Research Article - (2025) Volume 4, Issue 4
Expression of MicroRNA-133a and MicroRNA-208b in Egyptian STEMI Patients
2Microbiology, Immunology, and Biotechnology Department, German University in Cairo, Cairo, Egypt
Received Date: Nov 04, 2025 / Accepted Date: Dec 01, 2025 / Published Date: Dec 09, 2025
Copyright: ©2025 Sally I. Hassanein, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Hassanein, S. I., Kamal, D. O., Hegazy, A., Abou-Aisha, K., Gad, M. Z. (2025). Expression of MicroRNA-133a and MicroRNA-208b in Egyptian STEMI Patients. J Surg Care, 4(4), 01-10.
Abstract
Background: Myocardial microRNAs (Myo-miRs), such as miR-133 and miR-208, are specific to cardiac muscle development and function and are reported to increase after 14 hours of myocardial infarction. Early diagnostic potential of miRs in the acute episode of MI with ST-segment elevation (STEMI) is unknown. This study aimed to compare the fold changes in the peak time points of miRNAs in STEMI patients and compare them with the conventional markers; troponin I (TnI) and creatine kinase-MB (CK-MB).
Methods: Ten patients with new-onset episodes of STEMI and 10 healthy subjects were recruited for the study. Blood samples were collected following the onset of chest pain at 04, 08, 12, 24 and 48 hours, and the serum was separated to determine the levels of circulating miR-133 and miR-208 via quantitative PCR (qPCR) and the cardiac enzymes TnI and CK-MB.
Results: MiR-133a peaks at 8 hours, and miR-208b peaks at 12 hours from the onset of STEMI chest pain. In general, for both miRs, there was a significant difference between patients and healthy subjects.
Conclusions: Compared with TnI and CK-MB, miR-133a and miR-208b have good early diagnostic specificity during the first 4–12 hours for STEMI.
List Of Abbreviations
CVDs: Cardiovascular Diseases
ACS: Acute Coronary Syndrome
STEMI: Segment Elevation Myocardial Infarction
NSTEMI: Non-ST Segment Elevation Myocardial Infarction
cTn: Cardiac Troponin CK: Creatine Kinase
MI: Myocardial Infarction
AMI: Acute Myocardial Infarction
cTnI: Cardiac Troponin I
CK-MB: Creatine Kinase-MB
EGG: Electrocardiogram
IncRNA: Long Noncoding RNA
VF: Ventricular Fibrillation
Introduction
Cardiovascular diseases (CVDs) are among the leading causes of morbidity and mortality worldwide, including Egypt. Recent reports indicate that Egypt is an Eastern Mediterranean region with a high burden of CVD [1]. Acute coronary syndrome results from an inadequate supply of blood flow from the coronary arteries to the heart. Myocardial infarction (MI), which results from this shortage, can be classified into two types: ST segment elevation myocardial infarction (STEMI), a serious form of heart attack in which the coronary artery is completely blocked, resulting in cardiac injury and necrosis, and non-ST segment elevation myocardial infarction (NSTEMI), which results in less damage to myocardial tissue. Circulating biomarkers are essential for the diagnosis, prognosis, monitoring and treatment of MI patients along with electrocardiogram, echocardiography and coronary angiography [2].
Cardiac enzymes are useful for the diagnosis and prognosis of MI patients and act as conventional circulating biomarkers. Cardiac troponin (cTn) is currently considered the biomarker of choice for the diagnosis of MI because it is the most sensitive and specific available biochemical marker of myocardial necrosis [3]. Serum troponins are proteins that regulate muscle contraction. Two isoforms of these proteins, T and I, are specific to the myocardium. Cardiac troponin is expressed in both skeletal and cardiac myocytes. The diagnostic sensitivity of troponins is highly reliable at 12 h from the onset of acute MI episodes [4]. However, in the early hours following MI, myoglobin is more sensitive [3]. Creatine kinase (CK) is an enzyme that catalyzes the reversible transformation of creatine and ATP to creatine phosphate and ADP and is widely used for the diagnosis and prognosis of MI [4].
In humans, fewer than 2% of genes can be transcribed into messenger RNA, which encodes proteins. Most other genes encode noncoding transcripts. These RNAs can be divided into short noncoding RNAs (< 200 bp), which include microRNAs (miRNAs) and circular RNAs, and long noncoding RNAs (lncRNAs, > 200 bp). Currently, more than 2000 human miRNAs have been identified, which seem to regulate approximately 60% of human genes [5]. MiRNAs regulate cardiac development, remodeling and regeneration, endothelial function, vasculogenesis and neoangiogenesis through a variety of pathways [6]. In the blood, circulating extracellular miRNAs are packed in vesicles and are protected from endogenous ribonucleases to maintain their stability. Circulating miRNAs in blood can be detected as disease biomarkers. They are also suggested to be used as mediators of cellâ??cell communication [6].
MiRNAs that bind mRNA at the 3’-UTR prevent translation or promote degradation of the mRNA, thus negatively regulating gene expression at the posttranscriptional level [7]. MiRNAs are responsible for the formation of certain cellular signaling/ communication proteins. It is believed that acute onset of MI is associated with the detection of certain kinds of miRNAs that could be novel diagnostic markers for MI. MiR-133 inhibits the progression of cardiac hypertrophy and cardiac fibrosis, promoting the reprogramming of cardiac fibroblasts. Three miR-133 genes have been identified in the human genome, namely, miR-133a-1, miR-133a-2 and miR-133b, which are located on chromosomes 18, 20, and 6, respectively. MiR133a overexpression protects cardiac progenitor cells from apoptosis by targeting proapoptotic genes and inhibiting hypertrophy. Interestingly, this miRNA alleviates arrhythmia by regulating the expression of several ion channels. Moreover, it aggravates electrical activity disorders, causing atrial fibrillation (AF) and sudden death [8].
The diagnostic and prognostic value of serum miRNA133a was evaluated in stable coronary artery disease (CAD) and, particularly in acute coronary events, where miR-133 levels decrease in the myocardium and increase in the circulation proportionally to the extent of the infarcted area. The value of the circulating level of miR-133 as a diagnostic and prognostic biomarker in these patients during acute ischemic episodes and after primary revascularization has already been addressed [9].
MiR-133a and miR-1 play significant roles in arrhythmia, MI, diabetic cardiomyopathy, cardiac hypertrophy, cardiomyocyte differentiation and cell reprogramming. MiR-133a- is specifically expressed in heart and skeletal muscles. MiR-133a plays major roles in the development of heart cell differentiation into muscle tissue and in the later stages of cardiac morphogenesis. The levels of miRNAs (miR-1, miR-133a, amiR-499 and miR208) in the ischemic myocardium after MI are increased [10].
Many preclinical studies have shown that some miRNAs, such as miR-1, miR-133a/b and miR-208, are involved in heart development, regulate the heart and are dysregulated in hypertrophic and failing hearts and some CVDs, including MI. MiR-133 was recently shown to play a role in cardiovascular regenerative medicine by directing tissue repair and cellular differentiation. It was demonstrated that miR-133a is released into the blood from the injured myocardium in the early phase of AMI [11-13].
MiR-208 is one of the most important cardiac-enriched miRNAs that plays a crucial role in cardiovascular health and diseases. MiR-208 has two subfamily members, miR-208a and miR-208b, both encoded within an intron of the α-cardiac muscle myosin heavy chain gene (α-MHC, MYH6), which is required for cardiomyocyte hypertrophy and fibrosis. Expression of introns of the β-cardiac myosin heavy chain gene (β-MHC, MYH7) occurs in response to stress and hypothyroidism. Both of these are located on human chromosome 14 [11]. MiR-1, miR-133 and miR-208 have been found to be cardiac muscle-specific and thus have been called myo-miRs [12]. In this study, we determined the levels of circulating miR-133a and 208b in STEMI patients at different time intervals (4, 8, 12, 24, and 48 h) from the onset of infarction and compared them with conventional AMI biomarkers such as cTnI and CK-MB.
Methods
Subjects
Ten STEMI patients aged 35–60 years were recruited from the Cardiovascular Emergency Department of Ain-Shams University Hospital, Cairo. Patients with a history of previous treatment for MI, chronic diseases, or renal diseases were excluded from the study. Ten healthy age and sex-matched participants were also recruited. Informed consent was obtained from each subject before admission. Blood samples were collected following the onset of chest pain at 04, 08, 12, 24 and 48 hours, and the serum was separated for analysis of circulating miRNAs and cardiac enzymes. Blood samples were processed within 1 h of collection via two-step centrifugation. The supernatant was transferred to RNase/DNase-free cryotubes and stored at - 80°c. The serum characteristics of the subjects are displayed in Table 1.
Total RNA Isolation
Qiagen miRNeasy Serum/Serum Kit purification of total RNA was used to determine the levels of miRNA133a and miRNA208b in the serum samples. A C. elegans miR39 was used as a spike-in control for normalization and was injected into serum samples within the steps of RNA isolation to be used as an internal control. The purified RNA was then reverse transcribed via customized primers for both miRNAs via a TaqMan reverse transcriptase kit (Applied Biosystems # 4366596) and a TaqMan miRNA assay (Applied Biosystems cat. no. 4427975; assay IDs: 000200, 002246, and 002290 for the miR-39 mimic, miR-133a, and miR- 208b, respectively) according to the manufacturer’s protocol. The thermal profile for the reverse transcription reaction was 30 min at 16°C, 30 min at 42°c, and 5 min at 85°C. qRTâ??PCR was performed via Taqman master mix II (no UNG; cat. no. 4440043), and the probes were provided in the same kit according to the manufacturer’s protocol. The reactions were incubated at 95°c for 10 min, followed by 40 cycles of 95°c for 15 s and 60°c for 60 s. For each sample, a normalization factor was calculated by subtracting the mean spike from the median value of all the mean Cts in all samples. The normalization factor was then added to the raw Ct values obtained for each assay to obtain median-normalized Cts. Delta Cts were calculated by subtracting the mean Ct of the control samples from the mean Ct of the patient samples. The fold change was determined via the 2(−ΔCt) method.
CK-MB and cTnI Analysis
DRG® Troponin I Elisa (cTnI) (EIA-2952) and DRG® Creatine Kinase (MB-Isoform) (EIA-4112) kits were used for the quantitative determination of cTnI and CK-MB in human serum samples, respectively.
Data Analysis
The data collected were categorized into two groups: miRNAs and conventional blood biomarkers. The values are presented as the means ± SEMs. Separate tests were performed for each category. Receiver operating characteristic (ROC) curves and areas under the curve (AUCs) were determined to evaluate the associations between the two groups. ANOVA was employed to determine the statistical significance of the variables. Analysis of the data was performed via GraphPad Prism 9 software (GraphPad Software Inc., CA, USA).
Results
Serum Levels of Circulating miR-133a and miR-208b Over 48 Hours in STEMI Patients
Using median-normalized data, circulating miR-133a and miR- 208b showed similar time point-dependent concentration profiles. The miR-133a levels peaked after 8 hours and then started to decrease to reach low levels near the control levels after 48 hours, which were 2.65-fold higher than those in the controls. The miR- 208b levels peaked after 12 hours and remained high in the serum after 48 hours and were 26-fold higher in patients than in the controls (Figure 1 A and B)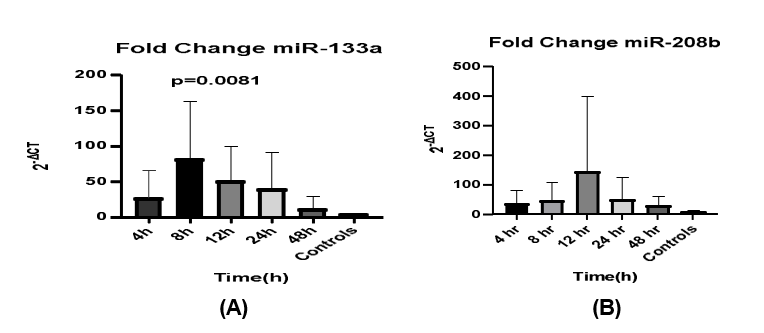
Figure 1: miR-133a (A) and miR-208b (B) in Serum Samples Collected from STEMI Patients (n = 10) at Different Time Intervals After the Onset of the Infarct. Error Bars Represent Mean ± SEM (n = 10). P Values Represent the Significant Difference from Controls
Although we initially attempted to generate receiver operating characteristic (ROC) curves to evaluate the limited sample size, these analyses were statistically unreliable. Consequently, ROC curves and associated metrics were excluded from the results.
Serum Cardiac Troponin I Levels in STEMI Patients
The cTnI levels measured after 4, 8, 12, 24, and 48 h of MI were 2.4, 12, 15, 8.8 and 4.1-fold greater than average control levels, respectively (mean ± SEM, 8.46 ± 2.35) (Figure 2).
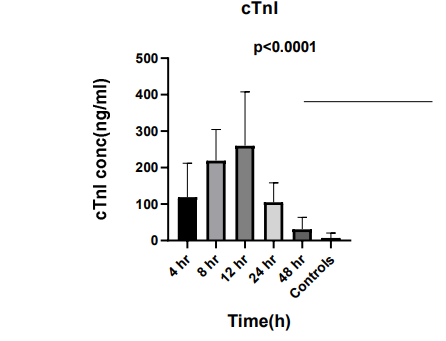
Figure 2: cTnI Serum Concentration (ng/ml) Measured at Different Time Points. Error Bars Represent Mean ± SEM. P Values Represent the Significant Difference from Controls
Serum CK-MB Levels Over 48 Hours in STEMI Patients
The CK-MB concentration also gradually increased at 4, 8, 12, 24 and 48 hours, with 21.7, 39.7, 46.1, 14.3- and 5.1-fold increases over average control level (25.36 ± 7.69), respectively (Figure 3).
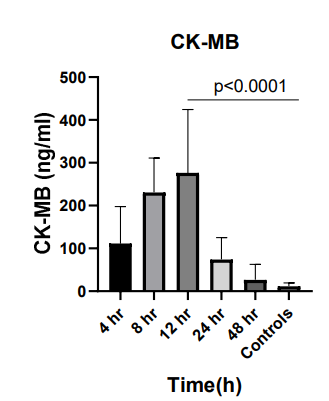
Figure 3: CK-MB Concentration (ng/ml) Measured at Different Time Points. Error Bars Represent Mean ± SEM. P Values Represent the Significant Difference from Controls
Correlation Analysis
We examined the potential correlations between circulating miRNAs (miR-133a and miR-208b) and conventional cardiac biomarkers, specifically cardiac troponin I (cTnI) and creatine kinase-MB (CK-MB), in STEMI patients.
Although a high correlation coefficient was observed between miR-133a and cTnI (R = 0.926, R² = 0.858), this association did not reach statistical significance (P = 0.235) due to the small sample size (Figure 4a). Similarly, the correlation between miR- 133a and CK-MB was weak and non-significant (R = 0.424, R² = 0.18, P = 0.47; Figure 4b).
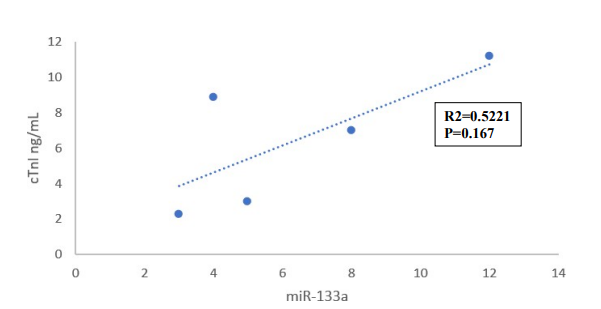
Figure 4a: Correlation Between Fold Change in Serum Levels of miR-133a and cTnI
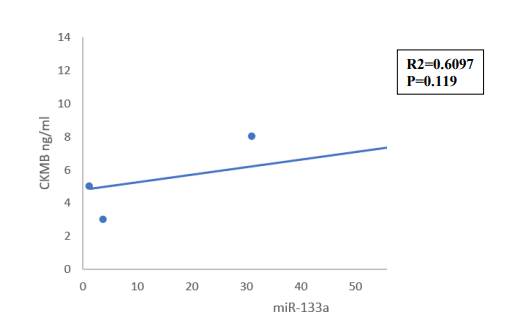
Figure 4b: Correlation Between fold Change in Serum Levels of miR-133a and CK-MB
In the case of miR-208b, there was also no statistically significant correlation with either cTnI or CK-MB (Figure 5a&5b). Additionally, while cTnI and CK-MB themselves demonstrated a moderately positive correlation (R = 0.732), this too was not statistically significant (Figure 6).
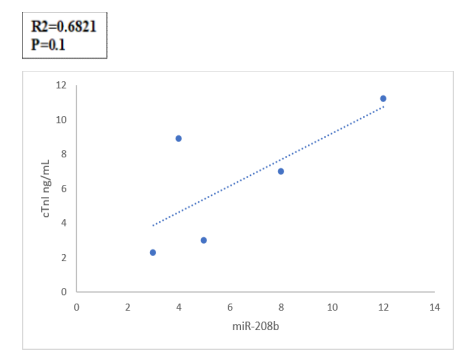
Figure 5a: Correlation Between fold Change in Serum Levels of miR-208b and cTnI
Figure 5b: Correlation Between fold Change in Serum Levels of miR-208b and CK-MB
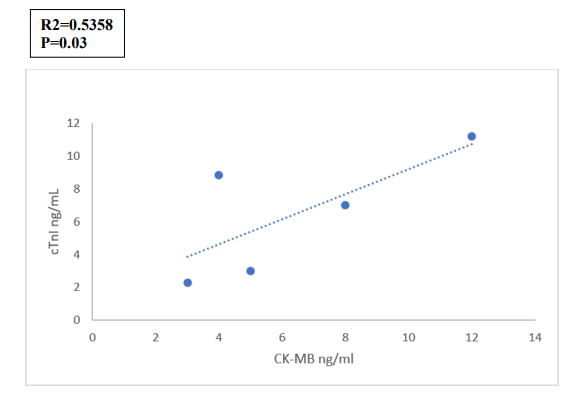
Figure 6: Correlation Between Fold Change in Serum Levels of CK-MB and cTnI
Overall, these findings suggest that although circulating miR- 133a and miR-208b exhibit distinct expression kinetics following STEMI, their levels do not significantly correlate with traditional cardiac biomarkers in this cohort.
Discussion
In this study, we investigated the temporal expression patterns of circulating miR-133a and miR-208b in patients with ST-elevation myocardial infarction (STEMI) compared to healthy controls, aiming to evaluate their potential utility as early biomarkers for acute myocardial infarction (AMI). Our findings revealed a distinct kinetic profile for each miRNA, with miR-133a levels peaking approximately 8 hours after symptom onset, whereas miR-208b reached its peak at around 12 hours. Both markers subsequently declined to near-baseline levels within 48 hours following clinical intervention. These results provide further evidence supporting the role of circulating miRNAs in the early detection of myocardial injury. The early rise of miR-133a observed in this study is consistent with previous reports suggesting its utility as a biomarker detectable before conventional cardiac markers, such as cardiac troponin I (cTnI) and creatine kinase-MB (CK- MB), become elevated. Wexler et al. performed a meta-analysis demonstrating that miR-133a levels significantly increase within the first few hours following AMI onset, potentially offering an earlier diagnostic window than traditional biomarkers [13]. Similarly, Widera et al. and Kuwabara et al. reported elevated levels of miR-133a in patients with acute coronary syndromes (ACS), further reinforcing its association with cardiac injury [14,15]. In contrast, the delayed peak of miR-208b in our study mirrors findings from previous investigations, which reported significant elevations in miR-208b within 12 to 24 hours of infarction [16,17]. Although miR-208b levels were markedly increased in our cohort, their later peak and variability may limit their utility as standalone early diagnostic markers.
Interestingly, in our analysis, neither miR-133a nor miR-208b demonstrated a consistent or statistically significant correlation with conventional markers such as cTnI or CK-MB. While previous studies have reported such associations, our results suggest that the relationship between circulating miRNAs and established biomarkers may be more complex and potentially influenced by patient heterogeneity, sampling timing, or other biological variables. This underscores the need for cautious interpretation and further validation in larger, well-controlled studies. The potential of circulating miRNAs to enhance early diagnosis of AMI remains compelling given the known limitations of traditional biomarkers. Cardiac troponins, while highly specific, typically require several hours post-injury to reach detectable levels. This delay can hinder timely intervention, which is crucial for minimizing myocardial damage and improving patient outcomes. Our findings, alongside those from prior studies, highlight the promise of miRNAs as complementary biomarkers that could facilitate earlier detection and treatment [18,19].
Despite these encouraging results, several limitations must be acknowledged. The most significant is our study’s relatively small sample size 10 STEMI patients and 10 healthy controls. Although the stringent inclusion criteria, including the requirement for five sampling time points per patient, ensured data quality and consistency, they inevitably restricted the overall cohort size. This limitation precluded the application of certain statistical analyses, such as receiver operating characteristic (ROC) curve generation, which requires larger sample populations to ensure robust and reliable interpretation. While we attempted to perform ROC analysis, the small cohort size rendered the results inconclusive and, therefore, unsuitable for inclusion. Future studies with larger patient populations are essential to validate these preliminary findings and to perform comprehensive statistical assessments, including sensitivity, specificity, and predictive values.
The broader landscape of miRNA research in cardiovascular disease highlights both the potential and challenges associated with these biomarkers. Several studies have demonstrated the diagnostic value of miRNAs, including miR-133a, miR-208b, and others such as miR-1 and miR-499, in various cardiac conditions [10,20,21]. However, variability across studies remains a critical concern. Differences in patient populations, sample collection times, detection methodologies, and confounding factors such as comorbidities and medication use can significantly impact miRNA levels. For instance, comorbid conditions like chronic kidney disease can elevate troponin levels independent of cardiac injury, potentially confounding diagnostic interpretations [22]. Similarly, miRNA levels may be influenced by systemic inflammation, renal function, and other non-cardiac factors, necessitating cautious interpretation and highlighting the need for multi-marker approaches.
In this context, the integration of miRNA profiling with conventional biomarkers and clinical parameters holds promise for improving diagnostic accuracy. Combining miRNAs with troponins, natriuretic peptides, and imaging modalities could create a more comprehensive diagnostic framework, capable of identifying AMI at earlier stages and distinguishing between different subtypes, such as STEMI and non-STEMI (NSTEMI). Moreover, miRNA panels, rather than individual markers, may offer enhanced sensitivity and specificity, as suggested by Wang et al., who demonstrated that combined miRNA signatures outperform single markers in diagnostic performance [18]. Beyond diagnosis, the prognostic potential of circulating miRNAs is an emerging area of interest. Several studies have suggested that miRNA levels correlate with infarct size, left ventricular remodeling, and clinical outcomes, including mortality [23,24]. In our study, while the small sample size limited our ability to assess prognostic implications, the observed expression patterns of miR-133a and miR-208b warrant further exploration in larger, longitudinal cohorts to evaluate their role in risk stratification and therapeutic monitoring.
Additionally, the mechanistic role of miRNAs in cardiac pathophysiology presents intriguing therapeutic possibilities. For example, miRNAs are involved in regulating apoptosis, fibrosis, and angiogenesis in the myocardium, making them potential targets for novel interventions aimed at mitigating ischemia/reperfusion injury and promoting cardiac repair [25,26]. The role of miR 221/222 in vascular homeostasis and myocardial remodeling, as highlighted by Askin, exemplifies the complex interplay between miRNAs and cardiovascular health, offering potential avenues for therapeutic modulation [26]. However, translating these insights into clinical practice requires further research to elucidate miRNA targets, refine delivery systems, and assess safety and efficacy in human populations. Finally, the role of extracellular vesicles, such as exosomes, in miRNA transport and signaling is an area of active investigation. These vesicles may facilitate targeted delivery of miRNAs to specific tissues, enhancing their diagnostic and therapeutic utility [21,25]. Understanding the dynamics of miRNA packaging, release, and uptake could unlock new strategies for non-invasive diagnostics and precision medicine.
In conclusion, our study reinforces the potential utility of circulating miR-133a as an early biomarker for AMI, with miR- 208b providing complementary kinetic information. However, the lack of significant correlation between these miRNAs and conventional markers in our data highlights the complexity of biomarker interplay and the need for further research. Future large- scale, multicenter studies with standardized protocols are essential to validate these findings and determine the clinical applicability of miRNA-based assays for AMI diagnosis and prognosis. The integration of miRNA profiling with established biomarkers and advanced imaging techniques may ultimately lead to more accurate and timely identification of myocardial infarction, improving clinical decision-making and patient outcomes.
Acknowledgements
Limitations No follow-up records are available for the recruited patients. Author contributions Concept—SIH, KA and MZG; design—SIH; supervision—KA, SIH and MZG; funding—Dina Osama and Ahmed Hegazy. ; materials—Dina Osama and Ahmed Hegazy; data collection and/or processing—Dina Osama and Ahmed Hegazy; analysis and/or interpretation—Dina Osama and Ahmed Hegazy. SIH and KA. Literature review—SIH, KA and MZG; writing—SIH; critical review—SIH, KA and MZG. Compliance with ethical standards Conflicts of interest. The authors declare that they have no conflicts of interest
Ethical Approval
All the subjects were informed of the nature of the study and thus provided written consent. Data and code abided by the Helsinki declaration. The study was approved by the local ethics committees of both Ain Shams, Faculty of Medicine, and German University in Cairo.
Competiting Interest
We certify that there are no potential conflicts of interest related to this article.
Author Contributions
Concept: SIH, KA and MZG
Study design: KA
Supervision: KA, SIH and MZG
Practical experiments: DOK and AH
Data collection: DOK and AH
Data analysis: DOK, AH. SIH and KA
Literature review: SIH, KA and MZG
Writing: SIH, DOK, AH, KA, MZD
Critical review— SIH, MZG and KA
Funding
No funding grants supported this study.
Dual Publication
Only some figures and results have been reprinted from the Research square
Authorship
I have read the Nature Portfolio journal policies and author responsibilities and submit this manuscript in accordance with those policies.
Data Availability
“All data generated or analyzed during this study are included within the manuscript file".
References
- El-Moselhy, E. A., Mohammed, A. S., Abd El-Aziz, A., Sadek, I., Hagrass, S. A., & Farag, G. A. S. (2018). Coronary artery disease among elderly Egyptian patients:I. Socio-demographic, lifestyle, psychosocial, medical, and biochemical risk factors. American Journal of Gerontology & Geriatrics, 1, 1006.
- Harjola, V. P., Parissis, J., Bauersachs, J., Brunner-La Rocca, H. P., Bueno, H., celutkiene, J., ... & Yilmaz, M. B. (2020). Acute coronary syndromes and acute heart failure: a diagnostic dilemma and high-risk combination. A statement from the Acute Heart Failure Committee of the Heart Failure Association of the European Society of Cardiology. European journal of heart failure, 22(8), 1298-1314.
- Wang, X. Y., Zhang, F., Zhang, C., Zheng, L. R., & Yang, J. (2020). The biomarkers for acute myocardial infarction and heart failure. BioMed Research International.
- Aydin, S., Ugur, K., Aydin, S., Sahin, ÃÂ?°., & Yardim, M. (2019). Biomarkers in acute myocardial infarction: Current perspectives. Vascular Health and Risk Management, 15, 1–10.
- Tornesello, M. L., Faraonio, R., Buonaguro, L., Annunziata, C., Starita, N., Cerasuolo, A., Pezzuto, F., Tornesello, A. L., & Buonaguro, F. M. (2020). The role of microRNAs, long non- coding RNAs, and circular RNAs in cervical cancer. Frontiers in Oncology, 10, 150.
- Condrat, C. E., Thompson, D. C., Barbu, M. G., Bugnar, O. L., Boboc, A., Cretoiu, D., Suciu, N., Cretoiu, S. M., & Voinea, S.C. (2020). miRNAs as biomarkers in disease: Latest findings regarding their role in diagnosis and prognosis. Cells, 9(2), 276.
- Gregorova, J., Vychytilova-Faltejskova, P., & Sevcikova,S. (2021). Epigenetic regulation of microRNA clusters and families during tumor development. Cancers, 13(6), 1333.
- Li, N., Zhou, H., & Tang, Q. (2018). miR-133: A suppressor of cardiac remodeling? Frontiers in Pharmacology, 9, 903.
- D'Alessandra, Y., Devanna, P., & Limana, F. (2015). MicroRNA-133: Biomarker and mediator of cardiovascular diseases. In V. R. Preedy & V. B. Patel (Eds.), Biomarkers in cardiovascular disease (pp. 287–313).
- Song, Z., Gao, R., & Yan, B. (2020). Potential roles of microRNA-1 and microRNA-133 in cardiovascular disease. Reviews in Cardiovascular Medicine, 21(1), 57–64.
- X., Wang, Y., & Sun, X., et al. (2020). The functions of microRNA-208 in the heart. Diabetes Research and Clinical Practice, 108004.
- Malizia, A. P., & Wang, D. Z. (2011). MicroRNAs in cardiomyocyte development. Wiley Interdisciplinary Reviews: Systems Biology and Medicine, 3(2), 183–190.
- Wexler, Y., & Nussinovitch, U. (2020). The diagnostic value of miR-133a in ST elevation and non–ST elevation myocardial infarction: A meta-analysis. Cells, 9(4), 793.
- Widera, C., Gupta, S. K., Lorenzen, J. M., Bang, C., Bauersachs, J., Bethmann, K., Kempf, T., Wollert, K. C., & Thum, T. (2011). Diagnostic and prognostic impact of six circulating microRNAs in acute coronary syndrome. Journal of Molecular and Cellular Cardiology, 51(5), 872–875.
- Kuwabara, Y., Ono, K., Horie, T., Nishi, H., Nagao, K., Kinoshita, M., Watanabe, S., Baba, O., Kojima, Y., Shizuta, S., Imai, M., Tamura, T., Kita, T., & Kimura, T. (2011). Increased microRNA-1 and microRNA-133a levels in serum of patients with cardiovascular disease indicate myocardial damage. Circulation: Cardiovascular Genetics, 4(4), 446–454.
- Corsten, M. F., Dennert, R., Jochems, S., Kuznetsova, T., Devaux, Y., Hofstra, L., Wagner, D. R., Staessen, J. A., Heymans, S., & Schroen, B. (2010). Circulating microRNA- 208b and microRNA-499 reflect myocardial damage in cardiovascular disease. Circulation: Cardiovascular Genetics, 3(6), 499–506.
- Gidlöf, O., Andersson, P., van der Pals, J., Götberg, M., & Erlinge, D. (2011). Cardiospecific microRNA plasma levels correlate with troponin and cardiac function in patients with ST elevation myocardial infarction. Cardiology, 118(4), 217– 226.
- Wang, B., Li, Y., Hao, X., Yang, J., Han, X., Li, H., Li, T.,Wang, D., Teng, Y., Ma, L., Li, Y., Zhao, M., & Wang, X. (2021). Comparison of the clinical value of miRNAs and conventional biomarkers in AMI: A systematic review. Frontiers in Genetics, 12, 668324.
- He, J. G., Li, S., Wu, X. X., Chen, X. H., Yan, D., Wang, X. J.,& Dang, Z. W. (2024). Circulating miRNA-21 as a diagnostic biomarker for acute coronary syndrome: A systematic review and meta-analysis. Cardiovascular Diagnosis and Therapy, 14(3), 328–339.
- Li, Y. Q., Zhang, M. F., Wen, H. Y., Hu, C. L., Liu, R., Wei,H. Y., Ai, C. M., Wang, G., Liao, X. X., & Li, X. (2013).Comparing the diagnostic values of circulating microRNAs and cardiac troponin T in patients with acute myocardial infarction. Clinics, 68(1), 75–80.
- Han, Z., Zhang, L., Yuan, L., Liu, X., Chen, X., Ye, X., Yang,C., & Yan, Z. (2015). Change of plasma microRNA-208 level in acute myocardial infarction patients and its clinical significance. Annals of Translational Medicine, 3(20), 307.
- Yu, H., Lu, Y., Li, Z., & Wang, Q. (2014). microRNA-133:Expression, function and therapeutic potential in muscle diseases and cancer. Current Drug Targets, 15(9), 817–828.
- Liu, X., Yuan, L., Chen, F., Zhang, L., Chen, X., Yang, C., & Han, Z. (2017). Circulating miR-208b: A potentially sensitive and reliable biomarker for the diagnosis and prognosis of acute myocardial infarction. Clinical Laboratory, 63(1), 101–109.
- Alavi-Moghaddam, M., Chehrazi, M., Alipoor, S. D., Mohammadi, M., Baratloo, A., Mahjoub, M. P., Movasaghi,M., Garssen, J., Adcock, I. M., & Mortaz, E. (2018). A preliminary study of microRNA-208b after acute myocardial infarction: Impact on 6-month survival. Disease Markers, 2018, 2410451.
- Boštjancic, E., Brandner, T., Zidar, N., Glavac, D., & Štajer,D. (2018). Down-regulation of miR-133a/b in patients with myocardial infarction correlates with the presence of ventricular fibrillation. Biomedicine & Pharmacotherapy, 99, 65–71.
- Askin, L., & Tanriverdi, O. (2023). Is the microRNA-221/222 cluster ushering in a new age of cardiovascular diseases? Cor et Vasa, 65(1), 65–67.