Research Article - (2020) Volume 2, Issue 2
Haemangioma of the Penis: A Review and Update of the Literature
Received Date: Feb 24, 2020 / Accepted Date: Feb 28, 2020 / Published Date: Apr 27, 2019
Copyright: ©Anthony Kodzo-Grey Venyo. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Haemangioma of the penis (HOP) is a rare benign vascular lesion which can affect the penis of males including infants, children, and adults of up to the over 80 year-olds. HOP does manifest as: asymptomatic lump or lesion on the penis; localised pain or tenderness anywhere within the penis; a tumoral mass anywhere on the penis; pain in the penis on erection; curvature of penis on erection; bright red compressible papule or papules on penis; nodules on penis; plaques on penis; ulceration on penis; haemorrhage on penis. HOP may be associated erectile dysfunction in some cases and could affect voiding if it is associated with the urethral meatus. HOP which tends to be solitary in most cases may mimic other diseases of the penis as well as may be associated with haemangioma in the scrotum and perineal region occasionally. Diagnosis can be established by the clinical features and this can be reaffirmed by radiology imaging features with the use of Doppler ultrasound scan, CT scan and MRI scan which show low-flow within the lesion. Majority of the lesions tend to be superficial and not associated with the corpora cavernosa and spongiosum. There is no consensus opinion on the best treatment options hence various treatment options for HOP have been successfully utilized including: complete surgical excision, sclerotherapy, laser treatment, cryotherapy, and other therapies. Most cases of HOP do not recur but following sclerotherapy and laser treatment of large HOPs repeat procedures tend to be required and there may be residual / recurrent lesion that may need to be treated again. Deep and large HOPs may require complete excision with insertion of tunica graft. Very large HOPs could be more effectively treated by one stage complete surgical excision but the choice of the patient is important. The diagnosis of the lesion can be confirmed by the histopathology features of the lesion and positive immunohistochemistry staining for CD31 (strong), Factor VIII, and HHF35 (myopericytes) and minimal staining for CD34. HOP needs to be carefully differentiated from epithelioid angiosarcoma of the penis and epithelioid haemangioendothelioma. A multi-centre trial of various treatment options for HOP would be required to provide treatment guidelines for HOP taking into consideration the size of the lesion and availability of the various treatment options.
Keywords
Haemangioma, Penis, Glans, Shaft, Root, Corpus Cavernosum, Corpus Spongiosum, Urethra, Meatus, Perineum, Surgical Excision, Sclerotherapy, Laser Therapy, Cavernous, Epithelioid, Capillary, Recurrence, Outcome
Introduction
Haemangiomas are benign vascular malformations of enlarged dysplastic vascular channels that contain abnormal growth of endothelial cells [1]. It has been iterated that haemangiomas had been classified into capillary, cavernous, arteriovenous, venous and mixed sub-types [1]. It has been stated that cavernous and mixed types of haemangiomas are the most common haemangiomas [1,2]. It has been stipulated that haemangiomas tend to be most commonly reported within the musculoskeletal system, the liver, and spleen [3]. It has additionally been documented that haemangiomas of the penis are very uncommon as well as the localization of haemangiomas upon the dorsum of the penis are even rarer [1,4]. It has been documented that on the whole haemangiomas of the penis tend to be small with regard to size, and clinically they tend to be irrelevant lesions except for the fact that complaints tended to be made about the aesthetic aspects of the lesion and the possibility of bleeding from the lesion during coital activity [1,2]. Various types of haemangioma of the penis have been reported including epithelioid haemangioma, capillary haemangioma, and cavernous haemangioma. Haemangiomas have been referred to as either congenital or infantile. The aetiology of dorsal haemangioma of the penis has not been clearly clarified or understood. Some people are of the opinion that haemangiomas should be considered as congenital vascular anomaly or a benign vascular neoplasm but other people have the opinion that haemangioma of the penis could emanate from herniation of the cavernous tissue and on the other hand other people are of the opinion that haemangiomas of the penis could grow up through the process of revascularization of previous haematoma of the penis [5-10]. Some of the documented treatment options that had been utilized for haemangioma of the penis include: surgical excision, electrofulguration, cryotherapy, sclerotherapy, and neodymium: yttrium aluminium garnet (Nd:YAG) laser application [11-13]. Intralesional sclerotherapy and laser fulguration have been utilized in the treatment of haemangioma of the penis. It has been stated that all non-operative treatment options tend to have their limitations, especially with regard to scenarios of attempts to resolve large haemangioma lesions on the dorsum of the penis that are greater than 1 cm in their diameter [3,8,14]. Radiology imaging including ultrasound scanning, computed tomography scanning, and magnetic resonance imaging is available in many developed countries to evaluate haemangiomas of the penis but in some developing countries radiology imaging is not readily available therefore clinicians would have to treat some haemangiomas of the penis based upon their clinical examination findings. Angiography techniques are available to evaluate haemangiomas of the penis in the developed countries but in many hospital centres in some developing countries angiography services are not available. Considering that haemangiomas of the penis are rare, it would be envisaged that many clinicians would not have encountered the lesion before and they may not be familiar with the management of the lesion. The ensuing literature review and update of the literature on haemangioma of the penis is divided into two parts (A) Overview and (B) Miscellaneous narrations and discussions from some case reports, case series and studies related to haemangioma of the penis with illustrations as well as a table of many reported cases.
Aim
To review and update the literature on haemangioma of the penis
Method
Internet data bases were used including: Google, Google Scholar, Yahoo, and PUBMED. The search words that were used included: Haemangioma of Penis; Penile haemangioma; Hemangioma of penis; penile hemangioma; Haemangioma of glans; Hemangioma of glans. Sixty three references were identified out of which 29 references were used to write the article which has been divided into two parts (A) Overview, and (B) Miscellaneous narrations from some case reports, case series and studies related to haemangioma of the penis. Further details related to the available experiences gained in the management of haemangiomas of the penis obtained from fifty one sources obtained from case reports, case series and studies related to haemangioma of the penis have been embodied into table 1 to provide additional details related to haemangioma of the penis that have not been covered in the main text.
Results
(A) Overview
Definition / general comments
• Haemangioma of the penis (HOP) is a very uncommon lesion of the penis
• Haemangioma of the penis can affect males of various age groups
Terminologies
• Some of the terminologies that have been utilized for haemangioma of the penis include: [15]
o Angioblastic (lymphoid) hyperplasia with eosinophilia
o Atypical haemangioma
o Inflammatory angiomatoid nodule
o Intravenous atypical vascular proliferation
Epidemiology
It had been stated that the reported ages of individuals who had been diagnosed as having haemangioma of the penis had ranged between 23 years and 75 years and the documented mean age of individuals diagnosed as having HOP was 45 years [15] Nevertheless HOP has been reported in neonates or in the under one year age group and in adults up to over the 80 years group.
Sites
• It has been stated that HOP mainly does affect the shaft of the penis but it also does affect the glans and root of the penis [15]
• HOP does affect the dorsum of the penis, the glans of the penis, the main shaft of the penis, the root of the penis, the urethral meatus, and on rare occasions the ventral aspect of the penis.
• HOP can also be diagnosed in association with its extension to the scrotum, the perineum, and the peri-anal region.
Aetiology
• It has been stated that HOP could possibly be a trauma related emanation [15]
• It has been documented that HOP could be a congenital vascular anomaly
• It has also been stated that HOP could be an emanation of revascularisation of a previous harmatoma
Clinical features
• A patient who has HOP could present with:
o Asymptomatic lump or lesion on the penis
o Localised pain or tenderness anywhere within the penis
o A tumoral mass anywhere on the penis
o Pain in the penis on erection
o Curvature of penis on erection
o Bright red compressible papule or papules on penis
o Nodules on penis
o Plaques on penis
o Ulceration on penis
o Haemorrhage on penis
Clinical examination findings
The general and systematic examinations would generally tend to be normal but at times the general and systematic examinations would reveal non-specific findings related to the other medical conditions the patient may have. Nevertheless, examination of the penis may reveal:
• Indurated sub-cutaneous nodules on the penis.
• Swelling on the penis
• An elevated irregular lesion on the penis which could be seen to have enlarged during erection.
• A bluish-red lesion with a smooth surface and irregular margin that is compressible and non- tender, as well as non-compressible on the penis.
• A large bosselated, soft bag of worm-like, superficial non-pulsatile swelling on the glans penis and shaft of the penis circumferentially and which could be seen as extending into the scrotum.
• Tender or non-tender palpable masses within the scrotum and shaft of the penis that may simulate a bag of worms.
• A firm nodule on the penis.
• An elevated, irregular bluish lesion on the glans and shaft of the penis, painless, non-pulsatile and compressible.
• A raised bluish purple lesion on the penis that has a smooth surface, irregular margin, compressible, non-tender, as well as non-pulsatile.
• Curvature of the penis
• Haemorrhage on the penis.
Laboratory investigations
Urine
Urinalysis, urine microscopy and urine culture are general tests that tend to be undertaken as part of the general assessment of individuals who have HOP but on the whole the results would tend to be normal and if there is any evidence of urinary tract infection it would be treated accordingly to improve upon the general condition of the patient as part of his general management.
Haematology blood test
Full blood count, erythrocyte sedimentation rate and coagulation screen are general tests that tend to be undertaken as part of the general assessment of all patients who have HOP but generally the results would tend to be normal and if any abnormality is detected it would be investigated and appropriate treatment would be provided to improve upon the general condition of the patients.
Biochemistry blood tests
Serum urea, creatinine, electrolytes, blood glucose, and liver function tests are general tests that tend to be undertaken as part of the general assessment of all patients who have HOP but generally the results would tend to be normal and if any abnormality is detected it would be investigated and appropriate treatment would be provided to improve upon the general condition of the patients.
Radiology imaging features
Ultrasound scan
Ultrasound scan of the penis would define the location of the lesion and its relationship to the corpora and mostly the HOPs would tend to be superficial and not connected to the corpora cavernosa or corpus spongiosum. Colour Doppler scan would tend to show low-flow within the penile lesion.
Computed tomography (CT) scan
CT scan of the penis would define the site, and the size of the lesion as well as its relation to the corpora cavernosa and corpus spongiosum and usually the lesion(s) would tend to be superficial and not connected to the corpora. In the scenario of a large lesion on the penis when the possibility of epithelioid angiosarcoma or epithelioid haemangioendothelioma need to be excluded CT scan of thorax, abdomen and pelvis would show no evidence of nodal disease or metastasis if required.
Magnetic Resonance Imaging (MRI) scan
MRI scan of the penis would define the site, and the size of the lesion as well as its relation to the corpora cavernosa and corpus spongiosum and usually the lesion(s) would tend to be superficial and not connected to the corpora. In the scenario of a large lesion on the penis when the possibility of epithelioid angiosarcoma or epithelioid haemangioendothelioma need to be excluded MRI scan of thorax, abdomen and pelvis would show no evidence of nodal disease or metastasis if required.
Positron Emission Tomography (PET/CT) scan
PET/CT scan is usually not undertaken in cases of HOP because majority of the lesions tend to be small and their clinical examination features tend to be illustrative of HOP and PET/CT would therefore not be undertaken.
Angiography
• Selective angiography and super-selective embolization of large HOPs could be undertaken especially with regard to HOPs that have extensions to the scrotum and the perineal region but this has not been reported because majority of the lesions tend to be small and can be treated by other forms of treatment that are less expensive and easier to undertake. Theoretically, this is a possible option to be used in the future for a very large HOP.
Diagnosis
• It has been iterated that diagnosis of haemangioma of the penis tends to be entirely a clinical decision [16].
• It had also been stated that ultrasound scan of the penis could illustrate low-flow state in cavernous haemangioma [16].
• It has additionally been iterated that computed tomography (CT) imaging as well magnetic resonance imaging (MRI) tends not to be able to show any clear demarcation between cavernous body and the angiomatous malformation [1]. Nevertheless, cavernosography tends to be useful with regard to locating the extension of the angiomatous lesion into the corpora cavernosa [1].
Treatment
Various options of treatment have been utilized with regard to the management of haemangioma of the penis (HOP) including:
• Conservative option -The conservative watch and wait option for very old and frail individuals who have extensive disease and severe co-morbidity well as those who do not want any treatment.
• Complete surgical excision of the lesion under general anaesthesia or local anaesthesia.
• Laser therapy
• Intralesional sclerotherapy with utilization of 3% sodium tetradecyl sulphate, 30% hypertonic saline and 2% polidocanol.
• Utilization of Itraconazole Oral solution (ITR) -Improvement/ reduction with regard to the size of congenital HOP has been experienced pursuant to utilization of Itraconazole Oral Solution (ITR) + Dermoscopy examination in a 2 month old baby who had HOP. This has only been reported in one case to the knowledge of the author.
• Cryotherapy can also be used as an option of treatment for HOP.
Macroscopic examination features
• It has been stated that HOP in majority of cases could be seen as solitary masses [15]; nevertheless, two or more lesions have been found on the penis in some cases of HOP.
• Hop tends to be seen on the dorsum of the penis, the glans, root of penis, urethral meatus, and ventral aspect of the penis and it may on rare occasions extend to involve the scrotum, perineal region, or peri-anal region.
• The reported size of HOP has ranged from less than 0.5 cm to 2.5 cm with a mean size of 1.2 cm [15]. Nevertheless HOPs have been reported with sizes greater than 2.5 cm
• HOPs could be compressible
• Ulcerations and bleeding could at times be visualised in cases of HOPs.
Microscopic examination features
• Some of the microscopic examination features of HOP include: [15]
o The findings of tumefactive proliferation of epithelioid endothelial cells that have nodular / lobular configuration which has been formed by immature but well-defined vessels [17].
o Microscopy examination of HOP does show that the proliferation does involve mainly epithelioid superficial tissues of the penis and it only on very rare occasions does involve the deep erectile tissues of the penis.
o Microscopy examination of HOP does illustrate epithelioid endothelial cells that are associated with abundance of amphophilic / eosinophilic cytoplasm, large nuclei, that contain open chromatin, delicate nuclear membranes, and distinctive nucleoli that are located centrally.
o Microscopic examination of HOP does demonstrate inflammatory infiltrate that contains lymphocytes and eosinophils.
o Microscopy examination of HOP does illustrate proliferation which quite often tends to be associated with a small arterial segment that shows intraluminal epithelioid endothelial cells as well as evidence of vascular damage which tends to involve thrombosis, fibro-intimal proliferation, duplication of internal elastic lamina, or mural duplication.
o Muscle-specific actin positive myopericytic cells tend to be seen that border the endothelial cells with regard to all cases of HOP.
o Microscopy examination of HOP specimens do show low mitotic rate and no evidence of atypical mitoses.
o Atypical / exuberant type of HOP does exist and with regard to the atypical / exuberant type of HOP, microscopy examination of the penile lesion would tend to show:
![]() A prominent area of the lesion which tends to be localized centrally that contains nests or sheet-like aggregates of epithelioid endothelial cells.
A prominent area of the lesion which tends to be localized centrally that contains nests or sheet-like aggregates of epithelioid endothelial cells.
![]() No vessel formation tends to be visualised within these central areas.
No vessel formation tends to be visualised within these central areas.
Immunohistochemistry staining study features
Positive staining
• It has been iterated that with regard to HOP, immunohistochemistry staining studies tend to show positive immunohistochemistry staining as follows [15]: o CD31 – strongly positive o Factor VIII - positive staining in myopericytes o HHF3 – positive staining in myopericytes o CD34 – minimal positive staining.
Negative staining
• It has been stated that with regard to HOP, immunohistochemistry staining studies would tend to show negative staining as follows [15]: o AE1/AE3 – negative staining but scattered positive staining cells could be present within the specimen.
Differential diagnoses
• Some of the documented differential diagnoses of HOP include [15]:
o Epithelioid angiosarcoma – In epithelioid angiosarcoma, the lesions tend to be located deeply within the penis and microscopic examination of the specimen does show marked nuclear atypia, destructive growth pattern, necrosis, high mitotic rate, as well as myxoid / hyalinized connective matrix, scarce / absence of inflammatory infiltrate, the lesion tends not to be associated with wall of arteries.
o Epithelioid haemangioendothelioma. – with regard to epithelioid haemangioendothelioma, microscopic examination of the specimen does show more nuclear atypia, infiltrative / destructive pattern of growth. Epithelioid haemangioendothelioma preferentially does affect veins, myxoid / hyalinized connective tissue matrix and scarce/ absent inflammatory infiltrate.
Outcome
• Generally the outcome following complete excision of the penile lesion, sclerotherapy of the penile lesion or laser treatment of the penile lesion, injection of hypertonic saline, the outcome does tend to be good.
• Incomplete excision of the HOP could lead to recurrence which would need to be re-excised or treated by sclerotherapy or laser treatment.
• At times sclerotherapy needs to be repeated for complete disappearance of the HOP
• With regard to very large HOPs sclerotherapy or laser treatment may lead to improvement or reduction in the size of the lesion but the cosmetic results may not be very good but only satisfactory.
• Spontaneous resolution / reduction in size of HOP could be experienced occasionally.
• Improvement / reduction in the size of congenital HOP has been experienced pursuant to utilization of Itraconazole Oral Solution (ITR) + Dermoscopy examination in a 2 month old baby who had HOP
• In an 80 year-old man who had Grade 5 adenocarcinoma of prostate associated with extensive HOP who was not provided any treatment and watched upon conservative management policy because of his age and co-morbidity no untoward effect developed and few other patients had been treated conservatively without any untoward effect.
(B) Miscellaneous narrations and discussions from some case reports, case series and studies related to haemangioma of penis
Srigley et al., reported two patients who were aged 36 years and 47 years and who had manifested with indurated subcutaneous nodules upon the penis which had been treated by means of local excision [18]. Histopathology examination of the lesions showed cords and nests of plump epithelioid cells that had been separated by a loose stroma which contained many lymphocytes and eosinophils. Light microscopy examination of the specimen revealed definite vasoformative (vessel formation) areas as well as solid regions that contained cytoplasmic vacuolization and nuclear atypia. Ultrastructural as well as immunohistochemistry staining studies for factor VIII-related antigen and Ulex europaeus agglutinin 1 showed that the lesions were of endothelial origin. Pursuant to excision of the nodules the two patients were reported to be free of disease five and half years and six and half years respectively. Srigley et al., stated that to their knowledge there had been only 1 case reported prior to their case report of haemangioma of penis (HOP) that had exhibited an epithelioid structure and this lesion should be considered a benign lesion as well as it should be treated by means of local surgical excision [18].
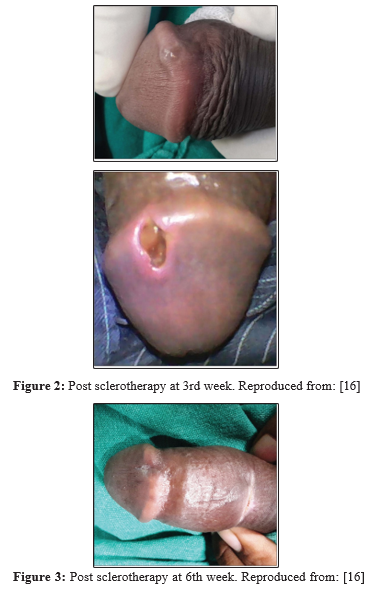
Mondal et al., reported a 22-year-old gentleman who had manifested with a swelling on his glans penis over the preceding 4 years [16]. The swelling did increase with regard to size upon erection. The lesion had been operated upon 1 year earlier but it did recur within 2 months. His clinical examination did reveal an elevated irregular lesion that measured 2 cm x 2 cm in its maximum dimension upon the dorsolateral part of his glans penis (figure 1). The lesion was bluish red with regard to colour with a smooth surface and irregular margin, and it was compressible, non-tender as well as non-pulsatile. He had colour Doppler ultrasound scan of his penis which showed low flow state within the lesion that was consistent with the diagnosis of cavernous haemangioma of the penis. He did not have any history of trauma from the lesion. The lesion was treated by means of intralesional instillation of 3% 1 ml of sodium tetradecyl sulfate. The instillation procedure was repeated one week later. During the third week the patient was seen and observed to have a scab at the site of the previous lesion (figure 2) which was removed spontaneously that resulted in an ulcer and by the 6th week the lesion had disappeared. At his 6th week follow-up, no telangectasia, scar, or ulcer was observed on his penis. At his 1-year follow-up he had not developed any recurrence. This case report does illustrate the usefulness of sclerotherapy in the treatment of HOP.
Gangkak et al., reported a 24-year-old gentleman who had manifested with painless peno-scrotal swelling of 3 years duration [19]. His clinical examination demonstrated a large bosselated, soft bag of worm-like, superficial non-pulsatile swelling on his glans penis and penile shaft circumferentially and which had extended into his scrotum (figure 4a). He had Colour Doppler scan of the lesion which demonstrated dilated tortuous vessels which upon angiography was shown as not demonstrating any connections to the corpora or major vessels in the penis in that the lesion was confined to the skin and subcutaneous tissue but not associated with the deeper tissues and corpora. He underwent surgical excision of the lesion under spinal anaesthesia during which he had urethral catheterization. A circum-coronal circumferential incision which was 1 cm from the coronal sulcus was undertaken and the dissection was carried deep up to bucks fascia and the haemangiomatous tissue was carefully dissected from the buck’s fascia and the skin of the shaft of the penis (figure 4 b). Additionally, via a 6 cm long elliptical incision that was centred over the median raphe, the haemangiomatous tissue was intussuscepted into the scrotum. No connection between the haemangioma and the corpora or main vessel of the penis was visualised. The Glanular lesion was left untreated. At the end circumcision and primary closure of the scrotal skin over a suction drain was undertaken (figure 4 c). Histopathology examination of the excised specimen showed dilated channels that were lined with endothelium containing red blood cells (RBCs). Presence of thick fibrous tissue in between the vessels was visualised, which was adjudged to be characteristic of cavernous haemangioma (figure 4 f). At his 1-month follow-up, he did not develop any recurrence and his wound was observed to have healed well (figures 4d and 4e). At his 6-month follow-up it was found that his glanular lesion had completely resolved with a resultant excellent cosmetic appearance. It would be argued that considering the fact that the haemangioma was a large one the best treatment option was appropriately utilised because the alternative options of sclerotherapy, and laser treatment would not be appropriate due to the fact that several treatment sessions would tend to be required and the possibility of residual haemangioma could possibly have been the end result. The fact that the lesion was completely excised and pathology examination of the specimen confirmed tissue diagnosis of cavernous haemangioma of the penis and scrotum would be re-assuring to the patient that he had a benign lesion.
Gangkak et al., reported a 24-year-old gentleman who had manifested with painless peno-scrotal swelling of 3 years duration [19]. His clinical examination demonstrated a large bosselated, soft bag of worm-like, superficial non-pulsatile swelling on his glans penis and penile shaft circumferentially and which had extended into his scrotum (figure 4a). He had Colour Doppler scan of the lesion which demonstrated dilated tortuous vessels which upon angiography was shown as not demonstrating any connections to the corpora or major vessels in the penis in that the lesion was confined to the skin and subcutaneous tissue but not associated with the deeper tissues and corpora. He underwent surgical excision of the lesion under spinal anaesthesia during which he had urethral catheterization. A circum-coronal circumferential incision which was 1 cm from the coronal sulcus was undertaken and the dissection was carried deep up to bucks fascia and the haemangiomatous tissue was carefully dissected from the buck’s fascia and the skin of the shaft of the penis (figure 4 b). Additionally, via a 6 cm long elliptical incision that was centred over the median raphe, the haemangiomatous tissue was intussuscepted into the scrotum. No connection between the haemangioma and the corpora or main vessel of the penis was visualised. The Glanular lesion was left untreated. At the end circumcision and primary closure of the scrotal skin over a suction drain was undertaken (figure 4 c). Histopathology examination of the excised specimen showed dilated channels that were lined with endothelium containing red blood cells (RBCs). Presence of thick fibrous tissue in between the vessels was visualised, which was adjudged to be characteristic of cavernous haemangioma (figure 4 f). At his 1-month follow-up, he did not develop any recurrence and his wound was observed to have healed well (figures 4d and 4e). At his 6-month follow-up it was found that his glanular lesion had completely resolved with a resultant excellent cosmetic appearance. It would be argued that considering the fact that the haemangioma was a large one the best treatment option was appropriately utilised because the alternative options of sclerotherapy, and laser treatment would not be appropriate due to the fact that several treatment sessions would tend to be required and the possibility of residual haemangioma could possibly have been the end result. The fact that the lesion was completely excised and pathology examination of the specimen confirmed tissue diagnosis of cavernous haemangioma of the penis and scrotum would be re-assuring to the patient that he had a benign lesion.
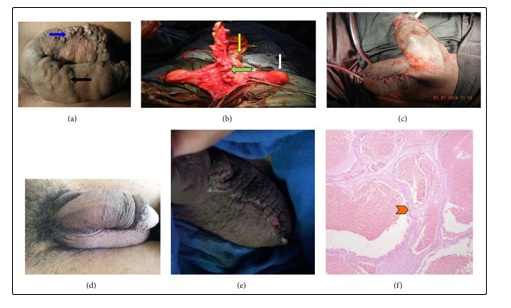
Figures 4 (a); 4(b); 4 (c); 4 (d); 4 (e); 4 (f).
Figure 4: (a) Showing preoperative photograph of the hemangioma: black solid arrow showing the penile component of hemangioma and blue solid arrow showing scrotal extension of hemangioma. (b) Showing intraoperative picture: green solid arrow showing the hemangioma tissue dissected from penile shaft, yellow solid arrow shows glans, and white solid arrow shows the testis (c) Final operative picture with suction drain in situ (d), (e) Postop picture at follow-up showing healed wound with an excellent cosmetic outcome. (f) Showing microscopic picture (H&E stain) endothelium lined vascular channels with intervening thick fibrous septa (red arrow head shows thick fibrous septa characteristic of cavernous hemangioma) [19].
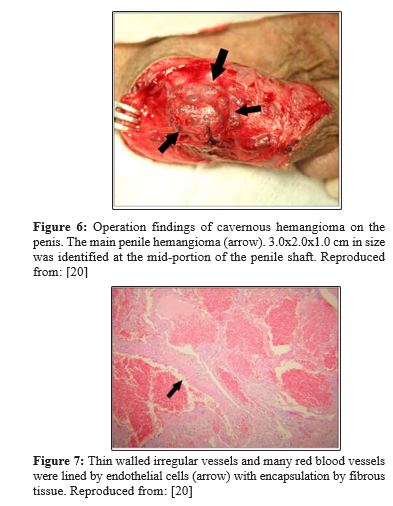
Lee et al., reported a 30-year-old gentleman who had manifested with painful palpable masses of his penis and scrotum which he had developed over the preceding 2 years [20]. He did not have any significant personal past or family medical history. His clinical examination revealed palpable masses on his scrotum, shaft of penis and glans of penis. The palpable masses within his scrotum and shaft of his penis were tender on palpation and thy mimicked a “bag of worms” (figure 5). The scrotal mass was noted to be single and it was separate from the mass within the penis as well as it did not trans-illuminate. The masses on the dorsum of the penis were located 0.5 cm below the coronal sulcus and they had extended onto the proximal shaft of his penis. Bluish tiny masses that were painless were visualised diffusively within four different areas of the glans penis. The lesions that were upon the shaft of his penis did cause a slight right sided curve of the penis laterally of less than 15 degrees during erection. The results of his routine urine tests, haematology and biochemistry blood tests were within normal range. The clinical examination features of the lesions were consistent with superficial lesions without any deep extensions. He underwent surgical exploration of his scrotal and penile lesions under spinal anaesthesia and during the procedure the area between his cutaneous tissue and Buck’s fascia was dissected up to the proximal shaft of his penis following a circumference incision 1.0 cm below the coronal sulcus. During the surgical procedure, two main masses were identified on the dorsal aspect of the shaft of his penis and 5 to 6 other masses were identified that had branched out on the lateral part of the shaft of his penis. The two masses within the penis looked like strawberry. The main masses within the penis were completely excised but other masses were partly excised in view of their adhesion the buck’s fascia. A separate vertical incision was made over the scrotal raphe and through this incision the scrotal mass was completely excised. The scrotal mass measured approximately 2.0 cm x 2.0 cm x 1.0 cm and it had a bumpy surface (figure 6). In view of the possibility of the mass within the glans penis as a result of a branched out mass of the shaft of the penis the masses of the glans penis were not excised but were left in situ. The masses within the scrotum and shaft of the penis were completely separated from each other. Microscopy examination of the specimens showed ectatic blood vessels that contained red blood cells and endothelial cells which had been consistent with the diagnosis of haemangioma. The examination also sowed encapsulated thickening fibrous tissues alongside the vessels which revealed typical characteristics of cavernous haemangioma (figure 7). At his 12-month follow-up he was well and satisfied with the cosmetic results of his operation and the haemangioma of his glans penis had completely resolved. With regard to this case report, it would be argued that the best treatment option was adopted and that procedures including laser therapy and sclerotherapy would not be appropriate procedures and that the other procedures would require several sessions of treatment and there would have been residual lesions after sclerotherapy and laser treatment. Additionally the specimens were examined by the pathologist who had confirmed the diagnosis of benign cavernous haemangioma which would reassure the patient.
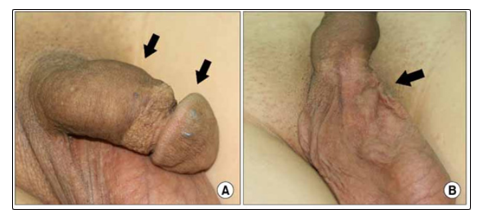
Figure 5: Cavernous hemangiomas of the glans penis, penis (A) and scrotum (B). Physical examination reveals a “bag of worms” (arrow) on the penis. Reproduced from: Reproduced from: [20]
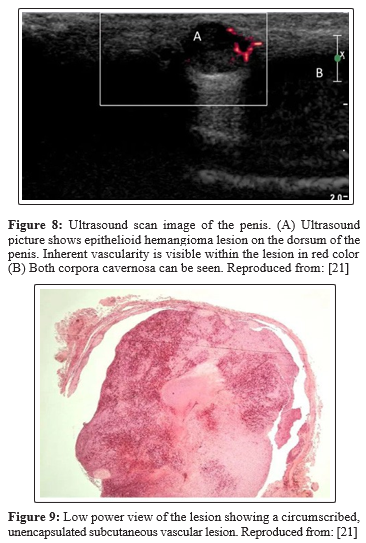
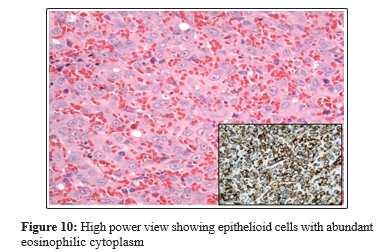
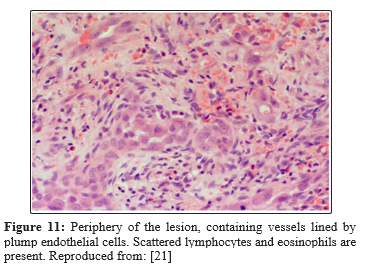
Ismail et al., reported a 50-year-old man who had presented with a painful nodule on the dorsum of his penis over the preceding 6 months which had been interfering with his coital activities [21]. A 5 mm, tender, firm nodule was palpated on the dorsal aspect of the midshaft of his penis and no evidence of lymphadenopathy was found. He had ultrasound scan of his penis which showed a well-circumscribed 6 mm x 8 mm lesion over the dorsal aspect of his penis within the sub-cutaneous tissue and superficial to the corpus cavernosum (figure 8) that was associated with an inherent blood flow. Under the suspicion of a vascular tumour of the penis, a local excision of the lesion was undertaken under general anaesthesia during which the lesion was found to be intimately involved with the neurovascular bundle. Feeding vessels were found which under magnification were ligated with preservation of the neuro-vascular bundle. Pathology examination of the specimen demonstrated a well-circumscribed, unencapsulated lesion (figure 9) which had consisted of central sheets of plump epithelioid cells that had scattered vessels at its peripheral aspect. The epithelioid cells contained abundant eosinophilic cytoplasm with large nuclei as well as distinct central nucleoli (figure 10). There was no evidence of significant cytological atypia and only few cells were observed in mitosis. Scattered capillary vessels that were lined by epithelioid cells were found at the periphery of the lesion (figure 11). Immunohistochemistry staining of the lesion showed strong positive staining for CD31 and CD34 and negative staining for S-100 protein and desmin (figure 11). The features of the lesion were consistent with the diagnosis of epithelioid haemangioma of the penis. At his 3-month follow-up his previous pain had resolved completely and he had normal erectile function as well as there was no evidence of lymphadenopathy found during his examination. This case report has illustrated the usefulness of complete local excision as treatment for epithelioid haemangioma of penis to confirm the diagnosis as well as to provide patient satisfaction.
Immunohistochemistry showed that the epithelioid cells were positive for CD34. Reproduced from: [21]
Pul and Pul reported a 6-month -old boy who had presented with a slowly enlarging penile mass over the preceding 2 months [22]. His clinical examination demonstrated an ovoid, solid, as well as cystic sub-cutaneous mass that measured 2 x 1, 5x1, and 5 cm on the right side of the shaft of his penis. The tumoral lesion was completely excised surgically and histopathology examination of the lesion confirmed the diagnosis of cavernous haemangioma of the penis. This case report has shown that cavernous haemangioma of the penis can affect neonates and infants and the lesion can successfully be treated by surgical excision.
Manerikar et al., reported a 22-year-old gentleman who had presented with a gradually increasing painless swelling of insidious onset over his glans penis over the preceding 10 years. He had been worried about the possibility of the lesion rupturing during coital activity [23]. His clinical examination showed an elevated irregular bluish red lesion that measured 2 cm x 1.5 cm upon the left dorsolateral part of his glans penis that was consistent with haemangioma of the glans of his penis. The lesion was compressible, painless as well as non-pulsatile. Colour Doppler ultrasound scan of the lesion revealed a well-defined, hypoechoic lesion that measured about 1.4 cm x 0.8 cm on the dorsal area of his glans penis within the subcutaneous plane. The scan also showed few feeding vessels that traversed the lesion and which had shown column flow on the Doppler imaging. The lesion was abutting the tunica albuginea but the corpora cavernosa were normal. There was no efflux or influx from the corpus spongiosum. The lesion was treated under local anaesthesia with utilization of 2% lidocaine penile block and 2 ml of 2% sodium tetradecyl sulfate. The lesion had resolved at his 5-week post sclerotherapy review. He was scheduled to undergo regular follow-up for 2 years. Lessons learnt from this case report include the fact that ultrasound scan of the penis can be used for the study of HOP in order to plan its treatment and also sclerotherapy under local penile block can be a useful treatment option for some cases of HOP.
Aydur et al., reported a 21-year-old man who had manifested with an enlarged giant haemangioma on his glans penis which had also caused him to have erectile dysfunction that responded partially to intracavernous injection stimulation test [24]. He had magnetic resonance imaging which had indicated that the lesion was a glandular haemangioma but he had a penile coloured Doppler ultrasound scan of the penis which had indicated invasion of the corpus cavernosum. Holmium laser coagulation of the lesion was undertaken in view of the cosmetic concerns of the patient. Nevertheless, the cosmetic results were not impressive and there was also no improvement with regard to the outcome of the intracavernosal injection stimulation tests. Aydur et al., concluded that the aplication of holmium laser should not be utilized for haemangiomas of the glans that are related to the corpus cavernosum, but further studies would be needed to reveal the effects of holmium laser application upon small haemiangiomas that are restricted to the glans penis [24]. With regard to this reported case some people could argue that the best treatment for such a case should be complete surgical excision of the haemangioma with insertion of tunica patch to the corpus carvernosum defect.
Deb and Sarkar reported a 28-year-old gentleman who had presented with gradually increasing swelling over his glans penis over the preceeding 5 years of insidious onset [25]. A raised bluish purple lesion that measured 1.5 cm x 1 cm over the right dorsolateral part of his penis was found that had a smooth surface, irregular margin, was compressible, non-tender, and non-pulsatile. He had Colour Doppler ultrasound scan which showed presence of a hypoechoic leion on the dorsal area of his glans penis with a low flow state. He was treated with utilization of 2 ml (60 mg) of sodium tetradecyl sulfate intralesional sclrotherapy with the aid of penile local anesthesia block. At his 6-months follow-up there was no recurrence following complete resolution of the lesion.
Savoca et al., reported a case of a large haemangioma of the glans penis that was treated via repeated injections of 2% polidocanol under local anaesthesia with utilization of 5% lidocaine-prilocaine cream [26]. No complications ensued after the procedure and a satisfactory aesthetic result was also achieved. At his 18-month follow-up, no recurrence had been encountered. Savoca et al., made the ensuing iterations: [26].
• A therapeutic reference standard for the treatment of haemangioma of the penis was still lacking in view of the rarity of the disease.
• Sclerotherapy did prove to be an effective, low-cost, and easy to undrtake procedure.
• Additionally, the procedure is repeatable in case of failure of the initial procedure.
López et al., reported a 28-year-old man who had presented with a lesion on his glans penis that had been present from his childhood [27]. He did not have any significant past medical history. His clinical eamination revealed a 1.5 cm, blue, compressible nodule, upon the left dorso-lateral area of his glans penis. A painful penile deformation was visualised during erection. He had magnetic resonance imaging which showed a vascular malformation. He had ultrasound scan of his penis which showed presence of a low vascular flow malformation. He refused to undergo surgical treatment. He received two treatments with Nd:YAG laser at one month interval with good cosmetic result and a slight scar depression. At his 1-year follow-up, he was well with no recurrence and he had resolution of his erectile function that he had earlier on. This case reports has shown that some small superficial haemangiomas of the penis can be successfully treated by laser therapy.
Norouzi and Shanberg reported their experience with the use of neodymium laser in the treatment of 4 patients who had 8 cavernous haemangiomas of penis [28]. The patients were aged 8 years, 16 years, 31 years, and 50 years. They were treated with neodymium:YAG laser at 5 to 15 watts of power for 3-second pulse durations. With regard to the outcome, all of the patients did have excellent cosmetic and functional outcome and their procedures were undertaken with no loss of blood and upon outpatient basis in the absence of any complication.
Zhang et al., reported 7 patients whose ages had ranged between 12 years and 32 years who had venous cavernous haemangioma of the penis between 2005 and 2011 [29]. All of the 7 patients had received treatments by means of percutaneous copper wires. With regard to the results, Zhang et al., reported that all of the operations were succesful [29]. All of the patients were satisfied with the aesthetic results of their procedures. They had a post-procedure follow-ups that that ranged between 1 year and 5 years and during this period recurrence was discovered in one patient who had the largest haemangioma of the corpus of the penis 2 months following his initial treatment. He underwent a secondary treatment procdure using the same technique and no recurrence was found at his subsequent follow-up assessments.
They did iterate the ensuing:
• The shortage of studies on this topic did prevent them from defining a therapeutic reference standard.
• The results of their study had confirmed that copper wire therapy for haemangioma of the penis is a simple, safe, useful option for the treatment of cavernous haemangioma of the penis.
Fetsch et al., described the clinical, histopathological, and the immunohistochemistry studies findings of 19 patients who had undergone treatment for haemangioma of the penis [17]. The patients had a median age of 45 years and their ages had ranged between 23 years and 75 years at the time of the initial resection of their tumours. Seventeen patients had manifested with a solitary penile mass, and two patients had manifested with two separate lesions that were closely approximated. The haemangioma process had involved: the glans penis with regard to 3 patients, the shaft of the penis with regard to 11 patients, the base of the penis with regard to 2 patients, and penis not otherwise specified with regard to 3 patients. The median size of the lesions was 1.2 cm and the sizes of the lesions had ranged between less than 0.5 cm to 2.5 cm in the greatest diameter. 11 of the haemangioma lesions were noted to be located on the dorsum of the penis and only one haemangioma of the penis was documented to be located on the ventral aspect of the penis. The commonest complaint of the patients was localised pain or tenderness which was documented with regard to 12 patients. The reported pre-oprative duration of the lesions had ranged between 5 days and 1 year and the median duration was 4.5 months. Microscopy pathology examination of the specimens did show that all the specimens had contained a tumefactive proliferation of epithelioid endothelial cells that were often in a nodular or lobular configuration and they also tended to be associated with an inflammatory infiltrate that contained lymphocytes as well as eosinophils. With regard to 14 cases, the vascular proliferation was associated with a small arterial segment and at times associated with mural damage and frequently in 13 cases it was associated with intraluminal epithelioid endothelial cells. Based upon the growth pattern of the epithelioid endothelial cells, 13 of the haemangiomas were considered “typical” and 6 of the haemangiomas were considred to be “atypical.” The atypical types of the haemangiomas had a prominent centrally located zone where nests or sheet-like aggregates of epithelioid endothelial cells had not formed discrete vessels. Data related to the immunohistochemistry staining studies of the haemangiomas was available for 15 tumours. The immunohistochemistry staining studies of the haemangiomas showed that the epithelioid endothelial cells usually had exhibited strongly positive reactivity for CD31, lesser reactivity staining for factor VIIIrAg, as well as minimal reactivity staining for CD34. With regard to 9 out of 12 cases, a small number of epithelioid endothelial cells did exhibit expression for keratins. With regard to all of the cases that had been tested, at least focal muscle-specific-actin-positive myopericytic cells were found present and they were bordering the endothelial cells which was specifically observed peripherally. The initial surgical interventional treatment had consisted of either a share biopsy which was undertaken in 1 case, excisional biopsy in 2 cases, or local excision with regard to 16 cases. A complete follow-up history was found with regard to 12 patients, and incomplete follow-up data was available for 4 other patients. One of the patients did develop a new epithelioid haemngioma at a site within his penis that was separate from the initial lesion; nevertheless, none of of the patients was found to have experienced a true metastasis or to have died of complications of the process. Fetsch et al., iterated that optimal management of epithelioid haemangoma of the penis does appear to be complete local excision with periodic follow-up visits in order to monitor for local recurrence [17].
Table 1: Summations of experiences gained in the management of haemangioma of the penis from case reports, case series and studies related to haemangioma of the penis from fifty one reported sources
|
Reference |
Age |
Side/Site/Size |
Treatment |
Histology/Immuno-histochemistry |
Outcome |
|
(1); [21] Ismail et al. [21] Epithelioid hemangioma pf penis: case report and review of literature J Med Case Rep. 2011 Jun; 5: 260 DOI: 10.1186/1752-1947-5-260 https://www.ncbi.nlm.nih.gov/pmc/articles/ PMC3141710/ |
50 years |
Dorsum of mid shaft penis 5 mm 6 x 5 mm on u/sound |
Local excision under GA + ligation of feeding vessel |
Well-circumscribed, encapsulated lesion; central sheets of plump epithelioid with scattered vessels in the periphery; cells with abundant eosinophilic cytoplasm, large nuclei,; central distinct central nucleoli; no significant cytological atypia; only a few cells in mitosis. CD31 strongly positive; CD 34 strongly positive; S-100 protein negative; Desmin negative Epithelioid Haemangima. |
Well with no recurrence |
|
(2) [30] Sharma G. Haemangioma of glans penis. The Internet Journal of Urology 2004; 3:1 http://ispub.com/iju/3/1/3235 |
22 years |
Glans penis from childhood; 2x1.2 cm |
Sclerotherapy with Sodium tetradecyl sulphate 3% |
No pathology results treatment based on ultrasound scan findings |
Minimal pigmentation after scab fell off at 8 weeks. |
|
(3) [31] Alter G J, Trengove-Jones Guy, Charles E H. Jr. Hemangioma of penis and scrotum. Urology 1993 Aug; 42(2): 205 – 208 https://doi. org/10.1016/0090-4295(93)90649-U https:// www.sciencedirect.com/science/article/ pii/009042959390649U#! |
Age not available |
Penis & scrotum |
Surgical excision |
Details not available |
Details not available |
|
(4) [1] Cheng G, Song N, Hua L, Yang J, Xu B, Li P, Yin C, Zhang W. Surgical Treatment of Hemangioma on the Dorsum of The Penis Journal of Andrology 2012 Sep-Oct; 33(5):921 – 926. https://onlinelibrary.wiley. com/doi/pdf/10.2164/jandrol.111.015685 |
6 patients with ages between 19 years to 42 years and mean age 23 years |
Dorsum of penis 2 small and < 1 cm; 4 large |
2 small ones – surgical excision; 4 surgical excision + insertion of tunica vaginalis graft. |
Pathology results not available. |
All 5 patients who were sexually active preceding their surgery continued to be sexually active within 3 months. No recurrence |
|
|
22 years |
Right dorsolateral part of Glans for 4 years; 2 cm x 2cm |
2 Injections of sclerosant 3% sodium tetradecyl sulfate at weekly intervals |
No pathology results diagnosis based upon Colour Doppler ultrasound scan |
Well with no recurrence at his 1 year follow-up. |
|
(5) [32] Kumar A, Goyal N K, Trivedi S, Dwivedi U S, Singh P B. Primary Cavernous Hemangioma of the Glans Penis: Rare Case Report with a Review of the Literature. Aesthetic Plastic Surgery. 2008; 32: 386 – 388. https://link.springer.com/ article/10.1007%2Fs00266-007-9076-8 |
23 years |
Glans penis |
Sclerotherapy |
Pathology not available |
Successful outcome. |
|
(6) [16] Mondal S, Biswal D K. Pal D P Cavernous hemangioma of the glans penis. Urology Annals 2015 Jul-Sep; 7(3): 399 - 401 DOI: 10.4103/0974- 7796.152037 https://www.researchgate. net/publication/276439793_Cavernous_ hemangioma_of_the_glans_penis europepmc.org/article/PMC/4518386 |
22 years |
Glans |
Sclerotherapy with 3% sodium tetradecyl sulfate |
Pathology not available |
Good outcome |
|
(7) [33] Ranganayakulu B, Reddy K S, Raju S. Hemangioma of penis. Indian Journal of Dermatology, Venearology, and Leprology. 1999; 65(2): 85 http://www.ijdvl.com/ article.asp?issn=0378-6323;year=1999;volu me=65;issue=2;spage=85;epage=85;aulast= Ranganayakulu |
30 years |
Dorsum proximal part of penis ; 3 cm x 3 cm |
Surgical excision under local anaesthesia. |
Vascular endothelial stroma with large spaces that stained dark brown. |
No follow data was reported |
|
(8) [17] Fetsch J F, Sesterhenn I A, Miettinen M, Davis C J Jr. Epithelioid hemangioma of the penis: a clininopathologic and immunohistochemical analysis of 19 cases, with special reference to exuberant examples often confused with epithelioid hemangioma and epithelioid angiosarcoma. DOI: 10.1097/00000478-200404000-00012 https:// |
19 cases aged 23 to 75 years with mean age 45 years. |
17 had single masses and 2 had 2 masses close together, Glans in 3 cases; Shaft in 11 cases; Base of penis in 2 cases; Penis not otherwise specified in 3 cases. 11 cases in dorsum; 1 case ventral |
Shave biopsy 1; excisional biopsy 2; local excision 3. |
Microscopy of the lesions showed that the lesions had contained a tumefactive proliferation of epithelioid endothelial cells, often in a nodular or lobular configuration and associated an inflammatory infiltrate that contained lymphocytes and eosinophils. With regard to 14 specimens, the vascular proliferation was associated with a small arterial segment, at times together with mural damage and frequently with intraluminal epithelioid cells that was observed in 13 cases. Based upon the growth pattern of the epithelioid endothelial cells, 13 cases were adjudged to be typical and were adjudged to be atypical or exuberant. The exuberant lesions had a conspicuous centrally located zone where nests or sheet-like aggregates of epithelioid endothelial cells did not form discrete vessels. Immunostains available for 15 cases: CD31 usually strongly positive; lesser positive reactivity for factor VIIIrAG; minimal reactivity for CD34.In 9 out of 12 cases the epithelioid endothelial cells stained for keratin. At least focal muscle-specific actin-positive myopericytic cells were present bordering the endothelial cells. |
Details of outcome not available |
|
(9) [22] Pul M, Pul N. Cavernous haemangioma of the penis in an infant. International Urology and Nephrology 1995 Jan; 27: 113 – 115. https://link.springer.com/ article/10.1007/BF02575229 |
6 months |
Right side of penile shaft; 2x1, 5x1, 5 cm |
Complete surgical excision |
Cavernous haemangioma demonstrated on microscopy |
|
|
(10) [34] Kishore M, Ahuja A, Bhardwaj M. Epithelioid hemangioma of penis mimicking malignancy: A rare case. Urology Annals 2017 Oct; 9(4): 397 https://www. researchgate.net/publication/320305150_ Epithelioid_hemangioma_of_penis_ mimicking_malignancy_A_rare_case |
64 years |
Glans penis painless bleeding mass. |
Local excision |
Epithelioid haemangioma was seen. CD31+. Smooth muscle actin+, cytokeratin negative. |
|
|
(10) [35] Hemal A K, Goswami A K, Sharma S K, Radotra B D, Malik N. Penile Venous Haemangioma. Australian and New Zealand Journal of Surgery. 1989 Oct; 59(10): 814 – 816. https://doi. org/10.1111/j.1445-2197.1989.tb07015.x https://onlinelibrary.wiley.com/doi/ abs/10.1111/j.1445-2197.1989.tb07015.x |
Not available |
Base of penis With peyronies type curvature with erection |
Surgical resection |
Venous haemangioma. 1st reported case of venous haemangioma |
|
|
(11) [36] Yoshikawa H, Ikeuchi T, Ohta M, Takanishi R, Matsumoto K, Kai Y. Hemangioma of the glans penis: A case report. Journal of the Showa Medical Association 1995; 55(6): 636 – 638. https://doi.org/10.14930/jsma1939.55.636 https://www.jstage.jst.go.jp/article/ jsma1939/55/6/55_6_636/_article |
41 years |
Dorsum of glans penis the 34th case reported in the Japanese literature. |
Surgical resection |
Venous haemangioma |
|
|
(15) [37] Freeman A, Damato S, Ismail M, Raj N. Epithelioid hemangioma of the penis: case report and review of literature. Journal of Medical Case Reports. 2011;5: 260 (1-4) https://link.springer.com/content/ pdf/10.1186/1752-1947-5-260.pdf |
50 years |
Dorsum mid shaft of penis 5 mm nodule; |
Local excision under general anaesthesia |
Well-circumscribed encapsulated lesion with central sheets of plump epithelioid cells that had scattered vessels at the periphery. The epithelioid cells had abundance of eosinophilic cytoplasm with large nuclei and distinct central nucleoli. no significant atypia and only few cells in mitosis. Scattered capillary cells lined by epithelioid cells at the periphery of the lesion. CD31 strongly +positive; CD34 strongly positive; S-100 negative; Desmin Negative. Diagnosis Epithelioid haemangioma |
Goodoutcome of surgery |
|
(16) [28] Norouzi B B, Shanberg A M. Laser treatment of large cavernous haemangioma of the penis. The Journal of Urology. 1998 Jul; 160(1): 60 – 62. https://doi.org/10.1016/ S0022-5347(01)63028-3 https://www. sciencedirect.com/science/article/abs/pii/ S0022534701630283 |
4 patients aged 8 years, 16 years, 31 years, 50 years |
Total of 8 haemangiomas of penis exact sites not available. |
YAG laser at 5 to 15 Watts of power for 3 second pulse duration. |
No histology |
Good cosmetic result. |
|
(17) [38] Ozdamar M Y, Zengin K, Tanik S, Albayrak S, Soyuer I. Cavernous hemangioma of the glans penis: Concomitant surgical excision with circumcision in an uncircumcised patient. Pediatric Urology Case Reports. 2014; 1(5): 1 – 4. http://oaji.net/ articles/2014/1334-1412274117.pdf |
9 years |
Left side glans penis |
Circumcision with total excision of lesion |
Cavernous haemangioma. |
Good cosmetic result. |
|
(18) [39] Singh A P, Gupta A K, Pardeshi R, Ansari M, Barolia D K. Diffuse hemangioma of penis, scrotum, perineum, and anal canal – A rare case report. Nigerian Journal of Plastic Surgery. 2018; 14(1): 12 – 14. http://www.njps.org/article. asp?issn=0794-9316;year=2018;volume=14 ;issue=1;spage=12;epage=14;aulast=Singh |
8 years |
Diffuse covering scrotal wall penis, perineum and right hemi-scrotum. Root of corpora cavernosa, corpora spongiosa, and around proximal penis |
Surgery was advised but he was lost to follow-up |
Ultrasound scan Doppler imaging and MRI scan confirmed diagnosis but no specimen obtained due to patient being lost to follow-up. |
He was lost to followup. |
|
(19) [19] Gangkak G, Mishra A, Shivam P, Tomar V.Large Genital Cavernous Hemangioma: A Rare Surgically Correctable Entity. Case Reports in Urology Volume 2015, Article ID 950819, 3 pages http:// dx.doi.org/10.1155/2015/950819 |
24 years |
Penis and scrotum (large) |
Surgical excision via circum-coronal scrotal excision. |
Cavernous haemangioma |
Good cosmetic result, no recurrence at 6-month follow-up. |
|
(20) [40] Akyol I, Jayanthi V R, Luquette MH. Verrucuous Hemangioma of the Glans Penis. Urology 2008 Jul 01; 72(1): https://www.deepdyve.com/lp/elsevier/ verrucous-hemangioma-of-the-glans-penis-OVpwJDyoJZ |
5 years |
Glans penis |
Various local excisions of 7 years with recurrences followed by second deep excision of recurrent lesion. |
Verrucous haemangiomas |
No further recurrence after 3 years follow-up. |
|
(21) [41] Kumar A, Goyal N K, Trivedi S, Dwivedi U S, Singh P. Primary Cavernous Hemangioma of the Glans Penis: Rare Case Report with A Review of the Literature. Aesthetic Plastic Surgery 2007. DOI: 10.1007/s00266-007-9076-8 |
23 years |
Glans |
Intra-lesional sclerotherapy. |
No specimen obtained and no histology |
Good cosmetic result. |
|
(22) [20] Lee J M, Wang J H. Kim H S. Multiple Cavernous Hemangiomas of the Glans Penis, Penis, and Scrotum. Korean J Urol. 2008 Jan; 49(1): 92 -94 https://doi. org/10.4111/kju.2008.49.1.92 |
Age not available |
In glans penis, penile shaft, and scrotum |
Surgical excision |
Cavernous haemangioma of glans penis, shaft of penis and scrotum |
At 12-months followup the protruding lesions of glans penis werenaturally relieved |
|
(23) [8] Senoh H, Ichikawa Y, Okuyama A, Takaha M, Sonoda Cavernous Hemangioma of Scrotum and Penile Shaft. Urol. Int. 1986; 41: 309 – 311 |
13 years |
3.5 cm x 2.5 cm x 1.5 cm not connected to corpus cavernosum |
Complete surgical excision At age 6 years he underwent excision of scrotal haematoma with histology finding of cavernous haemangioma. |
Haemangioma of shaft of penis |
Good surgical outcome |
|
(24) [42] Smart R H, Newton D E. Hemangioma of the penis with blue rubber bleb nevus. Journal of Urology. 1975; 113(4): 570 – 571. https://utsouthwestern. pure.elsevier.com/en/publications/ hemangioma-of-the-penis-with-blue-rubber-bleb-nevus-syndrome |
38 years |
Penis details not available |
Details not available |
Details not available |
Details not available |
|
(25) [43] Rastogi R. Diffuse Cavernous Hemangioma of the Penis, Scrotum, Perineum, and Rectum – A rare tumor. Saudi J Kidney Dis Transpl [serial online] 2008; 19: 614 – 618http:www.sjkdt.org/text. asp?2008/19/4/614/41323 |
18 years |
Entire penis and penoscrotal region |
Combination of laser ablation and surgical excision. |
Cavernous haemangioma. |
Long term outcome data not available but good post-operative outcome |
|
(26) [44] Echchaoui A, Zaouri H. Penile Cavernous Hemangioma. International Journal of Medicine and Surgery. 2015; 2(1):26 DOI: 10.15342/ijms.v2il.60 /https:// www.mbmj.org/index.php/ijms/article/ view/60/0 |
26 years |
Dorsum of penis 1 x 2 mm Ventral aspect of penis |
Neodymium YAG laser treatment was recommended |
Cavernous haemangioma on biopsy histology |
Details not published |
|
(27) [45] Rajender A, Phillips E, Munarriz R. Penile epithelioid hemangioma: A case report. Journal of Integrative Nephrology & Andrology 2015; 2(1): 38 – 40. DOI: 10.4103/2225-1243.150011 |
41 |
Dorsum of proximal shaft of penis |
Complete surgical excision |
Atypical variant of epithelioid haemangioma. CD31 positive; CD34 focally positive; Keratin AE1/3, & Ki-67 focally positive 10%, MSA (HHF-35) positive |
No recurrence at 1-month follow-up |
|
(28) [46] Ulker V, Esen T. Hemangioma of the Glans Penis Treated with Nd:YAG Laser. International Urology and Nephrology 2004; 37:95 – 96. https://www. semanticscholar.org/paper/Hemangioma-of-the-Glans-Penis-Treated-with-Nd%3AYAG-Ulker-Esen/844c08684e37ffe5e4317d11469 19d77f084c453 |
18 years |
Glans penis |
ND YAG Laser therapy |
No histology |
Excellent cosmetic result |
|
(29) [47] Zhou Y, McKay K, Shen D. Multifocal Epithelioid Hemangioma of the Penis. American Journal of Clinical Pathology 2014 Oct; 142(1): 256 https://doi. org/10.1093/ajcp/142.suppl1.256 |
27 years |
Four papules on Right of Glans penis; A lesion on urethral opening |
Visible portions of penile lesion excise surgically & lesion to right of urethral meatus superficially removed only. |
Diagnosis of epithelioid haemangioma was made. KI-67 index of 5% to 10%; CD31 positive, ERG positive; Fli-1 positive; INI-1 positive; CD34 negative; Cd30 negative; HHV-8 negative |
One month later residual palpable lesion deep to Glans epithelium on right, and oozing from small lesion 2 – 3mm at urethral meatus. |
|
(30) [23] Manerikar K, Singh G, Ali I. Intralesional Sclerotherapy in Hemangioma of the Glans Penis. International Journal of Scientific Study. 2015 Sep; 3(6): 207 – 209. Ijs_sep_cr03 |
22 years |
Glans Penis |
2ml of 2% sodium tetradecyl sulfate sclerotherapy |
No histology |
No reccurence at his 2-year follow-up |
|
(31) [18] Srigley J R, Ayala A G, Ordonez N G, Nostrand A W. Epithelioid hemangioma of the penis. A rare and distinctive vascular lesion. Archives of Pathology & Laboratory Medicine. 1985 Jan 01; 109(1): 51 – 54 |
Two patients 36 years & n 47 years |
Subcutaneous penile nodules. |
Excision of nodules. |
Factor VIII and Ulex europaus demonstrated the lesions were endothelial. |
No recurrence of lesion at Five and half years and six and half years later. |
|
(32) [24] Aydur E, Erol B, Tahmaz L, Irkilata H, Eken C, Peker A. Coagulation of giant hemangioma in glans penis with holmium laser. Asian Journal of Andrology 2008 Sep; 10(5): 819 – 821 |
21years |
Giant mass on Glans Penis |
Holmium laser coagulation |
No histology |
The cosmetic effect was not impressive |
|
(33) [48] Hemal A K, Aron M, Wadhwa S N. Intralesional Sclerotherapy in the Management of Hemangiomas of the Glans Penis. Journal of Urology. 1998; 159(2): 415 – 417. DOI: 10.1016/s0022- 5347(01):63936-3 |
Four patients Ages not available |
Glans penis |
30% hypertonic saline injections |
No histology |
All 4 patients satisfied with cosmetic result. |
|
(34) [49] Casale A J, Menashe D S. Massive Strawberry Hemangioma of the Male Genitalia. The Journal of Urology 1998 Mar 01; 141(3 Part 1): 593 - 594 https://doi. org/10.1016/S0022-5347(17)40904-9 |
Neonate |
Massive lesion involving scrotum and penis |
Conservative management of lesion and its complications of ulceration |
No histology |
Excellent outcome documented. |
|
(35) [50] Deb P P, Sarkar A. Penile Granular Hemangioma Treated With Intralesional Sclerotherapy Scholars Journal of Medical Case Reports (SJMCR) 2018 Apr; 6(4): 273 – 275. DOI: 10.21276/sjmcr.2018.6.4.16. |
28 years |
Glans penis |
Sclerotherapy 2 ml of 3% sodium tetradecyl sulfate. |
No histology |
Lesion resolved and no recurrence at 6 months. |
|
(36) [29] Zhang D, Zhang H, Sun P, Li P, Xue A, Jin X, A Creative Therapy in Treating Cavernous Hemangioma of Penis with Copper Wire. J Sex Med 2014; 11: 2605 - 2610 https://doi.org/10.1111/ jsm.12331 |
7 patients aged from 12 years to 32 years |
Penis |
Treatment with copper wire |
Cavernous haemangioma of penis |
All operations successful. Follow-up ranged from 1 year to 5 years. Recurrence was observed in 1 patient who had the largest lesion of corpus penis 2 months after treatment. Secondary procedure was undertaken with the same technique and no lesion were found later. |
|
(37) [51] Yigiter M, Arda I S, Hicsonmez A. An unusual cause of paraphimosis: hemangioma of the glans penis. Journal Of Pediatric Surgery 2008; https://doi. org/10.1016/jpedsurg.2007.10.052 |
Age not available |
Paraphimosis |
Treatment not available |
No histology available |
Not available |
|
(38) [52] Ergun O, Ceylon B G, Armagan A. Kaohsiung A Giant Scrotal Cavernous Hemangioma Extending to The Penis And Perineum: A Case Report. J Med Sci 2009 Oct; 25(10): 559 – 561. |
44 years |
Scrotal mass involving dorsal skin of penis and extending to perineum |
Complete surgical excision of lesion via the scrotal approach. |
Cavernous haemangioma. |
No recurrence at his 16-months follow-up |
|
(39) [53] Sumer F, Yildirim I, Aydur E, Irkilata C, Kocaoglu M, Peker A F. Penile Cavernous Hemangioma. TURK J Urol. 2008; 34(2): 269 – 271 |
Age not available (in Turkish) |
Penile Haemangioma extended from the corpora cavernosa at base of penis |
Intra-operative details in Turkish – details not available |
Details not available |
Details not available |
|
(40) [54] Toshihiko T, Takehisa I, Yoshinori I, Toshiya K, Kazunori K, Hiroyuki O. Cutaneous Hemangioma of the Penis Successfully Treated with Sclerotherapy and Ligation. International Journal of Urology. 1998 Jul 01; 5(4): |
18 years |
Proximal part of glans to prepuce |
Sclerotherapy |
No histology |
Successful treatment |
|
(41) [55] Javed A, Mahren S, Faig S . Penile hemangioma in a prostate cancer patient: a case report. J Dow Uni Health Sci. 2018; 12(3): 120 - 123 |
80 years with known prostate cancer had TURP and radiotherapy (Gleason 5) |
Swelling of shaft of penis with oval mass on right of shaft of penis possibly arising from the urethral epithelium and extending from the membraneous urethra to the mid penile epithelium with increased vascularity. |
Conservative management because of age of patient and clinical condition plus co-morbidity |
Radiology diagnosis only |
Doing well no untoward effect. |
|
(42) [56] Gite Venkat G A, Atul Singal Nicose Jayant V, Jain Hitesh M. Concealed Hemangioma: Rare Presentation Journal of Medical Science And Clinical Research. 2017 Nov; 5(11): 29920 - 29922 DOI: https://dx.doi.org/10.18535/jmscr/v5i11.18 |
71 years |
LUTS with phimosis and nodule underneath prepuce. 2 cm x 2cm |
Circumcision + concomitant excision of lesion. |
Haemangioma with thrombus and neovascularization. |
At 6-month followup no recurrence and patient satisfied with cosmetic results. |
|
(43) [57] Yang Q, Tang I Q, You Z M, Ran X, Yuping R. Itraconazole Oral Solution Cured a Case of Infantile Hemangioma and Penis with Ulcer: Monitoring by Demoscopy ECRONICON OPEN ACCESS E C PAEDIATRICS 2016;3(2): 346 – 349 |
2 months old |
Ulcer on haemangioma of left scrotum which involved penis |
Itraconazole Oral Solution (ITR) + Dermoscopy examination |
No histology |
After 60 days the lesion degraded and after 3 months the lesion had significantly regressed. |
|
(44) [58] Heidenreich A, Rothe H, Derschum W, von Vietsch H. Hemangioma of the glans penis. Its therapy by neodymium: YaG laser Urologe A. 1992 Mar; 31(2): 94 – 95 |
Young male (age not available) |
Glans |
YAG laser therapy under local anaesthesia |
No histology |
Good cosmetic results. |
|
(45) [59] Tsujii T Cutaneous Hemangioma of the Penis Successfully treated with sclerotherapy. Int J Urol. 1998 Jul;5(4): 396b – 397 DOI: 10.1111/j.1442-2042- 1998.tb00377.x https://www.ncbi.nlm.nih. gov/pubmed/9712455 |
18-years |
Proximal portion of glans to prepuce |
Sclerotherapy with polidocanol and interrupted ligation of haemangioma |
No pathology |
Successful outcome. |
|
(46) [1] Cheng G Surgical Treatment of Hemangioma on the Dorsum of the Penis 2012 Mar 06; |
6 patients ages between 19 years and 42 years mean age 23 |
Dorsal aspect of penis |
Surgical excision |
Haemangioma |
Follow-up 2 to 5 years median 2.5 years Good cosmetic results and good sexual function in 5 sexually active males |
|
(47) [60] Akin Y, Sarac M, Yucel Rare malformations of glans penis Arteriovenous malformation. Nigerian Journal of Clinical Practice. 2013; 16(3): 389-391 DOI: 10.4103/1119-3077.113471 |
41 years
& 2 years |
Glans penis
Glans lesion & genital area |
Surgical excision
Parents did not want surgery so observation |
Arterio-venous malformation |
Good outcome
no long term follow-up information for both patients |
|
(48) [27] Lopez V, Ricart J M, Lopez I, Martin J M, Marton D, Ortega C, Salvador C. Vascular malformation of the glans penis successfully treated with Nd:YAG laser. Dermatology Online Journal 2012; 18(10): 16 https://escholarship.org/uc/ item/019698rw |
28 years |
Glans penis |
Nd:YAG laser with 2 treatments 1 month apart. |
No histology |
Good outcome; no recurrence after one year and active coitally. |
|
(49) [61] Basu P, Cohen P R. Penile Angiokeratoma (Peaker): A Distinctive Subtype of Genital Angiokeratoma Cureus 2018nDec 28; 10(12): e3793 DOI: 10.7759/ cureus.3793 |
63 years |
2 x 2 mm papule on cutaneous region of corona of penis. |
No treatment |
Diagnosed as angiokeratoma therefore not haemangioma |
|
|
(50) [62] Balsarkar D, Gore M A. Venous malformations of glans penis: An unusual cause of hematuria. Indian Journal of Urology 2000; 17(1): 50 – 51 http://www.indianjurol.com/text. asp?2000/17/1/50/41016 |
17-years |
Vascular lesions on glans Penis extending to urethral meatus. |
0.5 ml solution of N-butyl cyanoacrylate mixed with 0.6 ml lipiodol injection therapy |
No pathology specimen |
At 2-years follow-up no recurrence and patient well. |
|
(51) [63] Shamsa A, Modaghegh H S, Asadpour A A. Glans Penis Hemangioma: A Rare case Report and its Treatment. Journal of Research in Urology 2016 Winter; 1(1): 31- 35 DOI: 10.21859/ruj-01013 |
26 years |
Glans |
3% sodium tetradecyl sulfate |
No histology |
Satisfactory results. |
Conclusions
Various types of haemangioma of the penis tend to be reported sporadically globally.
There are no globally adopted consensus opinion guidelines for the management of the lesion and the various commonly successfully utilized treatment options have included (a) surgical excision (b) sclerotherapy, (c) laser treatment. Other less commonly used options have included the conservative approach, wire therapy and potentially cryotherapy and embolization procedures as well as oral therapies which would need to be tried on a global multi-centre trial basis to define the best treatment options.
Some treatment facilities may not be available in some parts of the world especially laser treatment especially in developing countries and for this reason for large lesions especially, complete excision procedure should be under taken as a one stage procedure and small superficial lesions could be treated by sclerotherapy or any available local therapy.
The advantage of surgical excision is that histopathology examination would be undertaken to confirm the diagnosis.
In some parts of some developing countries there may not be facilities for radiology imaging scans including ultrasound scan, computed tomography scan, and magnetic resonance imaging scans therefore complete surgical excisions based upon clinical examination finding provisional diagnosis may tend to be enough to undertake surgical excision of the lesion as a one stage procedure and the pathology results would subsequently confirm the diagnosis.
Acknowledgements
Urology Annals and Wolters Kluwer – Medknow Publications for granting permission for reproduction of contents and figures from their journal article.
Case Reports in Urology Volume and Hindawi Publishing Limited for granting permission for reproduction of contents and figures from their journal article under Copyright © 2015 Goto Gangkak et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
J Med Case Reports for granting permission for reproduction of contents and figures from their journal article under copyright The authors which has granted to any third party, in advance and in perpetuity, the right to use, reproduce, or disseminate, the article, according to the BMC license agreement and licensed under the Creative Commons Attribution License 4.0. In line with BMC's Open Data Policy, data included in the article shall be made available under the Creative Commons 1.0 Public Domain Dedication waiver, unless otherwise stated. If the law requires that the article be published in the public domain, I/we will notify BMC at the time of submission, and in such cases not only the data but also the article shall be released under the Creative Commons 1.0 Public Domain Dedication waiver. For the avoidance of doubt it is stated that sections 1 and 2 of this license agreement shall apply and prevail regardless of whether the article is published under Creative Commons Attribution License 4.0 or the Creative Commons 1.0 Public Domain Dedication waiver.
References
- Cheng G, Song N, Hua L, Yang J, Xu B, et al., (2012) Surgical Treatment of Hemangioma on Dorsum of the Penis. Journal of Andrology 43: 921-926.
- Dinehart S M, Kinccanon J, Geronemus R (2001) Hemangiomas: evaluation and treatment. Dermatol Surg 27: 475-485.
- Lin Y, Sun G H, Yu D S, Wu C J, Chen H I, et al., (2002)Intrascrotal hemangioma. Arch Androl 48: 259-265.
- Jahn H, Nissen H M (1991) Haemangioma of the urinary tract:review of the literature. Br J Urol 68: 113-117.
- Lane J W (1953) Hemangioma of the glans penis. US Armed Forces Med J 4: 139- 140.
- Dehner L P, Smith G H (1970) Soft tissue tumors of the penis.Cancer 25: 1431-1447.
- Whitmore W F (1970) Tumors of the penis, urethra, scrotum and testis. In: Campbell M F, Harroson J M, eds. Urology, 3rd ed. Philadelphia, PA, 1190.
- Senoh K, Miyazaki T, Kikuchi I, Sumiyoshi A, Kohga A, et al., (1981) Angiomatous lesions of glans penis. Urology 17: 194-196.
- Mortenson H, Murphy L (1950) Angiomatous malformations of the glans penis. J Urol 64: 396-399.
- Gibson T E (1937) Hemangioma of the scrotum. Urol CutanRev 41: 43-44.
- Goldwyn R M, Rosoff C B (1969) Cryosurgery for largehemangioma in adults. Plast Reconstr Surg 43: 605-611.
- Yamazaki S, Kawashima T, Nishimura K, Sasaki M, Nakagawa T, et al., (1978) Penile hemangioma treated by cryotherapy: report of a case. Acta Urol 24: 325-332.
- Angulo J C, López J I, Iriate I, Loizaga A, Flores N, et al., (1993)Angioma of the glans, report of cases. Arch Esp Urol 46: 47-48.
- Jimenez-Cruz J F, Osca J M (1993) Laser treatment of glanspenis hemangioma. Eur Urol 24: 81-83.
- Chaux A, Cubilla A L (2019) Penis and scrotum Benign tumorsEpithelioid hemangioma.
- Mondal S, Biswal D K (2015) Pal D P Cavernous hemangiomaof the glans penis. Urology Annals 7: 399-401.
- Fetsch J F, Sesterhenn I A, Mettinen M, Davis C J (2004) Epitheliod hemangioma of the penis: a clinicopathologic and immunohistochemical analysis of 19 cases, with special reference to exuberant examples often confused with epithelioid hemangioendothelioma and epithelioid angiosarcoma. Am J Surg Pathol 28: 523 -533.
- Srigley J R, Ayala A G, Ordonez N G, Nostrand A W (1985) Epithelioid hemangioma of the penis. A rare and distinctive vascular lesion. Archives of Pathology & Laboratory Medicine 109: 51-54.
- GangKak G, Mishra A, Priyadarshi S, Tomar V (2015) Large Genital Cavernous Hemangioma: A Rare Surgically Correctable Entity. Case Reports in Urology 950819.
- Lee J M, Wang J H, Kim H S (2008) Multiple Cavernous Hemangiomas of the Glans Penis, Penis, and Scrotum. Korean J Urol 49: 92-94.
- Ismail M, Damato S, Freeman A, Nigam R (2011) Epithelioid hemangioma of penis: case report and review of literature J Med Case Rep 5: 260.
- Pul M, Pul N (1995) Cavernous hemangioma of the penis inan infant. International Urology and Nephrology 27: 113-115.
- Manerikar K, Singh G, Ali I (2015) Intralesional Sclerotherapy in Hemangiomas of the Glans Penis. International Journal of Scientific Study. Sep; 3: 207-209.
- Aydur E, Erol B, Tahmaz L, Irkilata H, Eken C, et al., (2008) Coagulation of a giant hemangioma in glans penis with holmium laser. Asian Journal of Andrology Sep; 10: 819-821.
- Deb P P, Sarkar A (2018) Penile Granular Hemangioma Treated With Intralesional Sclerotherapy. Scholars Journal of Medical Case Reports (SJMCR) 6: 273-275.
- Savoca G, De Stefani S, Buttazi L, Gattuccio I, Trombetta C, et al., (2000) Sclerotherapy of the glans penis. Urology 56: 153.
- López V, Ricart J M, López I, Martin J M, Marton D, et al., (2012) Vascular malformation of the glans penis successfully treated with Nd:YAG laser Dermatology Online Journal 18: 16
- Norouzi B B, Shanberg A M (1998) Laser Treatment of Large Venous Hemangiomas of The Penis. Journal of Urology 160: 60-62.
- Zhang D, Zhang H, sun P, Li P, Xue A, et al., (2014) A Creative Therapy in Treating Cavernous Hemangioma of Penis. J Sex Med 11: 2605-2610.
- Sharma G (2004) Haemangioma of glans penis. The Internet Journal of Urology 3:1.
- Alter G J (1993) Trengove-Jones Guy, Charles E H. Jr.Hemangioma of penis and scrotum. Urology Aug 42: 205-208.
- Kumar A, Goyal N K, Trivedi S, Dwivedi U S, Singh P B, et al., (2008) Primary Cavernous Hemangioma of the Glans Penis: Rare Case Report with a Review of the Literature. Aesthetic Plastic Surgery 32: 386-388.
- Ranganayakulu B, Reddy K S, Raju S (1999) Hemangioma of penis. Indian Journal of Dermatology, Venearology and Leprology 65: 85.
- Kishore M, Ahuja A, Bhardwaj M (2017) Epithelioid hemangioma of penis mimicking malignancy: A rare case. Urology Annals 9: 397.
- Hemal A K, Goswami A K, Sharma S K, Radotra B D, Malik N, et al., (1989) Penile Venous Haemangioma. Australian and New Zealand Journal of Surgery 59: 814-816.
- Yoshikawa H, Ikeuchi T, Ohta M, Takanishi R, Matsumoto K, et al., (1995) Hemangioma of the glans penis: A case report. Journal of the Showa Medical Association 55: 636-638.
- Freeman A, Damato S, Ismail M, Raj N (2011) Epithelioid hemangioma of the penis: case report and review of literature. Journal of Medical Case Reports 5: 260.
- Ozdamar M Y, Zengin K, Tanik S, Albayrak S, Soyuer I, et al., (2014) Cavernous hemangioma of the glans penis: Concomitant surgical excision with circumcision in an uncircumcised patient. Pediatric Urology Case Reports 1: 1-4.
- Singh A P, Gupta A K, Pardeshi R, Ansari M, Barolia D K, et al., (2018) Diffuse hemangioma of penis, scrotum, perineum, and anal canal – A rare case report. Nigerian Journal of Plastic Surgery 14: 12-14.
- Akyol I, Jayanthi V R, Luquette MH (2008) Verrucuous Hemangioma of the Glans Penis. Urology 72.
- Kumar A, Goyal N K, Trivedi S, Dwivedi U S, Singh P, et al., (2008) Primary Cavernous Hemangioma of the Glans Penis: Rare Case Report with A Review of the Literature. Aesthetic Plastic Surgery 32: 386-388.
- Smart R H, Newton D E (1975) Hemangioma of the peniswith blue rubber bleb nevus. Journal of Urology 113: 570-571.
- Rastogi R (2008) Diffuse Cavernous Hemangioma of the Penis, Scrotum, Perineum, and Rectum - A rare tumor. Saudi J Kidney Dis Transpl 19: 614 -618.
- Echchaoui A, Zaouri H (2015) Penile Cavernous Hemangioma. International Journal of Medicine and Surgery 2: 26.
- Rajender A, Phillips E, Munarriz R (2015) Penile epithelioid hemangioma: A case report. Journal of Integrative Nephrology & Andrology 2: 38-40
- Ulker V, Esen T (2004) Hemangioma of the Glans Penis Treated with Nd: YAG Laser. International Urology and Nephrology 37: 95-96.
- Zhou Y, McKay K, Shen D (2014) Multifocal Epithelioid Hemangioma of the Penis. American Journal of Clinical Pathology 142: 256.
- Hemal A K, Aron M, Wadhwa S N (1998) Intralesional Sclerotherapy in the Management of Hemangiomas of the Glans Penis. Journal of Urology 159: 415-417.
- Casale A J, Menashe D S (1998) Massive Strawberry Hemangioma of the Male Genitalia. The Journal of Urology 141: 593-594.
- Deb P P, Sarkar A (2018) Penile Granular Hemangioma Treated With Intralesional Sclerotherapy Scholars Journal of Medical Case Reports (SJMCR) 6: 273-275
- Yigiter M, Arda I S, Hicsonmez A (2008) An unusual cause of paraphimosis: hemangioma of the glans penis. Journal Of Pediatric Surgery 43: 31-33.
- Ergun O, Ceylon B G, Armagan A (2009) Kaohsiung A Giant Scrotal Cavernous Hemangioma Extending to The Penis And Perineum: A Case Report. J Med Sci 25: 559-561.
- Sumer F, Yildirim I, Aydur E, Irkilata C, Kocaoglu M, et al.,(2008) Penile Cavernous Hemangioma. TURK J Urol 34: 269-271.
- Toshihiko T, Takehisa I, Yoshinori I, Toshiya K, Kazunori K, et al., (1998) Cutaneous Hemangioma of the Penis Successfully Treated with Sclerotherapy and Ligation. International Journal of Urology 5: 396-397.
- Javed A, Mahren S, Faig S (2018) Penile hemangioma in a prostate cancer patient: a case report. J Dow Uni Health Sci 12: 120-123.
- Gite Venkat G A, Atul Singal Nicose Jayant V, Jain Hitesh M (2017) Concealed Hemangioma: Rare Presentation Journal of Medical Science And Clinical Research 5: 29920-29922.
- Yang Q, Tang I Q, You Z M, Ran X, Yuping R, et al., (2016) Itraconazole Oral Solution Cured a Case of Infantile Hemangioma and Penis with Ulcer: Monitoring by Demoscopy ECRONICON OPEN ACCESS E C PAEDIATRICS 3: 346-349.
- Heidenreich A, Rothe H, Derschum W, von Vietsch H (1992) Hemangioma of the glans penis. Its therapy by neodymium: YaG laser Der Urologe 31: 94-95.
- Tsujii T (1998) Cutaneous Hemangioma of the Penis Successfully treated with sclerotherapy. Int J Urol 5: 396-397.
- Akin Y, Sarac M, Yucel S (2013) Rare malformations of glans penis Arteriovenous malformation. Nigerian Journal of Clinical Practice 16: 389-391.
- Basu P, Cohen P R (2018) Penile Angiokeratoma (Peaker): ADistinctive Subtype of Genital Angiokeratoma Cureus 10: 3793.
- Balsarkar D, Gore M A (2000) Venous malformations of glans penis: An unusual cause of hematuria. Indian Journal of Urology 17: 50-51.
- Shamsa A, Modaghegh H S, Asadpour A (2016) Glans Penis Hemangioma: A Rare case Report and its Treatment. Journal of Research in Urology Winter 1: 31- 35.



