Review Article - (2020) Volume 5, Issue 1
Ejection Fraction and Espvr: A Study in the Mechanics of Left Ventricular Contraction
Received Date: Mar 31, 2020 / Accepted Date: Apr 06, 2020 / Published Date: Apr 30, 2020
Copyright: ©Rachad Mounir Shoucri. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The end-systolic pressure-volume relation (ESPVR) is the relation between pressure Pm and volume Vm in the heart left ventricle when the myocardium reaches its maximum state of activation during contraction near end-systole. Relations between the ejection fraction (EF), parameters describing the ESPVR and the areas under the ESPVR are derived in this study for a linear model of the ESPVR. An important feature of the model is the inclusion of the active pressure generated by the myocardium during an ejecting contraction (also called isovolumic pressure Piso) in the mathematical expression of the linear ESPVR. Criteria that can help in understanding the problem of heart failure with normal or preserved ejection fraction (HFpEF) are discussed. Applications to clinical data published in the literature are presented, the applications show the consistency of the mathematical formalism used. When ratios of pressures are used, the calculation can be carried out with clinical data measured in a non-invasive way (the ratio of pressures can be calculated). This study shows that the EF is just one index of several indexes that can be derived from the ESPVR for the assessment of the ventricular function, and that using bivariate (or multivariate) analysis of data is superior to univariate analysis for the purpose of classification and segregation between different clinical groups.
Keywords
Ventricular Function, Cardiac Mechanics, Contractility of the Myocardium, Pump Function of the Heart, Heart Failure with Preserved Ejection Fraction, HFpEF, End-Systolic Pressure- Volume Relation, ESPVR, Efficiency of Left Ventricle, Mathematical Physiology.
Introduction
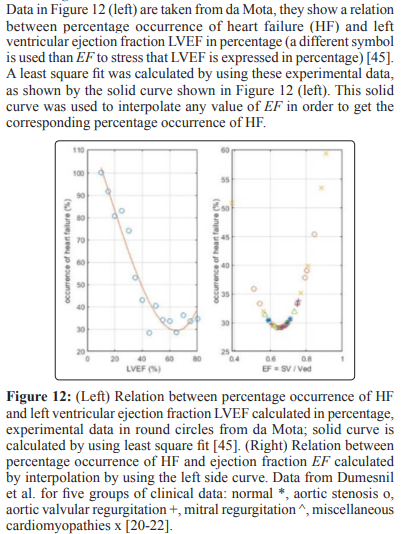
The relation between pressure Pm and volume Vm in the heart left ventricle when the mycocardium reaches its maximum state of activation during the contraction phase near end-systole is known as the end-systolic pressure-volume relation (ESPVR). In this study we assume that Vm Ves the left ventricular volume at end- systole (defined as the smallest left ventricular volume V, when the time derivative dV/dt = 0), Pm is the corresponding left ventricular pressure. Discussion of the properties of the linear ESPVR can be found in several publications [1-18], a review can be found in [5, 6]. Two important new features of the linear model of the ESPVR described in this study are the introduction of the active pressure generated on the inner surface of the myocardium (endocardium) (also called isovolumic pressure Piso by physiologists) in the mathematical formalism describing the ESPVR, and the inclusion of the total area under the ESPVR in the study of its properties [8-18]. The linear model of the ESPVR discussed in this study is a simplifued version of the mathematical formalism for a non-linear model of the ESPVR based on the theory of large elastic deformation of the myocardium. For small variations in Pm and Vm within the physiological range, the linear model is an accepted approximation to the non-linear model. Preliminary results by using the non-linear model of the ESPVR have been published [16-18]. Because those results are at a preliminary stage, this study is confined to the linear model of the ESPVR. The ejection fraction (EF) is defined as EF = SV/Ved, where Ved is the left-ventricular volume at end-diastole (defined as the largest ventricular volume V, when the time derivative dV/dt = 0), the stroke volume SV = Ved – Vm since we have assumed Vm Ves. The EF is widely used in the clinical practice because it can be calculated from non-invasive measurements. One should be careful in using EF that different methods of measurement can give different results [19]. For instance in one observed case, a measurement of EF = 0.67 by M-mode echocardiography was measured 0.54 by radionuclide angiography, the reason is damage in the aortic valve causing regurgitation. The EF is also sensitive to preload, afterload and ventriculo-arterial coupling, and consequently cannot be considered as an independent measure of the contractility of the myocardium. As observed by Dumesnil et al. the EF is influenced by intrinsic myocardial properties as well as by the geometry of the left ventricle, and that cases of aortic stenosis may have reduced longitudinal shortening whereas the EF appears normal [20-23]. The effect of intrinsic properties of the myocardium on its contraction is discussed in [24, 25]. The importance of the longitudinal axis dynamics in prognostic and diagnostic applications has been stressed by several authors, facilitated by the introduction of new techniques of measurement like Doppler tissue imaging or speckle tracking echocardiography [20-23, 26-29]. Particular attention was given to paradoxical low flow/low gradient aortic stenosis with preserved left ventricular EF according to the assumed definition EF > 0.5 [30-35]. It is now recognized that an early detection of signs of heart failure with preserved ejection fraction (HFpEF) or with normal ejection fraction (HFnEF) is an important clinical problem that may allow a better treatment of the underlying causes, delay may lead to complications with poor prognosis [36-39]. Descriptive terms like HFnEF, HFpEF, HFmrEF (mid-range 41% <= EF <= 50%), HFrEF (reduced EF <= 40%) are approximate and arbitrary cut- off values for classification of cases of HF that have overlapping but also dictinct characteristics (another definition of HFpEF is suggested in this study and explained in the discussion section at the end). Attention has also been given recently to pathophysiological mechanisms in HF studies [40-42]. The EF remains an important index used in the assessment of the ventricular function and it continues to be used in conjunction with other indexes in clinical applications. Experimental curves between percentage occurrences of HF and EF have been reported, they indicate that values of EF between 60%-65% are associated with the lowest mortality as discussed in what follows [43-46].
In this study the mathematical expression of the linear ESPVR is first introduced. Then relations between EF, the parameters describing the ESPVR and the areas under the ESPVR are derived and discussed, they give new insight into the mechanics of ventricular contraction. Applications to experimental and clinical data published in the literature shows the consistency of the mathematical formalism used [8-13, 47-56]. The mathematical formalism to be discussed in what follows can be applied to the four chambers of the heart, this study is limited to applications to the left ventricle [57-59].
Mathematical Formalism
The following variables are shown in Figure 1 and Figure 2.

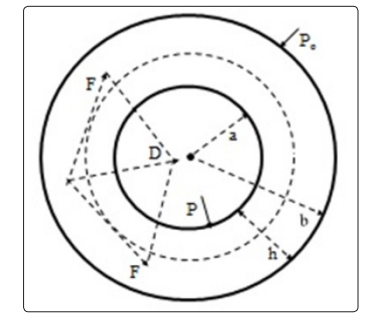
Figure 1: Cross-section of a thick-walled cylinder representing the left ventricle, D = active radial force/unit volume of the myocardium, P = left ventricular pressure, Po = outer pressure (assumed zero) on the epicardium, a = inner radius, b = outer radius, h = b – a = thickness of the myocardium. F = projection on the cross-section of the force generated by the myocardial fiber



Areas Under the Linear ESPVR



Stroke Volume


Maximum Stroke Work




Maximum Efficiency

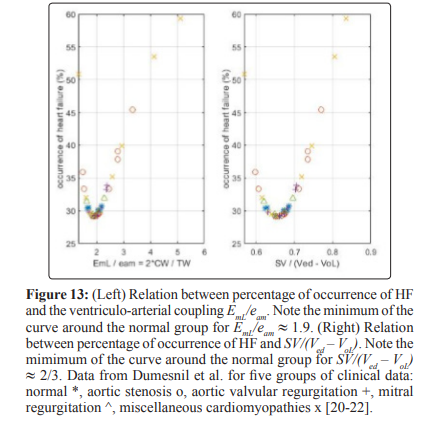
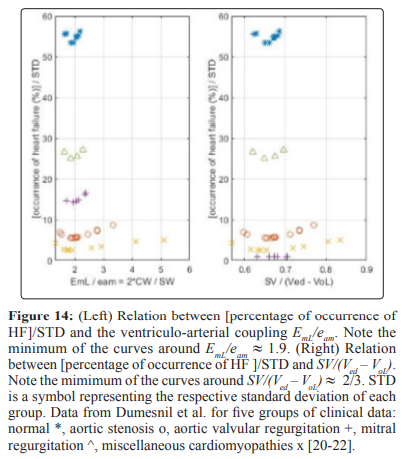
Percentage Occurrence of Heart Failure

Discussion
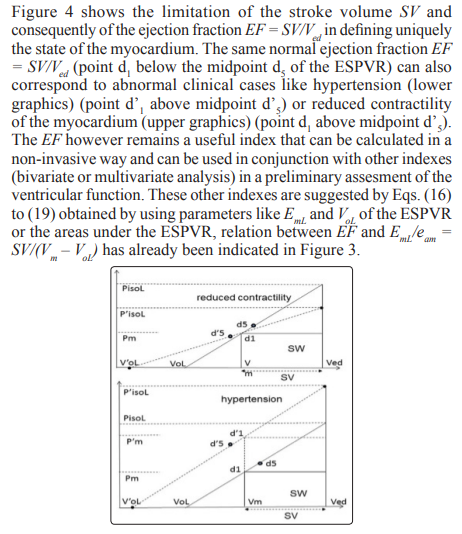
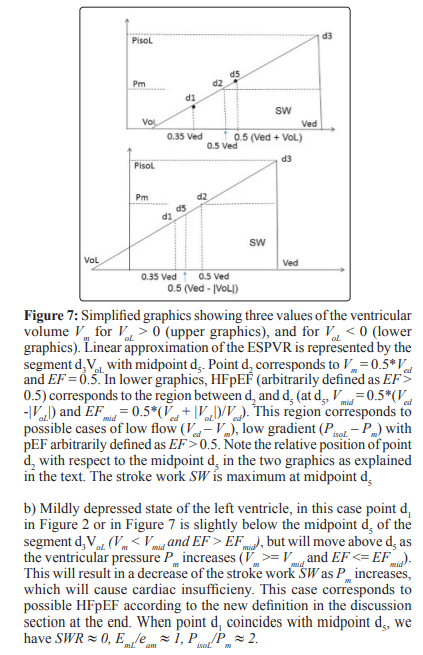
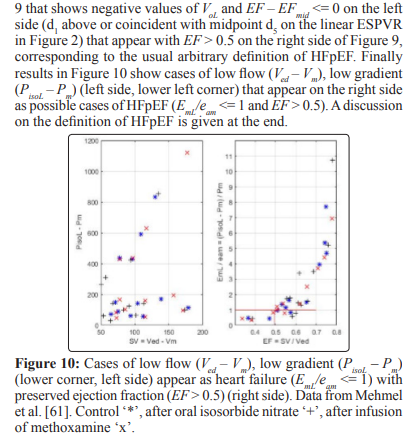
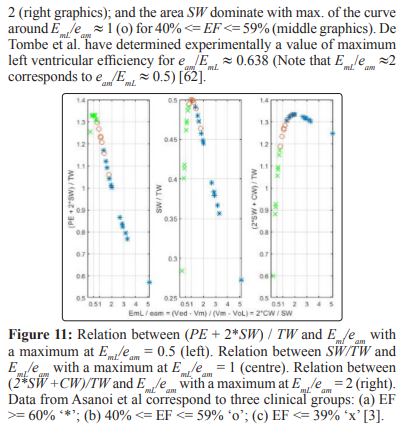
The results presented in this study show relations between the ejection fraction EF and the parameters VoL, EmL and PisoL describing the linear ESPVR, as well as possible clinical applications of these relations. The introduction of the maximum active pressure of the myocardium (also called peak isovolumic pressure PisoL) in the mathematical formalism describing the linear model of the ESPVR has allowed the use of the total area TW = PE + SW + CW in the study of the energetics of cardiac contraction (instead of the usual trend of using only the area PVA = PE + SW as in [4, 6, 60, 64]). The results of Figure 11 suggests that the study of the distribution of TW between the three areas PE, SW, CW under the ESPVR is a relevant factor for the understanding of the mechanics of cardiac contraction, whatever the physiological meaning of the areas PE and CW.
For simplicity in this study we have assumed that Pm is constant during the ejection phase, and that Vm » es the end-systolic ventricular volume. Applications to a variety of clinical and experimental data published in the literature and presented in this study show the consistency of the mathematical formalism used. The simplicity of the linear model should not hide the fact that the starting point of the mathematical derivation is based on the theory of large elastic deformation of the myocardium [47,48]. The linear model used in this study is a limiting case of a non-linear model of the ESPVR when the variations in Vm and Pm are relatively small and within the physiological range. Some preliminary results of the non-linear model of the ESPVR have been published, they need further studies for reliable application [16-18, 47, 48, 51].
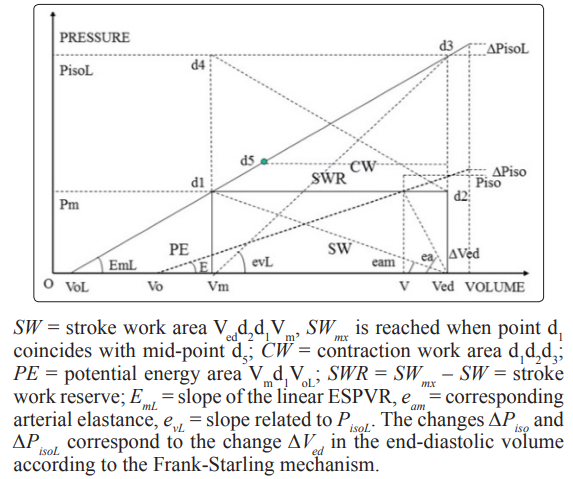
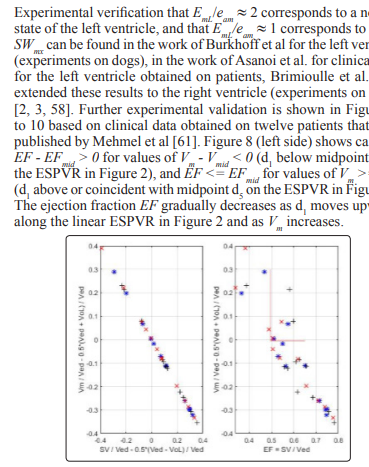
The ejection fraction EF of the left ventricle is one of several indexes that are related to the stroke volume SV = Ved – Vm. Equations (16) – (19) show some of the complex relations that exist between SV,and consequently EF = SV/Ved, and the parameters describing the ESPVR. In this study it was assumed that the parameters Vol , Eml and PisoL describing the linear ESPVR are constant when the point d1 in Figure 2 with coordinates (Vm, Pm) moves along the linear ESPVR. What actually happens is a complex mechanism that allows VoL, EmL or PisoL to vary in a way to allow the left ventricle to adapt to variation in load condition or inotropic changes in the myocardium, for instance by maintaining a stroke work reserve SWR = SWmx – SW > 0 (with point d1 on the ESPVR remaining below the midpoint d5 of the segment d3VoL), as explained in this study and in [8]. When this mechanism of adaptation fails then one can expect symptoms of heart failure, defined in this study as a reduction in stroke work SW when an increase in load demand occurs that correspond to an increase in the ventricular pressure Pm. This definition of heart failure is not unique, heart failure can be caused by other cardiac pathologies.
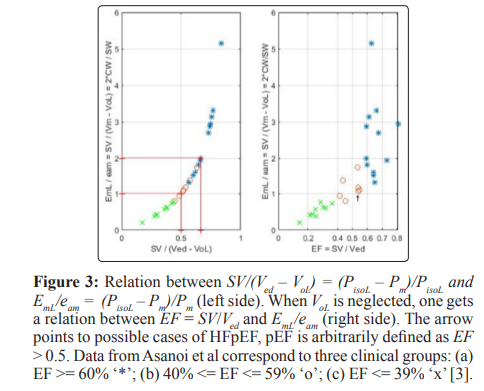
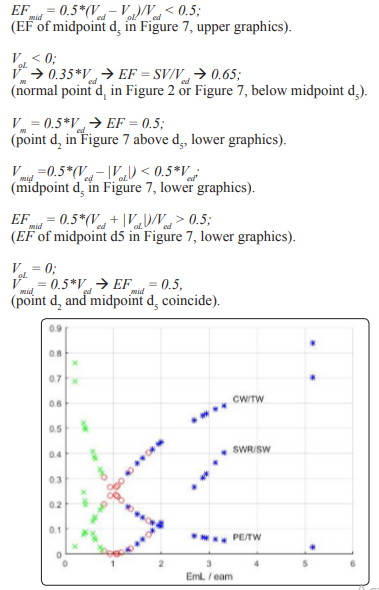
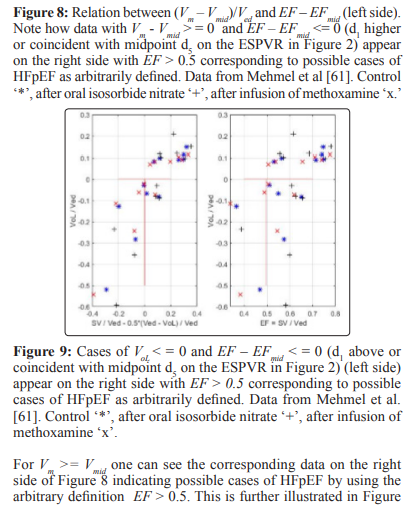
Two ways for the classification of the state of the left ventricle are indicated. One way is the ralation between percentage occurrence of heart failure and EF that can be extended to other indexes related to the EF as illustrated in Figs 12-14, these results show how useful the EF can be. The second way is a careful study of the position of the point d1 with coordidates (Vm, Pm) on the linear ESPVR with respect to the midpoint d5 with coordinates (Vmid = 0.5 (Ved + VoL), PisoL/2). As previously mentioned, the stroke work SW is maximum and equal to SWmx when point d1 coincides with midpoint d5 in Figure 2. When d1 is coincident or above the midpoint d5 on the ESPVR in Figure 2, an increase in Pm causes a decrease in the stroke work SW causing cardiac insufficiency. It has already been observed that there is a difficulty in defining HFpEF in terms of EF > 0.5 as explained in [65], and as discussed in this study in relation to Figure 7. A possible alternative definition of HFpEF is to refer to cases with d1 slightly below d5 on the ESPVR (Vm < Vmid, EF > EFmid) , but with d1 moving above d5 as a result of an increase in Pm (Vm >= Vmid, EF <= EFmid). Cases with Vm >= Vmid and EF <= EFmid = (Ved – Vmid)/Ved = 0.5*(Ved –VoL)/Ved correspond to cardiac insufficiency, agreeing with EF <= 0.5 only when VoL is zero.
Diastolic heart failure is linked to the Frank-Starling mechanism. As shown in Figure 2, normally an increase in the end-diastolic volume Ved by ![]() Ved is accompanied by an increase in the active pressure of the myocardium PisoL by
Ved is accompanied by an increase in the active pressure of the myocardium PisoL by ![]() PisoL. A good contractility of the myocardium is reflected in the way PisoL, EmL and/or VoL adapt to load condition in a way to maintain normal values for the ventriculo-arterial coupling EmL/eam. If an increase in load condition, as reflected by an increase in the ventricular pressure P , is not matched by adequate changes in m Vm, Ved and/or PisoL, the pressure gradient PisoL – Pm and/or the stroke volume SV» Ved - Vol may decrease when an increase is expected.As noted by Kitzman et al., congestive heart failure can occur in patients with preserved indexes of left ventricular systolic function, even in the absence of coronary and valvular heart disease, and that the patients’ ability to augment stroke volume SV by means of the Frank-Starling mechanism can be limited by abnormalities in the left ventricular diastolic function [66]. The ability of the ESPVR to adapt to the Frank-Starling mechanism is reflected on SV and PisoL by the relations given in Eqs (16) - (19).
PisoL. A good contractility of the myocardium is reflected in the way PisoL, EmL and/or VoL adapt to load condition in a way to maintain normal values for the ventriculo-arterial coupling EmL/eam. If an increase in load condition, as reflected by an increase in the ventricular pressure P , is not matched by adequate changes in m Vm, Ved and/or PisoL, the pressure gradient PisoL – Pm and/or the stroke volume SV» Ved - Vol may decrease when an increase is expected.As noted by Kitzman et al., congestive heart failure can occur in patients with preserved indexes of left ventricular systolic function, even in the absence of coronary and valvular heart disease, and that the patients’ ability to augment stroke volume SV by means of the Frank-Starling mechanism can be limited by abnormalities in the left ventricular diastolic function [66]. The ability of the ESPVR to adapt to the Frank-Starling mechanism is reflected on SV and PisoL by the relations given in Eqs (16) - (19).
The filling pressure Ped during the diastolic phase is small compared to PisoL or Pm and it is assumed zero in this study. The error in the calculation of the gradient PisoL – Pm = (PisoL - Ped) – (Pm - Ped ). cancels. The error in the calculation of the ratio (PisoL – Ped)/(Pm – Ped) = (PisoL /Pm )*[(1 – P ed/PisoL )/(1 – P ed/Pm )] » (P isoL /Pm )*[1 + P *(P oL mL – Pm)/(PisoL*Pm)] is small for relatively small Ped. The quantity Ped*(PisoL – Pm)/(PisoL*Pm) is usually much smaller than unity and can be neglected. Note that the linear model of the ESPVR gives a slight overestimate of PisoL.
Several topics have not been discussed in this study. For instance the influence of the geometry of the left ventricle on EF has been discussed by Dumesnil et al., possible influence of the geometry of the left ventricle on the ESPVR needs further consideration [20-22]. How coronary, valvular heart diseases or other cardiomyopathies affect the ESPVR is another subject that deserves attention [67-69]. The calculation of the parameters VoL and EmL of the linear model of the ESPVR in a non-invasive way for reliable clinical applications is also a topic of high priority [5,70]. Finally the reader may wish to compare the present study with Otto Frank’s original work on the pressure-volume relation in the left venbtricle as presented in a recent study [71].
Conclusion
Results presented in this study indicate that EF does not determine uniquely the state of the myocardium, bivariate (or multivariate) analysis of data appears a better way to assess the ventricular function as is evident from the figures presented in this study. New indexes derived from the ESPVR have been introduced, they do not replace, but complement and clarify the application of the EF in the study of cardiac mechanics. No one index can be used for classification or segregation between all cases of ventricular malfunction, some indexes are better than others depending on the clinical cases considered. An experimental relation between percentage occurrence of heart failure and EF has been used to derive similar relations with indexes derived from the ESPVR, it is an example of a useful use of the EF. Definitions of heart failure, HFpEF and diastolic heart failure in terms of the formalism used in this study have been introduced that need further evaluation. Extending the results of this study to a non-linear model of the ESPVR is an important topic for future research work.
Appendix
In this appendix the method of calculating the optimal efficiency given by Eq. (23) is explained. By using Eqs (9) and (13) we have
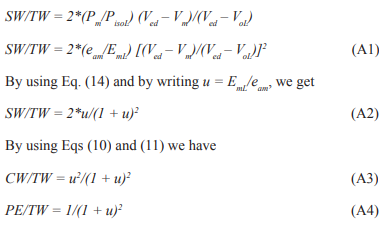

References
- Borow KM, Neumann A, Wynne J (1982) Sensitivity of end- systolic pressure-volume relations to the inotropic state in humans. Circulation 65: 988-997.
- Burkhoff D, Sagawa K (1986) Ventricular efficiency predicted by an analytical model. Am J Physiol 250: 1021-1027.
- Asanoi H, Sasayama S, Kameyama T (1989) Ventriculoarterial coupling in normal and failing heart in humans. Circ Res 65: 91-98.
- Sasayama S, Asanoi H (1991) Coupling between the heart andarterial system in heart failure. Am J Med 90: 14 -18.
- Kjørtad KE, Korvald C, Myrmel T (2002) Pressure-volume- based single-beat estimation cannot predict left ventricular contractility in vivo. Am J Physiol Circ Physiol 282: 1739-1750.
- Burkhoff D, Mirsky I, Suga H (2005) Assessment of systolic and diastolic ventricular properties via pressure-volume analysis; a guide for clinical, translational, and basic researchers. Am J Physiol Heart Circ Physiol 289: 501-512.
- Blaudszun G, Morel DR (2011) Relevance of the volume- axis intercept, Vo, compared with the slope of the end-systolic pressure-volume relationship in response to large variations in inotropy and afterload in rats. Exp Physiol 96: 1179-1195.
- Shoucri RM (1994) Clinical application of the end-systolic pressure-volume relation. Ann Biomed Eng 22: 212-217.
- Shoucri RM (1994) Possible clinical applications of the externalwork reserve of the myocardium. Jap. Heart J 35: 771-787.
- Shoucri RM (1997) Ventriculo-arterial coupling and the areas under the end-systolic pressure-volume relation. Jpn. Heart J 38: 253-262.
- Shoucri RM (1998) Studying the mechanics of left ventricular contraction. IEEE Eng Med Biol Mag 17: 95-101.
- Shoucri RM (1993) Theoretisher Beitrag zur Untersuchung der Druck-Volumen Beziehung im linken Ventrikel. Herz Kreislauf 25: 150-153.
- Shoucri RM (1995) Contribution théorique à l’étude de la relation pression-volume dans le ventricule gauche cardiaque. Innov Tech Biol Med 161-172.
- Perez E Riveros (2008) Análisis matemático de la curva presión- volumen del ventrículo izquierdo. Revista Col Anest 36: 265-268.
- Shoucri RM (2014) Basic relations between ejection fractionand ESPVR. Austin J of Clin Cardiol 1: 1-6.
- Shoucri RM (2011) A non-invasive method to calculate parameters of non-linear end-systolic pressure-volume relation. Inter J Cardiol 151: 389-391.
- Shoucri RM (2011) Calculation of parameters of end-systolic pressure-volume relation in the ventricles. Math and Comput.Modelling 54: 1638-1643.
- Shoucri RM (2013) Indexes derived from non-linear ESPVR for evaluation of ventricular performance, in: Modeling in Medicine & Biology X, edit. Kiss, R. & Brebbia, C. A., (Biomed 2013, Budapest) 17: 133-143.
- Dumesnil JG, Dion D, Yvorchuk K, Davies RA, Chan K, et al. (1995) A new, simple and accurate method for determining ejection fraction by Doppler echocardiography. Can. J Cardiol 11: 1007-1014.
- Dumesnil JG, Shoucri RM, Laurenceau JL, Turcot J (1979) A mathematical model of the dynamic geometry of the intact left ventricle and its application to clinical data. Circulation 59: 1024-1034.
- Dumesnil JG, Shoucri RM (1982) Effect of the geometry of the left ventricle on the calculation of ejection fraction. Circulation 65: 91-98.
- Dumesnil JG, Shoucri RM (1991) Quantitative relationship between left ventricular ejection and wall thickening and geometry. J Appl Physiol 70: 48-54.
- Pibarot P, Dumesnil JG, Leblanc MH, Cartier P, Métras J, et al. (1999) Changes in left ventricular mass and function after aortic valve replacement: A comparison between stentless and stented bioprosthetic valves. J Am Soc Echocardiogr 12: 981-987.
- Covell JW (2008) Tissue structure and ventricular wallmechanics. Circulation 118: 699-701.
- Coppola BA, Omens JH (2008) Role of tissue structure onventricular wall mechanics. Mol Cell Biomech 5: 183-196.
- Jones CJH, Raposo L, Gibson DG (1990) Functional importance of the long axis dynamics of the human left ventricle. Br Heart J 63: 215-220.
- Henein MY, Gibson DG (1999) Normal long axis function.Heart 81: 111-113.
- Aurigemma GP, Silver KH, Priest MA, Gaasch WH (1995) Geometric changes allow normal ejection fraction despite depressed myocardial shortening in hypertensive left ventricular hypertrophy. J Am Coll Cardiol 26: 195-202.
- Mignot A, Donal E, Zaroui A, Reant P, Salem A, et al. (2010) Global longitudinal strain as a major predictor of cardiac events in patients with depressed left ventricular function: a multicenter study. J Am Soc Echocard 23: 1019-1024.
- Pibarot P, Dumesnil JG (2010) Longitudinal myocardial shortening in aortic stenosis: ready for prime time after 30 years of research? Heart 96: 95-96.
- Dumesnil JG, Pibarot P (2012) Low-flow, low-gradient aortic stenosis with normal and depressed left ventricular ejection fraction. J Am Coll Cardiol 60: 1-9.
- Pibarot P, Dumesnil JG (2009) Aortic stenosis: look globally, think globally. J Am Coll Cardiol Cardiovasc Imaging 2: 400- 403.
- Baumgartner H, Otto CM (2009) Aortic stenosis severity: dowe need a new concept? J Am Coll Cardiol 54: 1012-1013.
- Clavel MA, Magne J, Pibarot P (2016) Low-gradient aortic stenosis. Eur Heart J 37: 2645-2657.
- Gozdzik AT, Jasinski M, Gozdzik W (2019) Echocardiographic evaluation of left ventricular strain in severe aortic stenosis with therapeutic implications and risk stratification. Adv Clin Exp Med 28: 1271-1279.
- Manisty CH, Francis DP (2008) Ejection fraction: a measure of desperation. Heart 94: 400-401.
- Sanderson JE (2007) Heart failure with normal ejection fraction.Heart 93: 155- 158.
- MacIver DH (2010) Current controversies in heart failure with a preserved ejection fraction. Future Cardiol 6: 97-111.
- Naing P, Forrester D, Kangaharan N, Mathumala A, Myint SM, et al. (2019) Heart failure with preserved ejection fraction, a growing global epidemic. Austr J Gen Pra 48: 465-471.
- Tromp J, Westenbrink BD, Ouwerkerk W, van Veldhuisen DJ, et al. (2018) Identifying pathophysiological mechanisms in heart failure with reduced versus preserved ejection fraction. J Am Coll Cardiol 72: 1081-1090.
- Pfeffer MA, Shah AM, Borlaug BA (2019) Circ Res 124: 1598-1617.
- Triposkiadis F, Butler J, Abboud FM, Armstrong PW, Adamopoulos S, et al. (2019) The continuous heart failure spectrum: moving beyond an ejection fraction classification. Eur Heart J 40: 2155-2163.
- Marwick TH (2018) Ejection fraction pros and cons. J Am CollCardiol 72: 2360-2379.
- Curtis JP, Sokol SI, Wang Y, Rathore SS, Ko DT, et al. (2003) The association of left ventricular ejection fraction, mortality, and cause of death in stable outpatients with heart failure. J Am Coll. Cardiol 42: 736-742.
- da Mota JPGF (2013) Intelligent modeling to predict ejection fraction from Echocardiographic reports, MSc thesis in Mechaniical Engineering, IST Técnico Lisboa, Portugal.
- Wehner GJ, Jing L, Haggerty CH, Suever JD, Leader JB, et al. (2020) Routinely reported ejection fraction and mortality in clinical practice: where does the nadir of risk lie? Eur Heart J 41: 1249-1257.
- Shoucri RM (1991) Non-linear pressure-volume relation in left ventricle. Jpn Heart J 32: 337-346.
- Shoucri RM (1991) Theoretical study of the pressure-volume relation in left ventricle. Am J Physiol Heart Circ Physiol 260: 282-291.
- Shoucri RM (1993) Theoretical study related to left ventricularenergetics. Jpn Heart J 34: 403-417.
- Shoucri RM (2000) Active and passive stresses in themyocardium. Am J Physiol Circ Physiol 279: 2519-2528.
- Shoucri RM (2010) Mathematical aspects of the mechanics of left ventricular contraction. Inter J Des Nature and Ecodyn 5: 173-188.
- Shoucri RM (2011) Numerical evaluation of the slope and intercept of end- systolic pressure-volume relation. In: Environmental Health & Biomedicine 15: 333-345.
- Shoucri RM (2013) Ejection Fraction and ESPVR: a study from a theoretical perspective. Inter Heart J 54: 318-327.
- Shoucri RM (2015) End-Systolic Pressure-Volume Relation, Ejection Fraction and Heart Failure: Theoretical aspect and clinical applications. Clin Med Ins Cardiol 9: 111-120.
- Shoucri RM (2010) ESPVR, Ejection Fraction and Heart Failure. Cardiovasc Eng 10: 207-212.
- Shoucri RM (2016) The pressure gradient across the endocardium. Computing in Cardiol 43: 181-184.
- Maughan WL, Shoukas AA, Sagawa K, Weisfeldt M (1979)Instantaneous pressure-volume relationship of the canine right ventricle. Circ Res 44: 309-315.
- Brimioulle S, Waulthy P, Ewalenko P, Rondelet B, Vermeulen F, et al. (2003) Single-beat estimation of the right ventricular end-sytolic pressure-volume relationship. Am J Physiol. Heart Circ Physiol 284: 1625-1630.
- Shoucri RM (1993) Pressure-volume relation in the rightventricle. J Biomed Eng 15: 167-169.
- Sunagawa K, Maughan WL, Sagawa K (1985) Optimal arterial resistance for the maximal stroke work studied in isolated canine left ventricle. Circ Res 56: 586-595.
- Mehmel HC, Stockins B, Ruffmann K, von Olshausen K, Schuler G, et al (1981) The linearity of the end-systolic pressure- volume relationship in man and its sensitivity for assessment of ventricular function. Circulation 63: 1216-1222.
- De Tombe PP, Jones S, Burkhoff D, Hunter WC, Kass DA, et al. (1993) Ventricular stroke work and efficiency both remain nearly optimal despite altered vascular loading. Am J Physiol (Heart Circ Physiol 33), 264: 1817- 1824.
- Kerkhoff PLM, Kresh JY, Li J KJ, Heyndrickx GR (2013) Left ventricular volume regulation in heart failure with preserved ejection fraction. Physiol Rep 1: 1-10.
- Chirinos JA (2013) Ventricular-Arterial coupling: Invasive andnon-invasive assessment. Artery Res 7: 2-14.
- Little WC (2008) Hypertension, Heart Failure, and EjectionFraction. Circulation 118: 2223-2224.
- Kitzman DW, Higginbotham MB, Cobb FR, Sheikh KH, Sullivan MJ, et al. (1991) Exercise intolerance in patients with heart failure and preserved left ventricular systolic function: Failure of the Frank-Starling mechanism. J Am Coll Cardiol 17: 1065-1072.
- Bastos MB, Burkhoff D, Maly J, Daemen J, den Uil CA, et al. (2020) Invasive left ventricle pressure-volume analysis: overview and practical clinical implications. Eur Heart J 41: 1286-1297.
- Li KJJ, Atlas G (2015) Left ventricle-arterial system interactionin heart failure. Clin Med Insights: Cardiol 9 : 93-99.
- Shoucri RM, Kerkhof LMP, Kozman H, Li KJJ (2015) Heart Failure: an exploration of recent advances in research and treatment. Clin Med Insights Cardiol 9: 143-146.
- Davidson H, Pretty C, Pironet A, desaive T, Janssen N, et al. (2017) Minimally invasive estimation of ventricular dead space volume through use of Frank-Starling curves. PLoS One 12: 1-11.
- Kuhtz Buschbeck JP, Drake-Holland A, Noble MIM, Lohff B (2018) Rediscovery of Otto Frank’s contribution to science. J Mol Cell Cardiol 119: 96-103.




