Research Article - (2019) Volume 2, Issue 1
Health Risk Assessment of Heavy Metal Contamination in Edible Fish Species from the Barekese Reservoir in Kumasi, Ghana
2Department of Chemistry, Kwame Nkrumah University of Science and Technology, Ghana
3Environmental and Safety Engineering, University of Mines and Technology, Ghana
Received Date: Nov 15, 2019 / Accepted Date: Dec 05, 2019 / Published Date: Dec 23, 2019
Copyright: ©Copyright: �2019 Eric Gyimah, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Heavy metal pollution and its resultant accumulation to toxic levels in the Barekese reservoir may threaten human health through the consumption of fish from the Reservoir. The presence and concentrations of arsenic and some other heavy metals (Hg, Pb, Ni, Cr, and Cd) were determined in muscle tissues of widely distributed and highly consumed fish species from the Barekese reservoir using Atomic Absorption Spectrophotometry. A total of 45 fish samples comprising three species (Oreochromis niloticus, Tilapia zilli and Heterotis niloticus) were analyzed for their metal levels. Heavy metals concentrations (mg/kg, w.w) showed varying trends of detection among muscle tissues of selected fish species. However, Hg, Ni, Cr and Cd were detected in muscle tissues of all fish species examined in the study. Indices for non-carcinogenic and carcinogenic risks were estimated for child and adult upon their consumption of fish from the Reservoir. Results of the study indicate a significant level of exposure to Hg, Ni, Cr and Cd through the consumption of fish species from the Reservoir. Health risk assessment also revealed that children are at a relatively higher non-carcinogenic risk through the consumption of metals in Heterotis niloticus (HQ = 2.95 × 10-1). Also, the consumption of all examined fish species from the Reservoir pose no significant cancer risk effects to humans. The present study, therefore, concludes that anthropogenic activities along the Offin River and within the Barekese reservoir’s catchments could lead to deteriorating the quality of fish from the Reservoir hence regular monitoring of such activities is needed.
Keywords
Accumulation, Barekese, Carcinogenic risk, Fish species, Hazard index, Health risk,Introduction
Contamination of the aquatic environment by heavy metals is a predisposing factor posing serious threat to the survival of aquatic organisms including fish [1]. Hence, the occurrence of toxic chemical pollutants especially heavy metals in fluvial ecosystems resulting mainly from anthropogenic activities such as discharge of untreated domestic waste, industrial waste and improper dumping of solid waste have become a problem of global concern [2]. Some heavy metals have biochemical functions which are essential for life activities in living organisms in regulatory amounts [3]. For example, Cr plays a vital role in maintaining carbohydrate and lipid metabolism at a molecular level. The biosynthesis of hydrogenase and the formation of enzymes for protein synthesis are some physiological benefits of Ni in organisms. Absence or below the threshold levels Cr in a biological system results in associated diseases such as glucose intolerance [4]. However, heavy metals like Hg, As, Cd and Pb have no known beneficial effects in humans and are toxic even at low concentrations hence chronic exposure to these toxic metals become detrimental to human health [5]. The WHO as well as the Food and Agriculture Organization of the United Nations emphasize the regular monitoring of these toxic metals in the environment [5].
The growing concern of the nutritional and therapeutic benefits of fish have increased its rate of consumption by humans [6]. Not limited to the obvious protein source of fish, fish is a major source of omega-3 fatty acids recommended for dietary intake to promote proper cardiovascular and brain functioning [7]. However, fish accumulate both essential and toxic metals from the aquatic ecosystem, hence regular monitoring of the levels of toxic metals in fish tissues becomes important in assessing food safety [8]. Heavy metals in aquatic ecosystems can be transferred into humans through the consumption of fish [9]. Toxicity and the subsequent human health risks of any contaminants are indeed a function of its concentration [10]. However, chronic exposure to toxic metals and metalloids at comparatively low levels can result in adverse health effects in humans owing to the accumulation of metals in living tissues [11]. Therefore the analysis of heavy metals levels in fish muscles helps to determine the direct transfer of heavy metals from contaminated aquatic ecosystem to human and their associated health risk implications [12].
Heavy metal contamination in the Barekese reservoir through unregulated artisanal mining activities along the offin River (the main feeder stream of the reservoir) can increase to toxic levels and pose threats to the ecosystem and humans who depend on the reservoir’s resource [13,14]. In spite of the possible sources of metals pollution in the reservoir, metal levels of fish in the reservoir and their humans’ health implications is still not well studied [15]. The present study seeks to determine the levels of heavy metals in muscle tissues of the most abundant and highly consumed edible fish species (Oreochromis niloticus, Tilapia zilli and Heterotis niloticus) from the Barekese reservoir and assess their carcinogenic and non- carcinogenic health risk implications to consumers which is pertinent with regards to food safety.
Materials and Methods
The Barekese reservoir stretches on latitude 6º44’N and longitude 1º42’Won the Offin River in the Ashanti Region [16]. The area has a major and minor rainfall occurring from March to July and September to mid-November respectively [17]. The Barekese reservoir provides pipe borne water to the Kumasi metropolis and its environs and a source of fish on commercial scale for human consumption [15,18]. However, the reservoir has seen persistent degradation through human activities such as the artisanal mining and farming along the Offin [18]. Also, the immerse use of agro- chemicals through subsistence farming within the reservoir’s catchment has been detrimental to the quality of the quality of the reservoir’s resources.
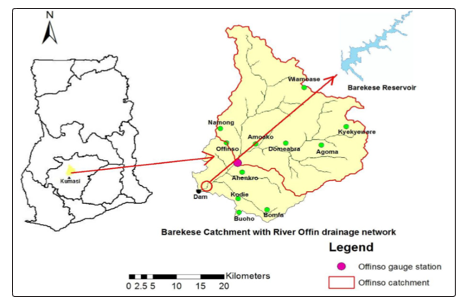
Figure 1: Location of Barekese Reservoir, dam and catchment area with Offin River drainage
Source: (Domfeh et al., 2015).
The geology of the Reservoir catchments consists of 97% Dahomeyan and 3% Upper Voltain with granitoid undifferentiated [17]. The Reservoir lies in a moist semi-deciduous forest and the forest vegetation provides livelihood for populate within the catchments through subsistence farming [14].
Sampling and sample preparation
Fifteen each of three edible fish species; Oreochromis nioticus (Nile tilapia), Tilapia zilli (Red belly tilapia) and Heterotis niloticus (Bony tongue) were purchased from fishermen at the landing site of the reservoir. The selection of the fish species was based on their abundant in the reservoir and their high consumption rate by human. The pre-treatment processes of collected fish samples are described in our previous study [15]. Briefly, portions of edible muscle tissues were removed from the dorsal portion into different sterile containers with the help of a sterile dissection knife. The content homogenized and a portion was taken for chemical analysis.
Sample digestion and analysis
Fish samples were digested for total metal determination by an open flask procedure develop at the National Institute for Minamata Disease (NIMD) in Japan by Akagi and Nishimura,1991 [19]. Briefly, 1.0 g each of homogenized fish sample was accurately weighed into digestion tubes, 2 mL of distilled water added followed by 2 mL HClO4-HNO3 (1:1 v/v) and 5 mL H2SO4. Each mixture was refluxed at 200 °C for about 30 min in a clean fume chamber. Complete digestion was observed when an initial yellowish vapor ceased. The digested fish samples were allowed to cool and filtered into 50 mL volumetric flasks and made to the mark with double distilled water and, transferred into a sterile screw capped plastic container, labeled, stored at 4 °C until needed for analysis.
Heavy metal analysis
Heavy metals (Hg, As, Cd, Cr, Ni and Pb) concentrations in the digested fish samples were determined using Perkin Elmer PIN Accle 900T Graphite Atomic Absorption Spectrophometer as previously described by [15]. The instrument was calibrated with series of standard solutions in accordance with the manufacture’s instruction.
Analytical performance
All chemicals and standard stock solutions were products of Sigma- Aldrich, Germany and conform to the specifications of the committee on Analytical Reagent grade of American Chemical Society unless otherwise stated. Also, double distilled water was used for the preparation of all solutions. Calibration of the analytical instrument is described in our previous study [15]. Briefly, calibration of the AAS was based on a linear five-point calibration curves with r2 values between 0.999 and 1.000 for each element. The method was validated using certified reference material (CRM), ISE 999, for Hg, As, Cd, Cr, Ni and Pb. Replicate analysis of the CRM showed a good accuracy with relative standard deviation ≤ 4% and a recovery rate ranged from 85% to 105%. Limit of detections (LOD) were determined using elemental standards in dilute aqueous solution. The LOD for each metal on the basis of 98% confidence in mg/L were as (0.03), Cd (0.002), Cr (0.004), Ni (0.07) and Pb (0.05). The LOD of Hg was 9.0 pg/L.
Human Health Risk assessment of Heavy Metals in Fish
Probable adverse health effects on humans upon exposure to heavy metals in fish were estimated using the USEPA risk assessment model and its threshold values [20].
Exposure Assessment
Exposure assessments of analyzed metals via the consumption of examined fish species were estimated. In this study, the metal’s concentrations in examined fish species were used for the estimation of average daily dose (ADD) in mg/kg/day via consumption of fish from the Reservoir using the models proposed by USEPA, 2012. Exposure assessments were separately estimated for children and adult using equation 2
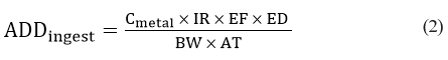
Where IR is the ingestion rate, EF is the exposure frequency (days/ year), ED is the exposure duration (years), BW is body weight in Kg and AT is the averaging time of exposure in years (life expectancy).
Reference dose (RfD) and Cancer slope factor (CSF) were used to estimate the non-carcinogenic and carcinogenic health risk effect respectively [20].
Table 1 shows the exposure parameters used for the exposure assessment via fish consumption by children and adult.
Non-carcinogenic effect
The non-carcinogenic effect of the individual metal is described by hazard quotient (HQ). The HQ was estimated using equation 3.

Where ADD and RfD are average daily dose and reference dose respectively. Hazard index (HI), is a combined toxic effects of metals on human health was calculated using equation 4 to estimated non-carcinogenic risk on humans (Akoto et al., 2014).

Where HQi represent the hazard quotient for the ith metal. Hazard Index (HI) > 1 denotes that the non-carcinogenic adverse health effect of the chemical should investigated further whereas HI ≤ 1 denotes a possible screened out without further investigation of adverse health effect associated with the contaminants.
Table1: Exposure parameters used for the health risk estimations through consumption of fish (USEPA, 2012)
|
|
|
value |
|
|
Parameter |
Unit |
Child |
Adult |
|
Body weight (BW) |
Kg |
15 |
70 |
|
Exposure frequency (EF) |
days/ years |
365 |
365 |
|
Exposure duration (ED) |
years |
6 |
30 |
|
Ingestion rate (IRfish) |
mg/day |
200 |
100 |
|
Average time (AT) |
days/ years |
|
|
|
For carcinogenic |
|
365 × 70 |
366 × 70 |
|
For non-carcinogenic |
|
365 × ED |
365 × ED |
Carcinogenic risk assessment
Carcinogenic risk assessment estimates the probability of an individual developing cancer over a lifetime due to exposure to the potential carcinogen. In this assessment, a cancer slope factor (CSF) was used to convert the ADD of the heavy metal over a lifetime of exposure to risk of an individual developing cancer [22]. Cancer risk was calculated using equation 5.
Cancer Risk = ADD×CSF (5)
Table 2 Reference dose and cancer slope factor for different heavy metals (USEPA, 2012)
|
Heavy metal |
Reference dose |
Cancer slope factor |
|
Hg |
3.0 × 10-4 |
NA |
|
Pb |
3.0 × 10-3 |
NA |
|
As |
3.0 × 10-4 |
1.50 |
|
Ni |
2.0 × 10-2 |
NA |
|
Cr |
3.0 × 10-3 |
NA |
|
Cd |
5.0 × 10-4 |
NA |
*NA: Not Available at the time of study
Statistical analysis
Statistical analyses of data were performed using IBM SPSS version 20.0 statistical software package. Data were, normalized by log transformation, expressed in mean ± standard deviation (SD). Evaluation of significant differences of analyzed metals levels within groups and between groups of examined species were estimated using inferential statistics (ANOVA). Results of testing were considered significant at P ≤ 0.05.
Results and Discussion
Heavy metal concentrations in fish
The coefficient of condition (K) obtained for all samples of the various fish Species indicate that all the fish samples used in the study were healthy (K > 1) as reported earlier in our previous studies [15]. It, therefore, infers that the fish species obtained from the Barekese reservoir used in the study were in healthy conditions in their habitats. The mean concentrations (mg/kg) wet weight ± SD of heavy metals in muscle tissues of the examined fish species are presented in Table 3. Mean concentrations were used to calculate the average daily dose for non-carcinogenic and carcinogenic risk effect.
Table 3: Mean Concentrations of heavy metals (mg/Kg) in muscles of fish species from the Barekese Reservoir (n=15)
|
Fish Species |
|||
|
Heavy Metals |
Oreochromis niloticus |
Tilapia Zilli |
Heterotis niloticus |
|
Hg |
0.56±0.03 |
0.91±0.51 |
1.21±0.66 |
|
Pb |
0.8±0.25 |
< 0.005* |
<0.005* |
|
As |
0.08±0.03 |
0.03±0.04 |
<0.003* |
|
Ni |
24.00±0.03 |
18.45±0.04 |
18.10±5.13 |
|
Cr |
7.00±1.50 |
6.93±2.23 |
5.68±1.13 |
|
Cd |
6.02±1.03 |
6.09±0.83 |
11.05±7.85 |
Values in * are the detection limit of the analytical instrument for the respective metals.
The mean concentrations (mg/kg) of Hg in examined species arranged in descending order as Heterotis niloticus > Tilapia zilli > Oreochromis niloticus (Table 3). However, all fish species analyzed in this study had mean total mercury concentrations above the FAO/ WHO permissible level of 0.5 mg/kg wet weight (FAO/WHO, 2013). High consumption of Hg results in irreversible damages, such as neurological impairment, lesions, behavioral and cognitive changes [23]. Variation of Hg levels detected in the muscle tissues of examined species were statistically significant at p < 0.05 level. Result obtained in this study is lower than that reported by Oppong et al (2010) in a study on total mercury in fish muscles from the River Pra Basin in Ghana [19]. The artisanal gold mining along the Offin River as well as the use of mercurial compounds in agricultural activities within the catchments of the Reservoir could be potential sources of Hg pollution in the Barekese reservoir.
The legislated regulations on permissible limits of Pb in fish according to EU (2006) is 0.2 mg/kg wet weight. Concentrations of Pb were not detected in the muscle tissues of Heterotis niloticus and Tilapia zilli in the study. However, mean Pb concentration 0.8±0.25 mg/kg wet weight recorded in Oreochromis niloticus exceeded the EU permissible limit. Orechromis niloticus is a benthopelagic species which feeds mainly on phytoplankton or benthic algae [24]. The concentration of Pb recorded in this study was lower than the findings of Akoto et al. (2014) where a high Pb concentration of 6.82±2.28 mg/kg was recorded in a study on concentrations of heavy metals in fish from the Fosu Lagoon in Ghana [21]. Lead deplete sulfhydryl containing antioxidants and enzymes in the cell hence increasing reactive oxygen species (ROS) production leading to various dysfunctions in lipids, proteins and DNA [23]. One might suffer these dysfunctions by eating Oreochromis niloticus from the Barekese Reservoir.
Arsenic residues were not detected in muscle tissues of Heterotis niloticus in this study. The recorded mean concentrations in Oreochromis niloticus and Tilapia zilli were 0.08±0.03 mg/kg and 0.03±0.04 mg/kg wet weight respectively. Variation in as concentrations in muscle tissues of Oreochromis niloticus and Tilapia zilli were statistically not significant at p < 0.05 level. The concentration of As recorded in the muscles of examined fish species in this study were relatively lower than that reported by Coffie (2014), a study on heavy metals assessment in muscles of Oreochromis niloticus form the Volta Lake in Ghana [25]. In contrast, the recorded mean as concentrations in muscle tissues of fish in this study is exceedingly higher than 0.009 mg/Kg wet weight in muscle tissues of Oreochromis niloticus reported by Kwansah-Ansah et al (2012), a study at the Volta Lake. Skin lesions, malfunctioning of renal and reproductive systems have been linked to too much intake of As [10].
Levels of Ni recorded among the fish species were in the order Oreochromis niloticus > Tilapia zilli > Heterotis niloticus with wet weight mean concentrations 24.00±0.03 mg/kg, 18.40±0.04 mg/kg and 18.10±5.13 mg/kg respectively (Table 3). The United States Food and Drug Administration, USFDA (1993) estimated the maximum guideline for Ni to be 70–80 mg/kg wet weight [26]. Based on this regulation, Ni concentrations recorded in all examined fish species reported in this study were within the USFDA set values. However, Ni concentrations in muscle tissues of all examined fish species reported in this study were far higher than that reported by Akoto et.al (2014), a study on the concentrations of heavy metals in fish from the Fosu Lagoon in Ghana [21]. Even though the concentrations of Ni recorded in muscle tissues of all examined fish species in this study were within the set USFDA values, regular monitoring of Ni residues in the Barekese reservoir is necessary in order to ensure food safety.
Chromium is biologically essential for the metabolism of carbohydrate [27]. The wet weight mean concentrations of Cr in muscle tissues of examined fish species were 7.00±0.50 mg/kg, 6.93±2.23mg/kg and 5.68±5.13 mg/kg for Oreochromis niloticus, Tilapia zilli and Heterotis niloticus respectively (Table 3). Variation in the mean concentrations of Cr in the muscle tissues of examined fish species were not statistically significant at p < 0.05 level among the different species. Concentrations of Cr in muscle tissues of all examined fish species recorded in this study were below the USFDA (1993) set limit of 12-13 mg/kg. Other studies have reported lower levels of Cr in muscles of fish in Ghana. For example, the concentration of Cr reported by Akoto et al (2014) in muscle tissues of fish from the Fosu Lagoon was below detection of the analytical instrument used in that study. Kwansah – Ansah et al. (2012) also reported a mean concentration of Cr ranging from 0.68–1.74 µg/g in examined fish species from the Volta Lake [28].
Cadmium is among the toxic metals that have no known biochemical importance to humans [21]. Variation in the mean concentrations of Cd in the examined fish species in this present study were statistically significant at p < 0.05 within the different species. Heterotis niloticos recorded the highest Cd mean concentration of 11.05±7.85 mg/kg wet weight whiles Oreochromis niloticus recorded the lowest value of 6.02±1.03 mg/kg wet weight (Table 3). Cadmium concentrations in all examined fish species in the study were exceedingly higher than the stipulated limit (0.05 mg/kg) set by the European Union (2006) [29]. Consumption of fish with high concentration of Cd (> 0.05 mg/kg) could pose threat such as Lung cancer, Osteoporosis and increased blood pressure in humans [30,31]. The result of the present study can be a basis for predicting severe chronic Cd poisoning via the consumption of Heterotis niloticos, Oreochromis niloticus and Tilapia zilli from the Barekese reservoir.
Health risk assessment
Non-carcinogenic risk
Table 4 presents the average daily intakes of Hg, As, Pb, Ni, Cr and Cd by both children and adults through consumption of the various fish species from the Reservoir. The ADD were remarkably below the Reference dose (RfD) values recommended by the USEPA (2012). Of all the estimated ADD for the metals considered in this study, Ni recorded the highest values. Although the concentrations of Hg and Pb detected in the muscle tissues of examined fish species exceeded their respective European regulation, the ADD calculations show that these metals do not pose risk to human since their estimated ADD values are below the RfD values (Table 2). However, the potential accumulation of heavy metals to toxic levels in fish makes it necessary for regular monitoring of metal contaminants in the Barekese reservoir. This could help contribute to food safety and facilitate the evaluation of possible health hazard to human via fish consumption from the reservoir.
Table 4: Average Daily Dose (mg/kg/day) of Heavy Metals in Fish Species for Non-Carcinogenic Risk Assessment
|
Average daily dose/ mg/kg/day |
||||||
|
|
Oreochromis niloticus |
Tilapia zilli |
Heterotis niloticus |
|||
|
Metal |
Child |
Adult |
Child |
Adult |
Child |
Adult |
|
Hg |
7.48E-06 |
1.60E-07 |
1.21E-06 |
2.60E-06 |
1.61E-05 |
3.46E-07 |
|
Pb |
1.04E-06 |
2.24E-07 |
- |
- |
- |
- |
|
As |
1.07E-06 |
2.29E-07 |
4.00E-07 |
8.57E-08 |
- |
- |
|
Ni |
3.19E-04 |
6.83E-05 |
2.41E-04 |
5.17E-05 |
2.41E-04 |
5.17E-05 |
|
Cr |
9.19E-05 |
1.97E-05 |
9.24E-05 |
1.98E-05 |
7.57E-05 |
1.62E-05 |
|
Cd |
8.02E-05 |
1.72E-05 |
8.62E-05 |
1.74E-05 |
1.47E-04 |
3.16E-05 |
The Hazard Quotients (HQs) of metals through the consumptions of fish species from the Barekese reservoir are presented in Table 5. The results showed that the HQ values of heavy metals in fish consumed by children and adult for all examined species were less than one (1). Hence there is no non-carcinogenic health risk to the population through the consumption of fish from the Barekese Reservoir. The additive effect of contaminants to the population for non-carcinogenic risk is necessary in predicting their possible effects on humans. The Hazard Index (HI) of the various metals considered in this study for all examined fish species revealed that all HI were less than 1. This indicates no adverse non-carcinogenic health risk to humans through the consumption of the metals in all examined fish species from the Barekese reservoir. However, Cd was the major contributor for HI among all examined fish species with an indicative that Children are at a relatively high non-carcinogenic risk via the consumption of Cd in Heterotis niloticus.
Table 5: Hazard Quotient of Heavy Metals through Consumption of Fish Species for Non- Carcinogenic Risk Assessment
|
Hazard Quotient |
||||||
|
|
Oreochromis niloticus |
Tilapia zilli |
Heterotis niloticus |
|||
|
Metal |
Child |
Adults |
Child |
Adults |
Child |
Adults |
|
Hg |
2.49E-02 |
5.34E-03 |
4.04E-02 |
8.67E-03 |
5.38E-02 |
1.15E-02 |
|
Pb |
3.48E-04 |
7.46E-04 |
|
- |
- |
- |
|
As |
3.56E-03 |
7.62E-04 |
1.33E-03 |
2.86E-04 |
- |
- |
|
Ni |
1.59E-02 |
3.42E-04 |
1.21E-02 |
2.59E-03 |
1.21E-02 |
2.59E-03 |
|
Cr |
3.06E-02 |
6.57E-03 |
3.08E-02 |
6.60E-03 |
2.52E-02 |
5.41E-03 |
|
Cd |
1.60E-01 |
3.44E-02 |
1.62E-01 |
3.48E-02 |
2.95E-01 |
6.31E-02 |
|
HI |
2.35E-01 |
4.82E-02 |
2.06E-01 |
4.43E-02 |
3.86E-01 |
8.26E-02 |
The present study, therefore, indicates a relatively higher potential health risk to humans via the consumption of Cd in the examined fish species from the Barekese reservoir.
Carcinogenic risk
The ADD for carcinogenic risk was calculated and presented in Table 6. Based on the carcinogenic risk of the calculated ADD for carcinogenicity (Table 6), cancer risk was calculated using the respective cancer slope factors of individual metals and the results presented in Table 7. Cancer risk regulation set by the USEPA ranged from 1.0 × 10-6 to 1.0 × 10-4 (USEPA, 2012). The results of the study showed that the estimated cancer risk for Pb and as through the consumption of examined fish species from the Barekese Reservoir were within the USEPA regulations. However, consumption of Cr from all examined fish species from the Reservoir could pose cancer risk effects to humans with adults being at a higher risk
Table 6: Average daily dose of Heavy Metals in Fish Species for Children and Adults for carcinogenic risk and cancer risk for arsenic
|
Average daily dose/ mg/kg/day |
||||||
|
|
Oreochromis niloticus |
Tilapia zilli |
Heterotis niloticus |
|||
|
Metal |
Child |
Adult |
Child |
Adult |
Child |
Adult |
|
Hg |
6.41E-07 |
3.21E-06 |
1.04E-06 |
5.20E-06 |
1.38E-06 |
6.91E-06 |
|
Pb |
8.95E-08 |
4.48E-07 |
- |
- |
- |
- |
|
As |
9.14E-08 |
4.57E-07 |
3.43E-08 |
1.71E-07 |
- |
- |
|
Ni |
2.73E-05 |
1.37E-04 |
2.07E-05 |
1.03E-04 |
2.07E-05 |
1.03E-04 |
|
Cr |
7.88E-06 |
3.94E-05 |
7.92E-06 |
3.96E-05 |
6.49E-06 |
3.25E-05 |
|
Cd |
6.87E-06 |
3.44E-05 |
6.96E-06 |
3.48E-05 |
1.26E-05 |
6.31E-05 |
|
Cancer risk |
|
|
|
|
|
|
|
As |
1.37E-07 |
6.86E-07 |
5.14E-08 |
2.57E-07 |
- |
- |
Carcinogenic risk of heavy metals is an additive effect of the individual metals contributing to the cancer risk. Results of this study indicates that the sum of cancer risks of the individual metals for the examined species could pose cancer risk effect to both children and adults through the consumption of examined fish species contaminated with Pb, As and Cr from the Barekese reservoir.
Conclusion
Heavy metals analysis showed that the mean concentrations of essential metals Ni and Cr in muscle tissues of all the examined fish species used in the study were within EU set limits. However, levels of toxic metals (Hg, Cd, Pb and as) in muscle tissues of all examined fish species from the Barekese reservoir were above stipulated limits. Human health risk assessment from heavy metal exposure through fish consumption from the reservoir for both children and adults showed no significant non-carcinogenic adverse health risk to humans. However, consumption of As, Cr and Pb in examined fish species from the reservoir could pose carcinogenic risk to both children and adults. The present study therefore reveals that the artisanal mining activity along the Offin River and the excessive use of agro-chemicals within the catchments of the Barekese reservoir are deteriorating the quality of fish from the reservoir [32,33].
Acknowledgement
The authors of the study are grateful to the Ghana Water Company Limited, Barekese for their kind courtesies and for permitting the use of their resources for this study. Also, the effort of Mr. Prince Owusu of Ecological laboratory, University of Ghana, Ghana for his assistance in the laboratory analysis of the fish samples is duly acknowledged.
References
- Coxon T, Odhiambo B, Giancarlo L (2016) The impact of urban expansion and agricultural legacies on trace metal accumulation in fluvial and lacustrine sediments of the lower Chesapeake Bay basin USA. Sci Total Environ 568: 402-414.
- Diop C, Dewaelé D, Cazier F, Diouf A, Ouddane B (2015) Assessment of trace metals contamination level, bioavailability and toxicity in sediments from Dakar coast and Saint Louis estuary in Senegal, West Africa. Chemosphere 138: 980-987.
- Akoto O, Bruce T, Darko D (2008) Heavy metals pollution profiles in streams serving the Owabi reservoir. African Journal of Environmental Science and Technology 2: 354-359.
- Mahurpawar M (2015) Effects of heavy metals on human health. International Journal of Research –GRANTHAALAYAH 530: 1-7.
- Nkansah MA, Ansah, JK (2014) Determination of Cd, Hg, As, Cr and Pb levels in meat from the Kumasi Central Abattoir. International Journal of Scientific and Research Publications 4: 1-4.
- El-Moselhy KM, Othman A , Abd El-Azem H, El-Metwally (2014) Bioaccumulation of heavy metals in some tissues of fish in the Red Sea, Egypt. Egyptian Journal of Basic and Applied Sciences 1: 97-105.
- Staniskiene B, Matusevicius P, Budreckiene R, Skibniewska K (2006) Distribution of Heavy Metals in Tissues of Freshwater Fish in Lithuania. Polish Journal of Environmental Studies 15.
- Murtala BA, Abdul WO, Akinyemi A (2012) Bioaccumulation of heavy metals in fish (Hydrocynus forskahlii, Hyperopisus bebe occidentalis and Clarias gariepinus) organs in downstream Ogun coastal water, Nigeria. Journal of Agricultural Science 4: 51.
- Karadede-Akin H, Ünlü E (2007) Heavy metal concentrations in water, sediment, fish and some benthic organisms from Tigris River, Turkey. Environ Monit Assess 131: 323-337.
- Lin Q, Liu E, Zhang E, Li K, Shen JJC (2016) Spatial distribution, contamination and ecological risk assessment of heavy metals in surface sediments of Erhai Lake, a large eutrophic plateau lake in southwest China. CATENA 145: 193-203.
- Morais S, Costa FG, Pereira M d (2012) Heavy metals and human health. Environmental Health – Emerging Issues and Practice 10: 227-246.
- El Nemr A, El-Said GF, Khaled A, Ragab S (2016) Distribution and ecological risk assessment of some heavy metals in coastal surface sediments along the Red Sea, Egypt. International Journal of Sediment Research 31:164-172.
- Jayaprakash M, Kumar R S, Giridharan L, Sujitha S, Sarkar S, et al. (2015) Bioaccumulation of metals in fish species from water and sediments in macrotidal Ennore creek, Chennai, SE coast of India. A metropolitan city effect 120: 243-255.
- Akoto O, Gyimah E, Zhan Z, Xu H, Nimako C,et al. (2019) Evaluation of health risks associated with trace metal exposure in water from the Barekese reservoir in Kumasi, Ghana.Human and Ecological Risk Assessment: An International Journal 1-15.
- Gyimah E, Akoto O, Mensah J K, Bortey-Sam N (2018) Bioaccumulation factors and multivariate analysis of heavy metals of three edible fish species from the Barekese reservoir in Kumasi, Ghana. Environ Monit Assess 190: 553.
- Blokhuis M, Brouwer R, Hulscher R, Thiadens A J D R P N R (2005) Feasibility study of Barekese water expansion project, Kumasi, Ghana 73.
- Domfeh M, Anyemedu F, Anornu G, Adjei K, Odai S, et al. (2015) Assessment of the water balance of the Barekese reservoir in Kumasi, Ghana. Journal of Science and Technology 35: 34-51.
- Kumasi T, Obiri-Danso K, Ephraim J (2007) Impacts of land-use change on the water quality of the main source of pipe borne water for Kumasi, Ghana (a case study of the Barekese reservoir catchment). Proceedings of conference on water management challenges in global change, Leicester, UK 243-248.
- Oppong S, Voegborlo R, Agorku S, Adimado A (2010) Total mercury in fish, sediments and soil from the River Pra Basin, southwestern Ghana. Bull Environ Contam Toxicol 85: 324- 329.
- Kamunda C, Mathuthu M, Madhuku M (2016) Health risk assessment of heavy metals in soils from Witwatersrand gold mining basin, South Africa. Int J Environ Res Public Health 13: 663.
- Akoto O, Bismark Eshun F, Darko G, Adei E (2014) Concentrations and health risk assessments of heavy metals in fish from the Fosu Lagoon. Int J Environ Res 8: 403-410.
- USEPA (2012) Edition of the Drinking Water Standards and Health Advisories. 2012 Edition of the Drinking Water Standards and Health Advisories 2-6.
- Authman MM, Zaki MS, Khallaf EA, Abbas HH (2015) Use of fish as bio-indicator of the effects of heavy metals pollution. Journal of Aquaculture Research & Development 6: 1.
- Froese R, Pauly D (2012) Fishbase (www database). World Wide Web Electronic Publications.
- Coffie J (2015) Assessment of physicochemical properties and heavy metals in water, sediment and fish (oreochromis niloticus) from the Upper Volta basin (stratum vii)Ghana.
- USFDA (1993) Food and drug administration. Guidance document for nickel in shellfish. DHHS/PHS/FDA/CFSAN/ Office of Seafood, Washington. D C.
- Pacheco M, Santos M, Pereira P, Martínez J, Alonso P (2013) EPR detection of paramagnetic chromium in liver of fish (Anguilla anguilla) treated with dichromate (VI) and associated oxidative stress responses-Contribution to elucidation of toxicity mechanisms. Comp Biochem Physiol C Toxicol Pharmacol 157: 132-140.
- Kwaansa-Ansah E, Akoto J, Adimado A, Nam D (2012) Determination of toxic and essential elements in tilapia species from the Volta lake with inductively coupled plasma–mass spectrometry. International Journal of Environmental Protection.
- Official Journal of the European Union (2006) Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs 364.
- ATSDR (2005) Toxicological Profile for Nickel. U.S Public Health Service, Agency for Toxic Substances and Disease Registry (August) 397.
- Saha N, Zaman M R (2013) Evaluation of possible health risks of heavy metals by consumption of foodstuffs available in the central market of Rajshahi City, Bangladesh. Environmental monitoring and assessment 185: 3867-3878.
- Gyampoh B, Amisah S, Idinoba M, Nkem J (2009) Using traditional knowledge to cope with climate change in rural Ghana. Center for International Forestry Research Center for International Forestry Research 60: 70-74.
- Zheng N, Wang Q, Zhang X, Zheng D, Zhang Z,et al. (2007). Population health risk due to dietary intake of heavy metals in the industrial area of Huludao city, China. Science of the Total Environment 387: 96-104.


