Research Article - (2026) Volume 2, Issue 1
Green Synthesis Characterization and Biological Assessment of Nickle based Microparticles using Justicia Adathoda L. Leaf Extract
2Lecturer of Botany Govt Degree College Gulabad, Kazakhstan
3Lecturer of Chemistry, Govt Degree College Lal Qilla Maidan, Kazakhstan
4Department of Chemistry, Govt Degree College Gulabad, Kazakhstan
Received Date: Nov 18, 2025 / Accepted Date: Dec 22, 2025 / Published Date: Jan 20, 2026
Copyright: ©2026 Aftab Alam, Noor Ul Islam, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ahmad, W., Alam, A., Islam, N. U., Wahab, A., Khan, M. I, et al. (2026). Green Synthesis Characterization and Biological Assessment of Nickle based Microparticles using Justicia Adathoda L. Leaf Extract. Arch Nucl Energy Sci Technol, 2(1), 01-09.
Abstract
Nickel-based oxide/hydroxide microparticles (NiO/Ni(OH)2 MPs) were synthesized via a green way utilizing Justicia adathoda L. leaf extract, serving as novel reducing and stabilizing agent. Plants phytochemicals such as alkaloids, flavonoids, and phenolics facilitated the eco-friendly formations of NiO/Ni(OH)2 MPs without toxic chemicals. Structure analysis through X-ray diffraction (XRD) confirmed the crystalline structure, while FTIR confirmed the involvement of functional groups in reduction and stabilizations. Morphological characterization by Scanning Electron Microscopy (SEM) revealed a granular dense structure microparticle of size about 1μm. Biological screening showed notable antibacterial and antifungal activities. NiO/Ni(OH)2 MPs show 60.24% inhibition against Staphylococcus aureus and 70.24% inhibition against Pseudomonas aeruginosa, while antifungal activity reached 95% against Aspergillus fumigatus. Compared to standard pharmaceutical agents such as Ofloxacin and Miconazole, the NiO/Ni(OH)2 MPs synthesized using Justicia adathoda L. leaf extract revealed potential antibacterial and antifungal activities. These results underline the value of Justicia adathoda as an eco-friendly and efficient biological resource for making functional NiO/Ni(OH)2 MPs. The synthesized microparticles show strong applications in biomedical fields, including therapeutic formulations, antimicrobial coatings, and presenting a sustainable unconventional to chemically synthesized micromaterials.
Keywords
Green Synthesis, Ni Based Microparticle, SEM, FTIR, XRD, Antimicrobial Activity.
Introduction
Green synthesis has become a major topic of interest in the field of Micro-nanotechnology [1]. Developments in micro-nanotechnology have led to the production of innovative materials, particularly ecofriendly micro-based-nanostructure synthetic procedures used to synthesize metal and metal oxide micromaterials of any shape and size [2]. Most physical, chemical, hybrid methods tend to be costly and detrimental due to the presence of harmful substances like solvents, reducing agents, and precursors [3]. In contrast, employing green techniques for the preparation of microparticles (MPs) is new economical, ecofriendly, and simpler to utensils. Consequently, such technique is increasingly utilized in the production of metallic based microparticles. A diverse span of natural deposit, inclusive of microorganisms, biomolecules, and plant extracts, was explored for green MP synthesis [4]. Although numerous studies have been conducted on the green synthesis of MPs utilizing biomolecules and microbes, plant extracts are often favored because they are cost-effective, efficient, and do not require the complex processes involved in isolating and maintaining microbial cultures [5]. Various plant species, their parts, and isolated compounds are effectively used to synthesize green MPs. Furthermore, the preparation of micromaterials can be made more efficiently by incorporating industrial and agricultural waste, in computation to being ecofriendly [6].
Phenolic chemicals can operate as capping agents in microparticles manufacturing, whereas biological molecules offer unique uses in micro-nanotechnology, making metal microparticles durable and trustworthy [7]. Recent studies have proved the effectiveness of using micromaterial’s in different aspects of life, including environmental control, energy storage, solar cell, biomarking, bio probes, tissue engineering, cancer therapy, cancer diagnosis, and drug delivery [8]. The method of green synthesis consists in obtaining particles using safe materials as reducing agents and stabilizers, for example, products of biological origin. For example, the method can be carried out using extracts of plants or their individual parts, as well as microorganism (bacteria, yeast, fungi) [9]. A plant contains various biochemicals and metabolites that can be used for the green synthesis of microparticles as they possess reducing and stabilizing properties. Green synthesis technologies are environmentally friendly, cost effective stable and nontoxic than traditional biological, physical, and chemical approaches [10]. Biosynthesized metal and metal oxide MPs have rising applications in the medical sector including immunotherapy, dentistry, wound healing, diagnosis, regenerative medicines, bio toxicology and bio-sensing platform and its antiviral, fungicidal and bactericidal potentials [11]. Microparticles can be variable in dimension, shape, size and architectures [12]. In the environmental field, the green synthesis of MPs has been used to control environmental pollution, such as with the degradation of organic dyes and chlorinated organic pollutants, heavy metal removal, in addition to the treatment of wastewater [13]. Justicia adathoda L. also known as Malabar nut is a well-known and important medicinally important plant of family Acanthaceae growing in different Asian countries including Pakistan, Sirilanka, India and Nepal [14]. Justicia adathoda L. has diverse applications in traditionally used Ayurvedic and Unani medicines. The local populace uses the plants' leaves to treat coughs, colds, and asthma. Justicia adathoda L. plant has several key properties, including antibacterial, antioxidant, antiulcer, antidiabetic, anti-allergic, anti-genotoxic, and anti-phlogiston. Adathoda vasica leaves contain phytochemicals including flavonoids, saponins, tannins, alkaloids and phenolic compounds. Justicia adathoda L. leaf extract contains the two most significant quinazoline alkaloids, vasicine and vasicione [15]. Phytochemicals are bioactive compounds naturally found in plants, and their composition varies greatly among different plant species and even within different parts of the same plant. The most common classes of phytochemicals include polyphenols (like flavonoids and tannins), terpenoids, alkaloids, carotenoids, and saponins. These compounds are involved in plant defense mechanisms and can also offer various health benefits to humans. Antibacterial agents are utilized to inhibit the proliferation of harmful bacteria. They help prevent illness and eradicate microorganisms [16]. Numerous antibiotics are sourced from plants and various chemicals to combat diseases. Bacteria are the pathogens responsible for poisoning. For this assay, we used five different strains of pathogens [17]. A disc diffusion method was employed to assess bacterial activity. Bacteria are classified into two categories: Gram-negative and Gram-positive. Staphylococcus aureus (S. aureus) is typically recognized as a Gram-positive bacterium and is frequently used in antibacterial studies due to its significant potential to cause infections in humans. Escherichia coli (E. coli), a Gram-negative bacterium, is established in contaminated water and along with as a pathogenic microbe that leads to human illness, as well as serving as an indicator of water quality. The antifungal test aims to eliminate microorganisms, especially fungi. Fungi pose significant risks and can lead to various illnesses. We utilize antifungal tests to identify infections caused by fungi. These organisms can damage fruits, vegetables, and plants [18]. Numerous plants are recognized for their antifungal properties. Unsanitary food and environments weaken the immune system, facilitating the spread of fungal infections. Antifungal medications are necessary to treat such infections [19]. Metallic nickel based microparticles are those that are used as the primary raw material without combining them with other metals, compounds or elements. The term nickel microparticle used in this review also refers to metallic nickel microparticle [20]. The successful production of Ni based MPs employing various plant extracts, as well as their enhanced biological potential, call for additional research into other medicinally essential plants for future enhancement [21]. Ni based MPs have gained much attention due to their unique magnetic, chemical, and physical properties as well as their potential applications in various technological fields such as catalysis, battery manufacture, incorporation in textile, and optical switches [22-25]. The current study aimed to synthesis Ni based MPs using Justicia adathoda L. leaf extract. The produced (NiO/Ni(OH))2 MPs) were examined by using modern analytical techniques, including X-ray diffraction (XRD), Fourier Transform Infrared Spectroscopy (FTIR), Scanning Electron Microscopy (SEM). Within inclusion, the antibacterial and antifungal potentials of the prepared Nickel based microparticles were measured.
Methodology
Chemicals and other Materials
The chemicals used were Distilled water and Nickel chloride. These chemicals were of analytical grade. Compounds with great clarity were purchased from Sigma-Aldrich and used without any further purification. Double distilled water was used throughout all the involved experiments as a solvent. The raw materials included NiCl, Ni (NO3)2-6H2O (98%, Sigma-Aldrich), salvia plant extract that was collected from the natural habitats and methylene blue (MB; 98%, Sigma-Aldrich) dye.
Synthesis of the Plant Extract
The leaves of Justicia adathoda L. were harvested in Khyber Pakhtunkhwa's Tehsil Timergara District Dir Lower. Following collection, the plant was identified and authenticated by the Flora of Pakistan. The collected plant leaves have been clean with filter water, shadow dried and grind within fine talc by utilizing pistil and motor. Approximately ten grams of powder were taken and 200 mL of distilled were added in a glass container and placed for 48 hours. The sample was then filtered through Whatman filter paper and leaf extract was obtained in a sterilized conical flask [26].
Green Synthesis of Ni based MPs
Ni based microparticles (NiMPs) were synthesized following the standard plant-mediated green reduction method [27]. The extract from Justicia adathoda L. was utilized as a bio reductant in the production of nickel based microparticles. To form a NiCl solution (2M Nickel chloride solution), 0.95g of nickel chloride was dissolved in 150 ml of distilled water. Initially, the Nickel chloride solution was stirred at room temperature for one minute before incorporating the Justicia adathoda L. leaf extract. The mixture was then agitated for one hour. Following the addition of the leaf extract, the solution transitioned from colorless to a brownish-black hue, indicating the formation of Ni based MPs. Subsequently, the samples underwent centrifugation for 40 min at 14,000 rpm at 60°C .The resulting remnant from the centrifugation be desiccated at standard temperature 60°C prior to characterization and biological assessment.
Characterization of the Synthesized Ni based MPs
Characterization of microparticles stands as the most important factor in comprehending and controlling the synthesis and application of microparticles. This vital factor can be achieved by utilizing a variety of procedures including X-ray diffraction (XRD, D8-Advance Bruker) [28]. NiO/Ni(OH)2 MPs was characterized by using X-ray powder diffraction using X-ray diffractometer [Model: Pert Pro (PAN Analytical). The XRD spectrum was taken over 2θ range of 5°-80°. Fourier transform infrared (FT-IR) analysis, using IR Prestige-21 Shimadzu 400-4000 cm-1. Fourier transform infrared (FT-IR) analysis, using IR Prestige-21 Shimadzu, was implemented to characterize the functional groups on the surface of the Ni MP. The scanned spectra were in the range 400 - 4000cm-1 at a resolution of 4cm-1 [29]. UV-Vis analysis was performed using perkin Elmer UV Win lab 7.3.0.340 /220126 to study the optical properties as well as band gap of microparticles [30], field electron scanning electron microscopy (FESEM)/ energy-dispersive X-ray spectroscopy (EDX, Tescan Brno-Mira3 Lmu), The purity of Ni MPs was confirmed by the means of EDX, which is capable of accurately measuring Ni along with displaying the Ni peaks without any impurities [31]. Elemental analysis was carried out by using EDX analysis to analyze the % amount of carbon, nickel and oxygen [32]. SEM was carried out to analyze the surface morphology of Ni based microparticles. The morphology of the prepared sample of Ni-MP was examined using a Scanning Electron Microscope SEM analysis (JEOL jsm-6480 LV) [33].
Antimicrobial Assay
The antimicrobial activity of the studied MPs was evaluated against 5 bacterial strains (Escherichia coli ATCC 25922, B. subtilis, Staphylococcus aureus, P. auriginosa, and Salmonella Typhi), belonging to Gram-positive and Gram-negative bacteria, and 7 yeast strains, Trichophyton rubrum, Candida albicans, Aspergillus niger, Microsporum canis, Fusarium lini, Candida glabirata, Aspergillus fumigatus. Were isolated from leaves extract of plant products [34]. Before use, bacterial strains were revivified with subcultures in tryptone soya agar (TSA, Biokar, Beauvais, France) at 37°C for 24 h, while yeast was revivified with subcultures in Sabouraud dextrose agar with chloramphenicol (Biokar, Beauvais, France) at 25°C for 48 h. Then, a microbial suspension equivalent to 0.5 McFarland (108 cfu/mL) was prepared in sterile physiological water (0.9% NiCL) and inoculated by swabbing on plates containing Mueller–Hinton agar (Biokar, Beauvais, France) [34]. On the surface of each plate, 10 μL of each MP solution at a concentration of 10 μg/mL was dropped on 6 mm diameter filter paper discs (Whatman Grade 4 Qualitative Filter Papers, Merck, Germany. Sterile distilled water (10 μL) was used as a negative control, while gentamicin (30 μg) was used as a positive control for bacteria and 10 μL of Canaflucan (fluconazole) Win® capsule 150 mg with a concentration of 0.6 μg/mL was used as a positive control for the yeast strain. The used plates were incubated at 37 â?? for 18–24 h for bacteria and 25°C for 44–48°C for yeast. After incubation, the inhibition diameter was measured in millimeters (disk included). The antimicrobial activity was classified into three levels based on the produced inhibitory diameter: weak (inhibition zone ≤ 12.0 mm), intermediate/moderate (12.1 mm ≤ inhibition zone ≤ 20.0 mm), and strong (inhibition zone ≥ 20.1 mm).
Statistical Analysis
Origin software and Microsoft Excel were used for the generation of figures and statistical analysis. In this study, the experiments were carried out in triplicate, and the difference between groups was determined by using the student t-test (P < 0.05).
Results and Discussion
NiO/Ni(OH)2 MPs Preparation and its Corroboration by SEM Spectroscopy
The SEM image of the resulting microparticles, as shown in the figure 1, displayed a crystalline, and slightly uneven shape, with a diameter 1 µm. Scanning electron microscopy became employed to scrutinize the physical dimensions with morphological characteristics of the biosynthesized Ni based microparticles [35]. The image shows that the microparticles have a highly crystalline as well as highly agglomerate and appear as cluster of microparticles with a size range of 1μm. The agglomeration of microparticles may be due to the exposure of microparticles to large volume of heat during calcination step or because of high surface tension of the ultrafine microparticles and high surface energy [36]. The SEM result is compatible with the result of who study the morphology of Ni based microparticles which are prepared by the sol-gel method using two different precursors nickel chloride hexahydrate and nickel nitrate hexahydrate. Our results are accurately similar to other studies in literature where it was revealed that Ni based MPs have important microparticles in other previous SEM analysis) [37].
Figure 1: Image of Synthesized NiO/Ni(OH)2 Microparticles
XRD Analysis
The XRD pattern was employed to characterize the structural features, crystalline information, and phase purity of the precursor NiCl.6H2O and the synthesized Ni based MPs. The results match well with the standard cards of pure NiCl2.6H2O JCPDS card (No. 025-10440) [38]. To determine the known phase of the Ni based MPs, an X-ray diffraction analysis was conducted. The XRD pattern of green produced Ni based microparticles has been displaced. The spectra revealed five distinct independent peaks at 2Ø = 10°, 24°, 27°, 29° and 32° corresponding to intensity planes (1900), (1200), (1200), (1700) and (1000) respectively. The highest intensity for the peak observed was at 2θ value 29°. The diffraction peak data calm coordinate with the FCC fabric report from the Joint Committee on Powder Diffraction Standards (JCPDS). Sprinkling small unexplained peaks were also seen, which could be due to bioorganic piece from the Justicia adathoda L. leaf extract crystallizing on the nickel surface [39].
Figure 2: Analysis of NiO/Ni(OH)2 Microparticles using XRD Spectroscopy
FTIR Analysis
FT-IR spectroscopy was used to study the functional groups present on the raw materials, NiCl, NiNO3 and the product Ni based MPs.
The FT-IR of the raw materials and products are summarized in figure [31]. FTIR spectroscopy in the spectral range of 400 cm−1 to 4000 cm−1 was performed to know about the possible involvement of different functional groups of plant extract in the reduction process of the Nickel based microparticles. The FTIR profile of NiMPs displayed vibration at 3309.75 cm 1 for-OH groups, at 2943.78 and 2831.77 cm 1 indicating the C-H stretching, while other multiple peaks at 1559.94, 1559.94, 1449.04,1415.19,1114.62 and 1021.53 cm−1 were related to C=C and C-O stretching for aromatic ring and polyphenols. The peak at 616.36 cm−1 displayed information about NiO in NiNO3. The presence of Ni¯ (OH)2 and NiO in the FT-IR spectra indirectly indicates the synthesis of Ni based MPs. Our results are meticulously similar to other studies in literature where it was revealed that Ni based MPs have important microparticles and related with other previous FTIR analysis [40]. The FTIR spectra displays functional groups exist in diverse locations. Figure shows displaced FTIR spectra of Ni based MPs and control leaf extract. Justicia adathoda L. leaf extract exhibits similar transmittance peaks at 3309 and 616.36 cm-1. The extensive and secure bands at 3309 cm-1 were caused by the leaf extract's bounded hydroxyl (-OH) or amine group (-NH). The C-H group can be examined by the peak screen at 2943cm-1. The tensile vibration of the C_O, C=C group caused the peaks at 1559.94 cm-1, 1449.04 cm-1, 1415.19 cm-1,1114.62 cm-1,1021.53 cm-1, and Ni based peak at 616.36 cm-1 [41].
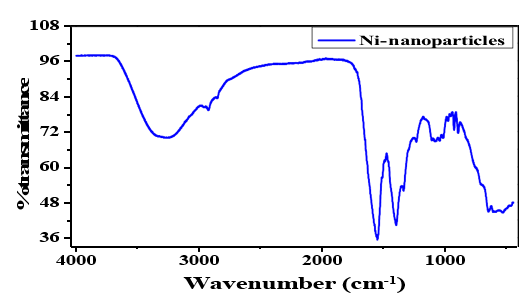
Figure 3: Analysis of NiO/Ni(OH)2 Microparticles
Antibacterial Activities of Justicia Adathoda L. Leaf Extract and NiO/(OH)2 MPs
We used the disc diffusion method to examine the antibacterial activity of Ni based microparticles biosynthesized from Justicia adathoda L. leaf extract. Six exertions in all were used to examine the biological activity. Three are gram negative (Escherichia. Coli), (Salmonella. Typhi) and (P. Aeruginosa) and three are gram positive (S. Aureus), (M. Letus) and (B. Subtilis). The finding was described in table. The consequences opposed five exertions were conflict with those prevail utilize streptomycin as the positive control. A region of hindering was seen after a day. Vernier callipers were used to measure the inhibition zone [42]. The concentration of Ni based MPs increased in tandem with the inhibition of bacterial growth. The sample's inhibition of gram-negative bacteria is 90.2%. 84.25% inhibition against gram-positive S. aureus bacteria and E. coli [42]. A previous study found that biosynthetic Ni based MPs derived from solanum nigrum leaf extract showed a correlation between the particle's size and antibacterial activity, significantly more antibacterial action found in smaller particles. The precise mechanism by which Ni based MPs inhibit bacterial growth is unknown, based on studies in the literature [43]. Sondi and Sondi, though, confirmed that the antibacterial activity of Ni based MPs against gram-negative bacteria depended on the Ni based MPs concentration and was directly associated to the establishment of pits in bacterial cell walls. Our results are meticulously similar to other studies in literature where it was revealed that Ni based MPs have important antibacterial activities against the verified bacterial strains [44].
|
S.no |
Bacterial species |
Percentage inhibition of sample |
Percentage inhibition of drug) ofloxacin) |
|
1 |
E. Coli |
55.3% |
90.2% |
|
2 |
B. subtilis |
No inhibition |
85.21% |
|
3 |
S. aureus |
60.24% |
84.25% |
|
4 |
P. auriginosa |
70.24% |
92.61% |
|
5 |
S. typhi |
No inhibition |
88.31% |
Table 1: Percent Inhibition of NiO/(OH)2 Microparticles
Figure 4: Anti-Bacterial Activity Of Nio/(Oh)2 Microoparticle
Antifungal Activity
Justicia adathoda L. leaf extract was utilize in antifungal research to assess the antifungal efficiency of biologically formed Ni based MPs against numerous fungus strains. The experiment was run, Table comprises the outcomes. Seven fungi such as Candia albicans, Microsprum canis, Trichophyton rubrum, Fusarium lini. Aspergillus fumigatus and Candida glabirata were used. In this experiment, diverse fungal strains were exposed to the antifungal possessions of green produced Ni based microparticles from Justicia adathoda L. leaf extract. The results showed hopeful antifungal activities, Aspergillus was strongly inhibited by Ni based MPs, which resulted in 95%. This indicates that Ni based MPs have a great deal of potential for managing this pathogenic fungus. 85% inhibition was shown by Trichophyton rubrum and 82% Candida albicans, while the remaining fungal species exabits i-e Candida glabitus, Aspergillus niger, Fusarium lini 75% inhibition [45]. Overall, the result demonstrates the antifungal properties of Ni based microparticles mediated from Justicia adathoda L. leaf extract and exhibit significant potential in fighting Candida albicans, Trichophyton rubrum, Aspergillus Niger, Fusarium lini, Microsporum canis, Aspergillus fumigatus, and Candida Glabirata Important repressive effects were also seen when associated to the other three strains. Micanazole and Amphotericin-B were utilized as encouraging control and distilled water was used as an adverse control [38]. A previous examination detected the antifungal features of Ni based microparticles made with extract from Camellia sinensis, or green tea [22].
|
S.no |
Fungal species |
Linear growth in sample in mm |
Percentage Inhibition |
Drug |
|
|
Sample |
Control |
||||
|
1. |
Trichophyton rubrum |
15mm |
100mm |
85% |
Miconazole |
|
2. |
Candida albicans |
18mm |
100mm |
82% |
Miconazole |
|
3. |
Aspergillus niger |
20mm |
100mm |
80% |
Amphotericin-B |
|
4. |
Microsporum canis |
25mm |
100mm |
75% |
Miconazole |
|
5. |
Fusarium lini |
40mm |
100mm |
60% |
Miconazole |
|
6. |
Candida glabirata |
35mm |
100mm |
65% |
Miconazole |
|
7. |
Aspergillus fumigatus |
5mm |
100mm |
95% |
Miconazole |
Table 2: Percent Inhibition of NiO/(OH)2 MPs
Figure 5: Antifungal Activity of NiO/Ni(OH)2 MPs
Conclusion
Recent study has focused on the green production of MPs utilize plant extracts. Plant metabolites promote the environmentally benign production of microparticles. Plant-produced MPs have been used in numerous medicinal and industrial submissions. In the recent study, Ni based MPs were formed biologically with Justicia adathoda L. extract as a caping agent, which is a maintainable and biologically friendly method that is a cheap alternate to other traditional procedures. The Ni based MPs and leaf extract derived from Justicia adathoda L. were also tested for antibacterial and antifungal activities. The aqueous extract of Justicia Aadathoda L. was shown to be less effective than Ni based MPs in terms of antibacterial and antifungal properties. The supplied plant extracts and Ni based MPs remained tested for MIC and MBC. Both the Ni based MPs, and the extract revealed probable antibacterial and antifungal properties when tested against the DPPH and ABTS radicals. The current study concluded that Justicia adathoda L.-based NiO/Ni(OH)2 MPs should be used as alternate treatments due to their antibacterial and antifungal capabilities. Though, additional vivo testing determination is required to expand their applications to biotic systems [46-50].
References
- Shebl, A., Hassan, A. A., Salama, D. M., Abd El-Aziz,M. E., & Abd Elwahed, M. S. (2019). Green synthesis of nanofertilizers and their application as a foliar for Cucurbita pepo L. Journal of Nanomaterials, 2019(1), 3476347.
- Fuku, X., Modibedi, M., & Mathe, M. (2020). Green synthesis of Cu/Cu2O/CuO nanostructures and the analysis of their electrochemical properties. SN Applied Sciences, 2(5), 902.
- Shaik, M. R., Albalawi, G. H., Khan, S. T., Khan, M., Adil,S. F., Kuniyil, M., ... & Khan, M. (2016). “Miswak” based green synthesis of silver nanoparticles: evaluation and comparison of their microbicidal activities with the chemical synthesis. Molecules, 21(11), 1478.
- Khan, F. A., Zahoor, M., Jalal, A., & Rahman, A. U. (2016). Green synthesis of silver nanoparticles by using Ziziphus nummularia leaves aqueous extract and their biological activities. Journal of Nanomaterials, 2016(1), 8026843.
- Khan, M., Karuppiah, P., Alkhathlan, H. Z., Kuniyil, M., Khan, M., Adil, S. F., & Shaik, M. R. (2022). Green synthesis of silver nanoparticles using Juniperus procera extract: their characterization, and biological activity. Crystals, 12(3), 420.
- Iftikhar, M., Zahoor, M., Naz, S., Nazir, N., Batiha, G. E. S., Ullah, R., ... & Mahmood, H. M. (2020). Green synthesis of silver nanoparticles using Grewia optiva leaf aqueous extract and isolated compounds as reducing agent and their biological activities. Journal of Nanomaterials, 2020(1), 8949674.
- Bar, H., Bhui, D. K., Sahoo, G. P., Sarkar, P., De, S. P., & Misra,(2009). Green synthesis of silver nanoparticles using latex of Jatropha curcas. Colloids and surfaces A: Physicochemical and engineering aspects, 339(1-3), 134-139.
- Mandal, G., & Ganguly, T. (2011). Applications of nanomaterials in the different fields of photosciences. Indian Journal of physics, 85(8), 1229-1245.
- Singh, J., Dutta, T., Kim, K. H., Rawat, M., Samddar, P., & Kumar, P. (2018). ‘Green’synthesis of metals and their oxide nanoparticles: applications for environmental remediation. Journal of nanobiotechnology, 16(1), 84.
- Mustapha, T., Misni, N., Ithnin, N. R., Daskum, A. M., & Unyah, N. Z. (2022). A review on plants and microorganisms mediated synthesis of silver nanoparticles, role of plants metabolites and applications. International Journal of Environmental Research and Public Health, 19(2), 674.
- Pandit, C., Roy, A., Ghotekar, S., Khusro, A., Islam, M. N., Emran, T. B., ... & Bradley, D. A. (2022). Biological agents for synthesis of nanoparticles and their applications. Journal of King Saud University-Science, 34(3), 101869.
- Machado, S., Pacheco, J. G., Nouws, H. P. A., Albergaria,J. T., & Delerue-Matos, C. (2015). Characterization of green zero-valent iron nanoparticles produced with tree leaf extracts. Science of the total environment, 533, 76-81.
- Saif, S., Tahir, A., & Chen, Y. (2016). Green synthesis of iron nanoparticles and their environmental applications and implications. Nanomaterials, 6(11), 209.
- Hossain, M. T., & Hoq, M. O. (2016). Therapeutic use of Adhatoda vasica. Asian Journal of Medical and Biological Research, 2(2), 156-163.
- Khadri, H., Alzohairy, M., Janardhan, A., Kumar, A. P., & Narasimha, G. (2013). Green synthesis of silver nanoparticles with high fungicidal activity from olive seed extract. Advances in Nanoparticles, 2(3), 241-6.
- Yao, J. D., & Moellering Jr, R. C. (2011). Antibacterial agents. Manual of clinical microbiology, 1041-1081.
- Balloux, F., & van Dorp, L. (2017). Q&A: What are pathogens, and what have they done to and for us?. BMC biology, 15(1), 91.
- Leyva Salas, M., Mounier, J., Valence, F., Coton, M., Thierry, A., & Coton, E. (2017). Antifungal microbial agents for food biopreservation—A review. Microorganisms, 5(3), 37.
- Das, D., Nath, B. C., Phukon, P., & Dolui, S. K. (2013). Synthesis of ZnO nanoparticles and evaluation of antioxidant and cytotoxic activity. Colloids and Surfaces B: Biointerfaces, 111, 556-560.
- Jaji, N. D., Lee, H. L., Hussin, M. H., Akil, H. M., Zakaria, M. R., & Othman, M. B. H. (2020). Advanced nickel nanoparticles technology: From synthesis to applications. Nanotechnology reviews, 9(1), 1456-1480.
- Hussain, S., Ali Muazzam, M., Ahmed, M., Ahmad, M., Mustafa, Z., Murtaza, S., ... & Imran, M. (2023). Green synthesis of nickel oxide nanoparticles using Acacia nilotica leaf extracts and investigation of their electrochemical and biological properties. Journal of Taibah University for Science, 17(1), 2170162.
- Bibi, I., Kamal, S., & Ahmed, A. (2017). Nickel nanoparticle synthesis using Camellia Sinensis as. environment, 7, 15.
- Cheng, Y., Guo, M., Zhai, M., Yu, Y., & Hu, J. (2020). Nickel nanoparticles anchored onto Ni foam for supercapacitors with high specific capacitance. Journal of nanoscience and nanotechnology, 20(4), 2402-2407.
- Jiao, M., Yao, Y., Pastel, G., Li, T., Liang, Z., Xie, H., ... & Hu,L. (2019). Fly-through synthesis of nanoparticles on textile and paper substrates. Nanoscale, 11(13), 6174-6181.
- Mary, A. R., Sandeep, C. S., Narayanan, T. N., Philip, R., Moloney, P., Ajayan, P. M., & Anantharaman, M. R. (2011). Nonlinear and magneto-optical transmission studies on magnetic nanofluids ofnon-interacting metallic nickel nanoparticles. Nanotechnology, 22(37), 375702.
- Ullah, I., Akhtar, S., Adnan, M., Nawab, J., Ullah, S., & Abdullah-Al-Wadud, M. (2025). Ethnobotanical knowledge and ethnomedicinal uses of plant resources by urban communities of Khyber Pakhtunkhwa, Pakistan: a novel urban ethnobotanical approach. Journal of Ethnobiology and Ethnomedicine, 21(1), 13.
- Sharma, V. K., Yngard, R. A., & Lin, Y. (2009). Silver nanoparticles: green synthesis and their antimicrobial activities. Advances in colloid and interface science, 145(1-2), 83-96.
- Velmurugan, G., Chohan, J. S., Paramasivam, P., & Maranan,R. (2024). Green synthesis of silver nanoparticles from southern Eucalyptus globulus: Potent antioxidants and photocatalysts for rhodamine B dye degradation. Desalination and Water Treatment, 320, 100687.
- Mohamed, M. A., Jaafar, J., Ismail, A. F., Othman, M. H. D., & Rahman, M. A. (2017). Fourier transform infrared (FTIR) spectroscopy. In Membrane characterization (pp. 3-29). elsevier.
- Behzadi, S., Ghasemi, F., Ghalkhani, M., Ashkarran,A., Akbari, S. M., Pakpour, S., ... & Mahmoudi, M. (2015). Determination of nanoparticles using UV-Vis spectra. Nanoscale, 7(12), 5134-5139.
- Sadeghi, M., Ghaedi, H., Yekta, S., & Babanezhad, E. (2016). Decontamination of toxic chemical warfare sulfur mustard and nerve agent simulants by NiO NPs/Ag-clinoptilolite zeolite composite adsorbent. Journal of environmental chemical engineering, 4(3), 2990-3000.
- Silva, A. R., Freire, C., De Castro, B., Freitas, M. M. A., & Figueiredo, J. L. (2001). Anchoring of a nickel (II) Schiff base complex onto activated carbon mediated by cyanuric chloride. Microporous and mesoporous materials, 46(2-3), 211-221.
- Bouremana, A., Guittoum, A., Hemmous, M., Martínez-Blanco, D., Gorria, P., Blanco, J. A., & Benrekaa, N. (2015). Microstructure, morphology and magnetic properties of Ni nanoparticles synthesized by hydrothermal method. Materials Chemistry and Physics, 160, 435-439.
- Ed-Dra, A., Filali, F. R., Presti, V. L., Zekkori, B., Nalbone, L., Bouymajane, A., ... & Giarratana, F. (2020). Chemical composition, antioxidant capacity and antibacterial action of five Moroccan essential oils against Listeria monocytogenes and different serotypes of Salmonella enterica. Microbial Pathogenesis, 149, 104510.
- Ni, S., Li, T., Lv, X., Yang, X., & Zhang, L. (2013). Designed constitution of NiO/Ni nanostructured electrode for high performance lithium ion battery. Electrochimica Acta, 91, 267-274.
- Ying, S., Guan, Z., Ofoegbu, P. C., Clubb, P., Rico, C., He, F., & Hong, J. (2022). Green synthesis of nanoparticles: Current developments and limitations. Environmental technology & innovation, 26, 102336.
- Wegner, S., Rutz, C., Schütte, K., Barthel, J., Bushmelev, A., Schmidt, A., ... & Janiak, C. (2017). Soft, wetâ?ÂÂchemical synthesis of metastable superparamagnetic hexagonal closeâ?ÂÂpacked nickel nanoparticles in different ionic liquids. Chemistry–A European Journal, 23(26), 6330-6340.
- Chandra, S., Kumar, A., & Tomar, P. K. (2014). Synthesis of Ni nanoparticles and their characterizations. Journal of Saudi Chemical Society, 18(5), 437-442.
- Li, N., Li, Y., Li, Q., Zhao, Y., Liu, C. S., & Pang, H. (2021).NiO nanoparticles decorated hexagonal nickel-based metal-organic framework: self-template synthesis and its application in electrochemical energy storage. Journal of Colloid and Interface Science, 581, 709-718.
- Thirumagal, N., & Jeyakumari, A. P. (2020). Structural, optical and antibacterial properties of green synthesized silver nanoparticles (AgNPs) using Justicia adhatoda L. leaf extract. Journal of Cluster Science, 31(2), 487-497.
- Luzala, M. M., Muanga, C. K., Kyana, J., Safari, J. B.,Zola, E. N., Mbusa, G. V., ... & Memvanga, P. B. (2022). A critical review of the antimicrobial and antibiofilm activities of green-synthesized plant-based metallic nanoparticles. Nanomaterials, 12(11), 1841.
- Dikshit, P. K., Kumar, J., Das, A. K., Sadhu, S., Sharma, S., Singh, S., ... & Kim, B. S. (2021). Green synthesis of metallic nanoparticles: Applications and limitations. Catalysts, 11(8), 902.
- Balogun, S. A., Abolarinwa, T. O., Adesanya, F. A., Ateba, C. N., & Fayemi, O. E. (2024). Spectroscopic and antibacterial activities of cobalt and nickel nanoparticles: a comparative analysis. Journal of Analytical Science and Technology, 15(1), 33.
- Álvarez-Chimal, R., García-Pérez, V. I., Álvarez-Pérez, M. A., Tavera-Hernández, R., Reyes-Carmona, L., Martínez-Hernández, M., & Arenas-Alatorre, J. Á. (2022). Influence of the particle size on the antibacterial activity of green synthesized zinc oxide nanoparticles using Dysphania ambrosioides extract, supported by molecular docking analysis. Arabian Journal of Chemistry, 15(6), 103804.
- Mohapatra, S., Hassan, N., Mirza, M. A., & Iqbal, Z. (2024). Antifungal Efficacy of Plant-Based Nanoparticles as a Putative Tool for Antifungal Therapy. In Advances in Antifungal Drug Development: Natural Products with Antifungal Potential (pp. 471-491). Singapore: Springer Nature Singapore.
- Egbuna, C., Mukherjee, M., Rao, G. N., Gido, L. J. F. J., & Tijjani, H. (2018). Introduction to phytochemistry. In Phytochemistry (pp. 3-36). Apple Academic Press.
- Jain, V. M., Karibasappa, G. N., Dodamani, A. S., Prashanth,V. K., & Mali, G. V. (2016). Comparative assessment of antimicrobial efficacy of different hand sanitizers: An: In vitro: Study. Dental research journal, 13(5), 424-431.. https:// doi.org/10.1177/2320206820939403.
- Hakeem, M. J., Feng, J., Nilghaz, A., Ma, L., Seah, H. C., Konkel, M. E., & Lu, X. (2020). Active packaging of immobilized zinc oxide nanoparticles controls Campylobacter jejuni in raw chicken meat. Applied and environmental microbiology, 86(22), e01195-20.
- Mariam, A. A., Kashif, M., Arokiyaraj, S., Bououdina, M., Sankaracharyulu, M., Jayachandran, M., & Hashim,U. (2014). Bio-synthesis of NiO and Ni nanoparticles and their characterization. Digest Journal of Nanomaterials and Biostructures, 9(3), 1007-1019.
- Schaechter, M. (2009). Encyclopedia of microbiology. Academic Press.



