Research Article - (2025) Volume 10, Issue 3
Efficacy of a Novel Medical Device to Aid in Ultrasound-Guided Intravenous Cannulation: Randomized Controlled Non-Clinical Trial
2Department of Obstetrics and Gynaecology, Madhipura Christian Hospital, India
3Department of Emergency Medicine, Father Muller Medical College, India
4Department of Emergency Medicine, Father Muller Medical College, India
Received Date: Jul 09, 2025 / Accepted Date: Aug 11, 2025 / Published Date: Sep 17, 2025
Copyright: © 2026 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Paul, I. J., Abraham, E. S., Shailaja, S., Sequeira, J. (2025). Efficacy of a Novel Medical Device to Aid in Ultrasound-Guided Intravenous Cannulation: Randomized Controlled Non-Clinical Trial. J Anesth Pain Med, 10(3), 01-10.
Abstract
Background: Peripheral intravenous (PIV) cannulation is a crucial and frequently performed medical procedure, but achieving successful venous access can be particularly challenging in patients with difficult venous access (DVA). This challenge often leads to procedural delays and increased patient discomfort, as well as higher risks of complications such as hematomas, phlebitis, and infection. The complexity of ultrasound-guided PIV (USPIV) procedures, particularly for novice practitioners, necessitates technological innovations to improve success rates and reduce cannulation time.
Objective: This study aimed to evaluate the efficacy of a novel medical device designed to aid in USPIV by stabilising the ultrasound probe and assisting with needle guidance. Specifically, we assessed it impact on reducing cannulation time and improving first-attempt success rates, particularly for novice users.
Methods: A randomized, controlled trial was conducted using a synthetic phantom model to simulate DVA. Participants were stratified by experience level into novice, experienced, and expert groups. The primary outcome measures were cannulation time and first-attempt success rate. Secondary outcomes included the overall success rate of cannulation and user feedback on the device’s usability.
Results: The device demonstrated a significant 46% reduction in cannulation time compared to traditional USPIV techniques. Novice users saw improvements in both cannulation speed and first-attempt success rates, which increased by 35%. Overall, the device improved first-attempt success rates by 29% across all participants. User feedback indicated that the device reduced procedural complexity and improved hand-eye coordination, especially for less experienced practitioners.
Conclusion: The novel stabilising device significantly enhances USPIV performance by reducing cannulation time and increasing first-attempt success rates. Its benefits are particularly notable for novice practitioners, offering the potential to reduce the learning curve associated with USPIV and improve clinical efficiency in high-stress environments such as emergency departments. Further clinical studies in real-world patient settings are warranted to confirm these findings and assess the long- term impact on training and healthcare costs.
Introduction
Peripheral intravenous (PIV) cannulation is one of the most fre-quently performed medical procedures across diverse clinical settings, including emergency departments, surgery, and general hospital wards. PIV cannulation involves inserting a catheter into a peripheral vein to administer medications, fluids, or blood prod¬ucts, making it a cornerstone of patient care. However, achieving successful venous access is not always straightforward, particular- ly in-patient populations characterised by difficult venous access (DVA), such as those with obesity, dehydration, chronic illness¬es, or a history of multiple venous access attempts. DVA affects a significant portion of hospitalized patients, and these populations face a heightened risk of PIV catheter failure, potentially leading to increased patient discomfort and procedural complications such as hematomas, phlebitis, infections, and thrombophlebitis [1,2].
Studies show that risk factors such as obesity, diabetes, and chemotherapy treatment contribute significantly to the likelihood of difficult PIV access. For example, Rodríguez-Calero et al. (2020) found that obesity was a significant risk factor, with an odds ratio of 1.48, indicating an increased likelihood of DVA in obese patients [3]. Non-visible veins and a prior history of difficult intravenous access were also identified as critical risk factors, further complicating the successful placement of PIV catheters [4]. These risk factors not only affect the success of the procedure but also delay treatment and lead to more serious complications.
Failed PIV cannulation often results in multiple insertion attempts, increasing patient discomfort and potentially leading to more se-vere complications, such as catheter-related bloodstream infec-tions (CRBSIs). Each failed attempt can also extend the time re-quired to secure venous access, delaying critical treatments [5]. Approximately 20% of PIV cannulations fail on the first attempt, with this rate increasing dramatically in DVA patients [6]. When PIV access fails in these populations, clinicians may resort to more invasive approaches such as central venous catheterisation (CVC). Although CVC offers reliable access for long-term or large-volume infusions, it comes with greater risks, including a higher incidence of infections, mechanical complications such as pneumothorax or arterial puncture, and increased healthcare costs [7,8].
From an economic standpoint, difficult venous access (DVA) represents a substantial burden on healthcare systems. A study by Gala et al. (2024) estimated that the total cost burden of DVA in emergency departments could reach up to $890,095 annually [9]. This cost includes not only additional clinician time and resources but also the potential need for midlines or central venous catheters when PIV access fails. Consequently, reducing DVA is critical for improving patient outcomes and minimising healthcare costs.
In recent years, ultrasound-guided peripheral intravenous (USPIV) cannulation has gained prominence as an effective alternative, particularly for patients with DVA. The use of ultrasound allows clinicians to visualize peripheral veins in real time, significantly improving both the accuracy and success rate of cannulation. Numerous studies have demonstrated that USPIV reduces failure rates, improves first-attempt success, and minimizes complications compared to traditional "blind" techniques [5,10]. Stolz et al. conducted a meta-analysis confirming that ultrasound guidance increased success rates in patients with DVA, while traditional techniques often led to repeated failures [11]. USPIV has been shown to increase success rates in paediatric populations, with up to 87% of children achieving successful venous access on the first or second attempt, even after previous failed attempts with conventional methods [5]. Similarly, studies in adult patients have highlighted the benefits of ultrasound guidance in reducing cannulation time and increasing the overall success rate [12].
While PIV cannulation is a common use case, ultrasound guidance is also integral to a variety of other medical procedures, such as biopsies, nerve blocks, and regional anaesthesia. These procedures share the challenges of maintaining stable hand-eye coordination and needle alignment while accurately visualising target structures [5]. Mastering ultrasound-guided techniques demands significant training and experience, and improper technique can lead to com¬plications [13]. For instance, in nerve blocks or regional anaesthe¬sia, poor needle placement can result in nerve injury or incomplete anaesthesia, while in biopsies, inadequate targeting can lead to misdiagnosis or repeated procedures [14]. Therefore, innovations that reduce the complexity of ultrasound-guided procedures, im¬prove accuracy, and decrease complication rates are of paramount importance across these various medical application [[15]. Despite its advantages, the adoption of USPIV remains limited in many healthcare settings due to the technical demands of the procedure. Successful USPIV requires a combination of ultrasound image acquisition and needle guidance skills, which can be difficult for novice practitioners to master. The need for precise hand-eye coordination presents a steep learning curve, contributing to the variability in procedural success. In addition to this, practitioner experience has been shown to directly influence the success of US¬PIV, with novice users taking significantly longer to perform the procedure compared to experienced practitioners [16].
In response to these barriers, technological innovations have focused on developing devices that assist with the stabilisation of the ultrasound probe and needle alignment. Such devices aim to simplify the procedure by reducing the manual dexterity required to maintain ultrasound visualisation while guiding the needle, potentially improving first-attempt success rates and reducing cannulation time. This study evaluates the efficacy of a novel device designed for this purpose, with particular emphasis on its impact on novice users.
Materials and Methods
Study Design and Participants
This study employed a prospective, randomized controlled trial design aimed at evaluating the efficacy of a novel stabilising device for ultrasound-guided peripheral intravenous (USPIV) cannulation. Participants were recruited from a cohort of healthcare professionals, including novice users, with varying levels of experience in ultrasound-guided procedures. All participants were trained in basic ultrasound operation and the standard USPIV technique prior to the study. The study was conducted in a controlled, simulation-based environment using synthetic vein phantoms to ensure standardized conditions across all trials.
Equipment
All participants used the same equipment for the study, which included the GE Logiq V2 portable machine and a GE 9L linear probe. Standardized settings for gain (90%) and depth (3 cm) were pre-configured on the ultrasound machine to maintain consistency across all trials. The participants performed ultrasound-guided needle placement into a vessel in a synthetic simulator called the branched 2-vessel Blue phantom. The same vessel on the phantom, located at a depth of 1 cm, was used by all participants to standardize the procedure.
Eligibility criteria for participation included healthcare providers with no prior experience using the stabilising device and at least a basic understanding of ultrasound image acquisition. Participants were excluded if they had extensive previous experience with USPIV cannulation, as this study specifically aimed to assess the device's utility for novice users. Ethical approval was obtained from the institutional review board, and all participants provided informed consent.
Device Description and Uniqueness
The device evaluated in this study is a novel, purely mechani¬cal, single-use stabilising guide designed to aid ultrasound-guid-ed peripheral intravenous (USPIV) cannulation. Unlike existing systems that primarily focus on aligning the needle, this device is universal — compatible with all standard linear ultrasound probes and needles — and provides simultaneous probe stabilisation di¬rectly over the target vessel. By preventing probe tilt and maintain¬ing a consistent imaging plane, it ensures continuous visualisation of the vein throughout the procedure. Its disposable, lightweight design allows for seamless integration into routine practice with¬out the need for additional electronic components, calibration, or probe-specific adaptors.
Study Intervention
Each participant performed two ultrasound-guided peripheral in-travenous (USPIV) cannulation procedures: one using the novel stabilising device and one without the device (control condition). The order in which participants performed the procedures was ran-domly assigned to avoid any learning or fatigue effects. This ran-domization determined whether participants used the device first or performed the unaided procedure first.
The novel device was designed to stabilize the ultrasound probe, ensuring consistent visualisation of the target vein and facilitating needle alignment throughout the procedure. This intervention aimed to reduce the manual dexterity required for accurate needle guidance, especially among less experienced practitioners.
All participants performed cannulation procedures on synthetic phantoms, ensuring that any differences in outcomes were attrib-utable to the device and not to variations in participant anatomy or vein accessibility.
Outcome Measures
The primary outcome measure was the time required to achieve successful cannulation, defined as the duration from the initial ultrasound image acquisition to the successful insertion of the catheter into the phantom vein. Time was recorded using a stopwatch and noted in seconds for each participant.
Secondary outcome measures included:
1. First-attempt Success Rate: The percentage of participants who successfully cannulated the vein on their first attempt.
2. Number of Attempts: The total number of needle insertion attempts required to achieve successful cannulation.
3. Qualitative Feedback: Open-ended questions in the survey allowed participants to provide qualitative feedback on their experience using the device, particularly focusing on perceived advantages or drawbacks compared to traditional USPIV techniques.
Training Protocol
Before beginning the trial, all participants underwent a standardized training session that included a 15-minute didactic lecture on the principles of ultrasound-guided cannulation, followed by a practical demonstration using both the standard technique and the novel stabilising device. Participants were allowed hands-on practice with the device until they demonstrated basic proficiency in its use. The training aimed to minimize variability in the skill level of participants, ensuring that differences in outcomes were attributable to the intervention rather than disparities in baseline ultrasound proficiency.
Data Collection and Statistical Analysis
Data on cannulation time, first-attempt success rate, number of attempts, and user satisfaction were collected and recorded for each participant. All statistical analyses were performed using SPSS software (Version 27.0). Descriptive statistics (means, standard deviations, frequencies) were calculated for each outcome measure. For primary and secondary outcomes, independent t-tests were used to compare mean cannulation times between the intervention and control groups. Chi-square tests were conducted to assess differences in first-attempt success rates and the number of attempts required for successful cannulation.
A significance level of p < 0.05 was used for all statistical tests. Additionally, qualitative data from participant feedback were coded and analysed to identify common themes regarding the usability and effectiveness of the device in improving procedural outcomes.
Results
Study Population and Follow-up
A total of 62 participants were recruited for this randomized controlled trial. Among them, 63% were female, and the mean age was 23.6 years (SD = 6.6). Most participants (93.6%) had no prior experience with peripheral intravenous (PIV) cannulation, while the remaining participants had up to six years of experience (Table 1). The participants’ demographics, including age distribution, are presented in Supplementary Fig A1.
|
Characteristic |
N = 62 |
|
Age (years), Mean (SD) |
23.6 (6.6) |
|
Sex, n (%) |
|
|
Female |
39 (63%) |
|
Male |
23 (37%) |
|
Experience (years), n (%) |
|
|
0 |
58 (93.6%) |
|
1 |
1 (1.6%) |
|
2 |
1 (1.6%) |
|
5 |
1 (1.6%) |
|
6 |
1 (1.6%) |
Table 1: Demographic and Baseline Characteristics of the Study Population Legend
Experimental Conditions
Across the control and test conditions, a total of 124 measurements were conducted. The median time to successful cannulation was 38 seconds (range: 3 to 719 seconds) for the control group and 14 seconds (range: 2 to 73 seconds) for the device group. An outlier in the control group displayed an extreme value of 719 seconds (Fig 1). Sensitivity analysis showed that excluding this outlier did not significantly affect the overall findings.
Fig 1
|
Characteristic |
Control (N = 62) |
Device (N = 62) |
|
Total time until success (seconds), Median (Range) |
38 (3, 719) |
14 (2, 73) |
|
Procedure failure, n (%) |
2 (3.2%) |
0 (0%) |
|
Number of attempts, n (%) |
|
|
|
1 |
21 (34%) |
50 (81%) |
|
2 |
21 (34%) |
11 (18%) |
|
3 |
7 (11%) |
0 (0%) |
|
4 |
7 (11%) |
0 (0%) |
|
5 |
4 (6.5%) |
0 (0%) |
|
6 |
2 (3.2%) |
0 (0%) |
|
7 |
0 (0%) |
1 (1.6%) |
Table 2: Experimental Measurements Across Control and Device-Assisted Conditions Legend
The control condition exhibited a broader distribution of attempts, whereas the device condition demonstrated greater concentration around fewer attempts (Table 2, Fig 2). Cannulation failures occurred in two participants (3.2%) in the control condition, while no failures were observed in the device condition. Supplementary Fig A2 provides further analysis on the relationship between attempts and experience.
Fig 2
Efficacy of the Novel Device
After removing participants who failed the cannulation procedure, the remaining dataset comprised 122 valid observations. Time to successful cannulation was log-transformed for the analysis, indicating a significant effect of the device on reducing cannulation time. The use of the device was associated with a 46% reduction in cannulation time compared to the control condition (β = 0.54, 95% CI 0.40 to 0.73; p < 0.001). On average, participants required 29.31 seconds (95% CI [23.30, 36.89]) in the control condition, compared to 15.96 seconds (95% CI [12.74, 19.98]) using the device, after adjusting for the number of attempts (Fig 3).
Fig 3
The number of attempts varied significantly between the groups, with the device group demonstrating higher success rates with fewer attempts (Supplementary Fig A2). Each additional attempt increased the total time required for cannulation by 62% (β = 1.62, 95% CI 1.42 to 1.85; p < 0.001).
|
Characteristic |
Beta |
95% CI |
p-value |
|
Experimental condition |
|
|
|
|
Control |
— |
— |
— |
|
Device |
0.54 |
0.40 to 0.73 |
< 0.001 |
|
Number of attempts |
1.62 |
1.42 to 1.85 |
< 0.001 |
Table 3: Statistical Analysis of Cannulation Time by Experimental Condition and Number of Attempt
Fig 3 demonstrates the predicted time to cannulation with 95% confidence intervals, adjusted for the number of attempts.
Observations and Limitations
An essential limitation of this study relates to the variability in participants' prior experience with PIV cannulation (Supplementary Fig A3). While most participants were inexperienced, the few participants with prior experience could have influenced the results. To mitigate this, all participants followed a standardized protocol, ensuring consistency in procedure steps. Future studies should consider further stratifying participants by experience to assess its direct effect on outcomes. Interestingly, as shown in Supplementary Fig A4, the number of attempts did not significantly vary based on the experience level of the participants.
Discussion
This study demonstrated that the use of a novel stabilising device significantly reduces the time required to perform ultrasound-guided peripheral intravenous (USPIV) cannulation. The device led to a 46% reduction in cannulation time and a higher success rate on the first attempt compared to traditional methods, underscoring its potential utility in clinical settings, especially for novice users. These findings are consistent with previous research highlighting the steep learning curve associated with USPIV techniques and the role of technical aids in improving procedural outcomes [15,17]. Given the increasing prevalence of difficult venous access (DVA) in various patient populations, including those with obesity, dehydration, and chronic illnesses, achieving rapid and successful cannulation remains a significant challenge [3]. This challenge is exacerbated in high-stress environments such as emergency departments and intensive care units, where delays in establishing venous access can result in delayed treatment, increased patient discomfort, and potential escalation to central venous catheterisation (CVC) [18]. The reduction in time to successful cannulation observed in this study is particularly relevant in such settings, where time is a critical factor in patient outcomes. By improving the efficiency of venous access, this device could help reduce procedural complications and potentially prevent more invasive procedures such as CVC, which carry higher risks of infection, thrombosis, and mechanical complications [7].
The increased success rate on the first attempt observed with the device also has significant clinical implications. Studies show that repeated cannulation attempts not only increase patient discomfort but also heighten the risk of complications such as hematomas, phlebitis, and catheter-associated bloodstream infections (CABSI) [5]. By reducing the number of attempts required for successful cannulation, this device could contribute to improved patient safety and overall satisfaction, particularly in populations prone to DVA. Furthermore, healthcare professionals often experience frustration and fatigue when repeated attempts are needed, which can impact job satisfaction and procedural confidence. Simplifying the process with this device could alleviate some of these pressures and promote better clinical outcomes.
Moreover, the device may have implications beyond the immediate procedural success. In the context of healthcare education, USPIV cannulation is a skill that typically requires extensive practice and guidance to master. For novice practitioners, maintaining hand-eye coordination while simultaneously managing the ultrasound probe and needle can be particularly challenging. The device tested in this study effectively addresses these challenges by stabilising the probe and guiding needle alignment, thus potentially reducing the learning curve associated with USPIV. This could have a significant impact on training programs, enabling faster and more efficient skill acquisition for new clinicians, particularly in resource-limited settings where mentorship and hands-on experience may be less readily available [19].
One of the key strengths of this study is the rigorous design, including a randomized controlled approach that provides robust evidence of the device’s efficacy. However, the study has limitations that should be addressed in future research. Most notably, the use of a synthetic phantom rather than real patients may not fully replicate the challenges encountered in clinical practice. While phantoms are valuable tools for procedural training and standardisation, they lack the physiological variability seen in patients with DVA, such as variations in vein size, tissue density, and movement [9]. Further validation in a clinical setting with diverse patient populations is necessary to confirm the generalizability of these findings.
Additionally, while the study demonstrated a significant reduction in cannulation time and improved first-attempt success rates, it did not evaluate the potential cost-effectiveness of the device. In clinical practice, adopting new technologies must be balanced against their costs. Future studies should explore whether the time saved and the potential reduction in complications associated with the use of this device could translate into cost savings for healthcare systems. For instance, by reducing the need for CVC placement and its associated complications, the device could offer significant financial benefits [11]. Cost-effectiveness analyses could also compare this device to other technological aids or traditional USPIV techniques to provide a comprehensive understanding of its economic impact.
Another area for future research is the long-term impact of the device on clinical training. While this study focused on the immediate benefits of the device in reducing cannulation time, further research could explore whether the device leads to sustained improvements in procedural proficiency among novice users. Does the device merely compensate for the learning curve, or does it facilitate a deeper understanding of the USPIV process that enhances long-term skills? Understanding the educational value of this device could inform its integration into training curricula for healthcare professionals [3].
Lastly, while the study results are promising, they may not fully capture the potential for patient-centered outcomes, such as patient satisfaction, anxiety reduction, and overall procedural comfort. Future studies should incorporate patient-reported outcomes to assess the device’s impact from the patient’s perspective, further informing its clinical utility.
Conclusion
In conclusion, this study provides compelling evidence that the novel stabilising device significantly enhances the efficiency and success of USPIV cannulation, particularly for novice users. By reducing the time required for venous access and increasing the likelihood of first-attempt success, the device could offer substantial benefits in high-stress clinical environments. However, further research is needed to validate these findings in real-world clinical settings, assess the long-term impact on healthcare training, and evaluate the cost-effectiveness of adopting this technology in routine practice.
Fig A1: Distribution of age in the study population. This figure shows the age distribution of participants enrolled in the study.
Fig A2: Number of cannulation attempts by condition and success rates. The figure depicts the number of attempts required to achieve successful cannulation in both control and test conditions.
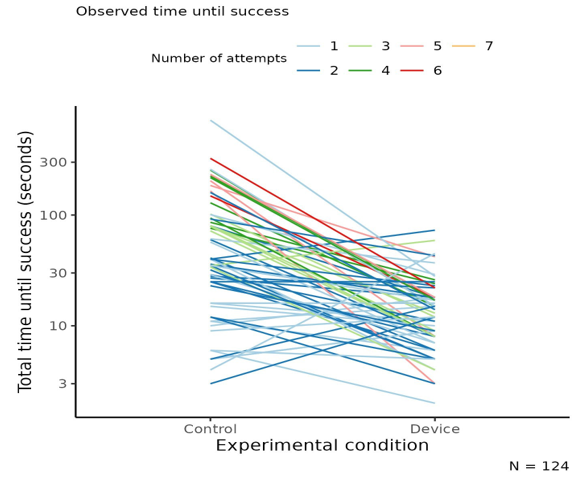
Fig A3: Experience levels among participants. This figure outlines the experience levels of the participants in the study, highlighting the number of participants with prior experience in ultrasound-guided cannulation.
Figure A4: Interaction effects in cannulation attempts. The figure illustrates nonparallel regression lines, highlighting the lack of interaction between experience and the number of attempts.
References
- Larsen, E. N., Marsh, N., O’Brien, C., Monteagle, E., Friese, C., & Rickard, C. M. (2021). Inherent and modifiable risk factors for peripheral venous catheter failure during cancer treatment: a prospective cohort study. Supportive Care in Cancer, 29(3), 1487-1496.
- Marsh, N., Webster, J., Larsen, E., Cooke, M., Mihala, G., & Rickard, C. M. (2018). Observational study of peripheral intravenous catheter outcomes in adult hospitalized patients:a multivariable analysis of peripheral intravenous catheter failure. Journal of hospital medicine, 13(2), 83-89.
- Rodríguez-Calero, M. A., Blanco-Mavillard, I., Morales-Asencio, J. M., Fernandez-Fernandez, I., Castro-Sánchez, E., & de Pedro-Gómez, J. E. (2020). Defining risk factors associated with difficult peripheral venous Cannulation: A systematic review and meta-analysis. Heart & Lung, 49(3), 273-286.
- Dat, M., JC, D. L. T. M., AC, N., Reis, R. K., MH, B., & SEM,T. (2021). Prevalence of and factors associated with difficult peripheral venipuncture in adult surgical patients. The Journal of Vascular Access, 22(3), 404-410.
- Vinograd, A. M., Zorc, J. J., Dean, A. J., Abbadessa, M. K. F., & Chen, A. E. (2018). First-attempt success, longevity, and complication rates of ultrasound-guided peripheral intravenous catheters in children. Pediatric emergency care, 34(6), 376-380.
- Lapostolle, F., Catineau, J., Garrigue, B., Monmarteau, V., Houssaye, T., Vecci, I., ... & Adnet, F. (2007). Prospective evaluation of peripheral venous access difficulty in emergency care. Intensive care medicine, 33(8), 1452-1457.
- Balsorano, P., Virgili, G., Villa, G., Pittiruti, M., Romagnoli, S., De Gaudio, A. R., & Pinelli, F. (2020). Peripherally inserted central catheter–related thrombosis rate in modern vascular access era—when insertion technique matters: a systematic review and meta-analysis. The journal of vascular access, 21(1), 45-54.
- Ullman, A. J., Kleidon, T. M., Turner, K., Gibson, V.,Dean, A., Cattanach, P., ... & Rickard, C. M. (2019). Skin complications associated with pediatric central venous access devices: prevalence, incidence, and risk. Journal of Pediatric Oncology Nursing, 36(5), 343-351.
- Gala, S., Alsbrooks, K., Bahl, A., & Wimmer, M. (2024). The economic burden of difficult intravenous access in the emergency department from a United States’ provider perspective. Journal of Research in Nursing, 29(1), 6-18.
- Rodríguez-Herrera, Á., Solaz-García, Á., Mollá-Olmos, E., Ferrer-Puchol, D., Esteve-Claramunt, F., Trujillo-Barberá, S., ... & Casaña-Mohedo, J. (2022, January). Use of the ultrasound technique as compared to the standard technique for the improvement of venous cannulation in patients with difficult access. In Healthcare (Vol. 10, No. 2, p. 261). MDPI.
- Stolz, L. A., Stolz, U., Howe, C., Farrell, I. J., & Adhikari,S. (2015). Ultrasound-guided peripheral venous access: a meta-analysis and systematic review. The journal of vascular access, 16(4), 321-326.
- Costantino, T. G., Parikh, A. K., Satz, W. A., & Fojtik, J.P. (2005). Ultrasonography-guided peripheral intravenous access versus traditional approaches in patients with difficult intravenous access. Annals of emergency medicine, 46(5), 456-461.
- Kanno, C., Murayama, R., Abe-Doi, M., Takahashi, T., Shintani, Y., Nogami, J., ... & Sanada, H. (2020). Development of an algorithm using ultrasonography-assisted peripheral intravenous catheter placement for reducing catheter failure. Drug discoveries & therapeutics, 14(1), 27-34.
- Mattox, E. A. (2017). Complications of peripheral venous access devices: prevention, detection, and recovery strategies. Critical care nurse, 37(2), e1-e14.
- Bahl, A., Johnson, S., Alsbrooks, K., Mares, A., Gala, S., & Hoerauf, K. (2023). Defining difficult intravenous access (DIVA): a systematic review. The journal of vascular access, 24(5), 904-910.
- Sultan, S. F., Shorten, G., & Iohom, G. (2013). Simulators for training in ultrasound guided procedures. Medical ultrasonography, 15(2), 125-131.
- Bahl, A., Pandurangadu, A. V., Tucker, J., & Bagan, M. (2016). A randomized controlled trial assessing the use of ultrasound for nurse-performed IV placement in difficult access ED patients. The American journal of emergency medicine, 34(10), 1950-1954.
- Au, A. K., Rotte, M. J., Grzybowski, R. J., Ku, B. S., & Fields, J. M. (2012). Decrease in central venous catheter placement due to use of ultrasound guidance for peripheral intravenous catheters. The American journal of emergency medicine, 30(9), 1950-1954.
- Piredda, M., Fiorini, J., Facchinetti, G., Biagioli, V., Marchetti, A., Conti, F., ... & De Marinis, M. G. (2019). Risk factors for a difficult intravenous access: a multicentre study comparing nurses' beliefs to evidence. Journal of Clinical Nursing, 28(19-20), 3492-3504.



