Research Article - (2025) Volume 9, Issue 1
Green Synthesis of Magnetic Nano Composite Ni0.1CO0.9Fe2O4@AC Using the Medicinal Plant Extract of Malva
Received Date: Feb 03, 2025 / Accepted Date: Mar 06, 2025 / Published Date: Apr 28, 2025
Copyright: ©�2025 Mohammad Kazazi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Einali, S., Kazazi, M. (2025). Green Synthesis of Magnetic Nano Composite Ni0.1CO0.9Fe2O4@AC Using the Medicinal Plant Extract of Malva. Adv Nanoscie Nanotec, 9(1), 01-05.
Abstract
One of the problems with synthesized nano adsorbents is their single-use nature. However, the synthesis of magnetic nano biocom- posites of Nickel /Cobalt/ Ferrite/ Iron and activated Carbon (AC) allows for multiple uses. The entry of wastewater containing dye molecules is a major issue that the world is facing, making wastewater treatment essential. With technological advancements, numerous solutions have been proposed to address this problem. Adsorption is a suitable method for water purification due to its low cost and high efficiency. Various materials are used as surface adsorbents, and magnetic nano composites are favored for their easy separation. In this study, magnetic nano biocomposites of Nickel /Cobalt /Ferrite/Iron and AC were synthesized and analyzed using FT- IR, XRD and SEM with results showing their nanometer scale.
Keywords
Synthesis, Magnetic Nano Biocomposite, FT- IR, SEM, Medicinal Plant Malva
Introduction
In recent years, the development of efficient green chemistry methods for the synthesis of metal Nanoparticles (Nps) has attracted significant attention from many researchers. Numerous efforts have also been made to promote the use of green chemistry methods and environmentally friendly
materials in Nps synthesis. The use of biological organisms such as microorganisms, plant extracts or plant biomass can replace chemical and physical methods for producing Nps in an environmentally friendly manner [1]. Among the common methods, the use of extracts from various plants has shown positive results in the preparation of Nps.
Generally, plant extracts, which act as stabilizers and capping agents to control crystal growth, are composed of various metabolites such as terpenoids, phenols, proteins or carbohydrates. These compounds are directly responsible for the extract ability to perform the redox reactions needed for Nps formation. Unlike time consuming chemical and physical methods that come with their own complexities, the green method is much simpler and safer.
In fact, green synthesis methods utilize principles and guidelines that can reduce or eliminate hazardous compounds in the production of chemicals [2].In recent years, the fabrication of Nps with magnetic properties has gained attention. Metal oxide Nps are of interest due to their unique properties in various optical and electrical applications. Changes in the size of semiconductors and metals lead to significant changes in their optical properties, such as color and energy gap. Iron Nps, which are expected to be non-toxic to humans, have garnered significant attention due to their unique properties, including high magnetic strength, biocompatibility and biodegradability. Nickel Nps are also highly regarded for their applications as catalytic materials in magnetic applications and in medical uses. Additionally, particle size, shape, and the synthesis route of Nickel Nps are key parameters in determining their catalytic behavior [3].
For this reason, magnetic Nps composed of Iron, Nickel and AC were simultaneously examined. For the first time, an aqueous of medicinal plant extract of Malva was used to prepare Nps with super magnetic properties in present study. This plant, with its unique medicinal properties, not only reduces Iron, Nickel and Cobalt to Nps but also imparts antibacterial properties to them. The proposed method in this study eliminates the use of chemical substances and solvents for synthesis of magnetic Nps of Iron, Nickel and Cobalt [4].
Emergence of nanotechnology in the science and industry of magnetic materials, has led significant improvement in the quality of magnets resulting in the creation of magnets with small dimensions and strong magnetic forces [5].
Biosynthesis of Nickel /Ferrite Nps prepared using an aqueous extract of Manna of hedysarum1 (Abdol Hossein Miri and et al., 2021) [6].Considering the importance and applications of Nickel / Ferrite Nps, this study aimed to synthesize these Nps using a cheap, non- toxic and fast method. Therefore, Ni0.1CO0.9Fe2O4 Nps were produced for the first time using the green synthesis method with the Malva plant [7].AC refers to a group of carbonaceous materials with high internal surface area and porosity which due to their significant internal area, porous and permeable structure, high absorption capacity, capability of surface reactivation, as well as low cost compared to inorganic absorbents such as Zeolites, are unique materials. Their important applications include the removal of odor, color, unwanted tastes from water in household and industrial operations, solvent recovery, air purification especially in restaurants, and in food and chemical industries [8, 9].
Experimental Methods Materials
Malva plant, Ni(NO3 )2 .6H2 O, CoCl2 .6H2 O, FeCl3 .6H2 O, Activated Carbon (AC), Distilled water.
Equipment
Magnetic Stirrer, Oven, Centrifuge, Electric Furnace, Digital Scale, Scanning Electron Microscope (SEM), Ultraviolet-Visible Spectroscopy (UV-Vis), Fourier Transform Infrared Spectroscopy (FT-IR) and X-Ray Diffraction Spectrometer (XRD).
Extraction of Malva Extract
The plant sample used in this study was obtained from the vicinity of Tuyserkan city in Hamedan province. After washing and drying, approximately 40 g of its powder was weighed and 200 ml of distilled water was added and was left aside for 48 h.
Synthesis of Magnetic Nano composite
Initially, solutions of Iron (III) Chloride hexahydrate, Cobalt (II) Chloride tetrahydrate, and Nickel(II) Chloride hexahydrate were prepared with a ratio of 0.1:0.9:4 and concentrations of 0.2, 0.09, and 0.05 M , respectively. These solutions were transferred to a reaction vessel and stirred for 30 min, then 1 g of AC was added to the reaction vessel to produce nano biocomposite.
In the next step, 100 ml of Malva extract solution was added to the stirring solution drop wise. The solution was left 1 h till reaction was completed and pH was controlled. Subsequently, the mixture treated by Ultrasonic for 30 min then washed with distilled water. Finally, the mixture dried in Oven at 100 °C for 2 h and calcined in Electric Furnace at 550 °C for 1 h.
Results & Discussion
FT-IR spectroscopy was used to identify functional groups. The observed peak at 3432 cm-1 corresponds to the O-H groups of water molecules as the active surface of Nps absorbs water. The observed peaks at 1632 cm-1, 1403 cm-1 are attributed to the carbonyl group , C-O bending group , respectively .The observed peaks at 570 cm-1 and 474 cm-1 are correspond to metal- oxygen bonds (M-O), as shown in Fig (1).
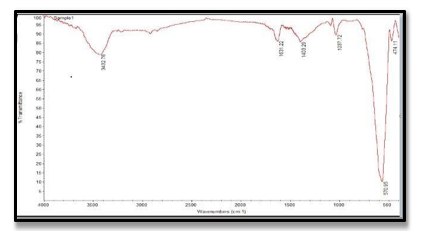
Figure 1: FT-IR spectrum of Magnetic Nano Biocomposite
XRD Spectrum of Magnetic Nano Biocomposite
The XRD spectrum of the magnetic nano biocomposite is shown in Fig (2), displaying the X-ray diffraction pattern of the AC /Nickel / Cobalt /Ferrite magnetic nano composite. Observed peaks at angles of 2ÃÂ?¨=27, 30, 31, 35, 45, 57 and 62 which are consistent with the crystalline structure of the nano composite. The identified peaks confirm the cubic spinel structure of the material, consistent with the research literature. The peak at approximately 27 indicates the presence of amorphous Carbon in the nano composite structure, while maintaining the crystalline structure of Nickel /Cobalt / Ferrite.
1 In Asian countries called it Taranjabin
The peaks at angles of 2ÃÂ?¨=62, 57, 35and 31 indicate the crystalline structure of magnetic Nps. The decrease in the number and intensity of peaks, as well as their shifting towards higher angles, indicates the filling of the space between the planes by AC. Additionally, the peak around 31 indicates the presence of amorphous Carbon and the successful reaction of AC. These peaks indicate that AC had no significant effect on the structure of Nickel/ Cobalt/ Ferrite
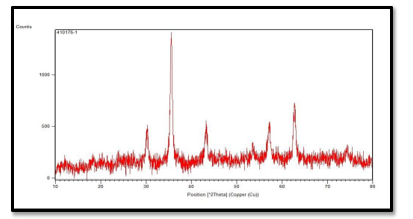
Figure 2: XRD Spectrum of Magnetic Nano Biocomposite
Field Emission Scanning Electron Microscopy (FESEM) Image
Field Emission Scanning Electron Microscopy (FESEM) was used to investigate the morphology of the synthesized Nps. SEM images were used to observe the surface morphology and calculate the size of the synthesized Nps using the Scherrer 2 equation. The particle size is approximately 40 nm. Results are provided at magnifications of 200, 500 nm and 1 μ, as shown in Fig (3).

Figure 3: Field Emission Scanning Electron Microscopy (FESEM) images
Effect of Time Variation on the Dye Adsorption Process
At first, a 100 ml solution of Methylene Blue (MB) with a specified concentration was prepared. Then, four samples of the solution were prepared, a control sample and the other three samples contained 20 ml of MB solution with a specified concentration and 0.1 g of the nano composite with stop times of 2, 4, and 8 h. Their absorption rates were measured using UV- VIS and results show a direct relation between dye removal rate and time duration, as shown in Fig (4).

Figure 4: Effect of Time Variation on the Dye Adsorption Process
Increasing surface area leads to enhanced photo catalytic properties of Ni0.1CO0.9Fe2O4@AC Nps due to active radicals (•OH) and consequently increased dye adsorption of MB. UV-Vis results show that the dye removal rate increases with the increasing amount of Nps, as shown in Fig (5).
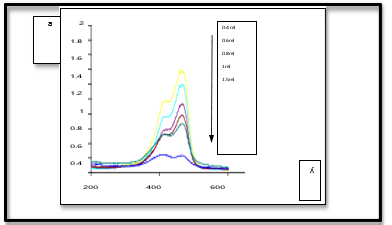
Figure 5: MB Adsorption by Ni0.1CO0.9Fe2O4@AC
The Effect of External Magnetic Field on the Separation of Nano Biocomposite
The magnetic separation of the nano biocomposite by an external magnetic field is shown in Fig (6). In addition to effective dye adsorption, the suitability of the magnetic properties of this nanomaterial indicates its application as a nano surface absorbent

Figure 6: The Effect of External Magnetic Field on Separation 0f Nano Biocomposite
Conclusions
The green synthesis of Nps offers advantages such as biocompatibility, low production cost, use of non-toxic solvents, ease of production and application that shows importance of green chemistry. Nps produced using green method showed better particle size distribution and stability compared to other methods.
A drawback of common synthesized nano adsorbents is their single- use nature, which can be addressed by synthesizing magnetic Nickel/ Cobalt /Ferrite / Iron and AC nano biocomposites, enabling multiple uses. On the other hand, a major problem facing the world is the disruption of the organs of living organisms due to the ingestion of carcinogenic and mutagenic dye molecules, as well as the entry of wastewater containing dye molecules, so wastewater treatment is essential .In this study, we synthesized magnetic nano biocomposites that do not pose environmental problems.
Recommendations
Considering the expanding applications of nanotechnology, we encourage scientists and researchers to conduct further research on synthesis of Nps using green chemistry.
References
- Iravani, S. (2011). Green synthesis of metal nanoparticles using plants. Green chemistry, 13(10), 2638-2650.
- Din, M. I., Nabi, A. G., Rani, A., Aihetasham, A., & Mukhtar,M. (2018). Single step green synthesis of stable nickel and nickel oxide nanoparticles from Calotropis gigantea: catalytic and antimicrobial potentials. Environmental Nanotechnology, Monitoring & Management, 9, 29-36..
- Fardood, S. T., Ramazani, A., & Moradi, S. (2017). A novel green synthesis of nickel oxide nanoparticles using Arabic gum. Chemistry Journal of Moldova, 115-118.
- Mollamahaleh, Y. B., Hosseini, D., Mazaheri, M., & Sadrnezhaad, S. K. (2011). Surfactant-free production of Ni- based nanostructures. Materials Sciences and Applications, 2(5), 444-452.
- Navaei Diva, T., Zare, K., Taleshi, F., & Yousefi, M. (2017). Synthesis, characterization, and application of nickel oxide/ CNT nanocomposites to remove Pb 2+ from aqueous solution. Journal of Nanostructure in Chemistry, 7, 273-281.
- Miri, A., & Sarani, M. (2021). Biosynthesis of Ferrit/Nikle NPs by aqueous extract of Manna of hedysarum.
- Chada, N., Romanos, J., Hilton, R., Suppes, G., Burress, J., & Pfeifer, P. (2012, February). Activated carbon monoliths for methane storage. In APS March Meeting Abstracts (Vol. 2012, pp. W33-012).
- Soo, Y., Chada, N., Beckner, M., Romanos, J., Burress, J., & Pfeifer, P. (2013, March). Adsorbed methane film properties in nanoporous carbon monoliths. In APS March Meeting Abstracts (Vol. 2013, pp. M38-001).


