Research Article - (2026) Volume 9, Issue 2
Green Synthesis and Characterization of Zinc Oxide Nanoparticles using Fragaria Anannsa Extract and their Antioxidant and Antimicrobial Activities
2Shifa College of Medical Technology, Shifa Tameer-E-Millat University, Islamabad, Pakistan
3Department of Material Science and Engineering. Zhejiang Sci-Tech University, Hangzhou, Zhejiang, China
4BPP university, UK
5Department of Biosciences, Comsats University, Islamabad, Pakistan
Received Date: Mar 10, 2026 / Accepted Date: Apr 03, 2026 / Published Date: Apr 15, 2026
Copyright: ©2026 Janeeta Raheel, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Owais, H., Bashir, M., Raheel, J., Tariq, T., Zaheer, M. U., et. al. (2026). Green Synthesis and Characterization of Zinc Oxide Nanoparticles using Fragaria Anannsa extract and their Antioxidant and Antimicrobial Activities. J Agri Horti Res, 9(2), 01-12.
Abstract
Background: The global threat of urinary tract infections and antimicrobial resistance towards urinary pathogens emphasizes the need for alternative treatments against evolving pathogens.This study focuses on synthesizing zinc oxide nanoparticles (ZnO NPs) using a simple, cost-effective, and eco-friendly green approach using extracts from Fragaria x ananassa (strawberry) for antimicrobial and antioxidant properties.
Objectives: To synthesize and evaluate Fragaria x ananassa-mediated zinc oxide nanoparticles for their antimicrobial and antioxidant activities.
Methods: Fragaria x ananassa ZnO nanoparticles were synthesized by blending 400mL of deionized water with 400mL of 400% (w/v) fruit extract at 30°C to prepare a 2M zinc acetate solution. Following filtration and drying at 80°C, the resulting powder was transferred into Eppendorf tubes for Characterization. The formation of ZnONPs was confirmed by ultraviolet- visible spectroscopy. The morphology and composition of prepared NPs were determined by Scanning electron microscopy(SEM), X-ray diffraction (XRD), Energy Dispersive X-ray analysis (EDX), and Fourier-transform infrared spectroscopy(FT-IR). The antioxidant activity was assessed using the DPPH scavenging assay. Finally, antimicrobial activities were examined against clinical isolates of urinary pathogens.
Results: The formation of ZnO nanoparticles was confirmed through the UV- Vis spectroscopy and a surface-plasmon resonance peak was observed at 205 nm. The particles were stable, closely packed, and irregular with a mean size of 14.92 nm. FTIR analysis showed a capping agent absorbed onto the surface of the nanoparticle. ZnO NPs also enhanced antioxidant activity due to the activation of reactive oxygen species (ROS). Overall, the outcomes of antimicrobial studies revealed that smaller particles demonstrated greater effects against pathogens.
Conclusion: Zinc Oxide Nano Particles were successfully synthesized using Fragaria x ananassa extract as an antimicrobial agent, reducing and capping agent.
Keywords
Antimicrobial Activities, Fragaria x Ananassa, Green Synthesis, Urinary Pathogens, ZnO Nanoparticles
Graphical Abstract
Introduction
The widespread occurrence of infectious diseases and the rise of antimicrobial resistance pose a global challenge to healthcare systems. At the same time, the increasing prevalence of Antimicrobial resistance worsens these issues [1]. The second most prevalent infection is urinary tract infections (UTIs) compared to respiratory, causing significant illness and healthcare expenses In 2019, 404.6 million and even more individuals suffered from UTIs globally, highlighting the considerable impact of this bacterial infection on public health. Compared to 1990 data, there has been a substantial rise of 60.40% in UTI cases, with 252.25 million cases reported in 1990 [2,3]. Approximately 80% of infection of the urinary tract are caused by E.coli [4]. It has a great potential to cause resistance via horizontal gene transfer and it is susceptible to major antibiotics [5]. other urinary pathogens like P. aeruginosa, K. pneumoniae, P.vulgaris are also common in UTI diagnosis [2].They can easily gain resistance through acquiring genes for extended-spectrum β- lactamases, carbapenemases, 16S rRNA methylases, mcr genes, and plasmid-mediated quinolone resistance [6]. That leads to an urgent need for alternative treatments. Nanotechnology has become the subject of growing interest among researchers [7]. Nanoparticle (NP)-based therapy gained considerable attention due to their potential applications in diagnostics, drug delivery, therapeutics, and alternative treatment for UTIs [8]. Silver (Ag), copper, titanium, zinc (ZnO), gold, and magnesium are widely used elements for NP synthesis [9] . Zinc oxide nanoparticles (ZnO NPs) have been widely studied for their antimicrobial activities against various pathogens [10]. Nanoparticles can be produced using several approaches, including chemical (such as sol-gel and hydrothermal synthesis), physical (such as laser ablation, and evaporation-condensation), and biological techniques (green synthesis), each with their benefits and drawbacks [11].
Moreover, green synthesis of nanoparticles from plant extracts, bacteria, or fungi has grabbed attention because of its eco-friendly nature, cost-effectiveness, less toxicity, and ability to produce nanoparticles of uniform size and shape [12]. Prior studies also indicate that it acts as a reducing and capping agent for humans and is environmentally friendly [13]. Phytochemicals obtained as fruits used in natural antimicrobials and natural consumption are naturally abundant in fruits [14]. Zinc oxide nanoparticles are being successfully synthesized from fruits such as Hylocereus undatus, Citrus sinensis, Gooseberry fruits, Myristica fragrans, Vaccinium Arctostaphylos, Punica granatum,Banana Peel [15,16]. In particular, Fragaria ananassa, commonly known as strawberry, is a member of the Rosaceae family, a low-growing plant. It contains bioactive compounds such as Anthocyanins, Propionic acid, Phenolic acids (Ellagitannins), Lignans (Hydroxymatairesinol (HMR) lignan), and flavonoids that contribute to their antimicrobial activity against urinary pathogens [17]. This study provides a good strategy to combat urinary tract infections. This paper explores the behavior of ZnO nanoparticles produced using a green method of synthesis. The biosynthesized ZnO NPs were confirmed and characterized by some analytical methods. These were UV-Vis spectroscopy, FTIR, XRD, SEM and energy-dispersive X-ray spectroscopy (EDS). Their antioxidant capacity was also determined. The nanoparticles were then evaluated against E. coli, S. aureus, P. aeruginosa, K. pneumoniae, and Proteus vulgaris using the antibacterial property. Furthermore, the antioxidant potential of the biosynthesized ZnO nanoparticles was evaluated using standard in vitro assays. The results of this study aim to provide insight into the potential application of green-synthesized ZnO nanoparticles as an effective, eco-friendly alternative for managing antimicrobial resistance and urinary tract infections.
Materials and Methods
Chemicals and Bacterial Strains
The research was performed at the Abasyn University Islamabad Campus and National University of Science and Technology, Islamabad. Zinc Acetate dihydrate [(CH3COO)2 Zn*2H20] (Sigma-Aldrich) was used for synthesis of zinc oxide nanoparticles. Mueller–Hinton agar (Sigma- Aldrich) were used as culture media. 1,1-diphenyl, 2-picrylhydrazyl (DPPH) radical (Sigma- Aldrich) were used in antioxidant scvanging assay. All of these chemicals were of analytical grade and purchased from Asia Scientific Trader Rawalpindi. The colonies of E. coli, S. aureus, P. aeruginosa, K. pneumoniae, and P. vulgaris isolated from clinical samples of Urine C/S from Capital Development Authority Hospital Islamabad.
Preparation of Plant Extract
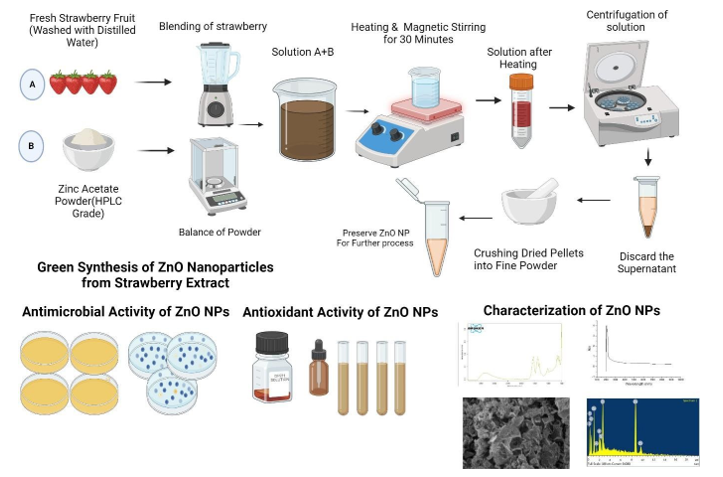
Fruits of Fresh Fragaria x ananassa have been collected in a local market of Islamabad and carried to the laboratory of Abasyn University (Islamabad Campus). The fruits were washed a few times with distilled water to get dust and other contaminants on the surface. After that, a Solution of 250gm of Fragaria x ananassa into 1000ml of Distilled water were obtained by using a blender. The solution were then be filtered using clean cloth and heated for 60 minutes. After completion, it were cooled to room temperature, then the solution were filtered using Whatman filter paper is used to obtain a purified extract [18].
Biosynthesis of ZnO Nanoparticles
A 400mL solution of 2M zinc acetate were prepared according to with slightly modification by using deionized water, which were blended with 400mL of 400% (w/v) Fragaria x ananassa fruit extract at a constant stirring temperature of 30°C using a magnetic stirrer. After an hour of stirring, the mixture were alkalinized to a pH of 12 with a 10M NaOH solution [18]. The resulting solution after stirring was collected and centrifuged at10,000 rpm for 10 min and the sediment of solution were collected. The separated sediment extract was dried in an oven at 80°C for 8 h and crushed with mortor and pestle and preserved in eppendorf tubes for further studies.
Characterization of ZnO Nanoparticles
The synthesized ZnO Nanoparticles were then characterized for further investigation. Molecular structures and crystalline nature of synthesized ZnO nanoparticles were determined using XRD while morphology and structure were determined using SEM. The elemental compositions were verified using EDS. FTIR analysis was performed for the determination of surface functional groups and the vibrations during stretching and bending. The formation and stability of nanoparticles was also determined by UV-Vis analysis. All these characterizations were made by National University of Science and Technology, Islamabad.
Antioxidant Activity
ZnO nanoparticles at 6.25, 12.5, 25, 50, and 100 mg/mL were dispersed in deionized water and sonicated for 10 min. Then, 1.0 ml of 100 mM DPPH and 1.0 ml of sample were vortexed and stored for 30 min at room temperature in the dark. the deep violet color of DPPH in solution, turns into pale yellow. The mixture was then centrifuged at 6,000 rpm at 10 min and the absorbance of the supernatant was determined at 517nm using a UV- spectrophotometer. Measurements were carried out in three replicates and the average values were computed. The reference antioxidant was ascorbic acid. The resulting values were then used to determine the IC50.
Antimicrobial Activity
ZnO nanoparticles were utilized to demonstrate the antimicrobial activity through disc diffusion technique. The nanobiotics of five different concentrations (6.25,12.5, 25, 50 and 100 µg/ml) were prepared for evaluation of antibacterial activity. Ciprofloxacin acts as a negative control for bacterial activity. Dimethylsulfoxide was utilized as a positive control. Muller-Hinton (MH) media is utilized for bacterial growth. Bacterial stocks were prepared in the same medium, without agar. Both Gram-positive and Gram-negative bacteria were used in research.
Preparation of Microbial Culture
The bacterial strain colonies of all the bacterial strains were stocked in various falcon tubes. A bacterial colony was placed in media to generate culture followed by development of cultures in 35°C until bacterial growth was realized. These growth stocks were identified by the growth of bacteria on the MH agar media in Petri plates.
Filter Disc Preparation
In each extract concentration, one dropped about 50 discs. Hot-air sterilized Petri dishes were filled with sterile paper discs. Following the labeling, the plates were streaked a number of times to produce a homogenous bacterial lawn. The surface moisture was dried up and the discs were placed on the agar. A sterile spatula was used to transfer discs and press them to contact the agar surface completely. Each plate was placed with seven discs: a DMSO disc, which served as a negative control, ciprofloxacin disc, which was an antibacterial control, and clotrimazole disc, which was an antifungal control. Plates were used after disc placement and inverted and incubated at 37 °C, 24 h.
Observation of Antibacterial Activity
All plates were analyzed after 24h of disc application. When the outcome was satisfactory and when it had formed a circular area of inhibition then the area of inhibition has been measured on a reading scale across the diameter of discs. Reading was observed after 48 h of application in the case of fungi.
Results
Characterization of ZNO Nanoparticles
UV–VIS Spectroscopy
UV–VIS absorption spectroscopy analysis was carried out with wavelengths ranging from 200 to 800 nm to confirm the formation of ZnONPs using Fragaria annasia. Figure 3.1 illustrates the spectra of reaction solution of reduced Zinc Acetate .The 10 μL of ZnO NP solution is transferred into a clean quartz cuvette for direct spectrum measurements. The ZnO NP colloid recorded the highest absorption at 205 nm in the UV-Vis spectrum. This characteristic band is attributed to ZnO NPs, which occurs due to zinc plasmon resonance (SPR).
Figure 1: UV-VIS spectroscopy for ZnONPs synthesized by Fragaria annansia. UV–visible spectroscopy confirmed the surface Plasmon resonance in synthesized ZnONPs at 205 nm
Fourier Transform Infrared Spectroscopy
FT-IR analysis of the synthesized zinc nanoparticles was utilized to figure out the different characteristic functional groups attached to the synthesized nanoparticles. The FTIR analysis revealed distinct peaks at 3380 cm-1, 1450 cm-1, 1000 cm-1, 680 cm-1, and 620 cm-1, each corresponding to different functional groups. The broad peak at 3380 cm-1 is attributed to the O-H stretching vibrations of hydroxyl groups, likely derived from the phenolic compounds in the Fragaria x ananassa extract. The presence of these hydroxyl groups suggests strong hydrogen bonding, contributing to the stability and capping of the ZnO-NPs. The 1450 cm-1 band is assigned to C-H bending, which indicates the presence of alkane-type groups of strawberry-derived organics bound to the surface of ZnO. These moieties can help in the interaction of the particles and enhance stability. The signal at around 1000 cm-1 indicates C-O stretching, which is also typical of alcohol, carboxylic acid and ester or ether groups existing in the extract, which probably act as capping agents during the formation of nanoparticles. These functional groups contribute to the reduction and capping processes during the synthesis of ZnO-NPs. Peaks at 680 cm-1 and 620 cm-1 are characteristic of Zn-O stretching vibrations, confirming the formation of zinc oxide nanoparticles. The observed peaks support successful formation of ZnO NPs through the green synthesis method utilizing Fragaria x ananassa extracts.
Figure 2: FTIR Spectrum of ZnONPs Synthesized by using FTIR Confirmed the Presence of Bond Stretching (OH–, C-H, C-O, and Zn-O) in the Synthesized ZnO NPs.
X-Ray Diffraction
Figures 3.3 shows the XRD pattern attained for the ZnONPs synthesized using the plant extract of Fragaria x ananassa . The XRD patterns revealed distinct diffraction peaks at 2θ values of 31.768°, 33.139°, 36.249°, and 59.041°. These peaks correspond to the characteristic planes of the hexagonal wurtzite ZnO structure, specifically the (100), (002), (101), and (110) planes, respectively. The XRD reflections are aligned to ZnO nanoparticles and indicate a highly crystalline phase. Additional peaks were not detected, indicating that the synthesized nanoparticles are phase-pure without any detectable impurities or secondary phases. The sharp and intense nature of the peaks suggests that the ZnO NPs possess a well-defined crystalline structure with a relatively small particle size,which is consistent with the nanoscale range. The mean size of crystallites in green synthesized ZnO nanoparticles is about 14.92 nm.
Figure 3: XRD Pattern of ZnONPs Synthesized from Fragaria x Ananassa. XRD Image Showing the Characteristic Hexagonal Wurtzite Crystal Structure.
Scanning Electron Microscopy
The particles were examined with scanning electron microscopy (SEM). It assisted them in estimating their shape, size, and surface features. A TESCAN VEGA 3 was used at the Institute of Space Technology, Islamabad, to conduct an SEM analysis. The micrographs reveal that the nanoparticles are irregular shaped and are densely clustered (Figure 4). Aggregates consist of a number of particles. The reason behind this clustering is probably associated with the high level of surface energy which enables particles to attract each other.

Figure 4: SEM Image of ZnO NPs Synthesized from Scanning Electron Microscopy Depicts the Irregular Nature of Synthesized ZnONPs
Energy Dispersive X-Ray Spectroscopy
The synthesized material was confirmed to display the elemental composition of the material using EDS. This was analysed using the Tescan VEGA 3. The EDS spectrum of the ZnO sample contained clear peaks of zinc and oxygen, which indicated the presence of the expected elements in appropriate quantities. It also detected a small carbon signal which could be attributed to the surface of the sample or the mounting medium. Figure 3.5 shows the results in a more detailed manner, giving a better idea of the elemental peaks and their relative intensities.
Figure 5: EDS Analysis of ZnO NPs Shows Elements Constituents.
|
ELEMENTS |
WEIGHT PERCENTAGE |
ATOMIC PERCENTAGE |
|
O |
40.1 |
73.3 |
|
Zn |
59.9 |
26.7 |
Table 1: Elements Constituting ZnO Nanoparticles
Antioxidant Activity
DPPH is a non-reactive free radical with a high UV-Vis absorption peak of 517 nm. To identify the free-radical scavenging ability of the biosynthesized ZnO nanoparticles, the DPPH assay was used. The test has a basic concept. When DPPH radicals are neutralized by the nanoparticles, the purple colour disappears and the absorbance increases at 517 nm reduces. This research analyzes the radical scavenging potential of ZnO nanoparticles that were prepared using Fragaria anan assays fruit extracts through this technique. Results revealed that ZnO NPs have promising antioxidant properties as shown in table 2. It is revealed from the table that the bioactive molecules present in plant extracts not only act as a reducing agent but also as a capping agent. The data includes absorbance values for ascorbic acid and ZnO nanoparticles across various concentrations and the percentage of radical scavenging activity (%RSA) and IC50 values. Ascorbic acid, a standard antioxidant, shows an increase in absorbance with increasing concentration, indicating its effective antioxidant properties. The ZnO nanoparticles, while exhibiting slightly lower absorbance values compared to ascorbic acid, also demonstrate significant antioxidant activity. The %RSA for ZnO nanoparticles is highest at 100 mg/mL (17.05%) and decreases with lower concentrations, reflecting a concentration-dependent increase in antioxidant activity. The IC50 values represent the concentration needed to inhibit 50% of the DPPH radicals, also increase with increasing concentration, highlighting the greater effectiveness of ZnO nanoparticles at higher concentrations.
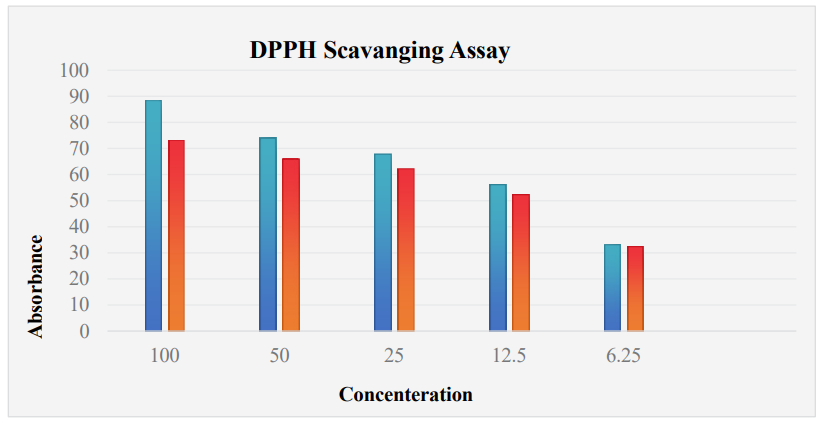
Figure 6: Free Radicle Scavenging Activity of Synthesized ZnO NPs of Concentration and Ascorbic Acid (as Positive Control) at Different Concentrations.
|
Calculation of %RSA and IC50 From DPPH Assay Absorbance Measurement Data |
||||
|
Concentration(mg/ml) |
Ascorbic Acid |
ZnO NPs |
%RSA |
IC50 |
|
100 |
88 |
73 |
17.04545 |
10.95mg/ml |
|
50 |
74 |
66 |
10.81081 |
|
|
25 |
58 |
62 |
8.823529 |
|
|
12.5 |
56 |
52 |
7.142857 |
|
|
6.25 |
33 |
32 |
3.030303 |
|
Table 2: Free Radicle Scavenging Activity of Different ZnO NPs:
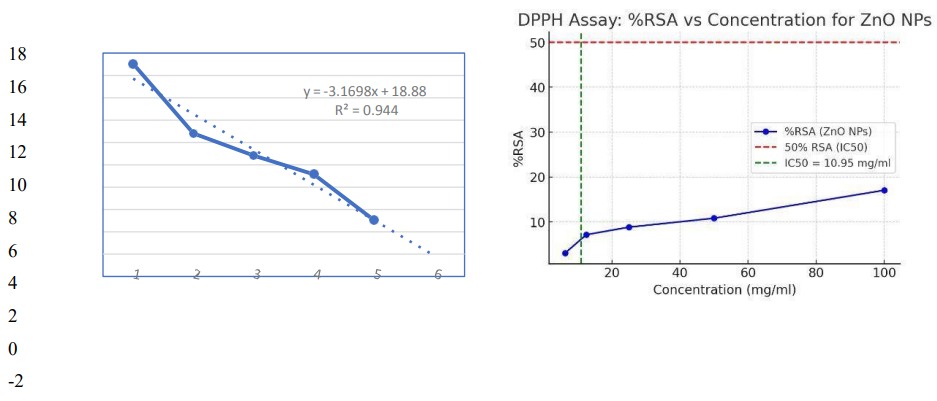
Figure 7: Calculation of %RSA & IC50 From DPPH Assay
Antimicrobial Activity
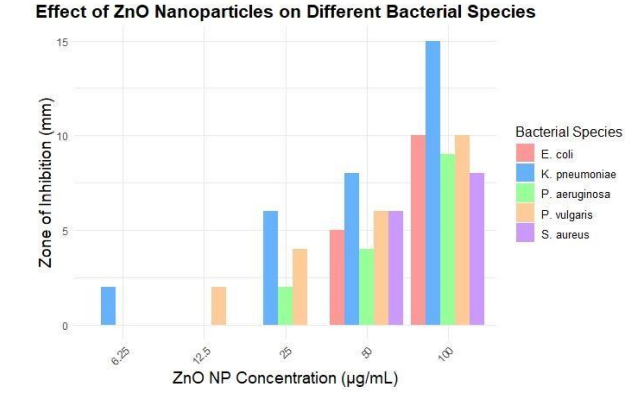
The disc diffusion method was applied to test the antibacterial activity of ZnO nanoparticles prepared with Fragaria ananassa fruit extract. Various concentrations were used (6.25, 12.5, 25, 50 and 100 ug/mL). The nanoparticles were tested on one Gram-positive (S. aureus) strain and on a number of Gram-negative urinary pathogens (P. aeruginosa, P. vulgaris, K. pneumoniae, E. coli). The positive control was Ciprofloxacin and the null/ negative control was DMSO. The presence of antibacterial activity was demonstrated by a distinct zone created around each disc. The presence of measurable zone of inhibition indicated that the synthesized ZnO nanoparticles possess antibacterial properties (Figure 3). In general, the disc containing 20 ug/mL ZnO nanoparticles provided a relatively larger inhibition zone than some of the other doses tested on the bacterial strains.
|
Bacterial Isolates |
Fragaria x ananassa ZnO NPs |
||||||
|
|
6.25 µg/ml |
12.5 µg/ml |
25 µg/ml |
50 µg/ml |
100 µg/ml |
Negative Control |
Positive Control |
|
E.coli |
0 mm |
0 mm |
0 mm |
5 mm |
10 mm |
0 mm |
10mm |
|
K.pneumoina |
2 mm |
0 mm |
6 mm |
8 mm |
15 mm |
0 mm |
12mm |
|
P.vulgaris |
0 mm |
02 mm |
04 mm |
06 mm |
10 mm |
0 mm |
15mm |
|
P.aeruogenosa |
0 mm |
0 mm |
02 mm |
04 mm |
09 mm |
0 mm |
10mm |
|
S.aureus |
0 mm |
0 mm |
0mm |
6mm |
8mm |
0 mm |
15mm |
Table 3: Antibacterial Assay of Nanoparticles of Nonpathogenic Strains: (Zone of Inhibition (ZOI) in (mm)
Discussion
Green synthesis has gained significant attention in the recent era because of its eco-friendly, non- toxic, and cost-effective properties. Plant extracts are widely used in green synthesis because of the presence of natural compounds like phenols, alkaloids, and terpenoids, which help reduce metal ions to nanoparticles [19]. By using plant extracts, we can alter the size and shape of nanoparticles in no time and without any complicated equipment as compared to the traditional methods [20]. Fruit extracts have been used for nanoparticle synthesis, as they possess antimicrobial and antioxidant properties. This makes nanoparticles effective against both pathogenic and oxidative stresses. Thus, they play a critical role in addressing antibiotic resistance by inhibiting microbial growth and reducing oxidative damage associated with antimicrobial treatments applied. Green synthesis of zinc oxide nanoparticles using Fragaria x ananassa extracts has several advantages because of being environmentally friendly, low-toxicity, and economical. This technique uses the chemicals present in the strawberry extracts, such as phenols, alkaloids, sugars, proteins, terpenoids, acids, and polyphenols, for metal ions reduction to nanoparticles. The synthesized ZnO NPs therefore show a wide arena of applications, mainly due to their antimicrobial and antioxidant properties. These features make it a potential candidate for fighting against Antimicrobial-resistant pathogens and oxidative stress-based problems. The properties of the ZnO NPs were confirmed with the help of different modern techniques. UV-Vis spectroscopy was used to identify the nanoparticles’ optical properties and confirm their formation. The characteristic UV maximum absorbance peak of the colloidal suspension of ZnO NPs was observed at 205 nm [21].
FTIR was utilized to identify the functional groups derived from the plant extract on the surface of the ZnO NPs. It is observed that peaks corresponding to O-H, C-H, C-O, and Zn-O stretching vibrations have been noted, thus verifying the existence of organic molecules on the surface of nanoparticles. An earlier work reported the same functional groups. These functional groups contribute towards the reduction and capping processes during the synthesis of ZnO-NPs [22]. ray diffraction analysis reflected the crystallinity of the ZnO NPs. Information on phase, structure, and crystallite size of about 14.92 nm for the green synthesized ZnO nanoparticles with a hexagonal wurtzite structure is gained from it. Previous works also show similar results. Therefore, these diffraction peaks represent a successful synthesis of ZnO NPs with highly crystalline phases, no extra peaks have been left, hence indicating that the synthesized nanoparticles are phase-pure and no detectable impurities or secondary phases found such sharp and intense peaks suggest a well-defined crystalline structure and small particle size of ZnO NPs, which fall under the nanoscale range [23].SEM was applied to investigate the surface morphology and size distribution, which showed an image that was irregular in shape and unevenly dispersed with some degree of aggregation. This irregularity in shape and size distribution is consistent with previous studies, which have reported similar findings for ZnO nanoparticles synthesized using plant extracts. Energy dispersive X-ray spectroscopy played a complementary role to SEM and offered data on the elemental composition of the material and proved the presence of zinc and oxygen with other elements from the plant extract [24].
The prepared ZnO NPs were also put under thorough study regarding their biological activities. Screening for antimicrobial activity against various strains of bacteria showed quite a potential against the development inhibition of both gram-positive and gram-negative bacteria. This antimicrobial activity is mainly due to the formation of ROS and the disruption of microbial cell membranes [25]. The free radical scavenging ability is tested with the use of the free radical compound DPPH. The ZnO nanoparticles depicted a remarkable anti-oxidant activity. In the case of ZnO nanoparticles, it is at its maximum at 100 mg/mL with a value of 17.05%. It decreases with lower concentration reflecting a concentration-dependent increase in the anti-oxidant activity. IC50 values are the concentrations needed to block 50% of DPPH radicals, also increased with increasing concentration, reflecting the greater effectiveness of ZnO nanoparticles at higher concentrations. This could therefore be an antioxidant activity, probably due to the synergistic effects of ZnO and the bioactive compounds from Fragaria x ananassa in neutralizing free radicals and mitigating oxidative stress.
Green synthesized ZnO NPs are known for their enhanced biocompatibility and reduced toxicity concerning those synthesized through chemical means. These studies have proved that plant- mediated ZnO NPs displayed excellent antimicrobial activity because they can generate ROS. The generated ROS could cause bacterial cell wall and DNA damage and leads to cell death. Further enhancement in this potential action of these nanoparticles is provided by the presence of phenolic compounds and flavonoids from plant extracts, which quench free radicals and act as free-radical scavengers, offering protection from oxidative stress. This is an environmentally benign and sustainable method to synthesize superior nanoparticles with enhanced antimicrobial and antioxidant activities, which would be a potential solution in medical and environmental applications.
Conclusions
The green synthesis approach is environmentally friendly, cost-effective, and can produce ZnO NPs of the appropriate size at room temperature. In this investigation, Fragaria ananassa fruit extracts were utilized as both reducing and stabilizing agents. The synthesized ZnO NPs were characterised using UV–Vis, XRD, FTIR, and SEM. UV-Vis spectral analysis validated the surface Plasmon resonance of green-synthesized Zinc nanoparticles. FTIR analysis confirms that biomolecules acted as both reducing and capping agents during ZnONP synthesis. SEM examinations indicated irregular, uniformed, and non-uniform-shaped zinc nanoparticles ranging in size of 14.92 nm. The XRD pattern revealed a hexagonal wurtzite crystal structure. The biosynthesized ZnONPs employed in this work were found to have strong antibacterial action against urinary infections. Because of their potent antibacterial properties. It showed dose-dependent inhibitory action against microbes. Biologically produced ZnONPs could be extremely useful in the medical field against harmful and resistant micro-organisms.
Funding: This research received no external funding.
Conflicts of Interest: No conflict of interest is declared by the authors.
References
- Salam, M. A., Al-Amin, M. Y., Salam, M. T., Pawar, J. S., Akhter, N., Rabaan, A. A., & Alqumber, M. A. (2023, July). Antimicrobial resistance: a growing serious threat for global public health. In Healthcare (Vol. 11, No. 13, p. 1946). MDPI.
- Mancuso, G., Midiri, A., Gerace, E., Marra, M., Zummo, S., & Biondo, C. (2023). Urinary tract infections: the current scenario and future prospects. Pathogens, 12(4), 623.
- Zeng, Z., Zhan, J., Zhang, K., Chen, H., & Cheng, S. (2022). Global, regional, and national burden of urinary tract infections from 1990 to 2019: an analysis of the global burden of disease study 2019. World Journal of Urology, 40(3), 755-763.
- Zhou, Y., Zhou, Z., Zheng, L., Gong, Z., Li, Y., Jin, Y., ... & Chi,M. (2023). Urinary tract infections caused by uropathogenic Escherichia coli: mechanisms of infection and treatment options. International journal of molecular sciences, 24(13), 10537.
- Tao, S., Chen, H., Li, N., Wang, T., & Liang, W. (2022). The spread of antibiotic resistance genes in vivo model. Canadian Journal of Infectious Diseases and Medical Microbiology, 2022(1), 3348695.
- Li, Y., Kumar, S., Zhang, L., Wu, H., & Wu, H. (2023).Characteristics of antibiotic resistance mechanisms and genes of Klebsiella pneumoniae. Open Medicine, 18(1), 20230707.
- Malik, S., Muhammad, K., & Waheed, Y. (2023). Emerging applications of nanotechnology in healthcare and medicine. Molecules, 28(18), 6624.
- Qindeel, M., Barani, M., Rahdar, A., Arshad, R., & Cucchiarini,M. (2021). Nanomaterials for the diagnosis and treatment of urinary tract infections. Nanomaterials, 11(2), 546.
- Asif, M., Yasmin, R., Asif, R., Ambreen, A., Mustafa, M., & Umbreen, S. (2022). Green synthesis of silver nanoparticles (AgNPs), structural characterization, and their antibacterial potential. Dose-response, 20(2), 15593258221088709.
- Sirelkhatim, A., Mahmud, S., Seeni, A., Kaus, N. H. M., Ann,L. C., Bakhori, S. K. M., ... & Mohamad, D. (2015). Review on zinc oxide nanoparticles: antibacterial activity and toxicity mechanism. Nano-micro letters, 7(3), 219-242.
- Altammar, K. A. (2023). A review on nanoparticles: characteristics, synthesis, applications, and challenges. Frontiers in microbiology, 14, 1155622.
- Hano, C., & Abbasi, B. H. (2021). Plant-based green synthesis of nanoparticles: Production, characterization and applications. Biomolecules, 12(1), 31.
- Okaiyeto, K., Gigliobianco, M. R., & Di Martino, P. (2024). Biogenic zinc oxide nanoparticles as a promising antibacterial agent: synthesis and characterization. International Journal of Molecular Sciences, 25(17), 9500.
- Suriyaprom, S., Mosoni, P., Leroy, S., Kaewkod, T., Desvaux, M., & Tragoolpua, Y. (2022). Antioxidants of fruit extracts as antimicrobial agents against pathogenic bacteria. Antioxidants, 11(3), 602.
- Vishnupriya, B., Nandhini, G. E., & Anbarasi, G. (2022). Biosynthesis of zinc oxide nanoparticles using Hylocereus undatus fruit peel extract against clinical pathogens. Materials Today: Proceedings, 48, 164-168.
- Gao, Y., Xu, D., Ren, D., Zeng, K., & Wu, X. (2020). Green synthesis of zinc oxide nanoparticles using Citrus sinensis peel extract and application to strawberry preservation: A comparison study. Lwt, 126, 109297.
- Mohd Yusof, H., Mohamad, R., Zaidan, U. H., & Abdul Rahman, N. A. (2019). Microbial synthesis of zinc oxide nanoparticles and their potential application as an antimicrobial agent and a feed supplement in animal industry: a review. Journal of animal science and biotechnology, 10(1), 57.
- Sutradhar, P., & Saha, M. (2016). Green synthesis of zinc oxide nanoparticles using tomato (Lycopersicon esculentum) extract and its photovoltaic application. Journal of Experimental Nanoscience, 11(5), 314-327.
- Selim, Y. A., Azb, M. A., Ragab, I., & HM Abd El-Azim, M. (2020). Green synthesis of zinc oxide nanoparticles using aqueous extract of Deverra tortuosa and their cytotoxic activities. Scientific reports, 10(1), 3445.
- Álvarez-Chimal, R., & Arenas-Alatorre, J. Á. (2023). Green synthesis of nanoparticles. A biological approach.
- Surendra, T. V., Roopan, S. M., Al-Dhabi, N. A., Arasu, M. V., Sarkar, G., & Suthindhiran, K. (2016). Vegetable peel waste for the production of ZnO nanoparticles and its toxicological efficiency, antifungal, hemolytic, and antibacterial activities. Nanoscale research letters, 11(1), 546.
- Gatou, M. A., Lagopati, N., Vagena, I. A., Gazouli, M., & Pavlatou, E. A. (2022). ZnO nanoparticles from different precursors and their photocatalytic potential for biomedical use. Nanomaterials, 13(1), 122.
- Haiouani, K., Hegazy, S., Alsaeedi, H., Bechelany, M., & Barhoum, A. (2024). Green synthesis of hexagonal-like ZnO nanoparticles modified with phytochemicals of clove (Syzygium aromaticum) and Thymus capitatus extracts: enhanced antibacterial, antifungal, and antioxidant activities. Materials, 17(17), 4340.
- Khan, Y., Sadia, H., Ali Shah, S. Z., Khan, M. N., Shah, A. A., Ullah, N., ... & Khan, M. I. (2022). Classification, synthetic, and characterization approaches to nanoparticles, and their applications in various fields of nanotechnology: a review. Catalysts, 12(11), 1386.
- Alfei, S., Schito, G. C., Schito, A. M., & Zuccari, G. (2024). Reactive oxygen species (ROS)-mediated antibacterial oxidative therapies: available methods to generate ROS and a novel option proposal. International Journal of Molecular Sciences, 25(13), 7182.


