Research Article - (2026) Volume 5, Issue 1
Green Synthesis, Characterization, and Biological Evaluation of Silver Nanoparticles Using Bassia Scoparia L. Leaves Extract
2Lecturer of Botany, Government Degree College, Pakistan
33Department of Chemistry, Nazarbayev University, Astana, Kazakhstan
4Department of Chemistry, University of Malakand, Kpk, Pakistan
5Department of Chemistry, Government Degree College Gulabad, Kpk, Pakistan
6Department of Materials Science, Beijing University of Chemical technology, China
Received Date: Jan 02, 2026 / Accepted Date: Jan 27, 2026 / Published Date: Feb 04, 2026
Copyright: ©2026 The authors. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Wahab, A., Alam, A., Ahmad, W., Ali, W., Khan, A., et al. (2026). Green Synthesis, Characterization, and Biological Evaluation of Silver Nanoparticles Using Bassia Scoparia L. Leaves Extract. J Traditional Medicine & Applications, 5(1), 01-09.
Abstract
Silver nanoparticles (AgNPs) are known for their unique properties and applications in fields such as medicine and environmental remediation. Recent environmental concerns and the need for sustainable technologies have spurred interest in eco-friendly synthesis methods, particularly green synthesis using plant extracts, which is a cost-effective and biocompatible alternative to traditional physical and chemical methods. Silver nanoparticles (AgNPs) were synthesized using an aqueous leaf extract of Bassia scoparia L. as a reducing and stabilizing agent, demonstrating a simple and sustainable method for nanoparticle production. Comprehensive characterization was performed using various techniques: Scanning Electron Microscopy (SEM) assessed surface morphology and size distribution, X-ray Diffraction (XRD) confirmed crystallinity, Fourier Transform Infrared Spectroscopy (FTIR) identified functional groups involved in synthesis, Ultraviolet-Visible (UV-Vis) Spectroscopy monitored surface plasmon resonance, and Zeta Potential analysis examined surface charge and colloidal stability of the AgNPs. The results confirm the successful biosynthesis of stable, crystalline silver nanoparticles, demonstrating the potential of Bassia scoparia L. as an effective plant source for green nanotechnology applications.
Keywords
Bassia Scoparia, AgNPs, XRD, SEM, FTIR, Unique Properties, Spectroscopy
Introduction
Chemistry, physics, pharmaceutical sciences, materials science, medicine, and agriculture are all included in the multidisciplinary discipline of nanotechnology. Precision farming, which maximizes resource utilization by examining crop production variability, is one way that its implementation has significantly improved agriculture. For precision agriculture to advance and environmental sustainability to be promoted, nanotechnology must be incorporated into farming operations [1]. Nanoparticles, due to their small size, high surface area, and unique optical properties, are utilized in agriculture for plant protection and nutrient delivery [2]. Nanoparticles, ranging in size from 1 to 100 nm, are composed of either dissolved, encapsulated, or integrated active compounds. They are fabricated into structures like Nano spheres and Nano capsules; the latter enclose substances within a non-toxic polymer shell, while the former allows for uniform distribution. Biodegradable polymer-based nanoparticles are particularly valuable for delivering proteins, genes, and peptides, especially those coated with hydrophilic polymers like polyethylene glycol (PEG). These serve as long-circulating particles. Nanoparticles are being investigated for targeted drug delivery and gene therapy due to their capability to target specific organs, sustain prolonged circulation, and assist with DNA transport [3].
Nanoscale metal oxide particles, particularly titanium dioxide (TiO2), are widely used in products such as toothpaste, cosmetics, sunscreens, and textiles. Silver (Ag) nano-powders are particularly noted for their strong antibacterial properties [4]. Metal oxides are preferred to organic antibacterial agents due to their greater safety and stability [5]. A Various physical and chemical methods are used to synthesize metal nanoparticles, but these are often costly, complex, and harmful to the environment. Consequently, there is a growing interest in sustainable and eco-friendly alternatives. Recent studies show that biological systems, particularly plants and algae, can effectively reduce inorganic metal ions to create metal nanoparticles. Among these methods, plant-based synthesis stands out for its higher production yields, safety, quicker synthesis, and lower cultivation costs [6].
Recent research highlights the significant role of microorganisms and biological entities in the synthesis of metal nanoparticles, moving away from traditional physical and chemical methods. The adoption of biological systems is favored for their simplicity, effectiveness, and alignment with green chemistry principles, as they avoid toxic and hazardous reagents [7]. Microorganisms with specific morphologies can create inorganic compounds at the nanoscale and demonstrate resistance to heavy metals through mechanisms like chemical detoxification and active ion transport via membrane proteins, such as ATPases, chemiosmotic cation channels, or proton antiporters. The solubility of these compounds is a key factor in microbial resistance [8,9]. Microbial systems effectively detoxify metal ions by converting soluble toxic ions into insoluble, non-toxic metallic nanoclusters through intracellular bioaccumulation and extracellular mechanisms like biomineralization and biosorption. Extracellular metal nanoparticle synthesis has significant industrial applications, with a focus on achieving monodispersity, as polydispersity is a major challenge. Fungi are highlighted as advantageous microbial candidates for intracellular nanoparticle synthesis due to their consistent sizing and reduced polydispersity.Their networks withstand flow stress, agitation, and other bioreactor conditions better than both plant-based systems and bacterial cells. Fungi are also slow-growing, easier to control, and simpler to cultivate. Additionally, they secrete more reductive proteins that aid extracellular synthesis, facilitating easier downstream processing. Because the nanoparticles precipitate outside the cell, they are free from intracellular contaminants, making them immediately suitable for diverse practical applications [10]. AgNPs, typically 1–100 nm in size, offer a higher surface area and enhanced reactivity compared to bulk silver. Their unique electrical, optical, and catalytic properties have driven research into their use for drug delivery, diagnostics, and imaging [11,12]. Most notably, AgNPs exhibit strong antibacterial activity even against multidrug-resistant pathogens [13,14]. This enhanced efficacy has facilitated the integration of silver nanoparticles (AgNP) in various healthcare and hygiene products, including surgical tools, cosmetics, dental materials, catheters, and dressings [15-18]. Their multiple mechanisms of action make them effective antibiotic alternatives by targeting various microbial structures simultaneously [19].
Silver nanoparticles (AgNPs) offer a promising alternative in the face of rising antibiotic resistance and the slow, costly development of new antibiotics, as they are effective in preventing and treating infections, decontaminating medical equipment, and addressing resistant microorganisms [20,21]. The increasing production of silver nanoparticles (AgNPs), now exceeding 500 tons annually, indicates a growing industrial demand. This surge has prompted greater attention to understanding their biological activity, mechanisms of action, and safety for both human and environmental health [22]. Bassia scoparia L. Voss, previously known as Kochia, is found in temperate and subtropical regions worldwide, though its exact Eurasian origin is unclear. It can be recognized by its annual life cycle, bushy form, flat leaves with petiole-like bases, leafy inflorescences, and distinctive fruiting perianth that may have tubercles or short wings. The species demonstrates considerable morphological variation, particularly in leaf shape, hair tufts at bract bases, and perianth structure [23,24].
Results and Discussion
Ultraviolet Visible Spectroscopy (UV-Vis spectroscopy) Analysis
The UV-Vis absorption spectra of nanoparticles synthesized from Bassia scoparia leaf extract reveal a strong peak at 405 nm and a secondary shoulder at 414 nm. The prominent 405 nm peak indicates the formation of small, stable spherical nanoparticles, while the 414 nm shoulder suggests variations in size or morphology.
Figure 1: UV-Vis Absorption Spectra of Green Synthesized Nanoparticles from Bassia Scoparia L. Leaf Extract
SEM Analysis
The SEM micrograph of synthesized silver nanoparticles shows predominantly spherical morphologies with an average diameter of approximately 30 nm, although some irregularities exist. Larger particles observed are likely due to the aggregation of smaller nanoparticles, highlighting the morphology of biologically synthesized silver nanoparticles. SEM was utilized to analyze the size, shape, and surface morphology of biologically synthesized silver nanoparticles (AgNPs). This electron microscopy technique provides high-resolution images, with magnification from 20X to 30,000X and a resolution of 50 to 100 nm. Earlier studies reported that silver nanoparticles made with Aloe vera extract ranged from 9 to 18 nm in size and typically exhibited hexagonal geometry, while zinc oxide nanoparticles from Glycosmis pentaphylla leaf extract were mostly spherical [28].
Figure 2: SEM Micrograph of Green Synthesized Nanoparticles from Bassia Scoparia Leaf Extract
XRD Analysis
X-ray diffraction (XRD) analysis identified the crystalline phase of green-synthesized silver nanoparticles (AgNPs), revealing six distinct peaks corresponding to various lattice planes at specific 2θ values. These peaks confirmed the face-centered cubic (FCC) structure of silver, as per JCPDS standards. Unassigned peaks were also noted, likely due to bio-organic compound crystallization from Phlomis extract on the nanoparticle surface.
Figure 3: XRD Pattern of Green Synthesized Nanoparticles from Bassia Scoparia Leaf Extract
Using X-ray diffraction, the structure, purity, and phase identity of green-produced AgNPs were ascertained [29]. A previous work using G. ofcinalisw plant extract where XRD peaks in degrees 2θ appear at 38.0946°, 41.4385°, 64.494°, and 77.349° this can be attributed to the planes (111), (200), (220), (311) and (222) sets of lattice planes of crystal [30]. The XRD pattern confirmed the crystalline nature of the synthesized silver nanoparticles, with sharp diffraction peaks indicating that the particles are within the nanometer size range, aligning with standard reference data from the JCPDS [31].
FTIR Analysis
he low-frequency region between 894 and 484 cm-1is associated with metal–oxygen linkages, confirming the interaction of silver with plant metabolites. These findings suggest that biomolecules such as polyphenols, proteins, and amines from Bassia scoparia leaf extract function both as reducing agents and as stabilizers during the nanoparticle synthesis process. The low-frequency region between 894 and 484 cm-1 indicates metal–oxygen linkages, confirming the interaction of silver with plant metabolites. Biomolecules from Bassia scoparia leaf extract, including polyphenols, proteins, and amines, act as reducing agents and stabilizers in nanoparticle synthesis.
Figure 4: FTIR Spectra of Green Synthesized Silver Nanoparticles from Bassia Scoparia Leaf Extract
Antibacterial Assay
We used the Microplate Alamar Blue Assay to assess the antibacterial properties of silver nanoparticles synthesized with Bassia scoparia L. leaf extract against five bacterial strains: Escherichia coli, Bacillus subtilis, Staphylococcus aureus, Pseudomonas aeruginosa, and Salmonella typhi. The results indicated that the AgNPs had the highest inhibition rates of 89% against E. coli and 69% against P. aeruginosa, while the standard drug showed 91% and 78% inhibition, respectively. No antibacterial activity was detected against B. subtilis, S. aureus, or S. typhi. These findings suggest that Bassia scoparia L. mediated AgNPs are selectively effective against specific gram-negative bacteria, notably E. coli and P. aeruginosa, but demonstrate limited or no activity against the tested gram-positive strains. AgNPs synthesized from Acer oblongifolium extract demonstrated strong antibacterial properties, evidenced by inhibition zones of 13–26 mm in disc diffusion assays against multiple strains [32]. Another study demonstrated that AgNPs from Cinnamomum tamala showed significant inhibitory effects against P. aeruginosa. This supports our results, highlighting that smaller AgNPs, capped with plant phytochemicals, have enhanced antibacterial efficacy, especially against gram-negative bacteria.
|
Name of Bacteria |
% Inhibition of Compound |
% Inhibition of Drug |
|
Escherichia coli |
89 |
91 |
|
Bacillus subtilis |
No activity |
89 |
|
Staphylococcus aureus |
No activity |
83 |
|
Pseudomonas aeruginosa |
69 |
78 |
|
Salmonella typhi |
No activity |
84.3 |
Table 1: Antibacterial Activity of Green Synthesized Nanoparticles from Bassia Scoparia Leaf Extract Compared with Standard Drug
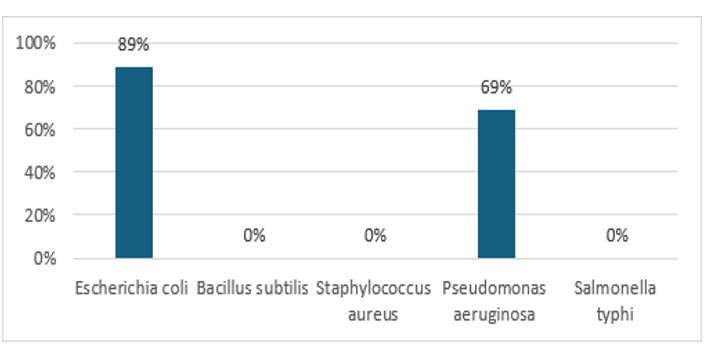
Figure 5: Comparative Antibacterial Activity of Green Synthesized Nanoparticles from Bassia Scoparia L. Leaf Extract and Standard Drug Against Selected Bacterial Strains
Antifungal Assay
An antifungal assay evaluated the effectiveness of biologically synthesized silver nanoparticles (AgNPs) using Bassia scoparia L. leaf extract against seven fungal pathogens: Trichophyton rubrum, Candida albicans, Aspergillus niger, Microsporum canis, Fusarium lini, Candida glabrata, and Aspergillus fumigatus. The inhibitory effect of AgNPs was measured by comparing the linear growth of fungal colonies with untreated controls. The results of this experiment are summarized in Table 2. The synthesized silver nanoparticles demonstrated significant antifungal activity, particularly against Aspergillus fumigatus (92% growth inhibition), Candida glabrata (89%), and Aspergillus niger (83%). Notable inhibition was also observed for Fusarium lini (75%), while moderate effects were noted against Trichophyton rubrum (62%), Microsporum canis (55%), and Candida albicans (50%). The study highlights that silver nanoparticles synthesized from Bassia scoparia L. leaf extract exhibit significant antifungal activity, particularly against Aspergillus fumigatus, Candida glabrata, and Aspergillus niger. Miconazole and amphotericin B served as positive controls, while untreated samples were negative controls, indicating the potential of green-synthesized AgNPs for biomedical and agricultural applications. The study explores the synthesis of silver nanoparticles using Bassia scoparia L. leaf extract for antifungal applications. It references similar research with Azadirachta indica (neem), which showed significant antifungal activity against strains like Aspergillus niger and Candida albicans, demonstrating that plant-derived silver nanoparticles can effectively inhibit pathogenic fungi, with results comparable to standard antifungal drugs [33].
|
Name of Fungus |
Linear Growth |
(mm) |
Inhibition% |
Standard Drug |
|
Sample (mm) |
Control (mm) |
|||
|
Trichophyton rubrum |
38 |
100 |
62% |
Miconazole |
|
Candida albicans |
50 |
100 |
50% |
Miconazole |
|
Aspergillus niger |
17 |
100 |
83% |
Amphotericin-B |
|
Microsporum canis |
45 |
100 |
55% |
Miconazole |
|
Fusarium lini |
25 |
100 |
75% |
Miconazole |
|
Candida glabrata |
11 |
100 |
89% |
Miconazole |
|
Aspergillus fumigatus |
8 |
100 |
92% |
Miconazole |
Table 2: Antifungal Activity of Green-Synthesized Nanoparticles from Bassia Scoparia Leaf Extract Compared with Standard Drug
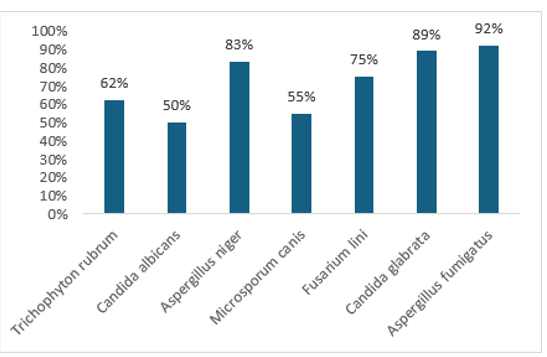
Figure 6: Comparative Antifungal Activity of Green Synthesized Nanoparticles from Bassia Scoparia L. Leaf Extract and Standard Drug Against Selected Bacterial Strains [34-37].
Methods And Materials
Collection of Plant Materials
The study involved the collection of a plant species from the Tazagram region in District Dir Lower, Khyber Pakhtunkhwa, Pakistan. The specimen underwent taxonomic identification and authentication at the Department of Botany, GDC Gulabad, where expert botanists verified its identity using morphological and taxonomic keys. A voucher specimen was prepared and deposited in the herbarium to facilitate future comparative studies and maintain scientific integrity [25].
Preparation of Plant Extract
After identification and authentication, Bassia scoparia L. leaves were washed with distilled water to remove contaminants. They were shaded and air-dried at room temperature for several days to preserve phytochemical constituents, then ground into a fine powder using a mechanical grinder the extraction process involved transferring powdered leaves into a sealed glass container, followed by the addition of distilled water in a specific ratio for aqueous extraction. The mixture was incubated in an orbital shaker at a controlled temperature for 24 to 48 hours to extract bioactive compounds. After incubation, the mixture was filtered through Whatman No. 1 filter paper to remove solid residues, and the clear filtrate was stored at 4°C for silver nanoparticles synthesis [26].
Biosynthesis of AgNPs Mediated by Bassia Scoparia Leaves Extract
The green synthesis of silver nanoparticles (AgNPs) was conducted by mixing Bassia scoparia L. leaf extract with an aqueous silver nitrate (AgNO3) solution. The leaf extract, containing phytochemicals, was added dropwise to the AgNO3 solution while continuously stirring at room temperature. This process allowed for the bioreduction of silver ions (Ag+) to metallic silver (Ag+) over several hours. A visual indicator of nanoparticle formation was the gradual color change of the solution from pale yellow to dark brown, serving as preliminary confirmation. This was further validated through spectroscopic and microscopic characterization techniques [26].
Characterization of the Synthesized AgNPs
UV-Vis Spectroscopy
Optical measurements were conducted with a Lambda 35 UV-Vis spectrophotometer, using distilled water as the reference solvent in quartz cuvettes. A 1 mL sample of the reaction mixture was diluted in 9 mL of water and sonicated for 15 minutes for UV measurements. Additionally, 2 mL of pure Ag NP stock solution was diluted with 8 mL of water for analysis, and a stock solution was prepared by dissolving 5 mg of silver nanoparticles in 5 mL of water and sonicating for one hour.
SEM Analysis
The surface morphology of the synthesized nanoparticles was analyzed using a Hitachi S-4500 scanning electron microscope. The sample underwent centrifugation at 14,000 rpm for 10 minutes, with the nanoparticle pellet resuspended in deionized water and recentrifuged three times, followed by an acetone wash. To achieve a uniform and stable suspension, the purified silver nanoparticles were sonicated for 30 minutes before drying the sample. A small dried sample was placed on a SEM grid to create a thin film, coated with gold via sputter coater, dried under a mercury lamp for 10 minutes, and SEM images were captured at various magnifications.
X-Ray Diffraction Spectroscopy Analysis
Cu Kα radiation (λ = 1.5418 Å) was used to create XRD patterns on an Ultima IV X-ray powder diffractometer (Rigaku, Tokyo, Japan).
Fourier-Transform Infrared Spectroscopy (FTIR)
A PerkinElmer 1000 FT-IR spectrometer was employed to acquire FT-IR spectra. The Ag NPs were purified with distilled water to remove free biomass and unbound extract. The final product was centrifuged for 30 minutes at 9000 rpm and dried. For measurement, the cleaned Ag NPs were mixed with KBr powder and pressed into a pellet, using a reference blank KBr pellet to adjust the background.
Antibacterial Assay
The antibacterial activity of the silver nanoparticle-synthesized extract was evaluated using the Microplate Alamar Blue Assay against various antibacterial strains. Sterilized Petri dishes with nutrient agar were prepared, and a standardized 24-hour bacterial culture was spread on the agar. Wells were created, and 50 µL of the extracts was introduced into each. After incubation at 37 °C for 24 hours, antibacterial activity was assessed by measuring the inhibition zones around the wells.
Antifungal Assay
The agar tube dilution method was utilized to assess the antifungal activity of silver nanoparticles (AgNPs) derived from the leaf extracts of Bassia scoparia L. Six fungal strains were tested: Trichophyton rubrum, Candida albicans, Aspergillus niger, Microsporum canis, Fusarium lini, and Candida glabrata. A mixture of 25 ml distilled water and 25 mg of the AgNPs pellet was prepared to achieve a final concentration of 1000 ppm. Fungal growth medium was prepared and sterilized by autoclaving at 121 °C for 20 minutes. Under aseptic conditions in a laminar flow hood, 4 mL of autoclaved Sabouraud Dextrose Agar was dispensed into test tubes, which were tilted to form slants. Fungal cultures were inoculated on the slants, with miconazole as a positive control and dimethyl sulfoxide (DMSO) as a negative control. The tubes were incubated at 27 °C for 7 days to evaluate antifungal activity using a percentage inhibition formula.
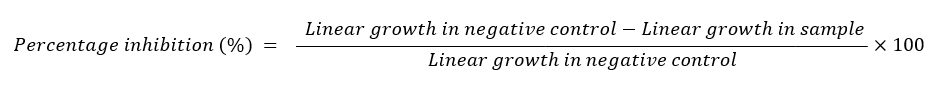
A negative control reading served as a reference for evaluating fungal growth, with percentage inhibition calculated using a specified formula [27].
References
- Spirescu, V. A., Chircov, C., Grumezescu, A. M., Vasile, B. È?., & Andronescu, E. (2021). Inorganic nanoparticles and composite films for antimicrobial therapies. International journal of molecular sciences, 22(9), 4595.
- Zhao, L., Lu, L., Wang, A., Zhang, H., Huang, M., Wu, H.,... & Ji, R. (2020). Nano-biotechnology in agriculture: use of nanomaterials to promote plant growth and stress tolerance. Journal of agricultural and food chemistry, 68(7), 1935-1947.
- Adeosun, S. O., Ilomuanya, M. O., Gbenebor, O. P., & Dada,M. O. (2020). Biomaterials for Drug Delivery: Sources,Classification. Advanced functional materials, 141.
- Babayevska, N., Przysiecka, Å., Iatsunskyi, I., Nowaczyk, G., Jarek, M., Janiszewska, E., & Jurga, S. (2022). ZnO size and shape effect on antibacterial activity and cytotoxicity profile. Scientific Reports, 12(1), 8148.
- Al-esnawy, A. A., Ereiba, K. T., Bakr, A. M., & Abdraboh,S. (2021). Characterization and antibacterial activity of Streptomycin Sulfate loaded Bioglass/Chitosan beads for bone tissue engineering. Journal of Molecular Structure,1227, 129715.
- Selim, Y. A., Azb, M. A., Ragab, I., & HM Abd El-Azim, M. (2020). Green synthesis of zinc oxide nanoparticles using aqueous extract of Deverra tortuosa and their cytotoxic activities. Scientific reports, 10(1), 3445
- Sarhadi, H., Shahdadi, F., Salehi Sardoei, A., Hatami, M., & Ghorbanpour, M. (2024). Investigation of physio-mechanical, antioxidant and antimicrobial properties of starch–zinc oxide nanoparticles active films reinforced with Ferula gummosa Boiss essential oil. Scientific reports, 14(1), 5789
- Dickinson, A. W., Power, A., Hansen, M. G., Brandt, K. K., Piliposian, G., Appleby, P., ... & Vos, M. (2019). Heavy metal pollution and co-selection for antibiotic resistance: A microbial palaeontology approach. Environment international, 132, 105117
- Gauba, A., Hari, S. K., Ramamoorthy, V., Vellasamy, S., Govindan, G., & Arasu, M. V. (2023). The versatility of green synthesized zinc oxide nanoparticles in sustainable agriculture: A review on metal-microbe interaction that rewards agriculture. Physiological and Molecular PlantPathology, 125, 102023.
- Kumar, D., Singla, R. K., Sharma, P., Kumar, L., Kaur, N., Dhawan, R. K., ... & Sharma, R. (2023). Phytochemistry and polypharmacological potential of Colebrookea oppositifolia smith. Current Topics in Medicinal Chemistry, 23(5), 334- 348.
- Silva, L. P., Silveira, A. P., Bonatto, C. C., Reis, I. G., & Milreu,P. V. (2017). Silver nanoparticles as antimicrobial agents: Past, present, and future. In Nanostructures for antimicrobial therapy (pp. 577-596). Elsevier.
- Yaqoob, A. A., Ahmad, H., Parveen, T., Ahmad, A., Oves, M., Ismail, I. M., ... & Mohamad Ibrahim, M. N. (2020). Recent advances in metal decorated nanomaterials and their various biological applications: a review. Frontiers in chemistry, 8, 341.
- Siddiqi, K. S., Husen, A., & Rao, R. A. (2018). A review on biosynthesis of silver nanoparticles and their biocidal properties. Journal of nanobiotechnology, 16(1), 14.
- Marambio-Jones, C., & Hoek, E. M. (2010). A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. Journal of nanoparticle research, 12(5), 1531-1551.
- Kulkarni, S. K., & Kulkarni, S. K. (2015). Nanotechnology: principles and practices. Springer.
- Tran, Q. H., & Le, A. T. (2013). Silver nanoparticles: synthesis, properties, toxicology, applications and perspectives. Advances in natural sciences: nanoscience and nanotechnology, 4(3), 033001.
- Argueta Figueroa, L., Arenas-Arrocena, M. C., Díaz-Herrera,., García-Benítez, S. V., & García-Contreras, R. (2019). Propiedades antimicrobianas y citotóxicas de un adhesivo de uso ortodóncico adicionado con nanopartículas de plata. Mundo nano. Revista interdisciplinaria en nanociencias y nanotecnología, 12(22), 0-0.
- Ge, L., Li, Q., Wang, M., Ouyang, J., Li, X., & Xing, M.M. (2014). Nanosilver particles in medical applications: synthesis, performance, and toxicity. International journal of nanomedicine, 2399-2407.
- Cheng, G., Dai, M., Ahmed, S., Hao, H., Wang, X., & Yuan, Z. (2016). Antimicrobial drugs in fighting against antimicrobial resistance. Frontiers in Microbiology, 7, 470.
- Natan, M., & Banin, E. (2017). From Nano to Micro: using nanotechnology to combat microorganisms and their multidrug resistance. FEMS microbiology reviews, 41(3), 302-322.
- Lee, N. Y., Ko, W. C., & Hsueh, P. R. (2019). Nanoparticles in the treatment of infections caused by multidrug-resistant organisms. Frontiers in pharmacology, 10, 1153.
- Ferdous, Z., & Nemmar, A. (2020). Health impact of silver nanoparticles: a review of the biodistribution and toxicity following various routes of exposure. International journal of molecular sciences, 21(7), 2375.
- Aellen, P. (1954). Ergebnisse einer botanisch-zoologischen Sammelreise durch Iran: Botanisch Ergebnisse IV:Chenopodiaceae (1): Kochia. BAUHINIA–Zeitschrift derBasler Botanischen Gesellschaft, 1(2), 4-16.
- Benson, K. M. (1955). Phenotypic Variations of Kochia Scoparia.
- Shuaib, M., Ahmed, S., Ali, K., Ilyas, M., Hussain, F., Urooj, Z., ... & Hussain, F. (2019). Ethnobotanical and ecological assessment of plant resources at district Dir, Tehsil Timergara, Khyber Pakhtunkhwa, Pakistan. Acta Ecologica Sinica, 39(1), 109-115.
- Logeswari, P., Silambarasan, S., & Abraham, J. (2013). Ecofriendly synthesis of silver nanoparticles from commercially available plant powders and their antibacterial properties. Scientia Iranica, 20(3), 1049-1054
- Sharmila, G., Thirumarimurugan, M., & Muthukumaran, C. (2019). Green synthesis of ZnO nanoparticles using Tecoma castanifolia leaf extract: characterization and evaluation of its antioxidant, bactericidal and anticancer activities. Microchemical Journal, 145, 578-587.
- Mohan, A. C., & Renjanadevi, B. J. P. T. (2016). Preparation of zinc oxide nanoparticles and its characterization using scanning electron microscopy (SEM) and X-ray diffraction (XRD). Procedia Technology, 24, 761-766.
- Naveed, M., Bukhari, B., Aziz, T., Zaib, S., Mansoor,M. A., Khan, A. A., ... & Alhomrani, M. (2022). Green synthesis of silver nanoparticles using the plant extract of Acer oblongifolium and study of its antibacterial and antiproliferative activity via mathematical approaches. Molecules, 27(13), 4226.
- Gonfa, Y., Tadesse, G., Kumar, S., Terfasa, T. T., & Tesfaye,M. Green Synthesis of Silver Nanoparticles Using Vernonia Amygdalina Plant Extract: Effect of Different Parameters on Synthesis and its Antimicrobial Activities. Available at SSRN 4415265.
- Mustapha, T., Ithnin, N. R., Othman, H., Abu Hasan, Z. I., & Misni, N. (2023). Bio-fabrication of silver nanoparticles using Citrus aurantifolia fruit peel extract (CAFPE) and the role of plant extract in the synthesis. Plants, 12(8), 1648.
- Ipe, D. S., Kumar, P. S., Love, R. M., & Hamlet, S. M. (2020).Silver nanoparticles at biocompatible dosage synergistically increases bacterial susceptibility to antibiotics. Frontiers in microbiology, 11, 1074.
- Ahmed, M., Marrez, D. A., Mohamed Abdelmoeen, N., Abdelmoneem Mahmoud, E., Ali, M. A. S., Decsi, K., & Tóth,Z. (2023). Studying the antioxidant and the antimicrobial activities of leaf successive extracts compared to the green- chemically synthesized silver nanoparticles and the crude aqueous extract froAzadirachta indica. Processes, 11(6), 1644
- Betts, J. W., Hornsey, M., & La Ragione, R. M. (2018). Novel antibacterials: alternatives to traditional antibiotics. In Advances in microbial physiology (Vol. 73, pp. 123-169). Academic Pres
- Yaqoob, A. A., Umar, K., & Ibrahim, M. N. M. (2020). Silvernanoparticles: various methods of synthesis, size affecting factors and their potential applications–a review. Applied Nanoscience, 10(5), 1369-1378.
- Sukhorukov, A. P. (2014). The carpology of the Chenopodiaceae with reference to the phylogeny, systematics and diagnostics of its representatives. Grif & Co., Tula, 400.
- Aminullah, F., Malek, N. A. N. N., & Jemon, K. (2021, May). Antibacterial activity of silver nanoparticles synthesized from Persicaria odorata (L.) sojak leaves extract. In AIP Conference Proceedings (Vol. 2353, No. 1, p. 030022). AIP Publishing LLC.