Research Article - (2025) Volume 8, Issue 1
“D-Neuron Protection / Activation (DPA) Therapy”: New Strategy for Treatment of Neuropsychiatric Illnesses
Received Date: Dec 18, 2024 / Accepted Date: Jan 15, 2025 / Published Date: Jan 23, 2025
Copyright: ©Â©2025 Keiko Ikemoto. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ikemoto, K. (2025).
Abstract
Recent progress of pharmacotherapy enabled anti-amyloid and anti-tau immunotherapy for Alzheimer disease. Yet, non-pharmacological factors, alimentation, sleep, exercise, stress-management or cognition stimulation, has also been emphasized. The D-neuron, that is β�?�phenylethylamine (PEA) neuron, or TAAR1 ligand neuron, localizes from D1 (spinal cord) to D18 (cerebral cortex) in caudo-rostral order of mammalian brains. In post-mortem brains with schizophrenia, D-neurons lacked in the nucleus accumbens (Acc, D16), a projection field of mesolimbic dopamine system. This lead to establish “D-cell hypothesis of mental illnesses”, and a novel therapy aimed at protection and activation of D-neuron, “D-neuron protection / activation (DPA) therapy”. As histochemical visualization of D-neurons is a no easy technique, D-neuron activity in the CNS should initially be evaluated indirectly by biomarkers, such as PEA, brain-derived neurotrophic factor (BDNF), tyramine, octopamine and β�?�endorphin, and biomarkers of inflammation, such as CRP, TNF and IL-6 in cerebrospinal fluid (CSF), serum or urine. Reliable biomarkers of mental / cognitive functions should also be employed. “DPA therapy” is a novel cellular level treatment of neuropsychiatric illnesses including dementia.
Keywords
TAAR1, Aromatic-L-Amino Acid Decarboxylase, β-Phenylethylamine, Dopamine, D-Neuron, Psychosis, D-Neuron Protection / Activation Therapy
Introduction
Clinicians note the influences of psychosocial impact on the onset and clinical course of mental illnesses including dementia in daily clinical examination.
More than 20 years ago, a double-blind test in clinical trial of Galantamine showed that the memory loss of a female outpatient with dementia dramatically recovered by a few months administration of a placebo, and that consequent Galantamine administration lead a sudden ambulatory worsening of physical symptoms. Since the Great East Japan Earthquake on March 11, 2011, the initial psychiatric treatment for about 500 suicide attempt inpatients through the nearest Ambulatory Center from the 1st Atomic Power Plant of Fukushima of Iwaki City General Hospital (now, Iwaki City Medical Center), a nuclear hospital, has been performed by a sole woman psychiatrist. This revealed various bio-psycho-socio-ethical factors related to mental health following the earthquake disaster. Notably, long-lasting influences of drug (alcohol, hypnotics, psychostimulants, etc.) dependence and / or sexual abuse on mental states and on behaviors of disaster victims made the disease state complex and / or treatment resistant [1]. The hospital stuffs have noticed that so-called “virgin killing”, being accepted in Iwaki region as virtue, despite a social taboo in the other regions, has been a trigger of women suicide (attempt). Though stress-induced neuroinflammation after disaster or abuse may be a risk factor of neurodegeneration which causes cognitive impairment, prospective studies on elderly neurocognitive impairment related to disaster of the Great East Japan Earthquake are neither known.
In spite of our great anticipation in elimination of amyloidβ(Aβ) protein from brains with Alzheimer disease of anti-Aβ immunotherapy, infusion reaction, and amyloid-related imaging abnormality (ARIA) in MRI findings, ARIA-E (edema/ effusion) and ARIA-H (hemosiderin deposit/hemorrhage) during administration of Lecanemab, Aducanumab (BioArctic AB and Eisai), or Donanemab (Eli Lilly) makes some clinicians hesitate in initiating or continuing prescription. Anti- microtubule binding region (MTBR) anti-tau immunotherapy to eliminate cerebrospinal fluid (CSF) MTBR-tau243 by E2814 (Eisai) administration was reported to proceeded to phase II clinical trial [2].
In addition to these powerful immunotherapies, necessity arises to revalue importance of non-pharmacological treatment and prevention of dementia, for example, alimentation, sleep, exercise, stress management or prevention of infection or inflammation [3].
In this context, protection of the D-neuron (trace amine neuron, type1) prior to disease onset, or D-neuron activation in clinical course of chronic neuropsychiatric diseases might be favorable method [4]. So, I propose “D-neuron protection / activation (DPA) therapy” for possible choice of the treatment and prevention of neuropsychiatric illnesses, such as neurocognitive disorders.
D Neuron and TAAR1
The trace amine (TA) neuron in the rat central nervous system (CNS) was described by Jaeger et al. in 1983 [5]. Initially, they defined “the non-monoaminergic aromatic L-amino acid decarboxylase (AADC)-containing cell”, and called the “D- cell” [5]. “D” means decarboxylation. AADC is an equivalent enzyme to dopa decarboxylase (DDC). The D-cell (=AADC-only cell) contains AADC but not dopaminergic nor serotonergic, which produces TAs, such as β-phenylethylamine (PEA), tyramine and tryptamine [5]. AADC is the rate-limiting enzyme for TA synthesis.
However, it is confusing that these TAs are also “monoamines”, as each one has one amino residue. It would be better to use the nomenclature of “TA cell, type 1” for D- cell, and “TA neuron, type 1” for D-neuron. There are other types of TAs which are not synthesized via AADC.
The D-neuron localizes in the mammalian CNS, from D1 (spinal cord) to D18 (cerebral cortex) in caudo-rostral order [5,6]. In the human nucleus accumbens (Acc), caudate nucleus (Ca) and putamen (Pu), there are D-neurons, though monkey homologous areas scarcely contained D-neurons (Figure, 1AB) [6-8]. In the ventral part of human Acc, the D-neuron localizes overlapping with the subventricular zone (SVZ), human NSC area, and regulates mesolimbic dopamine system (see review [6]). This neuronal circuit relates with reward, learning, addiction, substance abuse, paranoid-hallucinatory state and psychosis. Human mesolimbic dopamine D-neuron system, a part of PEA- dopamine system, may relate to higher mental function, including human faith or religion [9].
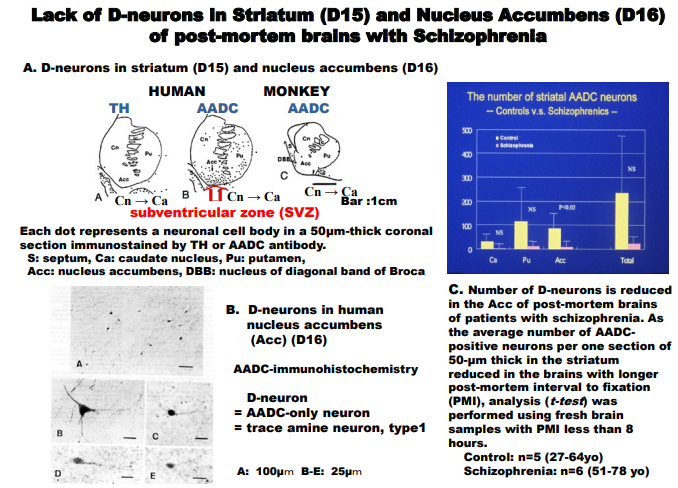
Figure Legends Figure. 1
A: Coronal sections through the caudate nucleus (Ca), putamen (Pu) and nucleus accumbens (Acc) of the human and monkey. The ventral margin of Acc coincides with the subventricular zone (SVZ) of lateral ventricle, NSC region.
B: AADC-immunostained coronal section through Acc of a post-mortem brain with no detectable neuropsychiatric illnesses.
C: Striato-accumbal D-neuron reduction in brains with schizophrenia.
The TA receptor, G-protein coupled receptor, located on chromosome focus 6q23.1 [10]. was cloned in 2001 [11,12]. The receptor has been shown to co-localize with DA or adrenaline transporters in monoamine neurons and to modulate the functions of monoamines [10]. TA receptors have 9 subtypes and among them 3 pseudogenes. PEA, tyramine, 3-iodothyronamine, 3-methoxytyramine, normetanephrine, and psychostimulants, for example methamphetamine and 3,4- methylenedioxymethamphetamine (MDMA) are TAAR1 ligands [13].
TAAR1 knockout mice showed schizophrenia-like behaviors with a deficit in prepulse inhibition [14]. TAAR1 stimulation increase of DA neurons in the midbrain ventral tegmental area (VTA) reduced firing frequency of VTA DA neurons [14,15]. This demonstrated a critical role of TAAR1 stimulation decrease for mesolimbic DA hyperactivity in schizophrenia.
Early in 1974, Sabelli and Mosnaim proposed “Phenylethylamine hypothesis of affective behavior”, indicating involvement of PEA in animal behaviours [16]. Level of PEA, a substrate for MAOB, elevated in the striatum of MAOB knockout mice, by 8-10 times of that of controls [17]. Indeed, MAOB enzyme active neurons, that is D-neurons, were densely packed in the Acc of mice which lack MAOA [18,19]. An initial clinical symptom frequently observed in first episode schizophrenia is disturbance of sleep- wake- rhythm, insomnia and daytime hypersomnia. A MAOB inhibitor, selegiline ameliorates daytime sleepiness of narcolepsy or other neuropsychiatric diseases, possibly by PEA increase via inhibition of PEA degradation.
D Cell Hypothesis of Schizophrenia
A novel theory, “D-cell hypothesis”, explains pathophysiology of mesolimbic DA hyperactivity of schizophrenia (Figure. 2A) [20]. By using pathological and legal autopsy brains of patients with schizophrenia and an immunohistochemical method, I showed lack of striato-accumbal D-neurons (D15, D16) in post-mortem brains of patients with schizophrenia (see Figure. 1C) [21,22]. The reduction of D-neurons was significant (t-test, p<0.05) in Acc of schizophrenia. I proposed “D-cell hypothesis of schizophrenia”, a link of dopamine hypothesis and NSC dysfunction hypothesis, showing NSC dysfunction-based D-neuron reduction being cellular basis of mesolimbic DA hyperactivity of schizophrenia, and predicted prospectiveness of TAAR1 agonists for treatment of schizophrenia. This hypothesis has finally been verified by effectiveness of a TAAR1 agonist in reducing symptoms of schizophrenia [23].
Figure 2:
A: Scheme of “D-cell hypothesis of mental illness”.
In a brain with schizophrenia, dysfunction of neural stem cells (NSC) in the subventricular zone (SVZ) of lateral ventricle causes D-neuron decrease in the striatum and nucleus accumbens (Acc). This induces TA (=β-phenylethylamine (PEA)) decrease in these nuclei and TAAR1 stimulation decrease into DA terminals of VTA DA neurons, causing firing frequency increase in VTA DA neurons.12, 13 This increases DA release and DA turnover in the Acc (mesolimbic DA hyperactivity). Striatal DA hyperactivity causes excessive D2 stimulation of NSC in the striatum and inhibits forebrain NSC proliferation, which accelerates D-neuron decrease and accelerates mesolimbic DA hyperactivity.
B: Novel strategies for treatment of mental illness, indicated by “D-cell hypothesis”
The cycle of disease progression is to be inhibited by intervention (shown by ×) with TAAR1 agonists, D2 antagonists, or neurotrophic substances, for example, brain-derived neurotrophic factor (BDNF), lithium, anticonvulsants or antidepressants. Stress, aging, and alcohol intake suppresses NSC functions, which may also cause psychotic state.
In brains of patients with schizophrenia, dysfunction of NSC in the subventricular zone of lateral ventricle (SVZ) causes D-neuron decrease in the striatum and Acc [24]. This induces TA decrease in these nuclei. Lateral ventricle enlargement seen in schizophrenia brain imaging is also due to NSC dysfunction [25,26]. TAAR1 stimulation decrease in DA terminals of VTA DA neurons, caused by TA decrease, increases firing frequency of VTA DA neurons [14,15]. This increases DA release and DA turnover in the Acc, resulting in mesolimbic DA hyperactivity (Figure. 2A). D2 stimulation of NSC in the striatum is shown to inhibit forebrain NSC proliferation [27]. Striato-accumbal DA hyperactivity may accelerate D-neuron decrease, which accelerates hyperactivity of mesolimbic DA system. D2 blocking agents in pharmacotherapy of schizophrenia block inhibition to forebrain NSC proliferations (Figure. 2B) [27]. This is consistent with clinical evidences that initial pharmacotherapy using D2 antagonists is critical for preventing progressive pathognomonic procedures of schizophrenia [28]. Duration of untreated psychosis is a predictor of long-term outcome of schizophrenia [28]. Importance of early intervention for first episode schizophrenia by using D2 antagonist has been emphasized [28]. D2 antagonists block disease progression (Figure. 2B).
D-cell hypothesis not only links DA hypothesis with NSC dysfunction hypothesis (Figure. 2A), but also explains the mechanisms of disease progression of schizophrenia (Figure. 2A). To inhibit the cycle of pathological progression, intervention indicated by × in Figure. 1B is effective. In animal studies, effectiveness of TAAR1 ligands for schizophrenia-like symptoms of schizophrenia model animals has been shown [15]. Recent clinical trial studies have shown the efficacy of a novel agent, SEP- 363856, TAAR1 full agonist and 5-hydroxytryptophan type 1A receptor partial agonist for treatment of schizophrenia [23]. Efficacy of TAAR1 full agonist to schizophrenia seems to be equivalent to that of DA agonist for treatment of Parkinson’s disease which lacks DA neurons in the midbrain substantia nigra.
Disease progression is inhibited also by neurotrophic substances (Figure. 2B), such as, brain-derived neurotrophic factor (BDNF) [36], lithium, anticonvulsants, or antidepressants. Neurotrophic effects of these substances activate NSC functions, and inhibit striato- accumbal D-neuron decrease. Stress, aging, and alcohol intake suppresses NSC functions, which may cause vulnerability to psychotic state.
“D-cell hypothesis” shows D-cell-involved etiological dynamism in also wide spectrum of psychotic states in neurological as well as psychiatric illnesses. NSC functions may effect D-neuron activities, clinical course and prognoses of neuropsychiatric illnesses.
Novel D Neuron Protection / Activation (DPA) Therapy
TAAR1 agonism, that is, non-D2-receptor binding mechanism, is focused on, in the field of medicinal chemistry of novel antipsychotics, such as SEP-363856 [23,29]. SEP- 363856 is trace amine-associated receptor 1 (TAAR1) full agonist and also 5- hydroxytryptamin 1A (5-HT 1A) receptor partial agonist [23]
Shang et al. and Liu et al. revealed detailed receptor signals of several TAAR1 agonists including β-phenylethylamine (PEA), methamphetamine, or SEP-363856. Where as, D-neuron signals have not yet been clarified [30,31].
However, it is clear that accumbal D-neuron reduction in the post-mortem brains with chronic schizophrenia is not only simply because pathophysiology of schizophrenia, but also mixed effects of lobectomy, insulin therapy, electro-convulsive therapy (ECT), long-term pharmacotherapy and aging-caused neurodegeneration. This lead us to consider a therapeutic strategy for neuroprotection so as to suppress degenerative pathophysiology, and to conserve and activate damaged neurons. A novel treatment aimed at protection and activation of D-neuron would be a prospective therapy.
As D-neurons visualization is a difficult histochemical technique, indirect evaluation of D-neuron activity in the CNS should be established. Biomarkers such as PEA, brain-derived neurotrophic factor (BDNF), tyramine, octopamine and βâ?ÂÂendorphin, and biomarkers of inflammation, such as CRP, TNF and IL-6 in cerebrospinal fluid (CSF), serum or urine, should be monitored, and reliable biomarker analysis of mental / cognitive functions should also be done.
Difficulty in Histochemical D Neuron Visualization
There are several reasons why D-neuron signals remain to be unknown. One reason is due to the paucity of PEA amount, produced by D-neurons in the mammalian brains. Another reason is species differences of localization and role of D-neurons in the animal CNS. In rodent striatum, AADC was expressed in astrocytes when immunestained by using old post-parafolmaldehyde (PFA)- fixed brain slices, after storage for more than 1 week (unpublished data). Additionally, MAOB was expressed in astrocytes of wild type rat striatum, being a confusing finding compared to results in mice, in which we found MAOB enzyme activity in neurons [18,32].
Human CNS D-neurons have been visualized by using immunohistochemical method and a specific antibody against AADC, a rate-limiting enzyme of TA synthesis, as a marker [21,22]. Another marker of D-neuron is MAOB, a degrading enzyme of TA. MAOB has been visualized by MAO enzyme histochemistry applied to brain samples of a MAOA knockout animal [18,19].
In humans, AADC immunohistochemistry is a sole reliable method in visualizing D-neuron. However, the limitation is to employ a PFA-fixed free-floating method for keeping antigenicity of AADC, and to visualize smaller amount of AADC in D- neuron, than that in serotonin neuron or dopamine neuron [21,22].
Due to disasters of the Great East Japan Earthquake from March 11, 2011 and COVID-19 pandemic, D-neuron research did not progress. AADC-immunostained glass slides of post-mortem brain specimens with or without clinical history of neuropsychiatric illnesses, obtained by legal or pathological autopsy, had been put under my repository manage for more than 10 years.
In post-COVID-19 period, it is timely to progress D-neuron research and to establish a novel “DPA therapy” from cellular level direction for treatment of neuropsychiatric illnesses such as dementia. The neuroprotection and activation of D- neuron would possibly delay the disease onset, and may also achieve sooner recovery from psychotic symptoms or neurocognitive impairment.
References
- Ikemoto, K. (2015). Factors of increase of women’s suicidal attempt after Great East Japan Earthquake. 6th World Congress on Women’s Mental Health (IAWMH2015), 86.
- Valiukas, Z., Ephraim, R., Tangalakis, K., Davidson, M., Apostolopoulos, V., & Feehan, J. (2022). Immunotherapies for Alzheimer’s disease—a review. Vaccines, 10(9), 1527.
- Berg-Weger, M., & Stewart, D. B. (2017). Non-pharmacologic interventions for persons with dementia. Missouri medicine, 114(2), 116.
- Ikemoto, K. (2016). So Called - D-Neuron. Journal of Cytology & Histology, 7(3).
- Jaeger, C. B., Teitelman, G., Joh, T. H., Albert, V. R., Park,D. H., & Reis, D. J. (1983). Some neurons of the rat central nervous system contain aromatic-L-amino-acid decarboxylase but not monoamines. Science, 219(4589), 1233-1235.
- Ikemoto, K. (2016). Involvement of so-called D-neuron (trace amine neuron) in the pathogenesis of schizophrenia: D-cell hypothesis. In Trace Amines and Neurological Disorders (pp. 295-307). Academic Press.
- Ikemoto K, Satoh K, Maeda T, Fibiger HC. (1995). Neurochemical heterogeneity of the primate nucleus accumbens, Experimental Brain Research 104(2): 177-190.
- Ikemoto, K., Satoh, K., Kitahama, K., Geffard, M., & Maeda,T. (1996). Experimental Brain Research study of dopaminergic structures in the medial subdivision of the monkey nucleus accumbens. Experimental brain research, 111(1), 41-50.
- Ikemoto, K., & Nakamura, M. (2004). Forced deprogramming from a religion and mental health: A case report of PTSD. International Journal of Law and Psychiatry, 27(2), 147-155.
- Lindemann, L., Meyer, C. A., Jeanneau, K., Bradaia, A., Ozmen, L., Bluethmann, H., ... & Hoener, M. C. (2008). Trace amine-associated receptor 1 modulates dopaminergic activity. Journal of Pharmacology and Experimental Therapeutics, 324(3), 948-956.
- Bunzow, J. R., Sonders, M. S., Arttamangkul, S., Harrison, L. M., Zhang, G. E., Quigley, D. I., ... & Grandy, D. K. (2001). Amphetamine, 3, 4-methylenedioxymethamphetamine, lysergic acid diethylamide, and metabolites of the catecholamine neurotransmitters are agonists of a rat trace amine receptor. Molecular Pharmacology, 60(6), 1181-1188.
- Borowsky, B., Adham, N., Jones, K. A., Raddatz, R.,Artymyshyn, R., Ogozalek, K. L., ... & Gerald, C. (2001). Trace amines: identification of a family of mammalian G protein-coupled receptors. Proceedings of the National Academy of Sciences, 98(16), 8966-8971.
- Zucchi, R., Chiellini, G., Scanlan, T. S., & Grandy, D.K. (2006). Trace amine-associated receptors and theirligands. British journal of pharmacology, 149(8), 967-978.
- Wolinsky, T. D., Swanson, C. J., Smith, K. E., Zhong, H., Borowsky, B., Seeman, P., ... & Gerald, C. (2007). The Trace Amine 1 receptor knockout mouse: an animal model with relevance to schizophrenia. Genes, Brain and Behavior, 6(7), 628-639.
- Revel, F. G., Moreau, J. L., Pouzet, B., Mory, R., Bradaia, A., Buchy, D., ... & Hoener, M. (2013). A new perspective for schizophrenia: TAAR1 agonists reveal antipsychotic-and antidepressant-like activity, improve cognition and control body weight. Molecular psychiatry, 18(5), 543-556.
- Sabelli, H. C., & Mosnaim, A. D. (1974). Phenylethylamine hypothesis of affective behavior. American Journal of Psychiatry, 131(6), 695-699.
- Grimsby, J., Toth, M., Chen, K., Kumazawa, T., Klaidman, L., Adams, J. D., ... & Shih, J. C. (1997). Increased stress response and β–phenylethylamine in MAOB–deficient mice. Nature genetics, 17(2), 206-210.
- Ikemoto, K., Kitahama, K., Seif, I., Maeda, T., De Maeyer, E., & Valatx, J. L. (1997). Monoamine oxidase B (MAOB)- containing structures in MAOA-deficient transgenic mice. Brain Research, 771(1), 121-132.
- Ikemoto, K., Kitahama, K., Maeda, T., Tokunaga, Y., Valatx, J. L., De Maeyer, E., & Seif, I. (1997). Electron- microscopic study of MAOB-containing structures in the nucleus accumbens shell: using MAOA-deficient transgenic mice. Brain Research, 771(1), 163-166.
- Ljungdahl, Fuxe, K., & Johansson, O. (1974). Dopamine nerve terminals in the rat limbic cortex: aspects of the dopamine hypothesis of schizophrenia. Science, 184(4133), 177-179.
- Kitahama, K., Ikemoto, K., Jouvet, A., Nagatsu, I., Sakamoto, N., & Pearson, J. (1998).Aromatic L-amino acid decarboxylase- and tyrosine hydroxylase-immunohistochemistry in the adult human hypothalamus. Journal of Chemical Neuroanatomy, 16(1), 43-55.
- Ikemoto, K., Amano, R., Ishibe, A., Nishimura, A., Nishi, K., & Nagatsu, I. (2000). Quantitative analysis of tyrosine hydroxylase-, aromatic L-amino acid decarboxylase-or phenylethanolamine-N-methyltransferase-immunoreactive neurons in the human medullary C1 region. Acta Histochemica et Cytochemica, 33(4), 259-265.
- Koblan, K. S., Kent, J., Hopkins, S. C., Krystal, J. H., Cheng, H., Goldman, R., & Loebel, A. (2020). A non–D2-receptor- binding drug for the treatment of schizophrenia. New England Journal of Medicine, 382(16), 1497-1506.
- Sanai, N., Tramontin, A. D., Quinones-Hinojosa, A., Barbaro,N. M., Gupta, N., Kunwar, S., ... & Alvarez-Buylla, A. (2004). Unique astrocyte ribbon in adult human brain contains neural stem cells but lacks chain migration. Nature, 427(6976), 740- 744.
- Degreef, G., Ashtari, M., Bogerts, B., Bilder, R. M., Jody, D. N., Alvir, J. M. J., & Lieberman, J. A. (1992). Volumes of ventricular system subdivisions measured from magnetic resonance images in first-episode schizophrenicpatients. Archives of General Psychiatry, 49(7), 531-537.
- Fernandes, B. S., Steiner, J., Berk, M., Molendijk, M. L., Gonzalez-Pinto, A., Turck, C. W., ... & Gonçalves, C. A. (2015). Peripheral brain-derived neurotrophic factor in schizophrenia and the role of antipsychotics: meta-analysis and implications. Molecular Psychiatry, 20(9), 1108-1119.
- Kippin, T. E., Kapur, S., & van der Kooy, D. (2005). Dopamine specifically inhibits forebrain neural stem cell proliferation, suggesting a novel effect of antipsychotic drugs. Journal of Neuroscience, 25(24), 5815-5823.
- Penttilä, M., Jääskeläinen, E., Hirvonen, N., Isohanni, M., & Miettunen, J. (2014). Duration of untreated psychosis as predictor of long-term outcome in schizophrenia: systematic review and meta-analysis. The British Journal of Psychiatry, 205(2), 88-94.
- Ikemoto, K. (2024). D-Neuron, Ligand Neuron of TraceAmine-Associated Receptor 1 (TAAR1): Key of Novel Non-D2 Receptor-Binding Antipsychotics. Journal of Biotechnology and Biomedicine, 7, 15-20.
- Liu, H., Zheng, Y., Wang, Y., Wang, Y., He, X., Xu, P., ... &Xu, F. (2023). Recognition of methamphetamine and other amines by trace amine receptor TAAR1. Nature, 624(7992), 663-671.
- Shang, P., Rong, N., Jiang, J. J., Cheng, J., Zhang, M. H., Kang, D., ... & Sun, J. P. (2023). Structural and signaling mechanisms of TAAR1 enabled preferential agonist design. Cell, 186(24), 5347-5362.
- Arai, R., Kimura, H., & Maeda, T. (1986). Topographic atlas of monoamine oxidase-containing neurons in the rat brain studied by an improved histochemical method. Neuroscience, 19(3), 905-925.


