Research Article - (2025) Volume 8, Issue 2
Effect of Vermicomposting on Growth, Physiology and Yield Traits of Mung Bean (Vigna radiata L.) Cultivar under Salinity Stress
Received Date: Jul 15, 2025 / Accepted Date: Aug 18, 2025 / Published Date: Sep 01, 2025
Copyright: ©©2025 Indra Jeet Chaudhary, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kalukhe, S. S., Chaudhary, I. J. (2025). Effect of Vermicomposting on Growth, Physiology and Yield Traits of Mung Bean (Vigna radiata L.) Cultivar under Salinity Stress. J Agri Horti Res, 8(2), 01-07.
Abstract
Salinity is one of the main environmental problems that causes yield loss by affecting physiological and morphological characteristics of plants. The vermicomposting plays a vital role in growth and development of plants. Despite that it’s also reduced harmful effects of various environmental stresses on plants. Therefore, the present study conducted for investigation of salinity stress and protective role of vermicomposting on various traits of Mung bean (Vigna radiata L.) cultivar. The findings suggest that 50 ppm salinity levels caused least impact than 100 ppm salinity levels. The application of vermicomposting drastically increased the growth and yield of mung bean cultivar. These results demonstrate the remarkable ability of vermicomposting to promote healthy mung bean growth even under challenging saline conditions. Based on these findings, the study strongly recommends the use of vermicomposting as a key strategy for optimizing the salinity level in the soil and crop productivity in saline soil. Additionally, the study suggests a potential long-term benefit of Vermicomposting provide immediate relief from salinity stress but also enhance the overall salinity tolerance of plants over time. This study highlights the importance of vermicomposting as a powerful tool for combating the harmful effects of salinity stress on plants.
Keywords
Salinity Stress, Vermicomposting, Mung Bean Plants, Plant Growth and Development
Introduction
Soil Salinity is one of the main environmental factors that limit plant growth and production. Salinity affects almost all the physiological and biochemical processes, as well as morphological characteristics, of plants [1]. Salinity stress affects almost all phases of the growth of leguminous plants, namely, the germination, vegetative and reproductive phases [2]. Salinity stress causes osmotic stress, nutrient imbalance, ion toxicity, increased reactive oxygen species (ROS) production, decreased photosynthesis and reduced plant productivity [2]. The increase in ROS damages biomolecules, such as lipid, protein and DNA; changes membrane integrity and ion transport; decreases enzyme activity; inhibits protein synthesis and leads to cell death. Salinity-induced oxidative stress in the form of ROS adversely affects plant growth and productivity. Plants trigger an antioxidant defence system through non-enzymatic compounds, such as ascorbic acid, glutathione, α-tocopherol, carotenoids and flavonoids.
Legumes, commonly referred to as pulses, are a valuable source of high-quality, readily available dietary protein. This affordability and nutritional significance have contributed to their nickname, "poor man's meat”. Beyond their protein content, pulses are a valuable source of complex carbohydrates, essential vitamins and minerals, omega-3 fatty acids, and dietary fiber. Mung bean is a nutritional powerhouse, boasting an impressive profile of high-quality protein, and essential amino acids, including a complete protein source, a diversity of fatty acids, dietary fiber, and a wealth of essential minerals and vitamins. The combined challenges of salinity and sodality are frequently encountered in irrigated agricultural systems, particularly within regions characterized by low precipitation and high evapotranspiration rates.
Plants experiencing salt (NaCl) stress invariably suffer potassium (K+) depletion alongside the increase in sodium (Na+) content. K+, a crucial macronutrient constituting 4-6% of a plant's dry matter, plays a critical role in determining crop yield and quality[3]. The photosynthetic process can be negatively affected by salinity, as reported by [2]. Salinity stress stands out as one of the most detrimental environmental factors limiting crop production globally. Salt toxicity unleashes a cascade of detrimental effects on plants, manifesting as tissue burning, root and shoot death, leaf shedding (abscission), reduced stomatal conductance, disrupted cellular metabolism, and a decline in photosynthetic activity. Salinity's adverse effects extend throughout a plant's lifecycle, negatively impacting all stages of development, from seed germination to robust vegetative growth. Salinity stress acts as a significant obstacle to seed germination and seedling development.
The application of organic fertilizers has emerged as a prominent strategy for the remediation of saline soils in recent decades. Recycling organic waste is a keystone of sustainable waste management, and biological treatment methods like composting and vermicomposting offer a powerful solution. Vermicomposting, a well-established biotechnology process, stands out as a highly effective and environmentally friendly technique for producing organic fertilizer from various organic waste streams [4]. Vermicomposting production relies on a fascinating collaboration between earthworms and microorganisms. The final product of this symbiotic process is vermicomposting, a nutrient-rich fertilizer created from the earthworm castings. Study on chickpea (Cicer arietinum L.), it was found that taking 3000 kg/ ha of vermicomposting significantly increases the biological function of plant and significant increase in K+ concentration. Therefore, the aim of the study is assessing salt impact on mung bean cultivar and protective role of vermicomposting. Based on physiology and morphology of plant study will suggest the effectiveness of vermicomposting. Therefore, this study will be helpful for sustainable agriculture with respect to application of vermicomposting against saline soil.
Materials and Methods
Experimental Design
This experiment was carried out on a Randomized Block Design with 2 replications at the terrace of the Department of Environmental Science, Savitribai Phule Pune University, Pune, Maharashtra (India) in January to April 2024. Pune experiences three seasons: summer, monsoon, and winter. Typical summer months are from March to June often extending till 15 June, with maximum temperatures sometimes reaching 42 °C (108 °F). The warmest month in Pune is between 20 April and 20 May; although summer doesn't end until May, the city often receives heavy dusty winds in May (and humidity remains high). Even during the hottest months, the nights are usually cool due to Pune's high altitude. The highest temperature ever recorded was 43.3 °C (109.9 °F) on 30 April 1897.The monsoon lasts from June to October, with moderate rainfall and temperatures ranging from 22 to 28 °C (72 to 82 °F). Most of the 722 mm (28.43 in) of annual rainfall in the city falls between June and September, and July is the wettest month of the year. (Pune Municipal Corporation Data).
The Mung (Vigna radiata) variety Jalgaon 781, plant is selected for the experiment. Usually, this plant is cultivated in the Kharif season in tropical, sub-tropical, and temperate regions. In India, mung is cultivated during the summer season due to its harness to water stress. The seed required for the experiment we acquired from the College of Agriculture, Pune. We chose genetically pure and clean seeds for the experiment. Mung is a salinity susceptible crop easily affected by it.
Composition of Vermicomposting and Treatments
This experiment was carried out in10 kg of soil we studied the effect of Vermicompost (125 gm) on Green Gram (Vigna radiata) under different doses of salinity (50ppm and 100 ppm). Chemical analysis of the Vermicompost as shown in the table 1. Analysis of soil after application of salt stress and the vermicompost is shown in the following table 2.
|
Sample |
Organic matter (%) |
pH |
C/N |
P (%) |
Ca (%) |
K (%) |
Na (%) |
Total N (%) |
|
Vermicompost |
35–40 |
8–8.5 |
16 |
1.5–2 |
3.8–4 |
0.9–1.5 |
0.6–0.9 |
1.3–1.6 |
Table 1: Chemical Composition of Vermicomposting
|
Parameters |
T1 |
T2 |
T3 |
T4 |
T5 |
|
pH |
7.4 |
7.45 |
7.45 |
6.4 |
6.7 |
|
EC |
0.2 |
0.3 |
0.3 |
0.6 |
0.6 |
Table 2: Analysis of Soil After Application of Salt and the Vermicomposting
Each experimental unit contained a pot with a height of 20 cm and 15 cm in diameter and 15 seeds were sown (8 Jan 2024) in each pot to ensure the emergence of the seedlings, then thinned plant in each pot. We maintained a sufficient quantity of moisture in the pot by applying a regular water supply. For maintenance of the salinity in the pots by regularly checking EC and pH. We designed the experiment as below (Table. 3).
|
T1 |
Controlled |
|
T2 |
Plant applied with 50 ppm salt |
|
T3 |
Plant applied with 100 ppm salt |
|
T4 |
Plant applied with 50 ppm salt and 250 gm of Vermicompost |
|
T5 |
Plant applied with 100 ppm salt and 250 gm of Vermicompost |
Table 3: Treatments
Sampling and Plant Analysis
Plant sample were taken at the juvenile stage (20 days after sowing) and vegetative stage (40 days after sowing) using standard sampling methods.
Growth and Biomass Analysis
For plant height, we measured the length of the root and shoot by meter scale and expressed it as cm/plant. For the leaf area, we used the graphical method placed a leaf on the graph paper and counted the squares occupied by the leaf and expressed in cm2/plant. For biomass estimation plants are washed and carefully and weighed for the fresh weight and for the dry weight estimation kept in the oven at 80°C for 24 hours.
Growth Indices
Biomass accumulation and allocation were measured using the following formula described in Hunt (1982).
Leaf area ratio (LAR) = LA / W (cm2 g-1)
Leaf area duration (LAD) = (LA1 + LA2) (T2 - T1) / 2 (days)
Root: Shoot ratio (RSR) = RW / SW (g g-1)
Absolute growth rate (AGR) = (H2 -H1 ) / (T1- T2 ) (cm day-1)
Net primary productivity (NPP) = Total biomass / Age of plant (g plant-1 day-1)
Where W = total biomass; RW = root dry weight; SW = shoot dry weight; LW = leaf dry weight; LA = leaf area; H = plant height; L = land area; WSF = dry weight of stem at flowering; WSM = dry weight of stem at maturity; WPM = dry weight of panicle at maturity; WPF = dry weight of panicle at flowering; (T2 = T1) = time interval and parameters subscript with 1 and 2 indicated that observations at time T1 and T2.
Total Chlorophyll and Carotenoids:
A 0.1 g leaf sample was chopped and mixed with 10 mL of an 80% acetone (v/v) solution. In a test tube, the solution was kept overnight at 4 C. The solution's OD (optical density) was measured at 663 nm and 645 nm. Total chlorophyll and carotenoids were calculated following the formula described by [5,6].
Estimation of Ascorbic Acid:
method was used for extraction and estimation of ascorbic acid content based on the reduction of 2, 6-dichlorophenolindophenol (DCPIP). 5 g fresh leaf sample was homogenized in 20 ml of extraction solution containing 5 g oxalic acid and 0.075 g EDTA in 100 ml distilled water, then centrifuged at 12,000x for 15 minutes.1ml of obtained supernatant was added with 5 ml of 2, 6-dichlorophenolindophenol which turned solution into pink colour [7]. At 520 nm, the absorbance of the pink colour solution (Es) was measured. To reduce DCPIP, a drop of ascorbic acid solution was added to the mixture, and the absorbance (Et) at the same wavelength was measured. Both absorbance was recorded against the blank (Eo) of 1 ml of extraction medium added with 5 ml of DCPIP. The total amount of ascorbic acid was calculated by a standard curve prepared from aqueous solution of ascorbic acid using the following formula: Ascorbic acid (mg g- 1 fresh leaf) = [{Eo- (Es- Et)} × V] / (v × W × 1000)
Yield Characteristics
At the maturity stage, out of each experimental plot, plants were randomly chosen and used for counting the number of grains as well as for estimating 10-grain weight. Additionally, at physiological maturity, the plants of each plot were manually harvested. Grain yield weight was also measured. For the number of pods, we calculated by counting the pods of the plant (60DAS). And then measure the length of the pods by meter scale in centimetres. After that, split the pod and collected and counted. Weighed the grains on the weighing balance and recorded them in grams.
Results and Discussions
As per the above analysis of the plant, it is clear that salinity adversely affected the plant. At the same time, we found that the vermicompost-applied plant performed better which ultimately means the vermicompost induced salinity tolerance into the mung plant.
Morphological Changes
Morphological changes of plant were analyses such as root, shoot and total plant height. The application of salt caused negative effect on root and shoot length of selected cultivar. While application of vermicomposting increased the plant height (Figure. 1). Leaf area was also reduced due to application of salt stress. Applied 100 ppm salt caused more severe effect than 50 ppm salt in both sampling periods. Present study shows that salinity reduced the growth of plants in terms of shoot height, root length, leaf area and biomass. Growth reduction was higher with increase in level of salinity treatment. Reduced growth and decline in biomass under soil salinity has been reported by a researcher in different plant species [2]. suggested growth retardation as an important aspect for assessing the extent of salinity-led destruction irrespective of the type of plant species. suggested inhibition in cell division and cell elongation induced by salt stress is the main reason of decreased growth, biomass and yield and the result of present study suggested higher sensitivity of spinach cultivar to salinity.
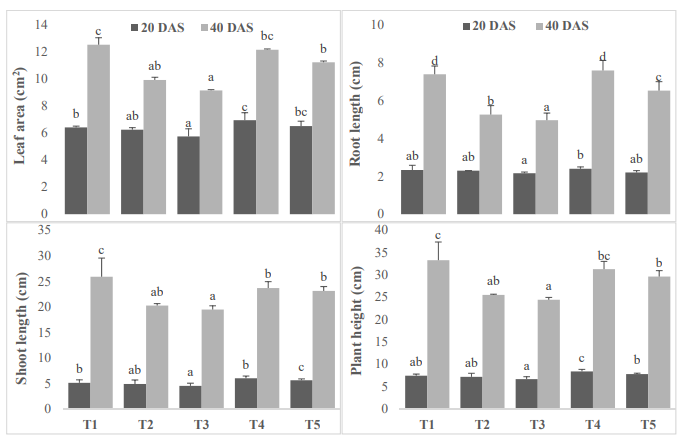
Figure 1: Effect of vermicomposting on leaf area (cm2), root length (cm), shoot length (cm) and total plant height (cm) of Mung bean (Vigna radiata L.) cultivar under two concentration of salt stress (Mean± standard deviation of three replicates are shown by thin vertical bars, Duncan’s Multiple Range Test showed value in column with same letter are not significantly different (p<0.05)).
Changes in Biomass Characteristics
Biomass is directly proportional to the photosynthetic activity of the plant, and also it's significant for the growth of the plant. In present study plant applied with the 50 ppm (T1) and 100 ppm(T2) salts shows a significant reduction in the biomass at both juvenile (20DAS) and vegetative state (40DAS). The application of vermicomposting in 50 ppm salt was more effective than 100 ppm salt. The higher growth was seen lower dose of salt with same concentration of vermicomposting application (Figure. 2).
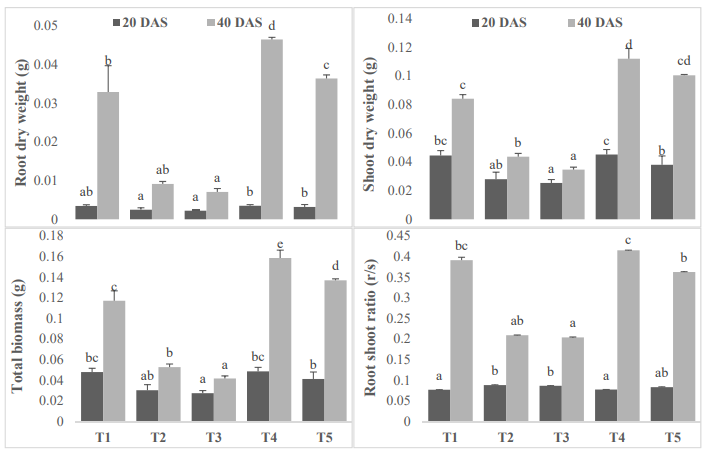
Figure 2: Effect of vermicomposting on root dry weight (g), shoot weight (g) and total plant biomass (g) of Mung bean (Vigna radiata L.) cultivar under two concentration of salt stress (Mean± standard deviation of three replicates are shown by thin vertical bars, Duncan’s Multiple Range Test showed value in column with same letter are not significantly different (p<0.05)).
Biochemical Changes
Salt stress negatively affects the mung cultivars are known to experience considerable metabolic alterations. Higher salt concentrations can alter protein and nucleic acid metabolism, membrane integrity, and nutritional absorption by causing osmotic stress, toxicity of ions, and oxidative damage []. The growth, development, and yield of plants may be adversely affected by these changes. However, it has been demonstrated that applying vermicomposting as a preventative can lessen the adverse effects of salt stress on mung cultivars. Presented study showed vermicomposting improves plant growth and stress tolerance by increasing biomass, photosynthetic and antioxidant activity. Applying vermicomposting can alter the expression and activity of several enzymes and metabolic pathways involved in stress response and growth control at the biochemical level [8,9]. Overall, the use of vermicomposting as a preventative in mung cultivars can result in significant biochemical alterations that enhance growth, stress tolerance, and yield under salt-stress conditions.
100 ppm salt concentrations can harm cells and prevent the production of chlorophyll, which lowers chlorophyll levels and reduces photosynthetic effectiveness [22]. However, it has been noted that the protective use of vermicomposting can lessen the adverse effects of salt stress on the chlorophyll content in moong bean plants [21]. Salt stress and vermicomposting treatment can have an impact on chlorophyll a, b, and total chlorophyll levels at the biochemical level. Overall, the use of vermicompost as a preventative in mung bean plants can cause considerable biochemical alterations that enhance chlorophyll content and photosynthetic efficiency under salt stress conditions [20].
Ascorbic acid controls the cell division cell elongation and cell differentiation and commonly known as vitamin C. It is an essential nutrient for humans, and also plays a crucial role in the plant kingdom. AsA is synthesized by plants through the d-mannose/l-galactose pathway that functions as a powerful antioxidant and protects plant cells from ROS generated during photosynthesis. AsA controls several key physiological processes, including: photosynthesis, respiration, and carbohydrate metabolism, either by acting as a co-factor for metabolic enzymes or by regulating cellular redox-status [10]. In present study, higher reduction of ascorbic acid at juvenile stage is 30.76% and at vegetative stage is 18.63% under 100 ppm salinity. However, antioxidants were improved by exogenously applied AA at both salinity levels. The results of the multivariate analysis were significant [11]. Expect in cultivars DAS and cultivars treatment in spinach to all the individual elements and their interplay. Compared to ascorbic acid, salt stress led to an increase in phenol content whereas exogenous protectants caused it to decrease in all experimental cultivars [19].
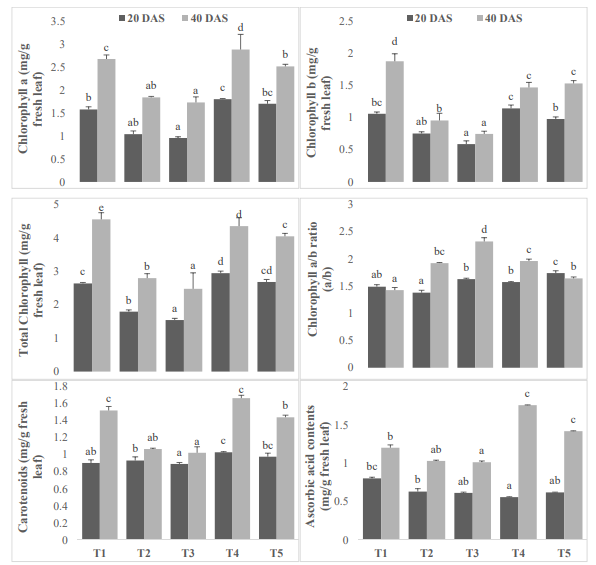
Figure 3: Effect of vermicomposting on chlorophyll a (mg/g fresh leaf), chlorophyll b (mg/g fresh leaf), total chlorophyll (mg/g fresh leaf), chlorophyll a/b ratio (mg/g fresh leaf), carotenoids contents (mg/g fresh leaf) and ascorbic acid contents (mg/g fresh leaf) of Mung bean (Vigna radiata L.) cultivar under two concentration of salt stress (Mean± standard deviation of three replicates are shown by thin vertical bars, Duncan’s Multiple Range Test showed value in column with same letter are not significantly different (p<0.05)).
Yield Characteristics
Salt stress is a key abiotic factor that can significantly reduce plant growth and yields [12]. There was a statistically significant effect of the Number of pods, and pod length of mung bean due to different doses of salinity [18]. The pod length of mungbean plants was significantly affected by the application of vermicompost. The number of seeds per pod were significantly impacted by the various salinity stresses [13]. There was a significant effect in the weight of 10 grains of mung bean due to vermicompost. The number of pods, Pod length, Number of grains, and grain weight are all characteristics that indicate the yield of plants that are negatively affected by the salt stress, and on the other hand, vermicompost increases positively [14,15]. Due to salinity exposure decline in RWC and chlorophyll content, which eventually results in reducing the photosynthesis rate and curtailed grain yield [16,17].
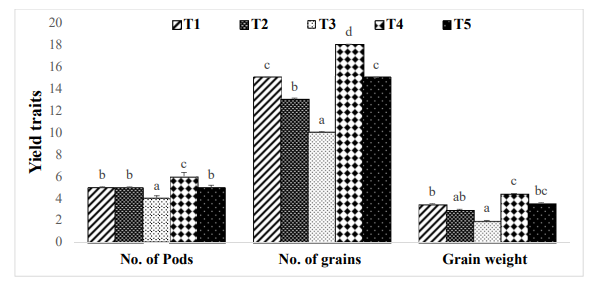
Figure 4: Effect of vermicomposting on number of pods (N plant-1), No. of grains (N plant-1) and grain weight (g plant-1) of Mung bean (Vigna radiata L.) cultivar under two concentration of salt stress (Mean± standard deviation of three replicates are shown by thin vertical bars, Duncan’s Multiple Range Test showed value in column with same letter are not significantly different (p<0.05)) [23].
Conclusion
This study investigated the impact of salinity stress on various physiological and biochemical processes within green gram (Vigna radiata L.) plants. The findings reveal a concerning trend: as salinity levels increase, the production of photosynthetic pigments – chlorophyll a, chlorophyll b, total chlorophyll, and carotenoids – significantly declines. These pigments play a critical role in capturing sunlight and fuelling plant growth. Their reduction weakens the plant's photosynthetic capacity, hindering its ability to generate essential energy. However, the study also identified a promising solution, the application of vermicomposting. This organic amendment, rich in nutrients and produced by the decomposition of organic matter through vermicomposting with worms, demonstrated remarkable effectiveness in mitigating the negative effects of salinity stress. Green gram, being highly sensitive to salinity, experiences stunted growth and alterations in its physiological traits when exposed to high salt concentrations.
References
- Patankar, A. V., Nigam, B., & Chaudhary, I. J. (2024). Effect of Salinity and Humic Acid on Growth, Biomass, and Yield of Mung Bean (Vigna Radiata L.) Cultivars. Insights Herb Med, 3(2), 01-14.
- Kale, S. S., Chauhan, R., Nigam, B., Gosavi, S., & Chaudhary,I. J. (2024). Effectiveness of nanoparticles in improving soil fertility and eco-friendly crop resistance: A comprehensive review. Biocatalysis and Agricultural Biotechnology, 56, 103066.
- Porter, J., Xie, L., Challinor, A. J., Howden, M., Iqbal, M. M., Lobell, D. B., & Travasso, M. I. (2014). Food security and food production systems.
- Berners-Lee, M., Kennelly, C., Watson, R., & Hewitt, C. N. (2018). Current global food production is sufficient to meet human nutritional needs in 2050 provided there is radical societal adaptation. Elem Sci Anth, 6, 52.
- Maclachlan, S., & Zalik, S. (1963). Plastid structure, chlorophyll concentration, and free amino acid composition of a chlorophyll mutant of barley. Canadian Journal of Botany, 41(7), 1053-1062.
- Yentsch, C. S., & Duxbury, A. C. (1956). Some of the Factors Affecting the Calibration Number of the Clarkeâ?Bumpus Quantitative Plankton Sampler 1. Limnology and Oceanography, 1(4), 268-273.
- Keller, T., & Schwager, H. (1977). Air pollution and ascorbic acid. European Journal of Forest Pathology, 7(6), 338-350.
- Ashraf, S. A., Siddiqui, A. J., Abd Elmoneim, O. E., Khan,M. I., Patel, M., Alreshidi, M., ... & Adnan, M. (2021). Innovations in nanoscience for the sustainable development of food and agriculture with implications on health and environment. Science of the Total Environment, 768, 144990.
- Chandrika, K. P., Pasala, R., Pandey, B. B., Sangeetha, B. M., Qureshi, A. A., Meena, S., & Yadav, P. (2024). Nanochelation: An efficient tool in plant nutrition management. In Nanofertilizer Delivery, Effects and Application Methods (pp. 15-33). Elsevier.
- Chandrika, K. P., Pasala, R., Pandey, B. B., Sangeetha, B. M.,Qureshi, A. A., Meena, S., & Yadav, P. (2024). Nanochelation: An efficient tool in plant nutrition management. In Nanofertilizer Delivery, Effects and Application Methods (pp. 15-33). Elsevier.
- Chaudhary, I., & Singh, V. (2020). Titanium dioxide nanoparticles and its impact on growth, biomass and yield of agricultural crops under environmental stress: A review. Res.J. Nanosci. Nanotechnol, 10(1).
- Chaudhary, I. J., & Rathore, D. (2021). Assessment of dose–response relationship between ozone dose and groundnut (Arachis hypogaea L) cultivars using Open Top Chamber (OTC) and Ethylenediurea (EDU). Environmental Technology & Innovation, 22, 101494.
- Chaudhary, I. J., & Rathore, D. (2021). Assessment of ozone toxicity on cotton (Gossypium hirsutum L.) cultivars: Its defensive system and intraspecific sensitivity. Plant Physiology and Biochemistry, 166, 912-927.
- Chaudhary, I. J., & Rathore, D. (2022). Effects of ambient and elevated ozone on morphophysiology of cotton (Gossypium hirsutum L.) and its correlation with yield traits. Environmental Technology & Innovation, 25, 102146.
- Helal, N. M., Khattab, H. I., Emam, M. M., NiedbaÅ?a, G., Wojciechowski, T., Hammami, I., ... & Hassan, H. M. (2022). Improving yield components and desirable eating quality of two wheat genotypes using Si and NanoSi particles under heat stress. Plants, 11(14), 1819.
- Kumar, D., Dhankher, O. P., Tripathi, R. D., & Seth, C. S. (2023). Titanium dioxide nanoparticles potentially regulate the mechanism (s) for photosynthetic attributes, genotoxicity, antioxidants defense machinery, and phytochelatins synthesis in relation to hexavalent chromium toxicity in Helianthus annuus L. Journal of Hazardous Materials, 454, 131418.
- Nile, S. H., Thiruvengadam, M., Wang, Y., Samynathan, R., Shariati, M. A., Rebezov, M., ... & Kai, G. (2022). Nano-priming as emerging seed priming technology for sustainable agriculture—recent developments and future perspectives. Journal of nanobiotechnology, 20(1), 254.
- Rai-Kalal, P., & Jajoo, A. (2021). Priming with zinc oxide nanoparticles improve germination and photosynthetic performance in wheat. Plant Physiology and Biochemistry, 160, 341-351.
- Rathore, D., & Chaudhary, I. J. (2021). Effects of tropospheric ozone on groundnut (Arachis hypogea L.) cultivars: Role of plant age and antioxidative potential. Atmospheric Pollution Research, 12(3), 381-395.
- Rhaman, M. S., Tania, S. S., Imran, S., Rauf, F., Kibria,M. G., Ye, W., ... & Murata, Y. (2022). Seed priming with nanoparticles: an emerging technique for improving plant growth, development, and abiotic stress tolerance. Journal of Soil Science and Plant Nutrition, 22(4), 4047-4062.
- Shelar, A., Singh, A. V., Maharjan, R. S., Laux, P., Luch, A., Gemmati, D., ... & Patil, R. (2021). Sustainable agriculture through multidisciplinary seed nanopriming: prospects of opportunities and challenges. Cells, 10(9), 2428.
- Simkin, A. J., Kapoor, L., Doss, C. G. P., Hofmann, T. A., Lawson, T., & Ramamoorthy, S. (2022). The role of photosynthesis related pigments in light harvesting, photoprotection and enhancement of photosynthetic yield in planta. Photosynthesis Research, 152(1), 23-42.
- Singh, H., Sharma, A., Bhardwaj, S. K., Arya, S. K., Bhardwaj, N., & Khatri, M. (2021). Recent advances in the applications of nano-agrochemicals for sustainable agricultural development. Environmental Science: Processes & Impacts, 23(2), 213-239.
- Tripathi, S., Mahra, S., Tiwari, K., Rana, S., Tripathi, D. K., Sharma, S., & Sahi, S. (2023). Recent advances and perspectives of nanomaterials in agricultural management and associated environmental risk: a review. Nanomaterials, 13(10), 1604.


