Research Article - (2024) Volume 9, Issue 2
Diagnostic Value of Left Atrial Strain in Heart Failure with Preserved Ejection Fraction
2Hematology and biology department, Internal Security Forces Hospital of Marsa, Tunisia
Received Date: Feb 01, 2024 / Accepted Date: Jun 05, 2024 / Published Date: Jun 14, 2024
Copyright: ©Ã??Ã?©2024 Ridha Fekih, et, al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Fekih, R., Antit, S., Abdelhedi, M., Dridi, K., Boussabeh, E., et al. (2024). Diagnostic Value of Left Atrial Strain in Heart Failure with Preserved Ejection Fraction. Cardio Open, 9(2), 01-08.
Abstract
Introduction: Despite different ultrasound parameters, left ventricular filling pressures (LVFP) assessment remains inconclusive in some cases. We aimed to determine the contribution of left atrial strain (LAS) in estimating of LVFP in patients suspected of having heart failure with preserved ejection fraction (HFpEF).
Methods: This was a monocentric study, carried out in the cardiology department of the Interior Security Forces Hos- pital of Marsa, between October 2021 and March 2022. Patients had a physical examination, a biological assessment and an ultrasound examination at rest and, if necessary, during exercise. We investigated the performance of LAS com- ponents (Peak atrial longitudinal strain (PALS), peak atrial contraction strain (PACS) and conduit function (FnC) in predicting LVFP rising.
Results: We enrolled 73 patients. The mean age was 61 ± 12 years old with a female predominance (57.5%). The me- dian PALS was 29.3% [21.4-32.4]. The mean PACS and FnC values were 13.4% ± 4.9 and 13.7% ± 4.7, respectively. Patients were categorized into two finals groups according to LVFP: Group A= high LVFP at rest or during exercise (25 = 34%) and Group B = not-high LVFP at rest and during exercise (48 = 66%). Patients with high LVFP had lower LAS parameters and higher NT-Pro BNP levels. LAS had negative correlations with E/e' ratio and NT-Pro BNP. PALS was an independent predictor of LVFP raising (HR = 0.711; 95% CI: 0.513 - 0.986; p = 0.041).
Conclusion: LAS is a simple, reproducible and sensitive ultrasound parameter for the diagnosis of HFpEF.
Keywords
Heart Failure with Preserved Ejection Fraction, Echocardiography, Left Atrial Strain, Left Ventricular Filling Pressures
Introduction
Heart failure (HF) is a major global health problem that continues to expand as the population ages with a proportional rise in its economic and social impact [1,2]. The diagnosis of HF is based on clinical, electrocardiographic, biological (natriuretic peptides) and echocardiographic data. It is simple in case of reduced left ventricular ejection fraction (LVEF) but more complex in case of preserved one (≥ 50%). Symptoms and signs of HF are neither sensitive nor specific especially in case of HF with preserved ejection fraction (HFpEF) in which even natriuretic peptides may be negative or moderately increased due to other comorbidities (such as old age, atrial fibrillation, renal failure). The ultrasound is the key test to distinguish between the different phenotypes of HF and to identify structural and functional abnormalities that leads to the diagnosis of HFpEF, such as a diastolic dysfunction and high left ventricular filling pressures (LVFP) [3]. Assessment of LVFP is an important step for both diagnosis and management of patients with HFpEF [4,5]. Although invasive procedures are considered as the "gold standard" for measurement of these pressures, conventional echocardiography is commonly used as a noninvasive alternative. Since none of the ultrasound methods used can measure them directly, several parameters and algorithms have been proposed by American and European associations initially in 2009 and then simplified in 2016 [6]. However, in some cases, the assessment of LVFP remains undetermined because of the absence of one of the required ultrasound parameters. Recently, the analysis of the longitudinal deformation of the left atrial by speckle tracking or LA strain (LAS), a new method developed to study atrial function, has been used to assess left diastolic ventricular function in several heart diseases [7-10]. Thus, we aimed to assess the contribution of LA strain in the estimation of LVFP in patients suffering from exertional symptoms and with preserved LVEF.
Methods
Patients and Study Design
This was a prospective mono-centric cross-sectional study,conducted from October 2021 to March 2022 in the Cardiology Department of the Internal Security Forces Hospital of Marsa Tunisia. The hospital’s ethics committee approved the study. All patients before participation in the study obtained informed verbal consent.
We included consecutive patients presenting exertional symptoms and without known structural heart diseases (significant valvular heart disease, prior valve replacement or repair, LVEF < 50%, infiltrative or hypertrophic cardiomyopathy, constrictive pericarditis, congenital heart disease, idiopathic pulmonary hypertension).
Patients with acute heart failure or with rhythm other than sinus at the time of the ultrasound examination, or with severe anemia and patients in whom one of the above-mentioned heart diseases was discovered on resting transthoracic echocardiography (TTE) or unable to perform exercise stress echocardiography (ESE) if indicated, were excluded from our study.
Patient demographic and anthropometric data, cardiovascular risk factors and comorbidities were collected. Symptoms (dyspnea, asthenia, reduced exercise tolerance) leading to the diagnosis of HF were noted. Systolic blood pressure (SBP), diastolic blood pressure (DBP), and heart rate (HR) at rest and during exercise were measured.
All patients performed a biological examination including hemoglobin (Hb), N terminal pro brain natriuretic peptide (NT- Pro BNP), and creatinine levels with calculation of creatinine clearance by the MDRD formula [11].
Echocardiographic Analysis
All patients underwent an ultrasound examination at rest using a Philips EPIQ 7C echocardiograph with simultaneously and continuous electrocardiographic tracing. All measurements were performed according to the recommendations of the American Society of Echocardiography and the European Association of Cardiovascular Imaging (ASE/EACVI) [12].
The following ultrasound parameters were collected at resting TTE: LVEF, left ventricular end-diastolic diameter (LVED), left ventricular mass index (LVMI), left ventricular end-diastolic volume index (LVEDV), maximum velocities of the E wave (E) and the A wave (A), E/A ratio, Septal velocity, Lateral velocity, tricuspid regurgitation (TR) velocity, left atrial volume index (LAVI), Average ratio and left ventricular global longitudinal strain (SLG).
LAS was measured as follow: Two loops of the left atrial were obtained in apical four and two-chamber views after moving the focus to the left atrial and reducing the size of the sector and emphasizing the distinction between the myocardium and extra cardiac structures, during a brief apnea and with a stable electrocardiogram recording and frame rate between 60 and 80 frames per second. Atrial strain curves were generated after semi-automatic tracing of the endocardial limits Figure 1.
Figure 1: Left Atrial Strain Measurement in Four and Two-Chambers Views
Abbreviations: PALS: Peak Atrial Longitudinal Strain; PACS: Peak Atrial Contraction Strain
Peak atrial longitudinal strain (PALS) is defined by the first peak of the positive atrial longitudinal strain at the beginning of the QRS complex. The second positive peak defines peak atrial contraction strain (PACS), lower than the first, which corresponds to the period before the atrial contraction (after the onset of the P wave on the electrocardiogram). The PALS and PACS values analyzed were averaged on apical four and two- chamber views. The conduit function (FnC) is the difference between PALS and PACS. The normal values of PALS, PACS an FnC were 39.4% (27.6 to 59.8%), 17.4% (14 to 25%) and 23% ± 2, respectively [13].
The LVFP at rest were assessed by applying the decision algorithm published by ESA/EACVI in 2016 with three possible profiles: normal, high, or indeterminate [6]. Those with normal or indeterminate LVFP at rest underwent ESE, according to the recommendations of the European Society of Cardiology and the following protocol: On a semi-supine bicycle, the patient should pedal with a speed of 60 rpm starting with a low workload at 15W and with a gradual increase of 5W every minute, until a submaximal target heart rate of 100-110/min (before the fusion of the E and A waves) or until the patient develops limiting symptoms. In both cases, the effort was considered as maximal and at this time the E wave, A wave, lateral e' velocity, septal e' velocity, and the ratio average E/e' ratio were measured [14].
Exercise LVFP was considered elevated if the average E/e' ratio at peak stress increases to ≥15. At the end of this assessment (at rest and during exercise), patients were classified, firstly, into three groups according to the estimated LVFP: Group 1 (high LVFP at rest), Group 2 (normal or indeterminate LVFP at rest but raised during exercise) and Group 3 (normal or indeterminate LVFP at rest and normal during exercise)
Statistical Analysis
Data were recorded and analyzed using IBM SPSS Statistics 23 software. The baseline characteristics were summarized using mean ± standard deviation (SD) or median ± 25-75 percentile levels, where appropriate, for continuous data, and counts with percentages for categorical data. The Kolmogorov-Smirnov test was used to evaluate whether the distribution of continuous variables was normal. Comparisons of means on independent series were performed by Student's t-test for independent series. Comparisons of percentages on independent series were made by the Pearson chi-square test. The links between two quantitative variables were assessed by Pearson's correlation coefficient (r). A correlation was considered strong if r ≥ 0.5 mildly if 0.3 ≥ r < 0.5 and weak if r < 0.3. The determination of threshold values of the studied parameters was done by analyzing their receiver operating characteristic (ROC) curves with comparison of the areas under the curve (AUC) using the Delong method. Binary logistic regression analysis was performed for the multivariate study. In all statistical tests, the significance level was set at 0.05.
Results
General Characteristics
A total of 73 patients were eligible for the study. Patient characteristics are summarized in Table 1. The mean age was 61 ± 12 years old. A female predominance was noted: 42 female patients (57.5%) and 31 male patients (42.5%). Hypertension and diabetes were the most common cardiovascular risk factors, being found in 81% (n = 59) and 51% (n = 37) of patients, respectively. Ten patients (14%) had coronary artery disease and 92% (n = 67) were either on overweight or obese. Exertional dyspnea (86%, n = 63) and poor exercise capacity (26%, n = 19) were the two most frequent complains.
|
|
General population (73=100%) |
Group A (25=34%) |
Group B (48=66%) |
P value |
|
Sex Male/Female (%) |
31 (42.5)/42 (57.5) |
10 (40)/15 (60) |
21 (44)/27 (56) |
0.807 |
|
Age (years) |
61 ± 12 |
67 ± 12 |
57 ± 10 |
< 0.001 |
|
Hypertension (n%) |
59 (81) |
24 (96) |
35 (73) |
0.017 |
|
Smoking (n %) |
12 (16) |
5 (20) |
7 (15) |
0.551 |
|
Dyslipidemia (n%) |
31 (42.5) |
8 (32) |
23 (48) |
0.192 |
|
Diabetes (n %) |
37 (51) |
16 (64) |
21 (44) |
0.140 |
|
CKD (n %) |
9 (11) |
8 (32) |
1 (2) |
0.001 |
|
Overweight/obesity (n %) |
67 (92) |
21 (84) |
46 (96) |
0.172 |
|
Coronary artery disease (n %) |
10 (14) |
3 (12) |
7 (15) |
0.974 |
|
SBP (mmHg) |
140 [130-145] |
145 [138-150] |
135 [125-140] |
0.008 |
|
DBP (mmHg) |
80 [77-90] |
85 [80-92] |
80 [75-84] |
0.005 |
|
Heart ate (b/m) |
80 [72-86] |
81 [76-86] |
77 [70-85] |
0.474 |
|
creatinine level (μmol/l) |
66 [56-86] |
86 [62-101] |
62 [55-77] |
0.002 |
|
creatinine clearance (ml/min/1.73m²) |
96 ± 33 |
79 ± 30 |
105 ± 31 |
0.001 |
|
Hemoglobin level (d/dl) |
12.9 ± 1.4 |
12.5 ± 1.9 |
13 ± 1.1 |
0.247 |
|
NT Pro-BNP (pg/ml) |
85 [50-195] |
282 [108-480] |
59 [35-101] |
< 0.001 |
|
LVFF (%) |
65 ± 6 |
64 ± 6 |
66 ± 5 |
0.089 |
|
LVED (mm) |
49 ± 5 |
50 ± 5 |
48 ± 5 |
0.134 |
|
LVEDV (ml/kg/m²) |
39 [34-47] |
38 [33-51] |
39 [35-44] |
0.919 |
|
LVMI (g/m²) |
89 ±23 |
102 ± 27 |
83 ± 19 |
0.002 |
|
LAVI (ml/m²) |
35 ± 12 |
45 ± 12 |
30 ± 7.3 |
< 0.001 |
|
Rest E/A ratio |
1.01 ± 0.43 |
1.2 ± 0.58 |
0.93 ± 0.28 |
0.002 |
|
Rest average e’ (cm/s) |
8.8 ± 2.4 |
7.3 ± 1.7 |
9.6 ± 2.3 |
0.001 |
|
Average E/e’ ratio at rest |
9.5 ± 3.6 |
13 ± 3.3 |
7.7 ± 2 |
< 0.001 |
|
TR velocity (cm/s) |
2.3 [2-2.7] |
2.9 [2.5-3.2] |
2.1 [2-2.4] |
< 0.001 |
|
GLS (%) |
-22 [-24_-21] |
-21.5 [-23_-19] |
-22 [-24_-21] |
0.127 |
|
PALS % |
29 [24-32] |
20 [16-23] |
31 [28-33] |
< 0.001 |
|
PACS (%) |
13.4 ± 4.9 |
9.4 ± 4 |
15.5 ± 3.9 |
< 0.001 |
|
FnC (%) |
13.7 ± 4.7 |
10 ± 3 |
15.5 ±4.2 |
< 0.001 |
|
Average E/e’ ratio at exercise |
9.9 ± 3.3 |
16 ± 1 |
8.8 ± 2.1 |
< 0.001 |
Data are presented as mean ± SD, median (interquartile range), or count and percentage of patients. Bold values indicate p < 0.05. Abbreviations: CKD: chronic kidney disease; SBP: systolic blood pressure: DBP: diastolic blood pressure; LVEF: left ventricular ejection fraction; LVED: left ventricular end-diastolic diameter; LVEDV: left ventricular end-diastolic volume index; LVMI: left ventricular mass index; LAVI: left ventricular atrial index; TR: tricuspid regurgitation; GLS: global longitudinal strain; PALS: peak atrial longitudinal strain; PACS: peak contraction longitudinal strain; Fnc: conduit function.
Table 1: Patient Characteristics According to Left Ventricular Filling Pressure
Mean hemoglobin value was 12.9 ± 1.4 g/dl, whereas medi- an NT-Pro BNP and creatinine values were 85 [50-195] pg/ml and 66 [56-86] μmol/l, respectively. Ten patients had creatinine clearance <60 ml/min. The mean LVEF was 65 ± 6.3%. The median GLS was -22% [-24_-21]. The median PALS was 29.3% [21.4-32.4]. The mean PACS and FnC values were 13.4% ± 4.9 and 13.7% ± 4.7, respectively. Assessment of rest LVFP classi- fied 17 patients (23%) as having high LVFP, 51 patients (70%) as having normal pressures and 5 patients (7%) as having inde- terminate pressures. Of the 56 patients (77%) who had not high LVFP at rest, only 8 patients (11%) had raised filling pressures on ESE Figure 2.
Figure 2: Protocol of Left Ventricular Felling Pressure Assessment
Abbreviations: LVFP: left ventricular filling pressure
Analytic Study
Based on these findings, patients categorized into two finals groups according to LVFP: Group A = high LVFP at rest or during exercise (25 = 34%) and Group B = not-high LVFP at rest and during exercise (48 = 66%) The comparison of clinical, biological, and ultra-sonographic characteristics between these groups reported in Table 1. Patients from group A were significantly older (p < 0.001) with a significantly higher prevalence of hypertension (p = 0.017) and chronic kidney disease (p = 0.001). SBP (p = 0.008) and DBP (0.005) were significantly higher in those patients.
Patients from group A had higher creatinine levels (p = 0.002) and lower clearance (p = 0.001). The NT Pro-BNP level was significantly higher in this group (p < 0.001). Given that the ASE/EACVI guidelines were followed for the evaluation of LVFP, the ultrasound parameters of diastolic function were significantly associated with LVFP including E/A ratio (p = 0.002), average E/e' ratio at rest (p < 0.001), TR velocity (p < 0.001), rest average e' velocity (p = 0.001), LAVI (p < 0.001), and average E/e' ratio at stress (p < 0.001). LVMI was significantly associated with higher LVFP (p = 0.002).
LAS analysis showed that PALS (p < 0.001), PACS (p < 0.001), and Fnc (p < 0.001) were significantly lower in-group A compared with group B. To further explore the relation between LAS components and the other parameters of HFpEF; we performed a bivariate analysis, as showed in Table 2.
|
|
PALS |
PACS |
FnC |
|
Rest average e’ |
R = 0.349; P= 0.002 |
R= 0.154; P= 0.093 |
R= 0.480; P< 0.001 |
|
Average E/e’ ratio at rest |
R = -0.647; P< 0.001 |
R= -0.539; P< 0.001 |
R= -0.513; P< 0.001 |
|
LAVI |
R = -0.624; P< 0.001 |
R= -0.476; P< 0.001 |
R= -0.526; P< 0.001 |
|
TR velocity |
R = -0.523; P< 0.001 |
R= -0.436; P< 0.001 |
R= -0.405; P< 0.001 |
|
NT Pro-BNP |
R = -0.551; P< 0.001 |
R= -0.440; P< 0.001 |
R= -0.419; P< 0.001 |
|
Average E/e’ ratio at stress |
R = -0.539; P< 0.001 |
R= -0.373; P= 0.005 |
R= -0.428; P= 0.001 |
Abbreviations: PALS: peak atrial longitudinal strain; PACS: peak contraction longitudinal strain; Fnc: conduit function; LAVI: left ventricular atrial index; TR: tricuspid regurgitation.
Table 2: Left Atrial Strain Components and the Other Parameters of Heart Failure with Preserved Ejection Fraction on Bivariate Analysis
Almost all of LAS components were significantly correlated to the ultrasound and biological elements of HFpEF. There was a strong and negative correlation between PALS and average E/e’ ratio at rest (r = -0.647; P < 0.001), LAVI (r = -0.624; P < 0.001), TR velocity (r = -0.523; P < 0.001), NT Pro-BNP (r = -0.551; P < 0.001) and average E/e’ ratio at stress (r = -0.539; P < 0.001). PACS was negatively and strongly correlated with average E/e’ ratio at rest (r = -0.539; P < 0.001) while a strong and negative correlation was found between FnC and average E/e’ ratio at rest (R = -0.513; P < 0.001) and LAVI (r = -0.526; P < 0.001).
We performed ROC curve analysis to assess the performance of LAS components in the prediction of high LVFP (group A) (Figure 3 and Table 3). All LAS components had a good perfor- mance to detect high LVFP (group A). PALS was the best pre- dictor of group A but this superiority was only significant com- pared to Fnc (p = 0.02). The ROC curves analyses were used to identify the cut-off values of LAS components predicting high LVFP. Thus, PALS < 24 (sensitivity of 82% and specificity of 85%) increased the likelihood of being from group A by 5.4 (RR = 5.4; 95% CI 2.21 – 13.5; p < 0.001). PACS < 13 (sensitivity of 80% and specificity of 78%) increased the likelihood of being from group A by 3.8 (RR = 3.8; 95% CI 1.73 – 8.56; p < 0.001). For FnC, a cut-off value < 14 (sensitivity of 85% and specificity of 70%) increased the likelihood of being from group A by 8.5 (RR = 8.5; 95% IC 2.24 – 32.9; p < 0.001).
Figure 3: Receiver Operating Characteristic Curves of Left Atrial Strain Components to Predict High Left Ventricular Filling Pressures
Abbreviations: PALS: peak atrial longitudinal strain; PACS: peak contraction longitudinal strain; Fnc: conduit function.
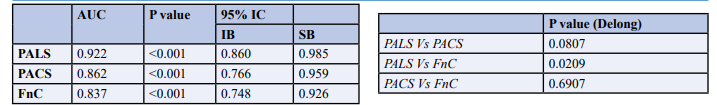
Abbreviations: PALS: peak atrial longitudinal strain; PACS: peak contraction longitudinal strain; Fnc: conduit function.
Table 3: Areas Under Curves of Left Atrial Strain Components to Predict High Left Ventricular Filling Pressures with De- long Test
Binary logistic regression analysis was performed for the multivariate study. Except for the ultrasound parameters used in the 2016 decision algorithm, all the other parameters for which the univariate analysis had p values <0.05 were included in the multivariate analysis. Only PALS (HR = 0.711; 95% CI: 0.513- 0.986; p = 0.041) was an independent predictor of high LVFP at rest or during exercise.
Discussion
The major findings of the present study were:
• LAS components are mildly to strongly correlate with the 2016 decision algorithm parameters.
• The cut-off values to predict high LVFP (at rest or during exercise) were 24, 13 and 14 for PALS, PACS and FnC, respectively.
• Only PALS was an independent predictor of high LVFP.
Left atrial function is articulated in three phases during each cardiac cycle: a “reservoir” phase (PALS) during ventricular systole, a “conduit” phase (FnC) during early diastole, and a “booster pump” phase (PACS) in late diastole. All phases are partially influenced by ventricular chamber spatial displacement of left ventricular systole with the downward movement of the mitral annulus and alongside with end-systolic volume, influence left atrial relaxation and compliance, which determine PALS. The latter is pivotal for successively left ventricular filling since the energy stored by left atrial during ventricular systole is then released after mitral valve opening, contributing to an adequate left ventricular stroke volume. Both left atrial and ventricular compliance influences FnC, which includes early left ventricular filling and diastasis. PACS is based on left atrial contractility, venous return, and left ventricular end-diastolic pressure [15- 17].
The findings of the present study agree with the results of previously published reports. Gaun et al. found that PALS and PACS were significantly correlated with E/e' ratio (r = -0.381 and -0.467, respectively, all with P < 0.013), in a population of 118 patients with HFpEF [18]. In a small cohort of 30 end- stage renal disease patients with HFpEF, Abid et al. investigated the relationship of PALS with elements of diastolic function, they found a negative correlation between PALS and E/e' ratio (r = -0.33, p = 0.049) and LAVI (r = -0.366, p = 0.047) while a positive correlation was found with average e' velocity (r = 0.557, p = 0.001) [19]. Lately, Fraydes et al. showed that PALS correlated with E/A ratio (r = -0.42, p< 0.01), LAVI (r = -0.35, p < 0.01), E/e' ratio (r = -0.42, p < 0.01), and TR velocity (r = -0.20, p < 0 .02), in a cohort of 300 patients diagnosed with HFpEF [8]. In line with this, some studies have shown that PALS has a stronger correlation with invasive LVFP at rest than LAVI [20,21]. In other studies, PALS and PACS were more correlated with invasive pulmonary capillary pressures measurement than the E/A ratio in patients with HFpEF [5,22].
In addition, many published reports assessed the relation between NT-Pro BNP and diastolic function and showed that it can be used to predict increased LVFP. Kurt et al. reported a negative and mildly correlation between PALS and PACS on the one hand and NT-Pro BNP on the other (r = -0.42, P = 0.001 and r = -0.37, P = 0.003, respectively) [20]. In a cohort of 74 patients with acute coronary syndrome, Topal et al. found a negative correlation between PALS and BNP (p = 0.001, r = -0.440) [23]. A similar finding was reported by other authors (r = -0.482, p = 0.007) [19]. Identical to our results, Aung et al. found a strong negative correlation between PALS and BNP (r = -0.567, p < 0.001) [24].
In the present study, LAS components were associated with high LVFP at rest. In this perspective, two recent studies should be mentioned. Inoue et al. included more than 300 patients with a median LVEF of 55% and showed that both PALS and PACS were associated with LVFP. The optimal cut-off to differentiate between normal and high LVFP was 18% for PALS and 8% for PACS [25]. Recently, Milijkovic et al. showed that PALS was significantly associated with the severity of diastolic dysfunction in hypertensive patients with HFpEF [7]. They found that a PALS value < 24.27% could predict high LVFP at rest with a sensitivity of 78.9% and a specificity of 84.6%. These results were equal to ours.
It is known that patients with HFpEF can elevate their filling pressures during exercise, despite normal resting LVFP. In a similar study, Ye et al. also evaluated the diagnostic value of LAS in detecting exercise LVFP raising in 669 patients with dyspnea assessed by stress echocardiography. They found that PALS and FnC were significantly lower in case of elevated LVFP during exercise, regardless of the grade of diastolic dysfunction at rest [9]. Telles et al. concluded that PALS and PACS were correlated with invasive pulmonary capillary pressures measured during exercise (r = -0.64 and r = 0.72, P < 0.001) and these parameters were remained independent predictors of HFpEF after multivariate analysis. A PALS value ≤ 33% predicted an invasively verified diagnosis of HFpEF with a sensitivity of 88% and specificity of 77% [26].
Left ventricular diastolic dysfunction is one of the major pathophysiological processes of HFpEF [27]. In the early stage, the left atrial enhances its pump function (PALS) to compensate for the obstruction of left ventricular filling caused by reduced left ventricular compliance. With the prolonged and worse of diastolic dysfunction, the compliance of the left atrial gradually impaired which results in a decrease in the reserve (PALS) of the left atrial, eventually leading to the enlargement and failure of the left atrial [28]. Therefore, left atrial strain reflects the cumulative adverse impact of chronically left ventricular diastolic dysfunction impaired on the left atrial [29].
The latest algorithms of EACVI guidelines for estimating diastolic dysfunction are based on several standard parameters and describe a precise scoring to quantify its severity [25]. The persistence of a "gray zone" of values in which quantification of diastolic dysfunction is not feasible, as well as an increase in the prevalence of HFpEF, has driven the research and the use of parameters with greater specificity and sensitivity: one of theme is LAS. The anatomy and mechanics of the left atrial are crucial to preserve the left ventricular function and the asymptomatic state of the patient. LAS, easily measured, correlates with LVFP raising both at rest and during exercise and therefore can be used as an alternative parameter in the diagnosis of HFpEF.
Limitation
Our study was monocentric and cross-sectional, used nonrandomized observational data, and is therefore subject to selection bias. However, we were careful to include consecutive patients. Secondly, we used echocardiographic variables to define high LVFP without hemodynamic validation. Besides, some degree of interobserver and intra-observer variability is known to be related to the analysis of the LAS. Finally, our study population was small compared with several larger-scale studies published in the literature. It is possible that the present study in some way is insufficient to accurately estimate the association between LAS and LVFP. Further longitudinal studies with a larger population are needed to confirm our results.
Conclusion
Our findings were concordant with studies that have recently underlined the predictive value of LAS components, essentially PALS, in predicting high LVFP, at rest or during exercise, and in providing a simple, reproducible, and sensitive ultrasound parameter for the diagnosis of HFpEF.
References
- Authors/Task Force Members, McMurray, J. J., Adamopoulos, S., Anker, S. D., Auricchio, A., Böhm, M., ... & Ponikowski, P. (2012). ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. European heart journal, 33(14), 1787-1847.
- Bhatia, R. S., Tu, J. V., Lee, D. S., Austin, P. C., Fang, J.,Haouzi, A., ... & Liu, P. P. (2006). Outcome of heart failurewith preserved ejection fraction in a population-based study.New England Journal of Medicine, 355(3), 260-269.
- McDonagh, T. A., Metra, M., Adamo, M., Gardner, R. S., Baumbach, A., Böhm, M., ... & Kathrine Skibelund,A. (2021). 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. European heart journal, 42(36), 3599-3726.
- Owan, T. E., Hodge, D. O., Herges, R. M., Jacobsen,S. J., Roger, V. L., & Redfield, M. M. (2006). Trends in prevalence and outcome of heart failure with preserved ejection fraction. New England Journal of Medicine, 355(3), 251-259.
- Mandoli, G. E., Sisti, N., Mondillo, S., & Cameli, M. (2020). Left atrial strain in left ventricular diastolic dysfunction: have we finally found the missing piece of the puzzle?. Heart Failure Reviews, 25, 409-417.
- Nagueh, S. F., Smiseth, O. A., Appleton, C. P., Byrd, B. F., Dokainish, H., Edvardsen, T., ... & Waggoner, A. D. (2016). Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Journal of the American Society of Echocardiography, 29(4), 277-314.
- Miljkovic, T., Ilic, A., Milovancev, A., Bjelobrk, M., Stefanovic, M., Stojšic-Milosavljevic A., ... & Petrovic,M. (2022). Left Atrial Strain as a Predictor of Left Ventricular Diastolic Dysfunction in Patients with Arterial Hypertension. Medicina, 58(2), 156.
- Frydas, A., Morris, D. A., Belyavskiy, E., Radhakrishnan, A. K., Kropf, M., Tadic, M., ... & PieskecKraigher, E. (2020). Left atrial strain as sensitive marker of left ventricular diastolic dysfunction in heart failure. ESC Heart Failure, 7(4), 1956-1965.
- Ye, Z., Miranda, W. R., Yeung, D. F., Kane, G. C., & Oh,J. K. (2020). Left atrial strain in evaluation of heart failure with preserved ejection fraction. Journal of the American Society of Echocardiography, 33(12), 1490-1499.
- Cameli, M., Mandoli, G. E., Loiacono, F., Dini, F. L., Henein, M., & Mondillo, S. (2016). Left atrial strain: a new parameter for assessment of left ventricular filling pressure. Heart failure reviews, 21, 65-76.
- Levey, A. S., Bosch, J. P., Lewis, J. B., Greene, T., Rogers, N., Roth, D., & Modification of Diet in Renal Disease Study Group. (1999). A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Annals of internal medicine, 130(6), 461-470.
- Lang, R. M., Badano, L. P., Mor-Avi, V., Afilalo, J.,Armstrong, A., Ernande, L., ... & Voigt, J. U. (2015). Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Journal of the American Societyof Echocardiography, 28(1), 1-39.e14.
- Pathan, F., D'Elia, N., Nolan, M. T., Marwick, T. H., & Negishi, K. (2017). Normal ranges of left atrial strain by speckle-tracking echocardiography: a systematic review and meta-analysis. Journal of the American Society of Echocardiography, 30(1), 59-70.e8.
- Pieske, B., Tschöpe, C., De Boer, R. A., Fraser, A. G., Anker,S. D., Donal, E., ... & Filippatos, G. (2019). How to diagnose heart failure with preserved ejection fraction: the HFA– PEFF diagnostic algorithm: a consensus recommendation from the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). European heart journal, 40(40), 3297-3317.
- TOMA, Y., MATSUDA, Y., MORITANI, K., OGAWA, H., MATSUZAKI, M., & KUSUKAWA, R. (1987). Leftatrial filling in normal human subjects: relation between left atrial contraction and left atrial early filling. Cardiovascular research, 21(4), 255-259.
- Hoit, B. D., Shao, Y., Gabel, M., & Walsh, R. A. (1994). In vivo assessment of left atrial contractile performance in normal and pathological conditions using a time-varying elastance model. Circulation, 89(4), 1829-1838.
- Hoit, B. D. (2014). Left atrial size and function: role in prognosis. Journal of the American College of Cardiology, 63(6), 493-505.
- Guan, Z., Zhang, D., Huang, R., Zhang, F., Wang, Q., & Guo, S. (2010). Association of left atrial myocardial function with left ventricular diastolic dysfunction in subjects with preserved systolic function: a strain rate imaging study. Clinical cardiology, 33(10), 643-649.
- Abid, L., Charfeddine, S., & Kammoun, S. (2016). Relationship of left atrial global peak systolic strain with left ventricular diastolic dysfunction and brain natriuretic peptide level in end-stage renal disease patients with preserved left ventricular ejection fraction. Journal of echocardiography, 14, 71-78.
- Kurt, M., Tanboga, I. H., Aksakal, E., Kaya, A., Isik, T., Ekinci, M., & Bilen, E. (2012). Relation of left ventricular end-diastolic pressure and N-terminal pro-brain natriuretic peptide level with left atrial deformation parameters. European Heart Journal–Cardiovascular Imaging, 13(6), 524-530.
- Morris, D. A., Takeuchi, M., Krisper, M., Köhncke, C., Bekfani, T., Carstensen, T., ... & Boldt, L. H. (2015). Normal values and clinical relevance of left atrial myocardial function analysed by speckle-tracking echocardiography: multicentre study. European Heart Journal-Cardiovascular Imaging, 16(4), 364-372.
- Hewing, B., Theres, L., Spethmann, S., Stangl, K., Dreger, H., & Knebel, F. (2017). Left atrial strain predicts hemodynamic parameters in cardiovascular patients. Echocardiography, 34(8), 1170-1178.
- Decirmenci, H., Bakc, E. M., Demirtac, L., Duman, H., Hamur, H., Ceyhun, G., & Topal, E. (2014). Relationship of left atrial global peak systolic strain with left ventricular diastolic dysfunction and brain natriuretic peptide level in patients presenting with non-ST elevation myocardial infarction. Medical science monitor: international medical journal of experimental and clinical research, 20, 2013- 2019.
- Aung, S. M., Güler, A., Güler, Y., Huraibat, A., Karabay, C. Y., & Akdemir, I. (2017). Left atrial strain in heart failure with preserved ejection fraction. Herz, 42(2), 194-199.
- Inoue, K., Khan, F. H., Remme, E. W., Ohte, N., García-Izquierdo, E., Chetrit, M., ... & Smiseth, O. A. (2022). Determinants of left atrial reservoir and pump strain and use of atrial strain for evaluation of left ventricular filling pressure. European Heart Journal-Cardiovascular Imaging, 23(1), 61-70.
- Telles, F., Nanayakkara, S., Evans, S., Patel, H. C., Mariani,J. A., Vizi, D., ... & Kaye, D. M. (2019). Impaired left atrial strain predicts abnormal exercise haemodynamics in heart failure with preserved ejection fraction. European journal of heart failure, 21(4), 495-505.
- Ponikowski, P., Voors, A. A., Anker, S. D., Bueno, H., Cleland, J. G. F., Coats, A. J. S., …… & ESC Scientific Document Group. (2016). 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J, 37(27), 2129-2200.
- Freed, B. H., & Shah, S. J. (2017). Stepping out of the left ventricle’s shadow: time to focus on the left atrium in heart failure with preserved ejection fraction. Circulation: Cardiovascular Imaging, 10(4), e006267.
- Obokata, M., Kane, G. C., Reddy, Y. N., Olson, T. P., Melenovsky, V., & Borlaug, B. A. (2017). Role of diastolic stress testing in the evaluation for heart failure with preserved ejection fraction: a simultaneous invasive- echocardiographic study. Circulation, 135(9), 825-838.




