Research Article - (2020) Volume 1, Issue 1
Estimation of Furosemide in Pharmaceutical Preparation Samples
Received Date: Nov 10, 2020 / Accepted Date: Nov 17, 2020 / Published Date: Nov 23, 2020
Copyright: ©Copyright: �2020 Nief Rahman Ahmed. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A simple, accurate, precise, rapid, economical and sensitive ultraviolet spectrophotometric method has been developed for the determination of Furosemide in pharmaceutical preparations, which shows maximum absorbance at 228 nm in. Beer’s law was obeyed in the range of 1 -10 μg/ ml, with molar absorptivity of 3.5×104 L.mol-1.cm-1, relative standard deviation of the method was less than 1.4%, and accuracy (average recovery %) was 100 ± 1.0. The method was successfully applied to the determination of Furosemide in some pharmaceutical formulations (tablets, Oral solution and injection) samples. The proposed method was validated by sensitivity and precision which proves suitability for the routine analysis of Furosemide in true samples.
Introduction
Furosemide chemically is [ 4-Chloro-2-[(furan-2-ylmethyl) amino]-5-sulfamoyl benzoic acid] [Figure 1]
C12H11ClN2O5S: 330.74
Furosemide is a potent diuretic with a rapid action. Like the other loop or high-ceiling diuretics it is used in the treatment of oedema associated with heart failure (below), including pulmonary oedema, and with renal and hepatic disorders and may be effective in patients unresponsive to thiazide diuretics. It is also used in high doses in the management of oliguria due to renal failure or insufficiency. Furosemide is also used in the treatment of hypertension, either alone or with other antihypertensive [1]
Furosemide should not be used to treat gestational hypertension because of the maternal hypervolemia associated with this condition, Furosemide acts within 1 hour of oral administration, (after IV peak effect within 30 minutes) diuresis complete within 6 hours [2, 3]. Analytical procedures for the estimation of Furosemide include titrimetric method, various visible spectrophotometric methods, high performance liquid chromatography (HPLC), voltammetry, Potentiometric sensor [4-12]. However, these methods are required expensive or sophisticated instruments and lack sensitivity and simplicity needed for routine analysis. The present method described a simple, economical, accurate, sensitive and reproducible new ultraviolet spectrophotometric method for the determination of Furosemide in pharmaceutical preparations samples (tablets, injections and solutions).
Experimental
Apparatus
Shimadzu UV- 1700 pharma spec (double beam) spectrophotometer with 1.0 cm quartz cells was used for absorption measurement.
Reagents
All chemicals used were of analytical or pharmaceutical grade and the Furosemide standard material was provided from state company for drug industries and medical appliance (HPI) Mosul-Iraq.
Furosemide Stock Solution (500 ppm)
This solution was prepared by dissolving 0.05gm of Furosemide in 100 ml 0.1 N Sodium hydroxide and diluting to 100 ml with 0.1 N Sodium hydroxide.
Furosemide Standard Solution 100ppm
This solution was prepared by diluting 2 ml of stock solution into 100ml by0.1 N Sodium hydroxide in a volumetric flask.
Sodium Hydroxide Solution (0.1N)
This solution was prepared by dissolving 4gm of Sodium hydroxide in 1000 ml of distilled water in calibrated flask.
Estimation of Absorption Maxima
The standard solution of Furosemide (8µg/ml) was scanned in the range of 220-350 nm which shows maxima located at 228 nm Figure 2. Therefore, 228 nm wavelength was selected for the construction of calibration curve.
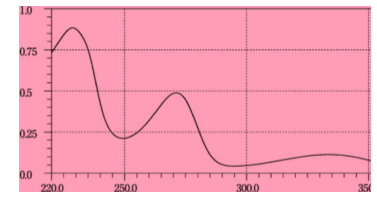
Figure 2:-Absorption spectra of 8 µg/ml Furosemide against blank.
Recommended procedure
From the absorption maxima, calibration curve was prepared in the concentration range of 1-10µg/ml. The absorbance was measured at 228 nm against 0.1 N Sodium hydroxide as a blank. The concentration of the sample solution can be determined by using the calibration curve.
Procedure for pharmaceutical preparations:
Tablets (40mg/tablet)
To minimize a possible variation in the composition of the tablets (containing 40mg of Furosemide / tablet were provided from AL-Hokamaa company for pharmaceutical industries (HPI) Mosul-Iraq). The mixed content of 10 tablets were weighed and grounded, then the powder equivalent to 10 mg of Furosemide in about 70 ml of 0.1N Sodium hydroxide was stirred well for 30 min and then filtered through whatman No. 42 filter paper and the filtrate solution was diluted to 100ml by 0.1N Sodium hydroxide and different volume of this solution was treated as described above under general procedure. The drug content of the sample was calculated by using regression analysis.
Injections (20mg/ampoule)
Ampoule containing 20mg of Furosemide (were provided from the state company of drug industries and medical appliance (NDI) Nineveh - Iraq). The content of 5 ampoules were mixed well in 250ml dried beaker. An aliquots equivalent to 10 mg of Furosemide was transferred into 100ml volumetric flask and diluted up to the mark with, 10 ml of this solution was diluted to 100ml with 0.1N Sodium hydroxide and an aliquot of this solution was treated as described above for recommended procedure.
Syrups (20mg/5ml)
Take a volume of syrups containing 10 mg of Furosemide in to 100 mL volumetric flasks and diluted with 0.1N Sodium hydroxide to the volume, and the amount of Furosemide was calculated by using regression analysis
Result and Discussion
UV visible spectrophotometry is still considered to be a convenient and low cost method for the determination of pharmaceuticals and has been increased considerably in recent years because of their importance in pharmaceutical analysis [13-16]. A new method has been developed for the spectrophotometric determination of Furosemide in pharmaceutical preparations samples was found to be sensitive, simple, accurate and reproducible. Beer s law was obeyed in the concentration range of 1-10 µg/ml. [Figure 3] with correlation coefficient of 0.9988, intercept of 0.0068 and slope of 0.1059. The conditional molar absorptivity was found to be 3.5×104 l/mol.cm.
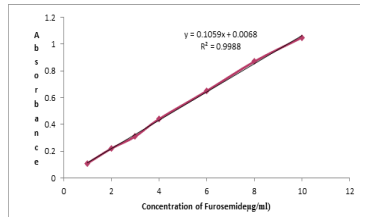
The accuracy and precision of the method, a pure drug solution was analyzed at three different concentrations, each determination being repeated six times. The relative error (%) and relative standard deviation values are summarized in table 1. From table 1 the values of standard deviation were satisfactory and the recovery studies were close to 100%. The RSD% value is less than 1.4 indicative of accuracy of the method.
Table I : Accuracy and precision of the proposed method.
|
Furosemide taken µg/ml)) |
Er (%)a |
RSD(%) |
|
2 |
2.04 |
1.3 |
|
6 |
6.05 |
1.1 |
|
10 |
9.95 |
1.3 |
a: Mean of six determination
The proposed method was compared with other reported UV spectrophotometric methods and found to be superior, high sensitive, more applications (Table 2).
Table 2: Comparison of the existing UV spectrophotometric methods with the proposed method for Furosemide
|
Parameters |
Method 1 |
Method 2 |
Method 3 |
|
Ref |
17 |
18 |
Proposed |
|
λ Max(nm) |
277 |
267 |
228 |
|
Solvents |
Ethanol |
Distilled water |
0.1N Sodium hydroxide |
|
Linear range µg/ml |
5-25 |
6.25-100 |
1-10 |
|
ÃÂ??(l/mol.cm) |
2.55x104 |
1.9x104 |
3.5×104 |
|
Application |
Tablets |
Tablets |
Tablets, Oral solutions and Injections |
Analytical Applications
The proposed method was satisfactorily applied to the determination of Furosemide in its pharmaceutical formulations. The results of the assay of the pharmaceutical formulations revels that there was closed agreement between the results obtained by the proposed method and the label claim. The results were also compared statistically by student t-test and by the variance ratio F-test with those obtained by official method at 95% confidence level [17-19]. The calculated t- and F- values did not exceed the theoretical values indicating that there were no significant differences between the precision of the proposed and official method as cited in Table 3.
Table 3: Determination of Furosemide in pharmaceutical formulations
|
Pharmaceutical formulations(HDI) |
Label amount, mg |
Official method [19] |
Proposed method * |
F- value |
t- value |
|
Lasix tablets |
40mg/tab |
39.9 |
39.8 |
1.03 |
1.15 |
|
Lazine injecting |
20mg/amp |
19.96 |
19.9 |
1.05 |
1.77 |
|
Lasix Syrups |
20mg/5ml |
19.92 |
19.96 |
1.09 |
1.85 |
*Mean value of ten determinations
t values (n=10, at 95% confidence level tabulated value 2.101).
F values (n1-1 and n2-1 =9, at 95% confidence tabulated value 3.18).
Conclusion
The developed method is found to be high sensitive, accurate, simple, precise and economical, and can be used for routine quality control analysis of Furosemide in pure form and pharmaceutical formulations preparations.
Acknowledgements
The author [Dr. Nief Rahman Ahmad] wishes to express gratitude to AL-hokamaa Company for pharmaceutical industries (HPI) Mosul-Iraq, for providing gift samples of Furosemide standard materials and pharmaceutical preparations.
References
- Sweetman SC (2009) editors. Martindale, ‘The Complete Drug Reference. 36th Edition. London: Pharmaceutical Press 2009: 1292.
- British National Formulary (2016) Royal Pharmaceutical Society 70: 221
- Caroline A, Ailean D (2019) The Renal Drug Hand book,5th Edition, CRC press 2019: 496.
- British Pharmacopoeia (2014) H.M. Stationery office,London, UK 1: 1020.
- Nief Rahman Ahmed, Fawas K Ibrahim (2006) ‘Spectrophotometric determination of frusemide in some pharmaceuticals via oxidative coupling reaction. J Educ Sci 18: 1-8.
- Tharpa K, Basavalah K, Vinay KB (2009) Spectrophotometric determination of furosemide in pharmaceuticals using permanganate. Jordan J Chem 4: 387-397.
- Mohauman Mohammad Al-Rufaie (2017) Modern kinetic spectrophotometric procedure for estimation of furosemide drug as bulk form and in pharmaceuticals preparations. Curr Issues Pharm Med Sci 29: 184-189.
- Kaynak MS, Sahin S (2013) Development and validation of a RP-HPLC method for determination of solubility of furosemide. Turk J Pharm Sci 10: 25-34.
- Patel H, Solanki S (2012) Simultaneous estimation of furosemide and spironolactone in combined pharmaceutical dosage form by RP_HPLC. Asian J Pharm Clin Res 5: 195-198.
- Solanki R (2011) Stability indicating RP_HPLC method for simultaneous determination of frusemide and amiloride hydrochloride in tablet dosage form. Int J Adv Pharm Anal 1: 16-23.
- Malode SJ, Abbar JC, Shetti NP, Nandibewoor ST (2012) Voltammetric oxidation and determination of loop diuretic furosemide at a multi-walled carbon nanotubes paste electrode. Electrochim Acta 60: 95-101.
- Santini AO, Pezza HR, Sequinel R, Rufino JL, Pezza L (2009) Potentiometric sensor for furosemide determination in pharmaceuticals, urine, blood serum and bovine milk. J Braz Chem Soc 20: 64-73.
- Nief Rahman Ahmed, Husam Waleed Yaseen (2018) Ultraviolet Estimation of Guaiphenesin in Pharmaceutical Preparations and Environmental Wastewater Samples. Research Journal of Pharmaceutical, Biological and Chemical Sciences 9: 39-45.
- Nief Rahman Ahmad, NM Ali (2006) Spectrophotometricdetermination of metclopromide in some pharmaceutical preparations via oxidative coupling reaction. J Edu Sci 18: 16-23.
- Nief Rahman Ahmed, Sheet NS, Essa MJ (2020) A Sensitive Ultraviolet Spectrophotometric Determination of Tamoxifen Citrate in Pharmaceutical Preparations and Environmental Wastewater Samples. Journal of Medical and Clinical Studies 3: 1-4.
- Nief Rahman Ahmed (2012) ‘Facial visible spectrophotometric determination of metformin hydrochloride in glucosam tablets and industrial waste water: Application to content uniformity testing, Iraq. Journal of Pharmacy 12: 75-85.
- Phalle Supriya, Patel SG, Dhobale SM (2018) Estimation of Frusemide in bulk and tablet formulation by UV spectrophotometric Area under Curve method. Int Res J of Science & Engineering A3: 96-100.
- Safila Naveed, Fatima Qamar, Syeda Zainab (2014) Simple UV spectrophotometric assay of Furosemide. Journal of Innovations in Pharmaceuticals and Biological Sciences 1: 97-101.
- British pharmacopeia (2009) Her Majesty, Stationary Office, London 2009: 8941-8943.


