metals were stable, immobile and not bioavailable. The mobility factor (MF) for Zn (27.60%); Cr (14.24%) Cd (18.82% and Pb (21.53%); indicating low percentage of metals found in mobile chemical form. Furthermore, the environmental risk assessment code (RAC %) value for all heavy metals in the sediment samples were ranged: 5.37 % - 8.51%, falling in low risk category (RAC=1-10%). Low mobility, bioavailability and risk assessment code may be attributed to slight alkaline pH, low organic matter, and caly dominated sediment particles. High residual fraction occurred due to clay particles which can form strong bounding matrix with metals. Thus, metals in the CW sediments may pose low toxicity to wetland plants and sediment living biota surrounding environment.
Keywords
Bioavailability, Heavy Metals Fractionation, Horizontal Subsurface Flow Constructed Wetland, Mobility, Sediment, Risk Assessment Code
Introduction
Some heavy metals are toxic to plants and animals, persistent in environment, tend to accumulate in living organisms over time, and eventually reach human through food chain and cause various health reprecussions [1]. When heavy metals accumulate in soil, water and plant tissues beyond allowable limits, it can impairs the normal growth of plants. The principal sources of heavy metals are often categorized into point and non-point sources. Points sources include industries like tannery, textile and metal processing facto- ries while non-point source encompasses agriculture (the applica- tion of agrochemical). Heavy metals generated either from point or non-point source are usually transported through water system and cause environmental contamination. The problem related to heavy metals contamination increasingly become a serious health issue in Ethiopia due to lack of wastewater treatment facilities, limit- ed regulatory control, and untreated or poorly treated industrial wastes are directly released into surface water. As a result, rivers like Beg Akaki River (BAR) and Little Akaki River (LAR) which are passing through the capital city (Addis Ababa City Adminis- tration) and receiving poorly treated and untreated wastewaters released from various industries. Some of the potential polluting industries include metal processing factories, tanneries, textile and garments, food processing, alcohol and beverage factories, vehicle battery maintenance, garage, painting and pigments [2, 3]. Metals can also generated from natural sources such as weathering of par- ent volcanic rocks materials.
In order to treat polluted river water and use the water for var- ious purposes, various technologies such as surface and subsur- face constructed wetland, oxidation, sand filter method, and other chemical treatment methods can be applied. Constructed wetland (CW) is usually viewed as a viable alternative technologies to treat polluted river water, because it is relatively low cost, ease to operate, effective in pollutants removal through various process- es [4]. Furthermore, the application of horizontal subsurface flow (HSSF- CW) technology allows the polluted river water to passes through the subsurface of the system and have enough contact time with wetland sediment (gravel), wetland plant roots, and micro- organisms, which all together contribute to remove contaminants through physical, chemical and biological processes [5]. In CW treatment system, sediment plays a critical role in the removal of heavy metals from the overlying water column through the process of settlement, precipitation and co-precpitation; hence, it is serving as a sink for heavy metals [6].
However, several studies have reported that determining total heavy metal concentrations in the sediment or soil provides infor- mation on overall pollution levels, but does not provide reliable information on heavy metal stability, transport, bioavailability, mobility and toxicity [6-9]. The distribution of metals in the CW sediments and their effects on sediment dwelling biota depends on their speciation and propensity for particular biogeochemical reac- tions [10]. According to John, metal mobility and its availability to living biota are closely associated with to metal speciation [11]. Several researchers have further confirmed that the transport, sta- bility, bioavailability, mobility, affinity, toxicity, and environmen- tal implications of heavy metals are governed by their speciation or chemical forms [4, 12-16]. The chemical forms of a specific metal in sediment or environmental media are referred to as heavy metal speciation [6]. Thus, speciation related to specific form of metal such as isotopic composion, molecular structure or oxidation state [10].
In order to determine heavy metals speciation, the European work- ing group has developed sequential extraction procedures usually known as the Community Bureau of Reference [17]. This method is ease to apply and to determine speciation of heavy metals found in environmental media like soil, sediment and sludge [1, 16, 18, 19]. Hence, it is a useful technique to determine mobility and bio- availability and risk of heavy metals to plants and animals, as well as, metals migration in soil, surface and ground water, and their contamination [20, 11].
Study reports have showed that heavy metals speciation in lake and river sediments, soils, sewerage and municipal sludge, and fish meat have been studied using this sequential extraction method [3, 8, 18, 21]. However, limited studies were conducted on heavy met- al speciation in CW sediments. Sullivan have conducted studies on the accumulation and fate of Zn, Pb, Fe and S in the sediments of constructed wetlands established for treating mine wastewater and they have reported that the majority of heavy metals and sulfre bound to residual fraction, mainly in the form of metal-sulfides [22]. Sundberg-Jones and Hassan have conducted studies on sed- iment associated Hg, Se and As using a pilot constructed wetland designed for treatment of simulated flue gas desulfurization waste- water, and investigated the distribution, characterization and tox- icity to Hyalella azteeca [10]. Thus, studies were not undertaken on HSSF-CW sediment treating polluted river water to investigate metals mobility, bioavailability, risk to environment and aquatic system. Thus, the study is a new attempt to investigate metals spe- ciation in CW sediments treating polluted river water and environ- mental implications.
The purpose of the studies were to (i) determine the concentration of trace metals (Zn, Cr, Cd, and Pb) in HSSF-CW sediments, (ii) determine heavy metals speciation, bioavailability, mobility factor, and metals immobilization or retention in the CW sediment, (iii) evaluate environmental risk metals and its implications for wet- land and surrounding environment and public safety. This kind of study is important to safeguard the environment and public health, because, contaminated sediment may affect water quality, wetland plants and microbes attached to gravel and plant roots, hence, pose metals toxicity to wetland biota and surrounding environment.
Materials and Methods
Experimental Study Site
The study was conducted in Addis Ababa City Administration. The pilot HSSF-CW was established nearby the LAR in Akaki Kalti Sub-administration of the City, at 38o44’50’’E and 8o 53’18’’N at an altitude of 2060 meter above sea level (Fig1). This site was se- lected for pilot HSSF-CW due to the high pollution load of heavy metals released into the river system from surrounding Akaki Kalti industrial zones and agrochemicals [3].
Figure 1: Map of the Experimental Site
Design and operation of HSSF-CW
The HSSF-CW with three treatment cells connected in series, and river water storage tank are the two primary components of the treatment system (Fig 2). The storage tank had a size of 9 m (length) x 5 m (width) x 0.9 m (height), and is used to store pollut- ed Akaki River water. The river water which was loaded with in- dustrial, municipal, and agro-chemicals was directly pumped into this tank, and then, continuously flow into the second component i.e. HSSF-CW treatment cell sediments under regulated flow con- dition. The HSSF-CW had three equal sized treatment cells (cell-1, cell-2, and cell-3) connected in series. The overall dimension of treatment cell is 6 m (length) x 4 m (width) x 0.6 m (depth). The treatment cells were filled with gravel (size: 2.0 mm - 31.5 mm diameter) to a depth of 0.6 m. After placing the sediment (gravel) plants (Typha latifolia) seedlings of equal size were collected and soil bound to their roots washed with clean water and planted at spacing of 0.3 cm x 0.3cm in the gravel layer. During planting, two spades of uncontaminated soil consist of 70% clay and 30% silt were carefully placed around the root of each plant to promote their survival and growth.
The CW was operated with theoretical hydraulic retention time (HRT) of 7 days with a hydraulic loading rate (HLR) of 0.033m/d and a mass loading rate (MLR) of 2 g. /m2.d.The polluted river wa- ter from the storage tank was constantly supplied into the HSSF- CW treatment cell at an average flow rate of 0.711m3/d.
Figure 2: Schematic Diagram Showing Akaki River Water, Water Storage Tank, HSSF-CW Treatment Cells and Sediment Sampling (SS1, SS2 and SS3) Locations
Collection of Sediment Samples and Analyses
Sample Collection
The sediment samples were collected in six rounds (once per two months) between January 2019 to March 2020. However, samples were not done during wet seasons (July-September) because of flood and inaccessibility. A composite sediment samples were col- lected from each treatment cell of HSSF-CW: Cell-1 (sediment sample (SS1); Cell-2 (sediment sample: SS2) and Cell-3 (sediment sample: SS3). From each cell, approximately 3000 gm of com- posite sediment samples were collected from 0–30 cm depth and placed in high-density polyethylene plastic bags, sealed, coded, and transferred to the Addis Ababa University laboratory, where they were promptly stored at 4oC until further processing [23].
Determining Sediment Samples pH, Particle Size Com- position, and Organic Matter
The pH of sediment samples determined following the procedures described in [16, 24, 25]. Accordingly, the sediment sample and deionized water were mixed at a ratio of 1: 5, and then, thoroughly stirred for 30 minutes and allowed to stay overnight. Then, pH of the suspended water was measured using a calibrated pH meter (model: PH PROBE, PHT-02 ATC, Taiwan).
The particle size composition of sediment samples were deter- mined following the procedures described in Uwah [26]. First, the sediment samples were oven dried using oven-dry (model: GX- 65B) at a constant temperature of 60oC. The dried samples were ground with a mortar and pestle and sieved with 63 μm sieve. The sediment grain size composition was determined by taking a 100 gm sieved sediment sample from each sample in such a way that particles that passed through the sieve and retained on the sieves were carefully collected and weighted. On the basis of sieve re- sults, the sediment particles were grouped as follows: clay = <0.002 mm, silt = 0.002–0.063 mm and sand =0.063–2 mm [27]. The percentage of grain size (%) was computed using the formula described in equation (1) [26]. The sediment texture classes were determined based on the percentage of grain size and a ternary diagram of Folk’s classification.
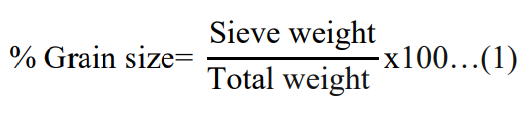
The organic matter (OM) content of sediment samples were de- termined by loss-in–ignition method [22, 28]. In this method, sediment samples were oven-dried at 105oC and weighted using an electronic balance (Model No. JD 210 - 4 CE) until constant weight were obtained. The dried samples were put in a ceramic crucible and placed in a muffle furnace (model: Sx-2.5 - 10) andignited at 600oC for 6 hrs, cooled and weighed. Finally, OM con- tent was computed as the difference in weight due to ignition [28].
Preparation Samples and Determination of Heavy Metal Concentration
The sediment samples were prepared in such a way that all de- bris, plant materials, fallen leaves, grass and weeds were removed from the samples. The sediment samples were oven-dried at 105°C until they reached a consistent weight [14]. The dried sediment samples were crushed with a mortar and pestle, homogenized, and sieved through a 45-μm stainless steel sieve. A sub-sample of 1 gm powdered sediment sample was digested using 12 mL aqua regia (3:1, HNO3 / HCl), in a microwave (Model: ETHOS ONE, MILE- STONE, Italy) according to the USEPA method (3051), soil and sludge digestion procedures. The digested samples were filtered using Whatmann filter paper No. 42, and the filtrate was made 50 mL by adding deionized water, sealed, and stored in refrigerator at 4oC until analyzed. The total concentrations of each heavy met- al were determined using an inductively coupled plasma optical emission spectrophotometry (ICP-OES, Model: ARCOS FHS12, Germany).
Before the analysis, ICP-OES was adjusted and calibrated accord- ing to the manufacturer's instructions. Standard solutions of 0.06, 0.11, 0.17, 0.56, 1.12, 1.68, 2.24, and 2.80 mg L-1 for Zn, while for other heavy metals: Cr, Cd, and Pb; 0.03, 0.06, 0.08, 0.28, 0.56, 0.84, 1.12, and 1.40 mg L-1 were used to calibrate the ICP-OES. For each metal, the total concentration of heavy metals was calcu- lated using a linear graph of the standard concentration and its cor- responding intensities. The calibration curves confirmed linearity for all measured metals in this experiment (R2 = 0.99944).
Determination of Heavy Metals Speciation
The modified Community Bureau of Reference (BCR) sequential extraction procedures were employed to determine heavy metals bound to different metal fractions. As described in Tokalioglu, the fractions of metal extracted in different operational steps include; water and acid soluble, exchangeable and bound to carbonates fraction (F1); bound to Fe–MN oxides (reducible) fraction (F2); metals bound to organic matter (oxidizable) fraction (F3), and metals bound to primary and secondary minerals crystal structures as a residual fraction (R) [16]. 1 gm of dried and powdered sed- iment samples were used for the fractionation analysis. In eachsuccessive extraction step, the suspended supernatant was careful- ly separated after centrifugation at 3000 rotation per minute (rpm) for 20 minutes following the procedures described in [17, 29]. The detailed steps applied in sequential extractions of (F1), (F2) and (F3), reagents used for each step, shaking time and temperature applied are summarized and presented in Table 1.
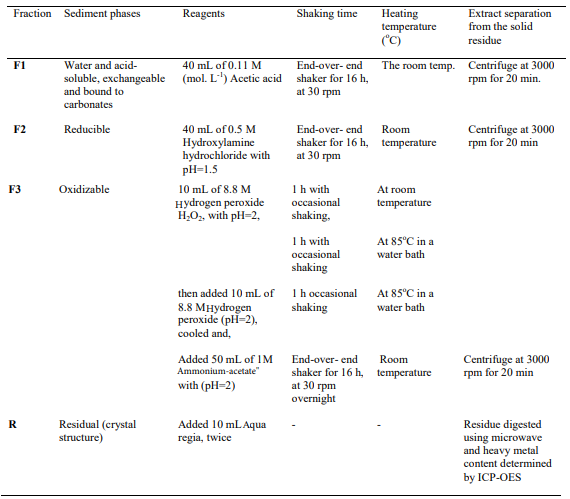
Table1: Sequential Extraction of Heavy Metals, Reagents Used, Shaking Time, Temperature and Extract Separation
The residual (R) fraction was digested following the procedures indicated in [3, 14, 16]. Following USEPA (3051) methods and the microwave-operating handbook for sludge and sediment analysis, the residual fraction was carefully separated from the supernatant and transferred to microwave vessels, where it was digested with 12 mL aqua- regia solution. The blank samples were also prepared and digested following the same procedures. The digest was fil- tered using What Mann filters paper No. 42, and the filtrate was made up to 50 mL with de-ionized water before being stored in the refrigerator at 4oC until analysis.
Evaluating Bioavailability and Mobility Factor (MF)
Bioavailability is usually described as the extents of heavy metals adsorbed or accumulated in biota [21]. extracted in the first step extraction (F1) are instable and highlybioavailable, easy to transport, transform and be absorbed by biota and cause toxicity [6, 16, 21, 30]. Heavy 13 metals occurred in reducible chemical form (F2) are also readily bioavailable. While metals occluded to organic matter (F3) are moderately bioavail- able [8]. Thus, metal fractions detected in the chemical forms of F1, F2 and F3 are considered to be in the bioavailable form.
The mobility of heavy metals is highly depend on the bounding strength of metals with a matrix. Accordingly, a weak bound oc- curs when metal found in the chemical form of (F1), less strong bound when it is occurred in reducible form (F2) and moderately strong bound occurs when it is bound to OM (F3) [13]. The mo- bility factor (MF) was determined by modifying equation stated in and applied as follows [13]:
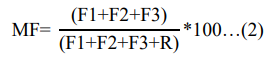
Where; (F1), stands for water and acid soluble, exchangeable and carbonate; (F2) stands for reducible fraction; (F3) stands for oxi- dizable fractions, and ( R) sands for residual fraction.
Risk Assessment Code (RAC)
The risk assessment code (RAC) is described as the percentage of metal, which is weakly occluded to water and acid soluble, exchangeable and carbonates (F1) in relation to total trace metal fractions [15]. RAC is a common ecological risk assessment index that based on the content heavy metal available in chemical form of (F1) [8]. The potential risk of heavy metals found in the sedi- ment can be assessed using risk assessment code (RAC) [31, 24]. According to RAC is expressed as [32].

The percentage RAC value indicating the levels of environmental risk, which classified as follows: RAC < 1% indicates no risk; RAC value ranged between: 1% < RAC <10%, indicates low risk; RAC value ranged 11% < RAC<30%, indicates medium risk; RAC val- ue ranged 31% < RAC <50% indicates high risk, and RAC value > 50% indicates very high risk [8, 32, 33].
Data Analysis and Quality Control
Descriptive statistics such as the mean and standard deviation of heavy metal concentrations in the sediment samples, the per- centage of metal fractions were calculated using Micro-soft excel version 10. The graphs were performed using Origin -Pro 2017 software. Paste software version 4.02 used to perform principal component analysis (PCA).
To ensure data quality, the digested sediment samples were an- alyzed in triplicate and the average concentrations were taken. Blank samples were prepared and run to check interferences. Spiked and non-spike samples were used to validate the method, and percentage recoveries for the four metals (Zn, Cr, Cd and Pb) were varied in between 106.30%-124.81%. The limits of detection (LOD) for each metal was determined based on three times the standard deviation and the results ranged from 0.05–0.06 mg kg-1 [16]. The limit of quantification (LOQ) of the each metal was cal- culated from the blank samples and ten times of the blank samples and results were ranged in between 0.16 - 0.19 mg kg-1. Moreover, glassware and bottles were socked in 10 mL HNO3 diluted water for 2 days, and then, thoroughly washed and rinsed with de-ion- ized water before use.
Results and Discussion
pH, particle size composition and organic matter content of sediment samples
The average pH value of sediment sample taken from cell-1, cell-2 and cell-3 were 7.69, 7.68 and 7.49, respectively, indicating slight- ly alkaline sediment. The results of pH analyses are shown in Fig 3. The sediment pH has a capacity to influence the properties of heavy metals. For instance, the solubility, mobility and bio-accu- mulation of heavy metals are dependent on the pH of sediment [25, 28]. At high pH, the solubility of the heavy metals decreases; hence, heavy metals are poorly subjected to dissolution from sedi- ment particles. Whereas, at low pH, the availability of heavy met- als can increase due to increase in metals solubility under acidic conditions. Dufresne have described that change in sediment pH may leads to change in the availability and concentration of heavy metals in the sediment through time [20]. In addition, the chemical forms or speciation of heavy metals are also influenced by pH val- ue of sediment. Mi have reported the concentration of exchange- able fraction (F1) of Cd and Pb increase with decreasing the pH of sediment [34]. In the contrary Gambrel described that sediment with pH value near neutral favor heavy metal immobilization [35]. However, in the present finding, the pH value for each cell was slightly alkaline, thus, favors immobilization of heavy metals in the sediment. This finding is comparable to the reported value for pH which is ranged 6.88-7.20 for CW sediment [10]
Figure 3: pH of the Sediment Samples
As shown in Fig 4, the particle size composition of the sediment samples were mainly consists of sand, silt and clay with average percentage of 14%, 22.67% and 63.33%, respectively. In all cells, the particle size of the sediment samples were predominantly com- posed of clay particles followed by silt particle may be due to the high clay content of soil used to promote wetland plant growth. The particle size composition of the sediment has an important effect on the chemical fractions of heavy metals. Mi have reported that the concentration of heavy metals such as Cd and Pb increase with decreasing particle size of sediment [34]. The particle size composition of sediments can affects heavy metals assembly and mobility in sediment; for instance, coarse textured sediment has low metal affinity so that metals may be mobile, while fine tex- tured sediments which may contains organic matter usually tends to bind and accumulate metals in the CW sediment hence, reduce metals mobility [35]
Figure 4: Particle Size Composition of Sediment Samples
Results of OM analysis of the sediment samples are presented in Fig 5. The OM content of sediment samples were ranged between 13.12% -14.55% with average value of 13.12%. Higher OM was found in the sediment of the first cell-1, and gradually decreased towards the second (cell-2) and third (cell-3) of HSSF-CW sedi- ments may be due to settling and decomposition of water carried debris, wetland plant leaves, stems, foliage by physical, chemical and biological processes. This finding is in agreement with Alayu and Leta, have reported a deceasing OM in organic matter content along the two – stage HSSF-CW system established to treat brew- ery infulents [36, 37]. Furthermore, cell-1 represents an inlet part of the CW and receives increased nutrient loading which promote plant growth leading to more availability of plant leaves, stems and foliage which decompose and form more OM, as compared to cell-2 and cell-3 sediments [38]. OM content of the sediment plays an impiortant role in sorption and binding of heavy metals. Hence, the bioavailability and mobility of heavy metals in sediment may change as a results of change in sediment OM content [20].
Figure 5: Organic Matter Content in the Sediment Samples along the Cells
Concetration of Heavy Metals in the Sediment
The results of total heavy metals concentration analyses of sedi- ment samples are presented in Table 2. The highest average con- centration was recorded for Pb (2.65 ± 0.58 mg kg-1). This result is may be due to Pb inputs from various sources such as used vehicle battery, used oil, fossil fuel, and garages found in the Akaki- Kalti Industrial Zone and agrochemicals [3, 39]. The average concentra- tion of Cr (1.30 ± 0.53 mg kg-1), Zn (1.22 ± 0.09 mg kg-1), and Cd (0.59± 0.10 mg kg-1), indicating their overall abundance in the sediment. Heavy metals like Cr largely generated from tanneries and metal works. Zn usually released from metal processing and agrochemicals, while Cd is generated from agrochemicals, metals works and solders [3]. Heavy metals can also be generated from natural source like volcanic rocks surrounding the Addis Ababa City transported by water and accumulated in the sediment. The decreasing order of avarge concentration of heavy metals in sed- iment samples was Pb > Cr > Zn > Cd. Thus, the study results showed that Pb and Cr were relatively more abundant metals in the sediments than Zn and Cd. Generally, the total concentration of heavy metals found in the sediment samples provides information on an overall pollution status of sediments of HSSF-CW, but not toxicity, mobility and bioavailability [6, 8, 9].
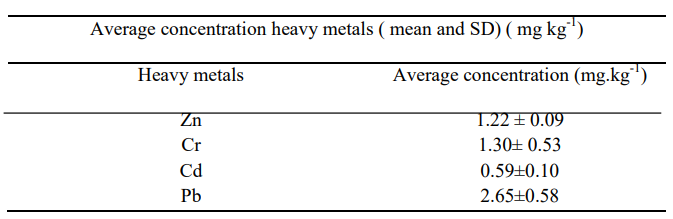
Table 2: The Concentration of Heavy Metals in the Sediment
Speciation of Heavy Metal in Sediment Samples
The chemical speciation of heavy metals in the sediment samples are presented in Table 3.
|
Concentration of Zn in extracted fractions (mg kg-1) |
||||||
|
Treatment cells |
F1: Soluble |
F2: Reduce able |
F3: Oxi- dize-able |
R: Residue |
Sum of frac- tions |
Total heavy metal ( THM) |
|
Cell-1 |
0.078 |
0.138 |
0.144 |
1.692 |
2.052 |
1.17 |
|
Cell-2 |
0.224 |
0.229 |
0.193 |
1.056 |
1.702 |
1.32 |
|
Cell-3 |
0.143 |
0.325 |
0.132 |
1.435 |
2.035 |
1.17 |
|
Av. |
0.148 |
0.231 |
0.156 |
1.394 |
|
|
|
SD |
0.073 |
0.094 |
0.032 |
0.320 |
|
|
|
Concentration of Cr in extracted fractions (mg.kg-1) |
||||||
|
Treatment cells |
F1: Soluble |
F2: Reduce able |
F3: Oxi- dize-able |
R: Residue |
Sum of frac- tions |
Total heavy metal ( THM) |
|
Cell-1 |
0.046 |
0.007 |
0.173 |
1.69 |
1.92 |
0.838 |
|
Cell-2 |
0.23 |
0.031 |
0.317 |
1.671 |
2.25 |
1.877 |
|
Cell-3 |
0.046 |
0.093 |
0.15 |
1.528 |
1.82 |
1.177 |
|
Av. |
0.107 |
0.044 |
0.213 |
1.630 |
|
|
|
SD |
0.106 |
0.044 |
0.091 |
0.089 |
|
|
|
Concentration of Cd in extracted fractions (mg.kg-1) |
||||||
|
Treatment cells |
F1: Soluble |
F2: Reduce able |
F3: Oxidize-able |
R: Residue |
Sum of frac- tions |
Total heavy metal ( THM) |
|
Cell-1 |
0.056 |
0.013 |
0.082 |
0.594 |
0.745 |
0.636 |
|
Cell-2 |
0.122 |
0.049 |
0.116 |
0.81 |
1.097 |
0.475 |
|
Cell-3 |
0.069 |
0.11 |
0.075 |
0.797 |
1.051 |
0.646 |
|
Av. |
0.082 |
0.057 |
0.091 |
0.734 |
|
|
|
SD |
0.035 |
0.049 |
0.022 |
|
|
|
|
Concentration of Pb in extracted fractions (mg.kg-1) |
||||||
|
Treatment cells |
F1: Soluble |
F2: Reduce able |
F3: Oxidize-able |
R: Residue |
Sum of frac- tions |
Total heavy metal ( THM) |
|
Cell-1 |
0.304 |
0.151 |
0.378 |
2.785 |
3.618 |
2.233 |
|
Cell-2 |
0.649 |
0.236 |
0.77 |
3.736 |
5.391 |
2.407 |
|
Cell-3 |
0.284 |
0.612 |
0.357 |
4.6 |
5.853 |
3.321 |
|
Av. |
0.412 |
0.33 |
0.502 |
3.707 |
|
|
|
SD |
0.205 |
0.245 |
0.233 |
0.908 |
|
|
Table 3: Concentration of Extracted Fractions of Metals in the HSSFCW Sediment
As shown in this Table 3, the concentrations of Zn fraction (F1: water and acid soluble, exchangeable and carbonate) in cell-, cell-2 and cell-3, ranged 0.078-0.224 mg kg-1; (F2) (reduce able fraction) ranged: 0.138-0.325 mg kg-1; F3 (oxidize able fraction) ranged: 0.132-0.193 mg kg-1and residue (R) ranged: 1.056-1.692 mg kg-1. The highest average Zn fraction (1.394 ± 0.320 mg kg- 1) was recorded for (R) which may attributed to metal adsorption to clay soil particles. Moving from cell-1 through cell-3, (F1) and (F3) showed a decreasing trend, may be due to slight decreasing OM content of sediment while (F2) showed slightly increasing may be due to a slight decreasing in the pH of sediment which cause release of metals bound to Fe-Mn oxides (Fig- 6(a).
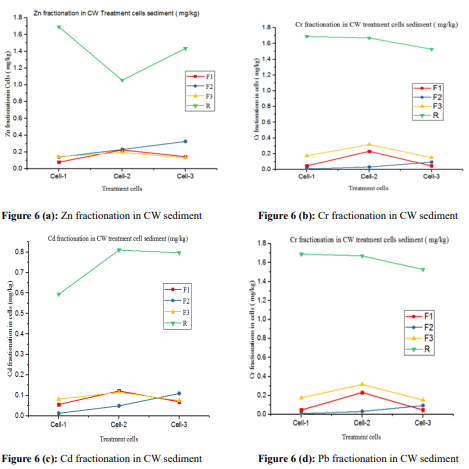
The residual fraction (R) showed decreasing trends between cell-1 and cell-2 may be due to Zn aborption to plant tissues and bound- ing to clay soil structure, while increasing from cell-2 to cell-3 may be due to decomposition of plant materials and release Zn in to sediment.
The Cr fraction that occurred in the chemical form of (F1), in cell- 1, cell-2 and cell-3, ranged 0.046-0.230 mg kg-1; (F2) (reduce-able fraction) ranged: 0.007-0.093 mk / kg-1; (F3) (oxidize- able frac- tion) ranged: 0.15-0.317 mg kg-1, and (R) (residue) ranged 1.528- Figure 6 (b): Cr fractionation in CW sediment Figure 6 (d): Pb fractionation in CW sediment 1.690 mg kg-1. The highest average Cr fraction (1.630 ± 0.089 mg kg-1) was recorded for (R). This result may attributed metal binding ability of small particles of clay and silt particles of the sediment. As shown in Fig-6 (b), moving from cell-1 to cell-3, the abundance of chemical fractions that occurred in the form of (F1), (F3) and (R) exhibits decreasing trends due to slight decreasing in OM content of sediment that bind metals in the sediment [35].
The concentration of Cd fraction that occurred in the chemical form of (F1) in cell-1, cell-2 and cell-3 were ranged: 0.059-0.122 mg kg-1; (F2) ranged: 0.013-0.110 mg kg-1; (F3) ranged: 0.075- 0.116 mg kg-1and (R) ranged: 0.594-0.810 mg kg-1. The chemical fractions of Cd which occurred in the form of (F1) and (F3) were showed slight decreasing trends between cell-2 to cell-3 (Fig 6 (c) may be due to decrease in OM content which can bound metals. On the contrary, (R) fractions were showed increasing trends be- tween cell-1 to cell-2, which may be due to high affinity of clay and silt particles for metals [34].
The chemical fractions of Pb that occurred in the form of (F1) in cell-1, cell-2 and cell-3 were ranged: 0.284-0.649 mg kg-1; (F2) ranged: 0.151-0.612 mg kg-1; (F3) ranged: 0.357-0.770 mg kg- 1and (R) ranged: 2.785-4.60 mg kg-1. Considering the average of three cells, the highest average Pb fraction occurred in the form of (R) (3.707± 0.908 mg kg-1), followed by (F3) (0.502 ± 0.233 mg kg-1) may due to metal affinity and bound to clay and silts, and met- al bound with OM respectively. As shown in Fig 6 (d), as moving from cell-1 through cell-3, the abundance of chemical fractions occurred in the form of (F2) and (R) exhibits slightly increasing trends may be due to Pb bounding to clay soil particle structure.
Generally, the highest average fraction for Zn (1.394±0.320 mg kg-1), Cr (1.630 ± 0.089 mg kg-1); Cd (0.734 ± 0.121 mg kg-1), and Pb (3.707± 0.908 mg kg-1) were found in the chemical form of a re- sidual fraction (R). As indicated in table-4, 71.67% of Zn; 82.20% of Cr ; 76.47% of Cd and 74.96 % of Pb were bound to residual fractions (R), while lower metals fraction occurred in the form of water and acid solubale and exchangeable (F1), reduce able form (F2) and oxidizable (F3). The high residual fractions of metals are attributed to the presence of silt and clay soil in the CW sediment, which have high affinity for heavy metals. Unlike (F1), (F2) and (F3), (R) fraction held with strong crystal structure and is forming very stable matrix in the CW sediment. Toxic metals bound to (R) fraction are immobile, not bioavailable, not cause metal toxicity; due to strong bounding with clay and silt structure; hence they have positive environmental and public health safety implication.
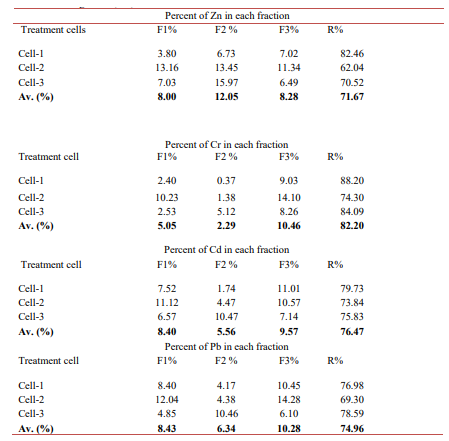
Table 4: Percentage of Zn, Cr, Cd and Pb Bound to Different Fractions in Each Treatment Cell Sediment
Bioavailability and mobility of heavy metals
Bio-available fractions of heavy metals in sediment samples
The bioavailable fractions of heavy metals in CW sediment sam- ples presented in Fig 7. The percentage of metals bound to water and acid soluble, exchangeable and carbonate (F1) were: Zn (8%); Cr (5.%); Cd (8.%) and Pb (8. %), indicating a low percentage of metals exists in readily bioavailable chemical forms. Heavy metals extracted in the first step extraction (F1) are instable and highly bioavailable [30, 6].
According to Nkinahamia, heavy metals that exist in the chemical form of (F1) are easily released into the environment [8]. Hence easy to transport, transform and are absorbed by biota and cause metal toxicity [21]. Thus, toxic heavy metals present in this form can be easily taken up by wetland plants or sediment inhibating biota; and cause hazardous to plants, animals and the ecosystem [6]. However, due to low percentage of metals that occurred in the chemical form of (F1), their adverse impacts on biota and environ- ment may not expected to be very serious.
Heavy metals bound to Fe and MN oxides form (F2) were: Zn (12%), Cr (3%), Cd (6%) and Pb (7%), are also indicating small percentage of heavy metals occurred in reducible chemical forms (F2). Meals occurred in this chemical form are less readily bio- available as compared to metals occurred in the chemical form of (F1). The percentage of metals bound to organic matter (OM) and occurred in the form (F3) were also low; Zn (8%), Cr (10%), Cd (9.57%) and Pb (10 %). Heavy metals that occluded to OM (F3) are moderately bioavailability as compared to metals bound to (F1) and (F2) fractions. Metals bound to OM are usually released when OM is decomposed or when acidic condition occurred.
Generally, low percentage of metals fractions occurred in the forms of (F1), (F2), (F3) could be attributed to the low percent- age of organic matter in the sediment [6]. Presence of clay and silt particles in the sediment scavenging trace metals and strongly bound the sediment and metals, and enhance, metals adsoption under alkaline pH condition [30, 11]. This result have environmen- tal implications; low bioavailable metal fractions decreases chance of uptake of toxic metals by plants and animals, and reduce envi- ronmental risks.
The highest percentage residual fraction ( R): Zn (72%), Cr (82%), Cd ( 76%), and Pb (75%), indicating that most heavy metals are not exist in bioavailable forms may be due to the presence of small size particle (clay and silt) sediment texture that can absorb metals [34].
In this respect, Klink reported that sediment that contains a higher percentage of small particles such as clay has a high surface area, and it has an affinity to bind metals in the sediments [33]. Authors have described that metals bound to residual fractions are not bio- available due to strong bound within the crystalline lattice structure of clay minerals by isomorphous substitution [12, 14, and 23]. The presence of heavy metals largely bound to residual fractions of the HSSF-CW sediment have positive implication for environment, because, toxic heavy metals are immobilized, stable and not reac- tive in terms of biological processes [40]. Klink have also reported that high percentage of metals that found in the chemical form of (R) implies that the CW sediment was relatively less unpolluted with heavy metals. In addition, heavy metals bound to (R) are not easily taken up by wetland plants because of strong bounding with clay structure [33].
Figure 7: Bioavailable Heavy Metal Fractions in the Sediment Samples
Mobility Factor (MF) of Heavy Metals in the Sediment Samples
The MF of heavy metals found in the HSSF-CW sediment samples are presented in Table 5.
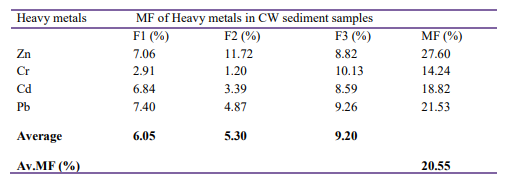
Table 5: Heavy Metals Mobile Factor for Sediment Sample
The MF of heavy metals in sediment samples were: 27.6% for Zn, 14.24% for Cr, 18.82% for Cd, and 21.53% for Pb. The average MF of all metals under the study was 20.55%, indicating low mo- bility of metals fractions. The decreasing order of heavy metals MF: Zn > Pb > Cd > Cr. Even though, the MF of all metal fractions are low, Zn and Pb are relatively more mobile than that of Cr and Cd. Hence, due to higher MF, Zn and Pb can move horizontally in the HSSF-CW sediment and cause sediment contamination, and ultimately, they pose adverse effects on wetland plants and other organisms found in the sediment.
Several authors have described that heavy metals which are weak- ly bound to water and acid soluble, exchangeable and carbonate (F1) and reducible fraction (F2) are considered to be highly mo- bile, while oxide-able fraction (F3) is moderately mobile [9, 13, 14, 34]. Thus, heavy metals which are available in the chemical forms of F1, F2, and F3 are mobile, migrate through soil and sed- iment may cause contamination of wetland treatment system and affects environmental quality when aged sediments (gravels and soils) of the HSSF-CW sediment is disposed into surrounding en- vironment [40]. However, in this study low average percentages of MF (20.55) was found. This result shows that the majority of heavy metals in sediments were not in mobile chemical forms and they are bound to residue (R) (table 4). Thus, due to the low mo- bility, toxic heavy metals may not cause high contamination of sediment, wetland plants, soil and surrounding environment.
Risk Assessment Code
The results of RAC of sediment samples are presented in Fig 8. The percent RAC for Zn, Cr, Cd and Pb in the sediment samples were: 7.67%, 5.37%, 8.51%, and 8.32%, respectively. According to Jakubus and Klink risk classification, the RAC % value for all metals fall in low risk (1% - 10%) category; indicating a lower risk to the environment [32, 33]. This implies that the majority of metals fractions found in the HSSF-CW sediments were in the chemical form of (R) farcation, and strongly bound to clay parti- cles and become immobilized. Thus, the HSSF-CW sediment may not be highly polluted with heavy metals.
Figure 8: Risk Assessment Code and Risk Classes of Sediment Samples
Principal Component Analysis
The principal component analysis (PCA) was applied to identify sources of pollutants and to describe the variations of the data set [40, 41]. The result of (PCA) is presented in Fig 9. The number of principal components (PC) to be retained was selected on the basis of scree plot [42, 43]. The extracted eigenvectors of PCA indicated that the first principal component (PC1) represents 99.7% varia- tion in the total data set of sediment samples with an eigenvalue of 3.99. The PC1 also showed that Cr, Cd and Pb were associat- ed with the residual fraction (Fig 9) may be due to high metals scavenging and strong binding capacity of clay soil. PC1 showed a high similarity with the loadings (> 0.50), indicating a similar source of Cr, Cd and Pb that generated from anthropogenic and geological source and transported through river water, but strongly bound to clay soil crystal structure in the sediment.
High loading of Zn was associated with reducible fraction, indi- cating anthropogenic source. Zn can be generated from galvanized metals, application of Zn oxide in rubber manufacturing industries and agrochemicals. The eigenvalue (0.01) of PC2 was lower than 1; hence, it has no significant contribution and ignored.
Figure 9: PCA of Sediment Samples
Conclusion
Heavy metals can be found in different chemical forms in con- structed wetlands sediments. The study conducted using HSSF- CW sediment showed that low concentration of heavy metals oc- curs in the chemical forms of (F1), (F2) and (F3) impliying that the bioavailability and mobility of metals in sediments is low. The high concetration of metals fraction bound to residucial fraction (R) indicating that most metals are strongly bound to clay soil and immobilized. The risk assessment code (RAC %) of heavy metals falls in the low risk category. Thus, it can be concluded that heavy metals in the CW sediment may not poses a serious adverse im- pacts on the surrounding environment.
Hence, heavy metals identified in HSSF-CW sediment are unlikely to cause significant sediment pollution. In general, the research has improved our understanding of heavy metal speciation in CW sed- iments or substrates, as well as, the environmental ramifications. It is therefore, highly recommended to use small amount of clay soil along with treatment media (gravel) to reduce contamination of sediment and local environment with heavy metals.
Acknowledgement
The author thank the Addis Ababa University for financial and lo- gistic support including laboratory facilities.
Funding
This research work was supported by Addis Ababa University, un- der the Thematic Research Project.
References
- Dapam, I. L., Ibrahim, E. G., & Dodo, J. D. (2018). Heavy metal speciation study of water and bottom sediments from River Wujam in Chip District of Pankshin local government area, Plateau State, Nigeria. Environmental Science and Pol- lution Research, 4, 242-245.
- Fitamo, D., Leta, S., Belay, G., Lemma, B., & Olsson, M. (2011). Phytoavailability of heavy metals and metalloids in soils irrigated with wastewater, Akaki, Ethiopia: A greenhouse study. Soil and Sediment Contamination: An International Journal, 20(7), 745-766.
- Mekuria, D. M., Kassegne, A. B., & Asfaw, S. L. (2021). As- sessing pollution profiles along Little Akaki River receiving municipal and industrial wastewaters, Central Ethiopia: Im- plications for environmental and public health safety. Heliy- on, 7(7).
- Knox, A. S., Paller, M. H., Nelson, E. A., Specht, W. L., Halv- erson, N. V., et al. (2006). Metal distribution and stability in constructed wetland sediment. Journal of Environmental Quality, 35(5), 1948-1959.
- O'Sullivan, A. D., Conlon, R., Moran, B., & Otte, M. (2005). Characterisation of constructed wetland substrates by chem- ical sequential extraction and X-ray diffraction analyses. In Biology and Environment: Proceedings of the Royal Irish Academy (Vol. 105, No. 2, pp. 87-94). Royal Irish Academy.
- Emurotu, J. E. (2020). Chemical speciation and potential mobility of some metals of selected farmland in Kogi state, North Central Nigeria. Nigerian Journal of Basic and Applied Sci- ences, 28(1), 48-55.
- Hanay, Ö., Hasar, H., Kocer, N. N., & Aslan, S. (2008). Evalu- ation for agricultural usage with speciation of heavy metals in a municipal sewage sludge. Bulletin of Environmental Con- tamination and Toxicology, 81, 42-46.
- Nkinahamira, F., Suanon, F., Chi, Q., Li, Y., Feng, M., et al. (2019). Occurrence, geochemical fractionation, and environ- mental risk assessment of major and trace elements in sewage sludge. Journal of Environmental Management, 249, 109427.
- Tytla, M., Widziewicz, K., & Zielewicz, E. (2016). Heavy metals and its chemical speciation in sewage sludge at differ- ent stages of processing. Environmental Technology, 37(7), 899-908.
- Sundberg-Jones, S. E., & Hassan, S. M. (2007). Sediment-As- sociated Elements in a Constructed Wetland Treatment Sys- tem: Distribution, Characterization, and Toxicity. Bioremedi- ation, Biodiversity and Bioavailability, 1(1), 41-55.
- Rieuwerts, J. S. (2007). The mobility and bioavailability of trace metals in tropical soils: a review. Chemical Speciation & Bioavailability, 19(2), 75-85.
- Abdallah, M. A. M. (2017). Chemical speciation and contam- ination assessment of Pb and V by sequential extraction in surface sediment off Nile Delta, Egypt. Arabian Journal of Chemistry, 10(1), 68-75.
- Abdu, N., Agbenin, J. O., & Buerkert, A. (2012). Fraction- ation and mobility of cadmium and zinc in urban vegetable gardens of Kano, Northern Nigeria. Environmental Monitor- ing and Assessment, 184, 2057-2066.
- Dabrowska, L. (2016). Fractionation of heavy metals in bot- tom sediments and sewage sludges using sequential extraction. Ecological Chemistry and Engineering. A, 23(1), 63-75.
- Jain, C. K. (2004). Metal fractionation study on bed sediments of River Yamuna, India. Water research, 38(3), 569-578.
- Tokalioglu, S., Kartal, S., & Gülteken, A. (2006). Investigation of heavy-metal uptake by vegetables growing in contaminated soils using the modified BCR sequential extraction method. Intern. J. Environ. Anal. Chem., 86(06), 417-430.
- European Commission (2001) BCR Information reference materials, the certification of the extractable content (mass fractions) of Cd Cr Cu Ni Pb and Zn in fresh water sediment following a sequential extraction procedures, BCR-701, print- ed in Belgium
- Yang, G. H., ZHU, G. Y., LI, H. L., HAN, X. M., & LI, J.M. (2018). Accumulation and bioavailability of heavy metals in a soil-wheat/maize system with long-term sewage sludge amendments. Journal of Integrative Agriculture, 17(8), 1861- 1870.
- Ryan, P. C., Hillier, S., & Wall, A. J. (2008). Stepwise effects of the BCR sequential chemical extraction procedure on dis- solution and metal release from common ferromagnesian clay minerals: a combined solution chemistry and X-ray powder diffraction study. Science of the Total Environment, 407(1), 603-614.
- Dufresne, K., Neculita, C., Brisson, J., & Genty, T. (2015, April). Metal retention mechanisms in pilot-scale constructed wetlands receiving acid mine drainage. In Proceedings of the 10th ICARD (International Conference on Acid Rock Drain- age)-IMWA (International Mine Water Association) (pp. 21- 24).
- Hei, L., Jin, P., Zhu, X., Ye, W., & Yang, Y. (2016). Charac- teristics of speciation of heavy metals in municipal sewage sludge of Guangzhou as fertilizer. Procedia Environmental Sciences, 31, 232-240.
- O'Sullivan, A. D., Moran, B. M., & Otte, M. L. (2004). Accu- mulation and fate of contaminants (Zn, Pb, Fe and S) in sub- strates of wetlands constructed for treating mine wastewater. Water, Air, and Soil Pollution, 157, 345-364.
- Peruzzi, E., Masciandaro, G., Macci, C., Doni, S., Ravelo,S. G. M., et al. (2011). Heavy metal fractionation and organ- ic matter stabilization in sewage sludge treatment wetlands. Ecological Engineering, 37(5), 771-778.
- Yang, K., Zhang, T., Shao, Y., Tian, C., Cattle, S. R., et al. (2018). Fractionation, bioaccessibility, and risk assessment of heavy metals in the soil of an urban recreational area amended with composted sewage sludge. International journal of envi- ronmental research and public health, 15(4), 613.
- Pöykiö, R., Watkins, G., & Dahl, O. (2019). Characterisation of municipal sewage sludge as a soil improver and a fertiliz- er product. Ecological Chemistry and Engineering S, 26(3), 547-557.
- Uwah, I. E., Dan, S. F., Etiuma, R. A., & Umoh, U. E. (2013).Evaluation of status of heavy metals pollution of sediments in Qua-Iboe River estuary and associated creeks, South-Eastern Nigeria. Environment and Pollution, 2(4), 110.
- Özkan, E. Y. (2012). A new assessment of heavy metal con- taminations in an eutrophicated bay (Inner Izmir Bay, Turkey). Turkish Journal of Fisheries and Aquatic Sciences, 12(1).
- Turek, A., Wieczorek, K., & Wolf, W. M. (2019). Digestion procedure and determination of heavy metals in sewage sludge—An analytical problem. Sustainability, 11(6), 1753.
- Usharani, B., & Vasudevan, N. (2016). Eco-friendly approach for leaching out heavy metals from sewage sludge. Chemistry and Ecology, 32(6), 507-519.
- Cao, L., Tian, H., Yang, J., Shi, P., Lou, Q., et al. (2015). Mul- tivariate analyses and evaluation of heavy metals by chemom- etric BCR sequential extraction method in surface sediments from Lingdingyang Bay, South China. Sustainability, 7(5), 4938-4951.
- Rumhayati, B., & Retnaningdyah, C. (2018). Integrative as- sessment of Pb and Cd pollution in Porong Estuaries using sediment chemistry, bioavailability, and bioconcentration fac- tor. Indonesian Journal of Chemistry, 18(3), 464-471.
- Jakubus, M. (2020). Changes in lead and chromium contents in sewage sludge evaluated using both single extractants and sequential method. Environmental Pollutants and Bioavail- ability, 32(1), 87-99.
- Klink, A., Dambiec, M., & Polechonska, L. (2019). Tracemetal speciation in sediments and bioaccumulation in Phrag- mites australis as tools in environmental pollution monitoring. International Journal of Environmental Science and Technol- ogy, 16(12), 7611-7622.
- Mi, Y., Zhan, F., Li, B., Qin, L., Wang, J., et al. (2018). Distribution characteristics of cadmium and lead in particle size fractions of farmland soils in a lead–zinc mine area in South- west China. Environmental Systems Research, 7(1), 1-10.
- Gambrell, R. P. (1994). Trace and toxic metals in wetlands—a review. Journal of environmental Quality, 23(5), 883-891.
- Alayu, E., & Leta, S. (2021). Performance efficiency and wa- ter quality index of a two-stage horizontal subsurface flow constructed wetland system polishing anaerobically treated brewery effluent. Journal of Water Process Engineering, 42, 102156.
- Alayu, E., & Leta, S. (2021). Post treatment of anaerobically treated brewery effluent using pilot scale horizontal subsur- face flow constructed wetland system. Bioresources and Bio- processing, 8, 1-19.
- Angassa, K., Leta, S., Mulat, W., Kloos, H., & Meers, E. (2019). Evaluation of pilot-scale constructed wetlands with Phragmites karka for phytoremediation of municipal waste- water and biomass production in Ethiopia. Environmental Processes, 6, 65-84.
- Modaihsh, A. S., Ai-Swailem, M. S., & Mahjoub, M. O. (2004). Heavy metals content of commercial inorganic fertil- izers used in the Kingdom of Saudi Arabia. Journal of Agri- cultural and Marine Sciences [JAMS], 9(1), 21-25.
- Usman, A. R., Almutairi, A. A., Elmaghraby, S., & Al-Far- raj, A. S. (2017). Levels, solid-phase fractions and sources of heavy metals at site received industrial effluents: a case study. Chemical Speciation & Bioavailability, 29(1), 78-88.
- Oketola, A. A., Adekolurejo, S. M., & Osibanjo, O. (2013). Water quality assessment of River Ogun using multivariate statistical techniques.
- Howladar, M. F., Al Numanbakth, M. A., & Faruque, M. O. (2018). An application of Water Quality Index (WQI) and multivariate statistics to evaluate the water quality around Maddhapara Granite Mining Industrial Area, Dinajpur, Ban- gladesh. Environmental Systems Research, 6, 1-18.
- Ouyang, Y. (2005). Evaluation of river water quality monitor- ing stations by principal component analysis. Water research, 39(12), 2621-2635.
- Boateng, T. K., Opoku, F., Acquaah, S. O., & Akoto, O. (2015). Pollution evaluation, sources and risk assessment of heavy metals in hand-dug wells from Ejisu-Juaben Munici- pality, Ghana. Environmental systems research, 4(1), 1-12.
- European Community (EC) (1998) Official Journal of the Eu- ropean Communities, Council Directive, on the protection of the environment, and in particular of the soil, when sewage sludge is used in agriculture, (86/278/EEC)
- Jakubus, M., & Czekala, J. (2001). Heavy metal speciation in sewage sludge. Polish Journal of Environmental Studies, 10(4), 245-250.
- Yang, J., Cao, L., Wang, J., Liu, C., Huang, C., et al. (2014). Speciation of metals and assessment of contamination in sur- face sediments from Daya Bay, South China Sea. Sustainabil- ity, 6(12), 9096-9113.


