Research Article - (2026) Volume 10, Issue 1
Facile and Eco-friendly One Step Low Temperature Synthesis of Very Large Scale ATiO3 (A = Ca, Sr, Ba and Cd)
2Department of Chemistry, Indira Gandhi National Tribal University Amarkantak, India
3Department of Chemistry, Kisan College Nalanda (Constituent unit of Patliputra University Patna), India
4Department of Chemistry, S B College Ara (Constituent unit of Veer, Kunwar Singh University Ara), India
Received Date: Dec 30, 2025 / Accepted Date: Jan 26, 2026 / Published Date: Feb 04, 2026
Copyright: ©2026 Bharat Kumar, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Pandey, P. K., Chaubey, R. R., Rao, G. K., Srivastava, S., Kumar, B., et al. (2026). Facile and Eco-friendly One Step Low Temperature Synthesis of Very Large Scale ATiO3(A = Ca, Sr, Ba and Cd). J App Mat Sci & Engg Res, 10(1), 01-10.
Abstract
The primary motivation of this work is to develop a low-cost and scalable methodology for the synthesis of industrially significant metal titanates. In this study, ATiO3 (A = Ca, Sr, Ba, and Cd) perovskite nanoparticles were successfully synthesized via a simple, one-step hydrothermal process carried out at 180 °C for 16 hours. By varying the titanium precursor from TiO2 to titanium isopropoxide, nanoparticles with different sizes and morphologies were obtained. This approach offers a general and practical route for the large-scale synthesis of ATiO3 perovskite materials.
Keywords
Hydrothermal, Titanium Isopropoxide, Titanates, Hollow
Highlights
Development of a low-cost methodology for the hydrothermal synthesis of titanates with varying size and morphology
![]() Low temperature general synthesis protocol for 4 titanates
Low temperature general synthesis protocol for 4 titanates
![]() Variation of reactant phase from solid to liquid and their effect of morphology
Variation of reactant phase from solid to liquid and their effect of morphology
![]() Hollow rectangle shape morphology of CaTiO3
Hollow rectangle shape morphology of CaTiO3
![]() Enhancement in the surface area of BaTiO3
Enhancement in the surface area of BaTiO3
![]() Scope of applications in various industry
Scope of applications in various industry
Introduction
Nanostructured materials such as nanoparticles, nano cubes, nanorods, nanotubes and nanowires are under intensive investigation due to their size and shape dependent properties and potential application in the all field of technology due to their thermal, electrical, mechanical and optical properties [1-5]. Among the nanostructured materials, perovskite with general formulae ABO3 constitute important classes of materials in solid state chemistry due their wide applicability in ferroelectrics, dielectric, piezoelectric nonlinear optics properties with their technology importance for fabricating the different class of devices [6-10]. On the commercial scale, these oxide powders are produced at high- temperature which’s chemical in homogeneity, reactivity problems and very little control on the size, shape and agglomeration. Due to these above problems, there is demand for the low temperature synthesis route to yield high-purity fine powders with controlled size and morphology for miniaturization of these materials to the nanometer scale for the next generation of electronics.
All the titanates are useful class of materials due to their industrial application in various technologies. Among them BaTiO3, SrTiO3, and CaTiO3 has more importance. BaTiO3 has ferroelectric property, high dielectric constant and positive temperature coefficient of resistivity (PTCR) used as multilayered ceramic capacitors (MLCCs), embedded decoupling capacitors (EDC), thermistor, waveguide modulators, IR detectors, microwave absorbers, dynamic random access memory and field - effect transistors [11-15]. SrTiO3 is an important n-type semiconductor with band gap between 3.2 – 3.4 eV have high dielectric constant, thermal and chemical stability. It has a wide application in field of thin-film capacitors, nonlinear optics, optical memories, oxygen gas sensors, photocatalyst and photoelectrode for splitting of water, solar cell and electrooptic modulators [16-19]. CaTiO3 crystallizes in orthorhombic crystal system with space group (Pbnm). It has various applications such as component of capacitors, immobilization of nuclear wastes, mineralogy, microwave dielectric applications (as resonators and filters), luminescent material and biomedical area [20-23]. CdTiO3 crystallizes in non- ferroelectric ilmenite rhombohedral crystal system. Below the 1000 0C annealing temperature is an intelligent material due to their application in optical fiber and sensor [24, 25]. It has also piezoelectric, pyroelectric, magneto restrictive and photostrictive properties [26-28].
There are various reports for synthesizing such titanates by solid state, sol – gel, hydrothermal, microwave hydrothermal method, spray pyrolysis, microemulsion, polymeric precursor and coprecipitation [29-37]. There are few reports for general methodology of the MTiO3 nanoparticles [38-41]. There are reports of the synthesis of SrTiO3 and BaTiO3 at low temperature however there is no any such report on CaTiO3 and CdTiO3 at low temperature [42-46]. There is no general approach reported for synthesizing the above four compounds by a single step on large scale.
In this letter, we report the synthesis of ATiOâ?? (A = Ca, Sr, Ba, and Cd) perovskite materials using two different titanium sources: titanium dioxide and titanium isopropoxide. The resulting compounds were thoroughly characterized using powder X-ray diffraction (PXRD), field emission scanning electron microscopy (FESEM), transmission electron microscopy (TEM), Brunauer– Emmett–Teller (BET) surface area analysis, and UV-Visible diffuse reflectance spectroscopy (DRS).
Synthesis and Characterstics
ATiO3 nanoparticles were synthesized by hydrothermal route. In typical synthesis process 3.43 m mol of A(NO3)2 was dissolve in 20 ml of double distilled water followed addition of 3.43 m mol of TiO2 (Titanium isopropoxide also). The above solution was stirred for 30 min and then 0.6 mol NaOH dissolved in 40 ml of water were added and stirred for 3 h. The obtained milky white solution was transferred to teflon beaker and putted inside the stainless- steel hydrothermal bomb and placed in oven at 180°C for 16 h. The schematic diagram for the synthesis of ATiO3 nanoparticles of different morphology is given in scheme 1.
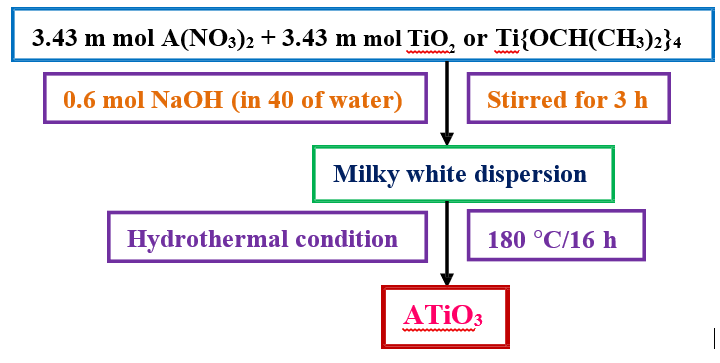
Scheme 1: General Procedure for the Synthesis of ATiO3
Powder X-ray diffraction studies (PXRD) were carried out using Ni filtered Cu-Kα radiation. Normal scans were recorded with a step size of 0.02° and step time of 1 s. The Kα2 reflections were removed to obtain accurate lattice constants. Field emission scanning electron microscopy (FESEM) was carried out by FEI quanta 3D FEG – FESEM operated at 10 kV. The pellet had been coated with gold. Transmission Electron Microscopy (TEM) studies were carried out using a Tecnai G2 20 electron microscope operated at 200 KV. TEM specimens were prepared by dispersing the sample in ethanol by ultrasonic treatment, dropping onto a porous carbon film supported on a copper grid, and then drying in air. Nitrogen adsorption–desorption isotherms were recorded at liquid nitrogen temperature (77 K) using a Nova 2000e (Quantachrome Corp.) equipment and the specific area was determined by the Brunauer–Emmett–Teller (BET) method. The powder sample was degassed at 150 ºC for 6 h prior to the surface area measurements. Diffuse-reflectance spectra (DR) spectra were recorded on Shimadzu UV-2450 spectrophotometer where the baseline was fixed using a barium sulfate reference.
Result and Discussion
The aim behind this study is to develop a low-cost methodology for the synthesis of important titanates with different morphology at very low temperature. Here we have developed a common one step synthesis protocol at very low temperature (180 ºC) to synthesize titanates at gram scale (~ 1g) level by hydrothermal route. Here we fixed the hydrothermal reaction time, temperatures, reactant and base concentration. We varied the one of the reactant (Ti source) phase from solid to liquid to see change in the morphology. Using a NaOH base, we obtained pure phase of the nanocrystalline titanates ATiO3 (Ca, Sr, Ba, Cd). Figures 1 (A, B, C, D) show the powder X-ray diffraction patterns of CaTiO3, SrTiO3, BaTiO3 and CdTiO3 respectively using both TiO2 and titanium isopropoxide as Ti source. The PXRD patterns confirm the formation of monophasic nature for all the eight titanates. All the observed diffraction patterns were index on the basis of their crystal structures. The lattice parameters were refined and it observed that in case of titanium isopropoxide as Ti source the values was lower as compared to TiO2 as Ti source due to decrease in the particles size of corresponding titanates.
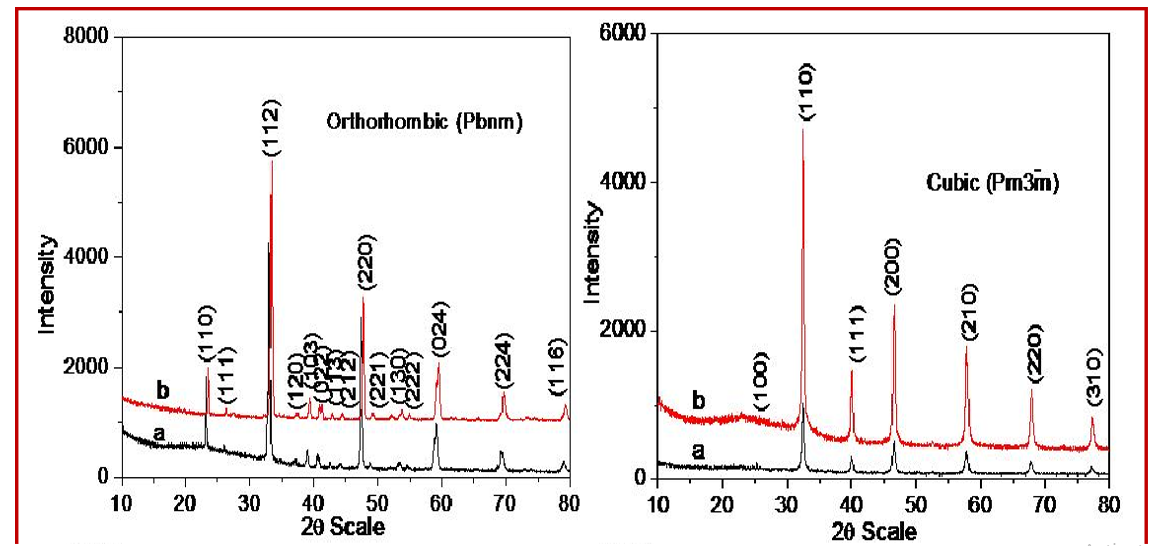
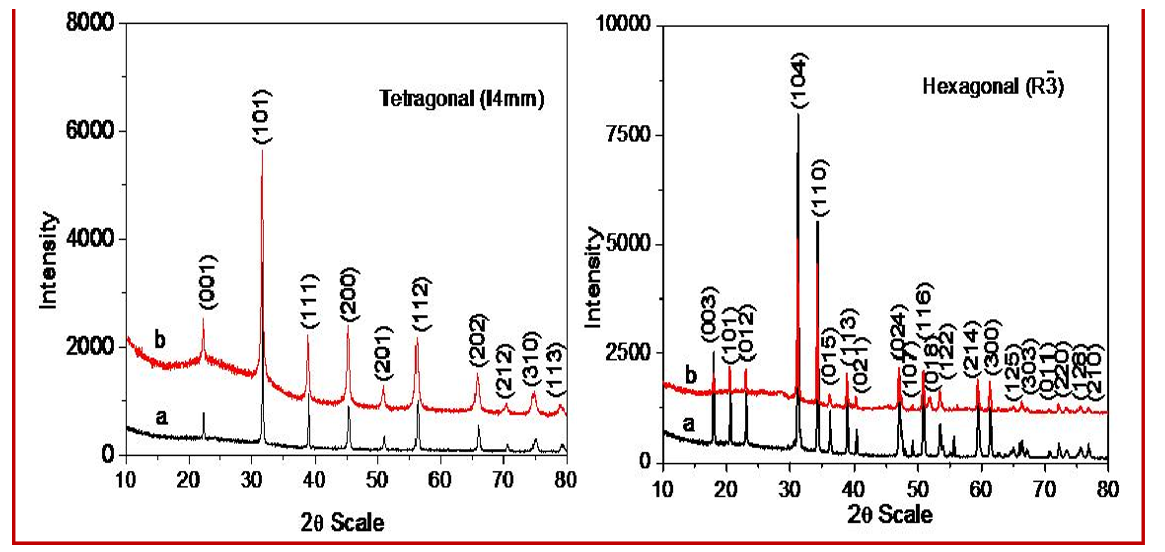
Figure 1: Powder x-ray Diffraction Pattern of (A) CaTiO3(B) SrTiO3(C) BaTiO3(D) CdTiO3
Field emission scanning electron microscopy was carried out to check the morphology of these synthesized titanates. Figure 2 and 3 show the field emission scanning electron micrograph of titanates. Figure 2a shows cube shape morphology of CaTiO3 obtained using TiO2 whereas in case of titanium isopropoxide (Figure 2b), rectangle hollow shape morphology was obtained. This result clearly indicates that with change in source phase from solid (TiO2) to liquid (titanium isopropoxide) the morphology was changed. This hollow morphology type’s materials are very interesting for various applications such as drug delivery and catalysis [47-49]. The SrTiO3 (Figure 2c and 2d) the particles are spherical in nature having two different sizes for the two different titanium sources. For the titanium isopropoxide source, the particles are smaller as compared to titanium dioxide. In the case of TiO2 source some of these nanoparticles (30 nm size) are aggregated and form cube shaped structures. The BaTiO3 nanoparticles size distribution (Figure 3b) is very uniform in case of titanium isopropoxideas Ti source and it is found to be very small in size (further confirm by TEM) as compared to TiO2 sources. The BaTiO3 obtained from TiO2 (Figure 3a) shows the formation of mixed morphology i.e. spherical and cube. For CdTiO3, sheet like morphology was obtained in both the cases. The size of sheet is smaller and broken in case of liquid phase sources. These sheets like structures are very useful for the formation of 2-D materials for practical applications.


Figure 2: FESEM Micrograph of (a, b) CaTiO3 (c, d) SrTiO3 using TiO2 and Ttitanium Isopropoxide as Ti Source Respectively

Figure 3: FESEM Micrograph of (a, b) BaTiO3 (c, d) CdTiO3 using TiO2 and Titanium Isopropoxide as Ti Source Respectively
Transmission electron microscopy (Figure 4) was carried out to confirm the particle size of SrTiO3 and BaTiO3 nanoparticles. The SrTiO3 nanoparticles (Figure 4a and 4b) are spherical in nature having average particles size of 30 nm and 12 nm for titanium dioxide and titanium isopropoxide respectively. There are very few reports of SrTiO3 having particles of size less than 15 nm but none of the reports claim synthesis at gram scale level [50, 51]. The BaTiO3 nanoparticles are uniformly distributed and spherical in nature in case of titanium isopropoxide as titanium sources (Figure 4d). The average diameter of the particles was found to be 5 nm. In case of metal titanates obtained from TiO2 source showed mixed distribution of particle size (Figure 4c). There are only some reports in literature of BaTiO3 of such small size nanoparticles however the synthesis method not showed a general protocol [42- 46, 51].

Figure 4: TEM Micrograph of (a, b) SrTiO3 (c, d) BaTiO3 using TiO2 and Titanium Isopropoxide as Ti Source Respectively
N2 adsorption-desorption measurement was carried out for surface area measurements of the nanoparticles (Table 1). It was observed that as titanium source changed from solid phase (TiO2) to liquid phase (titanium isopropoxide) the surface area of the titanates enhanced along with decrease in particle size. In case of CaTiO3 and BaTiO3, the surface area is more enhanced due to the hollow shape particles (CaTiO3) and small in BaTiO3 in case of titanium isopropoxide as Ti sources. Figure 5 shows the diffuse reflectance spectra of the all the titanates. It was observed that as titanium source changed from TiO2 to titanium isopropoxide the band gap of the titanates increased due to decrease in particle size. All the titanates have band gap in the UV and close to the reported value. The optical properties (band gap) of nanomaterials are size and morphology dependent [52, 53]. For several cases, it is observed that a change in size and morphology can alter the band gap. The band gap of all the titanates is given in Table 1.
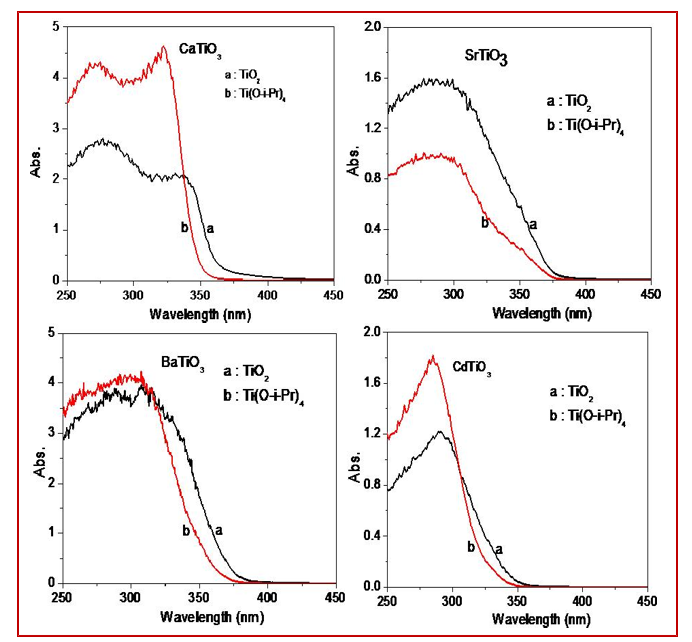
Figure 5: Diffuse Reflectance Spectra of(A) CaTiO3(B) SrTiO3(C) BaTiO3(D) CdTiO3
|
S. No. |
Compound |
Ti Source |
Surface area (g/m2) |
Band Gap (eV) |
|
1 |
CaTiO3 |
TiO2 |
49 |
3.4 |
|
Ti(O-i-Pr)4 |
152 |
3.5 |
||
|
2 |
SrTiO3 |
TiO2 |
55 |
3.3 |
|
Ti(O-i-Pr)4 |
64 |
3.35 |
|
3 |
BaTiO3 |
TiO2 |
58 |
3.3 |
|
Ti(O-i-Pr)4 |
184 |
3.4 |
||
|
4 |
CdTiO3 |
TiO2 |
67 |
3.6 |
|
Ti(O-i-Pr)4 |
95 |
3.7 |
Table 1: Surface Area and Band Gap of Synthesized Titanates
Conclusions
We report a general and cost-effective one-step hydrothermal protocol for the low-temperature synthesis of CaTiO3, SrTiO3, BaTiO3, and CdTiO3 nanoparticles. The morphology and particle size are strongly influenced by the titanium precursor, with liquid- phase titanium isopropoxide producing significantly smaller and more uniformly distributed nanoparticles than TiO3; in particular, BaTiO3 nanoparticles as small as ~5 nm were obtained. The size and morphology can be further tuned by varying hydrothermal parameters such as reaction time, temperature, base, and reactant concentration. The successful synthesis of ultra-small titanate nanoparticles at low temperatures and in large quantities demonstrates the scalability of this approach and highlights its potential for commercial production of nanostructured metal titanates.
Acknowledgements
PKP and BK thanks ANRF for providing support through the grant no SUR/2022/004717. The authors also thank IIT Delhi India for some of reaction and characterization facility.
Author Contributions
Pramendra Kumar Pandey and Revati Raman Chaubey: Synthesis and draft writing, Gyandeshwar Kumar Rao, Suman Srivastava and Arvind Kumar Singh: Characterization, Bharat Kumar: Concept, synthesis modification, final draft writing and final submission. All the authors reviewed the manuscript.
Funding
Funding is not applicable
Declarations
Clinical Trial
Clinical trial is not applicable in the manuscript.
Consent to Publish Declaration
Not applicable
Ethics and Consent to Participate Declarations:
Not applicable
Competing Interests:
The authors declare no competing interests.
Data Availability
The authors declare that the data supporting the findings of this study are available within the paper file. Should any raw data files be needed in another format they are available from the corresponding author upon reasonable request.
References
- He, Y. B., Li, G. R., Wang, Z. L., Su, C. Y., & Tong, Y. X. (2011).Single-crystal ZnO nanorod/amorphous and nanoporous metal oxide shell composites: Controllable electrochemical synthesis and enhanced supercapacitor performances. Energy & Environmental Science, 4(4), 1288-1292.
- Merchan-Merchan, W., Saveliev, A. V., & Taylor, A. M. (2008). High rate flame synthesis of highly crystalline iron oxide nanorods. Nanotechnology, 19(12), 125605.
- Zhang, S., Shan, J., Zhu, Y., Nguyen, L., Huang, W., Yoshida, H., ... & Tao, F. (2013). Restructuring transition metal oxide nanorods for 100% selectivity in reduction of nitric oxide with carbon monoxide. Nano letters, 13(7), 3310-3314.
- Wu, Y., Xiang, J., Yang, C., Lu, W., & Lieber, C. M. (2004). Single-crystal metallic nanowires and metal/semiconductor nanowire heterostructures. Nature, 430(6995), 61-65.
- Okada, K., Ricco, R., Tokudome, Y., Styles, M. J., Hill, A. J., Takahashi, M., & Falcaro, P. (2014). Copper conversion into Cu (OH) 2 nanotubes for positioning Cu3 (BTC) 2 MOF crystals: controlling the growth on flat plates, 3D architectures, and as patterns. Advanced functional materials, 24(14), 1969- 1977.
- Meyer, B., Padilla, J., & Vanderbilt, D. (1999). Theory of PbTiO3, BaTiO3, and SrTiO3 surfaces. Faraday Discussions, 114, 395-405.
- Lebedev, A. I. (2009). Ab initio calculations of phonon spectra in A TiO3 perovskite crystals (A= Ca, Sr, Ba, Ra, Cd, Zn, Mg, Ge, Sn, Pb). Physics of the solid state, 51(2), 362-372.
- Demirors, A. F., & Imhof, A. (2009). Batio3, srtio3, catio3, and ba x sr1− x tio3 particles: A general approach for monodisperse colloidal perovskites. Chemistry of materials, 21(13), 3002- 3007.
- Im, B., Jun, H., Lee, K. H., & Lee, J. S. (2011).Fabrication of nanoporous MTiO 3 (M= Pb, Ba, Sr) perovskite array films with unprecedented high structural regularity. CrystEngComm, 13(24), 7212-7215.
- Deng, H., Qiu, Y., & Yang, S. (2009). General surfactant- free synthesis of MTiO 3 (M= Ba, Sr, Pb) perovskite nanostrips. Journal of Materials Chemistry, 19(7), 976-982.
- Lin, Z. H., Yang, Y., Wu, J. M., Liu, Y., Zhang, F., &Wang, Z. L. (2012). BaTiO3 nanotubes-based flexible and transparent nanogenerators. The journal of physical chemistry letters, 3(23), 3599-3604.
- Lin, M. F., Thakur, V. K., Tan, E. J., & Lee, P. S. (2011). Dopant induced hollow BaTiO 3 nanostructures for application in high performance capacitors. Journal of MaterialsChemistry, 21(41), 16500-16504.
- Yasui, K., & Kato, K. (2012). Dipole–dipole interaction model for oriented attachment of BaTiO3 nanocrystals: a route to mesocrystal formation. The Journal of Physical Chemistry C, 116(1), 319-324.
- Qin, S., Liu, D., Zuo, Z., Sang, Y., Zhang, X., Zheng, F., ... & Xu, X. G. (2010). UV-irradiation-enhanced ferromagnetism in BaTiO3. The Journal of Physical Chemistry Letters, 1(1), 238-241.
- Yang, J., Zhang, J., Liang, C., Wang, M., Zhao, P., Liu, M.,... & Che, R. (2013). Ultrathin BaTiO3 nanowires with high aspect ratio: a simple one-step hydrothermal synthesis and their strong microwave absorption. ACS applied materials & interfaces, 5(15), 7146-7151.
- Da Silva, L. F., Avansi, W., Andrés, J., Ribeiro, C., Moreira,M. L., Longo, E., & Mastelaro, V. R. (2013). Long-range and short-range structures of cube-like shape SrTiO 3 powders: microwave-assisted hydrothermal synthesis and photocatalytic activity. Physical Chemistry Chemical Physics, 15(29), 12386-12393.
- Zheng, Z., Huang, B., Qin, X., Zhang, X., & Dai, Y. (2011). Facile synthesis of SrTiO3 hollow microspheres built as assembly of nanocubes and their associated photocatalytic activity. Journal of colloid and interface science, 358(1), 68- 72.
- Souza, A. E., Santos, G. T. A., Barra, B. C., Macedo Jr, W.D., Teixeira, S. R., Santos, C. M., ... & Longo, E. (2012). Photoluminescence of SrTiO3: influence of particle size and morphology. Crystal growth & design, 12(11), 5671-5679.
- Hu, Y., Tan, O. K., Pan, J. S., & Yao, X. (2004). A new form of nanosized SrTiO3 material for near-human-body temperature oxygen sensing applications. The Journal of Physical Chemistry B, 108(30), 11214-11218.
- Wang, D., Liu, Y., Hu, H., Zeng, Z., Zhou, F., & Liu, W. (2008). Electrochemical characterization of the solution accessibility of CaTiO3 microstructures and improved biomineralization. The Journal of Physical Chemistry C, 112(41), 16123-16129.
- Li, J., Zhang, Y. C., Wang, T. X., & Zhang, M. (2011).Low temperature synthesis and optical properties of CaTiO3 nanoparticles from Ca (NO3) 2· 4H2O and TiO2 nanocrystals. Materials Letters, 65(11), 1556-1558.
- Ohtsu, N., Sato, K., Yanagawa, A., Saito, K., Imai, Y., Kohgo, T., ... & Hanawa, T. (2007). CaTiO3 coating on titanium for biomaterial application—Optimum thickness and tissue response. Journal of Biomedical Materials Research Part A: An Oficial Journal of The Society for Biomaterials, The Japanese Society for Biomaterials, and The Australian Society for Biomaterials and the Korean Society for Biomaterials, 82(2), 304-315.
- Huang, C. L., & Weng, M. H. (2001). Improved high Q value of MgTiO3-CaTiO3 microwave dielectric ceramics at low sintering temperature. Materials Research Bulletin, 36(15), 2741-2750.
- Phani, A. R., Passacantando, M., & Santucci, S. (2000). Synthesis and characterization of cadmium titanium oxide powders by sol-gel technique. Journal of materials science, 35(21), 5295-5299.
- Imran, Z., Batool, S. S., Jamil, H., Usman, M., Israr-Qadir, M., Shah, S. H., ... & Willander, M. (2013). Excellent humidity sensing properties of cadmium titanate nanofibers. Ceramics International, 39(1), 457-462.
- Z, Imran., M, A. Rafiq., M, Ahmad., K, Rasool., S, S. Batool.,M, M. Hasan. (2013). AIP Adv.3, 032146-032159
- Karunakaran, C., & Vijayabalan, A. (2013). Electrical and optical properties of polyethylene glycol-assisted sol–gel solid state reaction-synthesized nanostructured CdTiO3. Materials science in semiconductor processing, 16(6), 1992-1996.
- Mohammadi, M. R., & Fray, D. J. (2009). Low-temperature perovskite-type cadmium titanate thin films derived from a simple particulate sol–gel process. Acta materialia, 57(4), 1049-1059.
- Hou, R. Z., Ferreira, P., & Vilarinho, P. M. (2011). Fabrication of BaTiO3–Carbon Nanocomposite and Porous BaTiO3. Crystal growth & design, 11(12), 5215-5220.
- Maxim, F., Ferreira, P., Vilarinho, P. M., & Reaney, I. (2008). Hydrothermal synthesis and crystal growth studies of BaTiO3 using Ti nanotube precursors. Crystal Growth and Design, 8(9), 3309-3315.
- Bai, H., & Liu, X. (2013). Low temperature solvothermal synthesis, optical and electric properties of tetragonal phase BaTiO3 nanocrystals using BaCO3 powder. Materials Letters, 100, 1-3.
- Li, H. L., Du, Z. N., Wang, G. L., & Zhang, Y. C. (2010).Low temperature molten salt synthesis of SrTiO3 submicron crystallites and nanocrystals in the eutectic NaCl– KCl. Materials Letters, 64(3), 431-434.
- Yang, S., Kou, H., Wang, H., Cheng, K., & Wang, J. (2010). Preparation and band energetics of transparent nanostructured SrTiO3 film electrodes. The Journal of Physical Chemistry C, 114(2), 815-819.
- Huang, Y. J., Chiu, H. T., & Lee, C. Y. (2009). Growth of CaTiO 3 dendrites and rectangular prisms through a wet chemical method. CrystEngComm, 11(9), 1904-1909.
- Wang, D., Guo, Z., Chen, Y., Hao, J., & Liu, W. (2007).In situ hydrothermal synthesis of nanolamellate CaTiO3 with controllable structures and wettability. Inorganic chemistry, 46(19), 7707-7709.
- Yang, L. Y., Feng, G. P., Wang, T. X., Zhang, J. M., & Lou, T.J. (2011). Low temperature preparation and characterization of CdTiO3 nanoplates. Materials Letters, 65(17-18), 2601- 2603.
- Yang, M., & DiSalvo, F. J. (2012). Template-free synthesis of mesoporous transition metal nitride materials from ternary cadmium transition metal oxides. Chemistry of Materials, 24(22), 4406-4409.
- Wang, Y., Xu, H., Wang, X., Zhang, X., Jia, H., Zhang, L., & Qiu, J. (2006). A general approach to porous crystalline TiO2, SrTiO3, and BaTiO3 spheres. The Journal of Physical Chemistry B, 110(28), 13835-13840.
- Demirors, A. F., & Imhof, A. (2009). A general approach for monodisperse colloidal perovskites, Chemistry of Materials. Chemistry of Materials, 21, 3002-3007.
- Deng, H., Qiu, Y., & Yang, S. (2009). General surfactant- free synthesis of MTiO 3 (M= Ba, Sr, Pb) perovskite nanostrips. Journal of Materials Chemistry, 19(7), 976-982.
- Dong, W., Li, B., Li, Y., Wang, X., An, L., Li, C., ... & Shi, Z.(2011). General approach to well-defined perovskite MTiO3 (M= Ba, Sr, Ca, and Mg) nanostructures. The Journal of Physical Chemistry C, 115(10), 3918-3925.
- Su, K., Nuraje, N., & Yang, N. L. (2007). Open-Bench Method for the Preparation of BaTiO3, SrTiO3, and Ba x Sr1-x TiO3 Nanocrystals at 80° C. Langmuir, 23(23), 11369-11372.
- Qi, J., Li, L., Wang, Y., & Gui, Z. (2004). Preparation of nanoscaled BaTiO3 powders by DSS method near room temperature under normal pressure. Journal of crystal growth, 260(3-4), 551-556.
- Qi, J. Q., Wang, Y., Chen, W. P., Li, L. T., & Chan, H. L.W. (2005). Direct large-scale synthesis of perovskite barium strontium titanate nano-particles from solutions. Journal of solid state chemistry, 178(1), 279-284.
- Beier, C. W., Cuevas, M. A., & Brutchey, R. L. (2008). Roomâ?ÂÂTemperature Synthetic Pathways to Barium Titanate Nanocrystals. Small, 4(12), 2102-2106.
- Qi, J. Q., Peng, T., Hu, Y. M., Sun, L., Wang, Y., Chen, W. P., ... &Chan, H. L. W. (2011). Direct synthesis of ultrafine tetragonalBaTiO3 nanoparticles at room temperature. Nanoscaleresearch letters, 6(1), 466.
- Yang, X., Fu, J., Jin, C., Chen, J., Liang, C., Wu, M., & Zhou,W. (2010). Formation mechanism of CaTiO3 hollow crystals with different microstructures. Journal of the American chemical society, 132(40), 14279-14287.
- Zhang, Q., Li, X., Ren, Z., Han, G., & Mao, C. (2015).Synthesis of CaTiO3 nanofibers with controllable drug-release kinetics. European journal of inorganic chemistry, 2015(27), 4532-4538.
- W, Dong., Q, Bao., X, Gu., G, Zhao., J, Cerac. (2015). Soc. Jpn. 123, 643-648.
- Fujinami, K., Katagiri, K., Kamiya, J., Hamanaka, T., & Koumoto, K. (2010). Sub-10 nm strontium titanate nanocubes highly dispersed in non-polar organic solvents. Nanoscale, 2(10), 2080-2083.
- Niederberger, M., Garnweitner, G., Pinna, N., & Antonietti,M. (2004). Nonaqueous and halide-free route to crystalline BaTiO3, SrTiO3, and (Ba, Sr) TiO3 nanoparticles via a mechanism involving C− C bond formation. Journal of the American Chemical Society, 126(29), 9120-9126.
- Kumar, B., Saha, S., Ganguly, A., & Ganguli, A. K. (2014). A facile low temperature (350 C) synthesis of Cu 2 O nanoparticles and their electrocatalytic and photocatalytic properties. Rsc Advances, 4(23),12043-12049.


