Research Article - (2025) Volume 9, Issue 1
Green Synthesis of Magnetic Iron Oxide Nanoparticles Using Quince Leaf Extract Coated with SiO2 Using Ultrasonic Treatment
Received Date: Feb 03, 2025 / Accepted Date: Mar 06, 2025 / Published Date: Apr 28, 2025
Copyright: ©©2025 Mohammad Kazazi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Souri, S, Kazazi, M. (2025). Green Synthesis of Magnetic Iron Oxide Nanoparticles Using Quince Leaf Extract Coated with SiO2 Using Ultrasonic Treatment. Adv Nanoscie Nanotec, 9(1), 01-05.
Abstract
Today, one of the most effective methods for synthesizing Nanoparticles (Nps) is through plant extracts. The chemical synthesis of Nps is expensive and causes environmental pollution. In this study, quince leaf extract from a tree was used for the green syn- thesis of magnetic iron oxide Nps. The magnetic iron oxide Nps were coated with silicon dioxide Nps and the color change in the solution and the brown precipitates, confirmed the successful green synthesis of magnetic iron oxide Nps. Analyses including Vi- brating Sample Magnetometer (VSM), Fourier Transform Infrared Spectroscopy (FT-IR), X-ray Diffraction (XRD) and Scanning Electron Microscopy (SEM) were performed. The average size of the crystalline magnetite (Fe3O4) Nps was calculated using the Debye-Scherrer formula, which is in the nanometer scale.
Keywords
Green Synthesis, Environmental Pollution, Silicon Dioxide Nps, Brown Precipitates, Crystalline Nps.
Introduction
Particles with a high surface-to-volume ratio exhibit new and unique properties [1]. Nanotechnology involves the study of materials that have at least one dimension smaller than 100 nm, such that the physical, chemical and biological properties of these particles which fundamentally differ from their original source [2]. Iron oxide Nps are a class of nanomaterials composed of iron and oxygen atoms arranged in a crystalline structure at the nanoscale, typically ranging in size from 1 to 100 nm [3].
One of the most common forms of iron oxide Nps is magnetite (Fe3O4), which is the most magnetic form of iron oxide and is composed of both Fe²âº and Fe³âº ions. Some iron oxide Nps, such as magnetite and maghemite, exhibit superparamagnetism, meaning they can be easily magnetized. The high surface area of iron oxide Nps increases their reactivity and adsorption capacity, making them useful in various applications [4].
Iron oxide Nps are generally considered biocompatible and are widely used for biomedical applications, such as drug delivery, magnetic resonance imaging (MRI) contrast agents and hyperthermia treatment. Due to their high surface area and redox properties, iron oxide Nps can act as catalysts in various chemical reactions. These Nps have a wide range of applications in fields such as biomedicine, environmental remediation, catalysis, energy storage, and electronics. These properties make them valuable in areas such as targeted drug delivery, magnetic separation, water purification and energy storage devices [5].
Magnetic Nps provide suitable substrates for performing heterogeneous catalytic reactions due to their easy recoverability from the reaction environment, typically achieved using a strong magnet. Although various magnetic materials can be used in the fabrication of magnetic nanocomposites, iron magnetic oxides are preferred due to their non-toxic nature, low cost and prominent magnetic properties. Additionally, magnetic Nps, due to their high specific surface area and strong magnetic dipole interactions, can easily agglomerate and oxidize. Even under mild oxidative conditions (such as exposure to atmospheric oxygen), rapid oxidation of the nanoparticle surfaces occurs, leading to the formation of oxide layers, which in turn alters the properties of the samples [6]. One of the well-known methods to preserve the unique magnetic properties of these Nps and protect them is through this oxidation reaction [7]. Coating Nps can be achieved using various compounds such as carbon, silica, precious metals, certain metal oxides, organic polymers and surfactants [8-12]. Silica is one of the very suitable coating agents. The process of coating magnetic Nps is relatively straightforward and the resulting coatings are environmentally friendly, exhibit good stability and low toxicity [13].
The silica layer coating Fe3O4 prevents the agglomeration of these Nps and effectively creates a protective layer for Fe3O4 Nps. Furthermore, the chemical inertness of the silica coating enhances the biocompatibility of the magnetic Fe3O4 Nps [14]. The silanol groups (OH-Si) present on the surface of the silica coating provide an easy pathway for the attachment of various functional groups [15]. Therefore, different metal oxides can be deposited onto the silica surface by forming bonds between the silanol OH groups and the metal oxides. The synthesis of metal oxide Nps using microorganisms and plants, known as the green synthesis method, has gained attention due to its cost-effective, simple and more environmentally friendly synthetic conditions compared to conventional chemical and physical methods [16].
In addition to various biological materials such as yeasts, bacteria, fungi, mosses and eggshells that are used for these purposes, plant extracts have also attracted significant attention due to their green characteristics and easy availability [17]. Plant extracts contain antioxidants such as polyphenols, reducing sugars, nitrogenous bases and amino acids [18-20]. Since the production of Nps based on physical and chemical methods is costly and causes serious harm to the environment, there is currently a notable shift towards biological and green approaches for nanoparticle synthesis. These methods are considered suitable alternatives to chemical and physical methods due to their efficiency, cost-effectiveness, environmental compatibility and safety [21,22]. In this approach, biological systems such as fungi, algae, bacteria and plant extracts are utilized for the synthesis of Nps [23-37].
The plant known scientifically as cydonia oblonga is native to Iran, Southern Europe and Asia Minor and is now cultivated globally in countries such as Turkey, China, Uzbekistan, Iran, Argentina and New Zealand. It is a tree from the rose family, belonging to the apple family, with hairy trichomes of its leaves [38]. The aim of this research is to use quince extract as a reducing and stabilizing agent in the synthesis of iron magnetic Nps.
Experimental Methods Materials
Iron (II) chloride hexahydrate, Iron (III) chloride tetrahydrate, Tetraethyl orthosilicate (TEOS),
Quince leaf extract.
Equipment
Oven, X-Ray Diffraction (XRD) spectrometer, Scanning Electron Microscope (SEM), Fourier- Transform Infrared spectrometer (FT-IR), Magnetic heater, Ultrasonic device, Vibrating Sample Magnetometer (VSM) and Energy Dispersive X-ray (EDX) spectrometer.
Removing quince leaf extract
First, 50 g of powdered quince leaves were added to 200 ml of distilled water, then placed it on a magnetic heater at temperature of 100 °C and the stirring speed of 900 rpm for 30 min,. After that, mixture was placed in the ultrasonic bath to fully remove the extract, for 30 min.
Green synthesis of magnetic iron oxide nanoparticles
First, 1 g of iron (II) chloride hexahydrate was added 100 ml of distilled water. Next, 2 g of iron (III) chloride tetrahydrate was added then placed on a magnetic heater to homogenize. Finally, 100 ml of the extracted quince solution was added dropwise, until a dark-colored solution was obtained, resulting in the precipitation of Nps.
Coating of magnetic iron oxide nanoparticles with SiO2
First, 1 g of iron magnetic oxide added to 50 ml of distilled water and then the solution was treated by ultrasonic for 20 min to homogenize. Finally, 30 ml of ethanol, a few drops of ammonia and 5 ml of tetraethyl orthosilicate were added to the solution and treated by ultrasonic for 60 min.
Results and Discussion
The analyses conducted, including X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FT-IR), scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDX), and vibrating sample magnetometer (VSM), were performed at the Central Laboratory of Sharif University of Technology and the Central Laboratory of Shahid Beheshti University.
Fourier transform infrared spectroscopy of magnetic iron oxide Nps with SiO2
FT-IR spectroscopy is used to identify functional groups and the types of their bonds. The strong and broad stretching peak observed around 3083 cm-1 corresponds to the OH group of water and the silanol group. The strong peaks around 1208 cm-1 and 1100 cm-1 are related to Si-O bonds, while the peaks at 430 cm-1, 473 cm-1 and 760 cm-1 are correspond to Fe-O bonds ,as shown in Figure (1).
Figure 1: FT-IR Analysis of Magnetic Iron Oxide Nps with SiO2
Scanning electron microscopy analysis of magnetic iron oxide Nps with SiO2
SEM spectroscopy is used to investigate the morphology and structure of the Nps. In present study, magnifications of 200 nm, 500 nm and 1 μ were used. The images show that the iron oxide Nps are dispersed on silica plates and the average particle size is consistent with the values obtained from XRD calculations. The results indicate that the iron oxide Nps coated with SiO2 are in the nanoscale, as shown in Figure (2).

Figure 2: SEM Analysis Of Magnetic Iron Oxide Nps with SiO2
X-ray Diffraction Of Magnetic Iron Oxide Nps Coated with SiO2
XRD analysis was used to determine the phase and examine the crystalline structure, types of structural defects and size of the iron oxide Nps coated with SiO2. The horizontal and vertical axes represent the diffraction angle (2θ) and the intensity of the X-ray, respectively. The width of the peaks in the diffraction pattern indicates the nanometric size of the iron oxide particles coated with silicon dioxide and the results are consistent with the XRD standard card ( JCPDS 75-1609),as shown in Figure ( 3).
Figure 3: X-ray Diffraction of Magnetic Iron Oxide Nps Coated with SiO2
Vibrating Sample Magnetometer of Magnetic Iron Oxide Nps Coated with SiO2
VSM analysis is the primary method for studying the magnetic properties of materials. The result of this analysis is the acquisition of a hysteresis curve or remanence loop of the materials, which allows for the determination of data such as coercivity, saturation magnetization, magnetic permeability and the magnetism classification of material, such as ferromagnetic, paramagnetic and superparamagnetic. Remanence loop of the iron magnetic oxide Nps coated with SiO2 is in fields ranging from -15000 to +15000 Oersted. The hysteresis curve indicates that the coated sample exhibits soft magnetic properties, as shown in Figure (4).
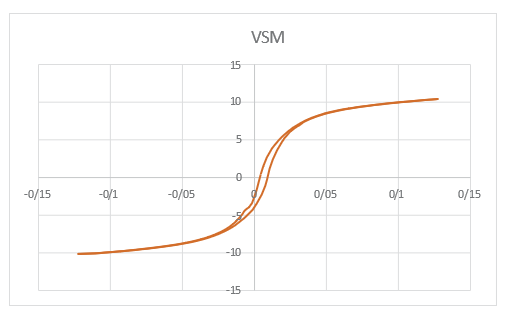
Figure 4: VSM Spectrum Of Magnetic Iron Oxide Nps Coated with SiO2
Energy Dispersive X-Ray Spectroscopy of Magnetic Iron Oxide Nps Coated with SiO2
EDX is an analytical method used for the structural analysis or chemical properties of a sample. This method relies on the interaction between an x-ray excitation source and a sample, allowing for the determination of the percentage of elements present in the sample, as shown in Figure (5).
Figure 5: EDX Spectrum Of Magnetic Iron Oxide Nps Coated with SiO2
Conclusion
Magnetic iron oxide Nps were successfully synthesized using the extract of the quince leaves and coated with tetraethyl orthosilicate as a source of silicon dioxide. Finally, the relevant analyses confirmed the structure and nanometric size of the composite.
Recommendations
Due to the widespread applications of iron oxide Nps and the non-toxicity of their coating with silicon dioxide, further research should be conducted in this field.
References
- Shen, Y. F., Tang, J., Nie, Z. H., Wang, Y. D., Ren, Y., & Zuo,L. (2009). Preparation and application of magnetic Fe3O4 nanoparticles for wastewater purification. Separation and purification technology, 68(3), 312-319.
- Liu, J., Liu, G., Zang, L., & Liu, W. (2015). Calcein- functionalized Fe 3 O 4@ SiO 2 nanoparticles as a reusable fluorescent nanoprobe for copper (II) ion. Microchimica Acta, 182, 547-555.
- Guo, C., Lu, W., Wei, G., Jiang, L., Yu, Y., & Hu, Y. (2018).Formation of 1D chain-like Fe3O4@ C/Pt sandwich nanocomposites and their magnetically recyclable catalytic property. Applied Surface Science, 457, 1136-1141.
- Hou, S., Li, X., Wang, H., Wang, M., Zhang, Y., Chi, Y., & Zhao, Z. (2017). Synthesis of core–shell structured magnetic mesoporous silica microspheres with accessible carboxyl functionalized surfaces and radially oriented large mesopores as adsorbents for the removal of heavy metal ions. RSC advances, 7(82), 51993-52000.
- Wolf, M., Fischer, N., & Claeys, M. (2018). Surfactant-free synthesis of monodisperse cobalt oxide nanoparticles of tunable size and oxidation state developed by factorial design. Materials chemistry and physics, 213, 305-312.
- Jokandan, S. F., Badi, M. Y., Esrafili, A., Azari, A., Tarhandeh, H., & Kermani, M. (2019). Investigation of the eficiency of powder activated carbon magnetized with Fe3O4 nanoparticles in the removal of catechol from aqueous solutions by response surface methodology.
- Maleki, S., Falaki, F., & Karimi, M. (2019). Synthesis of SDS micelles-coated Fe 3 O 4/SiO 2 magnetic nanoparticles as an excellent adsorbent for facile removal and concentrationNanostructure in Chemistry, 9, 129-139.
- Rambabu, K., Bharath, G., Banat, F., & Show, P. L. (2021). Green synthesis of zinc oxide nanoparticles using Phoenix dactylifera waste as bioreductant for effective dye degradation and antibacterial performance in wastewater treatment. Journal of hazardous materials, 402, 123560.
- Peng, X., Xu, F., Zhang, W., Wang, J., Zeng, C., Niu, M., & Chmielewská, E. (2014). Magnetic Fe3O4@ silica–xanthan gum composites for aqueous removal and recovery of Pb2+. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 443, 27-36.
- Wolf, M., Fischer, N., & Claeys, M. (2018). Surfactant-free synthesis of monodisperse cobalt oxide nanoparticles of tunable size and oxidation state developed by factorial design. Materials chemistry and physics, 213, 305-312.
- Chen, M., Yamamuro, S., Farrell, D., & Majetich, S. A. (2003). Gold-coated iron nanoparticles for biomedical applications. Journal of applied physics, 93(10), 7551-7553.
- Piranoul., Q. F, Bavorsi, F & Dadashian. (2017) Production of core/shell microstructure of Fe3O4/SiO2/TiO2 and investigating their structural properties, New Processes in Materials Engineering, No 4, pp.143-150.
- Pattanayak, M., & Nayak, P. L. (2013). Ecofriendly green synthesis of iron nanoparticles from various plants and spices extract. International Journal of Plant, Animal and Environmental Sciences, 3(1), 68-78.
- Jeong, U., Teng, X., Wang, Y., Yang, H., & Xia, Y. (2007). Superparamagnetic colloids: controlled synthesis and niche applications. Advanced Materials, 19(1), 33-60.
- Gholamrezapor, E., & Eslami, A. (2019). Sensitization of magnetic TiO2 with copper (II) tetrahydroxylphenyl porphyrin for photodegradation of methylene blue by visible LED light. Journal of Materials Science: Materials in Electronics, 30(5), 4705-4715..
- Kharissova, O. V., Dias, H. R., Kharisov, B. I., Pérez, B. O., & Pérez, V. M. J. (2013). The greener synthesis of nanoparticles. Trends in biotechnology, 31(4), 240-248.
- Njagi, E. C., Huang, H., Stafford, L., Genuino, H., Galindo, H. M., Collins, J. B., ... & Suib, S. L. (2011). Biosynthesis of iron and silver nanoparticles at room temperature using aqueous sorghum bran extracts. Langmuir, 27(1), 264-271.
- Ahmad, N., Sharma, S., Alam, M. K., Singh, V. N., Shamsi, S. F., Mehta, B. R., & Fatma, A. (2010). Rapid synthesis of silver nanoparticles using dried medicinal plant of basil. Colloids and Surfaces B: Biointerfaces, 81(1), 81-86.
- Kasthuri, J., Veerapandian, S., & Rajendiran, N. (2009). Biological synthesis of silver and gold nanoparticles using apiin as reducing agent. Colloids and Surfaces B: Biointerfaces, 68(1), 55-60.
- Panigrahi, S., Kundu, S., Ghosh, S., Nath, S., & Pal, T. (2004). General method of synthesis for metal nanoparticles. Journal of nanoparticle Research, 6(4), 411-414.
- Liu, T., Wang, L., Yang, P., & Hu, B. (2008). Preparation of nanometer CuFe2O4 by auto-combustion and its catalytic activity on the thermal decomposition of ammonium perchlorate. Materials Letters, 62(24), 4056-4058.
- Jamzad, M., Kamari Bidkorpeh, M., & Naderi, F. (2019). (Short paper) Potential evaluation of the aqueous extract of Portulaca oleracea L. in the synthesis of Iron oxide nanoparticles. Eco- phytochemical Journal of Medicinal Plants, 7(2), 117-128.
- Ardakani, L. S., Alimardani, V., Tamaddon, A. M., Amani, A. M., & Taghizadeh, S. (2021). Green synthesis of iron-based nanoparticles using Chlorophytum comosum leaf extract: methyl orange dye degradation and antimicrobial properties. Heliyon, 7(2).
- Lohrasbi, S., Kouhbanani, M. A. J., Beheshtkhoo, N., Ghasemi, Y., Amani, A. M., & Taghizadeh, S. (2019). Green synthesis of iron nanoparticles using Plantago major leaf extract and their application as a catalyst for the decolorization of azo dye. BioNanoScience, 9, 317-322.
- Cheera, P., Karlapudi, S., Sellola, G., & Ponneri, V. (2016). A facile green synthesis of spherical Fe3O4 magnetic nanoparticles and their effect on degradation of methylene blue in aqueous solution. Journal of Molecular Liquids, 221, 993-998.
- Singh, K. K., Senapati, K. K., & Sarma, K. C. (2017). Synthesis of superparamagnetic Fe3O4 nanoparticles coated with green tea polyphenols and their use for removal of dye pollutant from aqueous solution. Journal of environmental chemical engineering, 5(3), 2214-2221.
- Bibi, I., Nazar, N., Ata, S., Sultan, M., Ali, A., Abbas, A., ... & Iqbal, M. (2019). Green synthesis of iron oxide nanoparticles using pomegranate seeds extract and photocatalytic activity evaluation for the degradation of textile dye. Journal of materials research and technology, 8(6), 6115-6124.
- Pantidos, N., & Horsfall, L. E. (2014). Biological synthesis of metallic nanoparticles by bacteria, fungi and plants. Journal of Nanomedicine & Nanotechnology, 5(5), 1.
- Pattanayak, M., & Nayak, P. L. (2013). Green synthesis and characterization of zero valent iron nanoparticles from the leaf extract of Azadirachta indica (Neem). World Journal of Nano Science & Technology, 2(1), 06-09.
- Quintana, N., Weir, T. L., Du, J., Broeckling, C. D., Rieder,J. P., Stermitz, F. R., ... & Vivanco, J. M. (2008). Phytotoxic polyacetylenes from roots of Russian knapweed (Acroptilon repens (L.) DC.). Phytochemistry, 69(14), 2572-2578.
- Ramesh, A. V., Rama Devi, D., Mohan Botsa, S., & Basavaiah,K. (2018). Facile green synthesis of Fe3O4 nanoparticles using aqueous leaf extract of Zanthoxylum armatum DC. for efficient adsorption of methylene blue. Journal of Asian Ceramic Societies, 6(2), 145-155.
- Rasaee, I., Ghannadnia, M., & Honari, H. (2016). Antibacterial properties of biologically formed chitosan nanoparticles using aqueous leaf extract of Ocimum basilicum. Nanomed. J, 3(4), 240-247..
- Sari, I. P., & Yulizar, Y. (2017, April). Green synthesis of magnetite (Fe3O4) nanoparticles using Graptophyllum pictum leaf aqueous extract. In IOP Conference Series: Materials Science and Engineering (Vol. 191, No. 1, p. 012014). IOP Publishing.
- Singh, N., Jenkins, G. J., Asadi, R., & Doak, S. H. (2010). Potential toxicity of superparamagnetic iron oxide nanoparticles (SPION). Nano reviews, 1(1), 5358.
- Song, J. Y., & Kim, B. S. (2009). Rapid biological synthesis of silver nanoparticles using plant leaf extracts. Bioprocess and biosystems engineering, 32, 79-84.
- Barzinjy, A. A., Hamad, S. M., Abdulrahman, A. F., Biro, S. J., & Ghafor, A. A. (2020). Biosynthesis, characterization and mechanism of formation of ZnO nanoparticles using Petroselinum crispum leaf extract. Current Organic Synthesis, 17(7), 558-566..
- Bajpai, N., Tiwari, A., Khan, S. A., Kher, R. S., Bramhe, N., & Dhoble, S. J. (2014). Effects of rare earth ions (Tb, Ce, Eu, Dy) on the thermoluminescence characteristics of sol–gel derived and γâ?irradiated SiO2 nanoparticles. Luminescence, 29(6), 669-673.
- http://www.loghatnaameh.com/dehkhodaworddetail- 0971383fc0414578b30202cc3a5d25ef-fa.html .


