Research Article - (2023) Volume 6, Issue 1
Development of Techniques for Analysis of Nutraceuticals, With Specific Reference to Glucosamine and Coenzyme Q10
Received Date: Oct 06, 2023 / Accepted Date: Oct 27, 2023 / Published Date: Nov 09, 2023
Copyright: ©Â©2023 Dr. Rehan Haider. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Haider, R., (2023) Development of Techniques for Analysis of Nutraceuticals, With Specific Reference to Glucosamine and Coenzyme Q10. Toxi App Pharma Insights Research, 6(1), 94-103.
Abstract
The reasoning of nutraceuticals, in the way that glucosamine and Coenzyme Q10 (CoQ10) hold paramount significance on account of their potential strength benefits, This abstract argues for the development of techniques for the inclusive reasoning of these nutraceuticals, expounding their concentrations and kinds in miscellaneous formulations. Glucosamine, a widely used and easily consumed supplement, is famous for its allure in supporting joint fitness. CoQ10, in another way, is famous for its alluring antioxidant features and its part in the basic strength result. Reliable examination methods guarantee the productiveness and security of production containing these compounds. This study focused on the growth and addition of methods to measure and characterize glucosamine and CoQ10. Various chromatographic orders, including extreme-efficiency liquid chromatography (HPLC) and vapor chromatography (GC), have been used to obtain the correct measurement of these compounds in different matrices. Spectroscopic methods, including UV-Vis and bulk spectrometry, have been used to identify and prove the demeanor of glucosamine and CoQ10 in complex formulations. Special consideration has been given to sample preparation to extract and detach these compounds from different forms, such as capsules, tablets, and working foods. Additionally, confirmation of the grown forms following worldwide directions has been performed to guarantee veracity, accuracy, subtlety, and reproducibility.
Keywords
Nutraceuticals, Glucosamine, Coenzyme Q10 (CoQ10), Analysis methods, Quantification, Chromatographic plans, High-act liquid chromatography (HPLC), Gas chromatography (GC), Spectroscopic methods, UV-Vis spectroscopy, Mass spectrometry.
Introduction
Over the past few decades, all have acquired a growing interest in the function of weight-reduction plans, vitamins, and digestive dietary supplements within the stop and scenario of diseases [1]. This has resulted in an exciting boom in the use of commercially unfastened nutraceuticals and their foods [2]. This massive use of nutraceuticals has aroused any of the examining methods to reveal their identification and levels in vacant green fabrics, easy extracts, planned devices, organic fluorides, and any of the different casts.
This evaluation covers the off-direction range of techniques developed for the reasoning of remaining typically intentional nutraceuticals there are any of the following: motives for the development of better-examining methods, particularly inside the volume of supplementary knowledge on ailment states and their situations, particularly the fads of the operation of specific nutraceuticals. Different primary reasons include forecast of disease states and agreement the analyzing processes secondhand for labeling and calculation of nutraceuticals are getting more cosmopolitan, indicating modern examining advances. Producers, free scientists, and offering preparations such as Consumer Lab.com regularly produce particularized records of approximate ranges of live elements, in two together, the unaffected subjects and the formulated commodity. Clinical investigators are greater reality-locating organic fluids in clinical U.S. country assessments and in vivo research in an attempt to determine the organic destiny of nutraceuticals.
There are few written reviews of inspection strategies for nutraceuticals. There has been a restrained survey of 26 examples written in 2007 [3]. A study of refined texts written in 2002 however, for the maximum, skilled are few loose methods [4, 5]. Monographs, to some extent the ones being produced for the FDA and the Office of Nutrition Supplements of the countrywide Institutes of Fitness of the United States, list techniques for pattern improvement and starting place, chromatographic spoil-up, discovery pill with stimulant die, and quantitation strategies [3]. In addition, those brochures' focal points the roomy range of techniques is increasingly being used secondhand for their identification and quantification in collectively made merchandise and natural fragrances. HPLC, which uses a variety of diverse detectors, has been used for the majority of nutraceuticals, but newer techniques are also becoming widespread.
The phytoestrogen from soy and flax, n-3 PUFAs and CLA, carotenoids, lycopene, zeaxanthin, astaxanthin, and lutein have been the subject of the latest edited e-book on difficulty [4]. The opposite text, with the aid of Ho and colleagues, reviews the techniques to be had for cocoa, chocolate, cranberry, and guggul (Commiphora Wight ii) materials [5]. Recent courses have reviewed the analysis of soy and tea polyphenols [6, 7]. This study will focus on the techniques used for two vital and extensively used nutraceuticals, the analysis of which, specifically glucosamine and CoQ10, has not been reviewed previously. Bankruptcy will also explain the improvement in the techniques used in the analysis of such nutraceuticals. Glucosamine is extensively used to improve joint health, especially osteoarthritis and rheumatoid arthritis, and pores and skin fitness. It is also widely used to treat joint disorders in domestic animals. CoQ10 has been suggested to be involved in cardiovascular health, cancer prevention, breathing and skin illnesses, and animal illnesses.
Techniques FOR Analysis of Glucosamine
There are many challenges in the evaluation of glucosamine. First, the hydrophilic nature of the molecule makes the extraction with natural solvents from plasma ineffectual. Together with the drug, a selection of endogenous compounds with a chemical structure similar to glucosamine, which include glucose, galactose, and different sugars and amino sugars might additionally be extracted from an organic sample [8, 9]. To avoid this, lengthy sample guidance strategies are frequently used. Second, numerous ingredients are regularly observed in commercial glucosamine systems. Chondroitin, additionally a glycosaminoglycan with a molecular weight of 5,000–55,000 is typically located in such formulations, and this compound has the potential to interfere with the quantitative evaluation of glucosamine [10]. Evaluation of chondroitin has not evolved as much as that of glucosamine.
Radiolabeling
Preceding analytical strategies for biological samples trusted radiolabeling However, the use of radioactivity to quantify glucosamine can also have doubtless confounding effects because a discernible drug molecule cannot be differentiated from its degradation products and metabolites suggest that there may be a need for specific analytical techniques to study the glucosamine content in both commercial products and organic fluoride [10-12]. Both areas present distinct problems in terms of evaluation. A variety of strategies have been counseled in an attempt to conquer such demanding situations, with every technique having its packages, boundaries, and advantages, so one can be discussed underneath.
HPLC Separation
It's been advised that, of all the techniques, HPLC is the most extensively used and a touchy approach for glucosamine analysis [8]. Glucosamine poses several challenges for the evaluation of liquid chromatography. Highly-Pharmaceutiques and numerous strategies have been used and studied. The first demanding situation was created with the aid of the glucosamine molecule (Figure 7.1). Because sugars and amino sugars (together with glucosamine) are noticeably polar molecules, they are no longer retained on common hydrophobic HPLC column-packing substances (e.g., RP C18), making separation difficult [13]. A possible solution to this problem is to use h amino columns or the complexation of glucosamine through its amino group with a hydrophobic compound [14]. The chemical shape of glucosamine additionally lacks the shapeable chromophore or fluorophores, which absorb within the wavelength range useful for High-performance liquid chromatography (HPLC) with UV or fluorescence detection [10, 15].
Precolumn Derivatization retailers for UV/Fluorescent Visualization
To conquer this trouble, glucosamine is often derivatized to incorporate a suitable chromophore or fluorophore into its structure to improve its detection by UV techniques and such agents may be sure to glucosamine via amino acid organization [10]. It has been shown that the pre-column derivatization steps can be lengthy. Making the time of analysis for such techniques unsuitable for recurring use [10]. Problems with the stability of glucosamine-derivative complexes have also been addressed. Monitored the UV absorption of glucosamine spin-off phthalaldehyde-3-mercaptopropionic acid (OPA- MPA) over time to illustrate the stability problems [10]. The derivatization reaction was accomplished with an OPAMPA molar ratio of 1:50, according to a published technique in the U.S. Pharmacopeia. The 1:50 ratio was proven to improve boom balance and became the chosen technique, and the maximum absorption of the glucosamine-OPA-MPA by-product in borate buffer (80 mM, pH 9.5) was determined to arise at 335 nm. Absorbance was measured value for 4.5 hours after mixing the reagents. Showed that the UV absorbance reduced to approximately 1/2 the maximum over a length of 4 h. This instability ought to result in inaccuracy in evaluation if the term varies between acting the derivatization reaction and injection of the sample into the HPLC machine several sellers have been recommended as suitable to make detectable complexes with glucosamine [10]. Studies investigated the usefulness of every agent for a variety of packages, such as the glucosamine content in a business product or biological fluid judgment. Manageable precolumn derivatization entrepreneurs include OPA and phenyl is thiocyanate (PITC) [15, 16, and 3].
N-(9-fluorenyl-methoxycarbonyloxy) succinimide and nine- fluorenyl-methyl chloroform ate (FMOC-Cl) [11, 12]. All of these dealers may have complexed the accompanying glucosamine pre-column earlier than a breakup used OPA- MPA as a precolumn derivatization agent for glucosamine [15]. The derivatization acid-base indicator was changed to normal oblique by dissolving 5 mg of OPA in 900 l of intoxicating, 100 l of borate buffers, and 10 l of MPA solutions. The separation was completed using a 250 mm × 4 mm RP C18 pillar. The field segment was exchanged into isocratic and contained flammable liquid sodium phosphate (pH 6.5, 12.5, 10+ 90, extent/volume [v/v]) and flammable liquid tetrahydrofuran (90 seven+3, v/v) for 20 short terms at a flow rate of man or woman ml/min with capacities of 8 5+15. Detection can easily be accomplished using a fluorescence indicator. A measurement curve became buxom with the usage of requirements offset from 5 to 200 % of the insignificant assay consideration of 10 g/ml glucosamine HCl, which proved extremely good time with an equivalence coefficient of nothing.9980. In my opinion, the lower limits of detection (LOD) and quantification (LOQ) were 0.009 and not 027 g/ml [15]. Nemati et al. determined that derivatization accompanying OPA-MPA is extraordinarily easy and sturdy, with recurring extra sensitivity, compared to various derivatization methods. The derivatization reaction took an awful lot less than five brief terms. Nevertheless, the category of electricity is suitable for computerization because professionals no longer need to throw away extra derivatization electricity, and the solution now does not require a dissolution step, which might reduce human mistakes. OPA also has the advantage that UV and fluorescence detectors may be used collectively, bestowing it miles greater off-path applicability. Balance issues accompanying OPA have existed for a long time; for example, glucosamine-OPA complexes rot through the ages [15, 11]. Assessment of merciless glucosamine fluids entails singular analytical strategies and is essential for understanding the physiological role of glucosamine, absorption, pharmacokinetics, outcome, and manner of motion. Huang and others examined using FMOC-Cl as a derivatization power for the evaluation of glucosamine content in fabric, pores, and skin. The levels of glucosamine in merciless plasma are remarkably decreased, presenting a greater analytical challenge.
Consequently, a suitable inspection process needs to be sensitive, smooth, and brief. The derivatization acid-base indicator consisted of 0.8 mM FMOC-Cl in acetonitrile. Separation was achieved using a C18 inspection column (150 × x 4.6 mm vital width, 5 m). Elution improvements were obtained by utilizing the slope steps of solvents a (acetonitrile) and B (water):30:70 (A/B) for 10 brief terms and before 98:2 for 15 short terms, at a flow rate of one ml/min. The LOD was more advantageously situated at 15 ng/ml (signal-to-noise ratio >=three), which is efficaciously below the anticipated drug knowledge in purple body fluid samples from a troubled character, probably recuperation doses of glucosamine sulfate. Huang decided that the use of FMOC-Cl as a derivatization agent had impressionable, speedy (able to be traced to faster pattern course times), smooth, and honest analytical consequences. Furthermore, it has been proven that there are no cohesion questions with FMOC-Cl- glucosamine aggregates because the samples had been validated and predicted to be forceful for twenty-four hours at 4 °C [9]. Consequentially, the method may apply to the reappearing evaluation of glucosamine for pharmacokinetics, bioavailability, or bioequivalence studies [11].
Post column Agents Used for Indirect Fluorescent Detection Another method used to overcome the issue of limited UV or fluorescence detection attributed to glucosamine is to attach a visualizing agent after column separation. Exploited an indirect fluorescence detection method for the analysis of glucosamine in dietary supplements [10]. This method avoids the stability and time issues associated with pre-column derivatization and thus could be advantageous. This method is based on the fluorescent signal of L-tryptophan (L-TRP) or DL-5-methoxytryptophan (5-MTP). The post-column was added to either compound as a copper complex; when bound to the copper (II) ion, the fluorescent signal of these compounds was quenched, resulting in no absorbance. Glucosamine is capable of complexing with the copper (II) ion and thus displacing some fraction of L-TRP or 5-MTP, recovering their fluorescent signal. The amount of glucosamine present can be calculated indirectly from the intensity of the signal produced by displaced L-TRP or 5-MTP. Shen, Yang, and Tomellini used the following chromatographic conditions: mobile phase, 1.6 mM sodium borate, pH 9.0; flow rate, 1 ml/min; post-column interaction component 2 x 10-5 MCU (L-TRP)2 in 40 mM sodium borate at pH 9.0, or 2 x 10-5 M Cu (5-MTP)2 in 40 mM sodium borate at pH 8.4; flow rate, 1 ml/min; column, strong anion-exchange column, PRP-X100 (250 x 4.1 mm, 10?m) with a fluorescence detector [10].
The main disadvantage is the lower sensitivity of the method compared to pre-column derivatization. The detection limit found using post-column derivatization corresponded to a glucosamine concentration of 3.2. G/ml compared to the detection limit of 0.009? G/ml and 0.075? G/ml for pre-column derivatization with OPA-MPA and PITC, respectively [15, 17]. However, the technique would still be an acceptable alternative if detection limits were not an issue (e.g., in the analysis of glucosamine in commercial products) because it avoids time-consuming derivatization reactions and the possibility of stability issues [10].
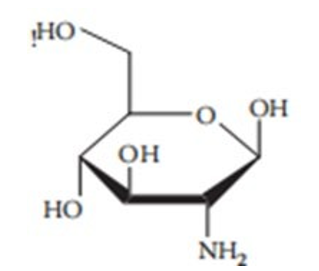
Figure 7.1 The structure of glucosamine
HPLC Using Electrospray Ionization-MS Detection Because of the possible limitations in the sensitivity of the methods involving precolumn and postcolumn derivatization followed by UV or fluorescence detection, researchers investigated the possibility of HPLC separation followed by MS detection [11, 13]. A method developed by Huang et al. used pre-column derivatization with PITC, followed by electrospray ionization (ESI)-MS detection. Huang used an RP C18 column, and, because of the high polarity of glucosamine, the elution was too fast; thus, suitable derivatization was used to help facilitate HPLC component separation. PITC was chosen over other agents because it gave selective mass spectra. Elution was performed using 0.2% glacial acetic acid (A) and methanol (B) at a flow rate of 0.3 ml/min. Gradient HPLC was used with a changing solvent ratio from 80:20 to 10:90 over 8 min, and then 80:20 for 13 min for A and B, respectively. Quantification was achieved by MS in positive ionization mode with ESI as an interface, a drying gas flow rate of 10 L/min, a drying gas temperature of 350 °C, a nebulizer pressure of 50 psi, a capillary voltage of 4,000 V, and fragmentation energy of 130 V. This method obtained good precision, accuracy, and speed with a LOQ of 0.1 g/ml and a LOD of 35 ng/ml (signal-to-noise ratio of 3), and a time of 8 min per analysis. It was concluded that this method could be used for the analysis of glucosamine in human plasma, including analysis of basal glucosamine plasma levels [11]. This technique allows information to be gathered on the physiological role of endogenous glucosamine and its involvement in disease processes. Another method studied by used HPLC, followed by ESI-MS/MS detection [13]. In this study, a polymer-based amino column was used to increase the retention of glucosamine, thereby preventing the need for derivatization. This was beneficial for several reasons. First, the analysis speed was increased. Second, the derivatization reactions are specific and can derive other plasma components, resulting in increased "noise" in the separation and detection steps.
The separation of components on the column was achieved using a gradient composed of Milli-Q water (A) and acetonitrile (B) at a flow rate of 0.3 ml/min. The gradient elution program used was as follows % A for 7 min; 7-8 min: a linear increase from 20 to 50% A; 50% A for 8 min; 16–17 min: a linear decrease from 50 to 20% A; and 10 min: 20% A. Detection was performed using a triple quadrupole mass spectrometer that was set in the positive ionization mode, with quantification performed in the multiple reaction monitoring mode. This method achieved a LOQ and LOD of 10 and 5 ng/ml, respectively, without the need for a derivatization step. Roda et al. concluded that this method was suitable for the measurement of endogenous glucosamine plasma levels due to its high sensitivity. This method is said to be advantageous over other techniques because no pre-analytical derivatization step is required, resulting in an analysis that can be performed quickly and reduces analytical variability [13]. Both methods using HPLC coupled with ESI-MS have high sensitivity and accuracy. However, criticisms exist regarding the use of HPLC and ESI-MS methods for the routine analysis of glucosamine. Some have claimed that LC-MS technology is not yet widely available in laboratories, thus making the method unsuitable for widespread analysis. It has also been claimed that precolumn derivatization with FMOC-Cl with UV detection provides the same sensitivity at a lower cost than the LC-MS technique [8].
The Use of HPLC with Alternative Detection Methods
HPLC with pre-column derivatization methods is cheap and effective. They offer a suitable level of accuracy for the analysis of glucosamine products, and derivatization with FMOC- Cl may offer suitable sensitivity for the analysis of biological samples. The derivatization step is involved in many criticisms of this method. The initial derivatization reaction may be time- consuming, increasing the analysis time and rendering the methods unsuitable for routine use. Once prepared, the derivative- glucosamine complexes could have stability issues that may confound the results. Finally, the derivatives could negatively affect the performance of the assay. To avoid these issues, HPLC ESI-MS/MS can be performed using an amino column that circumvents the use of derivatization agents. However, this method is more expensive and less readily available but offers good sensitivity. The last HPLC option is indirect fluorescence detection with L-TRP or 5-MTP. This method avoids the derivatization step and is inexpensive and accessible; however, it lacks suitable accuracy for the analysis of biological samples. Overall, the HPLC method of choice depends on the analytical function to be performed.
Gas Chromatography
Glucosamine has been determined in soil using gas chromatographic methods. This technique is said to be suitably sensitive, but the need to make solutes volatile through complicated sample preparation procedures can be time-consuming and can result in multiple peaks for a single component [18]. This makes this method unsuitable for routine use. Looked at the determination of glucosamine in soil samples, and, thus, its relevance to the quantification of glucosamine in commercial products or plasma samples may be questionable. High-Performance Thin-Layer Chromatography a quantitative densitometric high-performance thin-layer chromatographic method was developed by for the determination of glucosamine in dietary supplements [19]. This method was sought because no rapid, simple, or selective HPLC method was reasonably available for the quantitative determination of glucosamine from its complex matrix. Ester et al. achieved separation on 20 × 10 cm silica gel 60 F254 high-performance thin-layer chromatographic plates with a mobile phase consisting of a 2-propanol/ethyl acetate/ammonia solution (10:10:10 v/v/v). As glucosamine lacks a suitable chromophore, the plates were immersed in a visualization reagent solution consisting of diethyl ether, glacial acetic acid, anisaldehyde, and sulfuric acid (136:91:1.2:20 v/v/v/v). The plates were then processed, and glucosamine appeared as brownish-red zones on a colorless background. The densitometric determination of glucosamine was performed at 415 nm using reflectance scanning. The amount of glucosamine was determined by the intensity of the diffusely reflected light. This method is advantageous because it circumvents the tedious and time-consuming sample preparation steps necessary for the HPLC techniques discussed above. Concluded that this method was reliable, repeatable, and accurate [19].
Capillary Electrophoresis
Determination Both HPLC with UV/fluorescence and ESI- MS detection include particular disadvantages that are discussed above, including a relatively high cost. Capillary Electrophoresis (CE) is considered a suitable alternative because of its high efficiency, fast speed, small sample size, simplicity, and flexibility [16]. For these reasons, CE has been widely used for the rapid analysis of biomolecules such as carbohydrates, amines, and amino acids, providing high resolution and short separation times of approximately 3 min [20]. Similar to the HPLC method with pre-column derivatization, the main drawback of CE is its low detection sensitivity to glucosamine, which has to be improved by the incorporation of a suitable UV-absorbing group onto the glucosamine molecule [18, 20]. Capillary Electrophoresis with Dansylation of Glucosamine under Microwave Irradiation appreciated that a major drawback of derivatization is its increase in the time of analysis [18]. To obviate this problem, they investigated the use of dansyl chloride as a suitable labeling agent. To accelerate the labeling process of glucosamine, they performed the translation reaction in a microwave oven, which gave labeling speeds up to 50 times faster than common methods. Derivatization was achieved by mixing 10 mg of dansyl chloride with 10 ml of acetone and the glucosamine solution (tablets dissolved in 80 mM borate buffer at pH 9.5). The solution was placed in a microwave oven and irradiated at 385 W for 6 minutes. CE was performed using a bare fused-silica capillary of 75 mm inner diameter x 57 cm. Samples were injected at 0.5 psi for 2 s and separated at +18 kV at 20 °C. The separated bands were detected by UV absorption at 214 nm. This technique gave reasonable sensitivity with an LOD of 1 g/ml. Qi et al. concluded that the use of CE using accelerated labeling of glucosamine with dansyl chloride under microwave irradiation was a reliable, accurate, quantitative, and highly applicable method for the determination of glucosamine content within commercially available glucosamine tablets but was not sufficiently sensitive for the determination of glucosamine in biological samples [19]. Microchip Capillary Electrophoresis with Fluorescamine Labeling for Anomeric Composition Determination Glucosamine can exist as either an alpha or beta anomer, with an anomer being an epimer that is a stereoisomer of saccharides, differing only at the reducing carbon atom. Because of this, there is a need to assess the Anomeric composition of glucosamine products and the inter-conversion rates between the two anomer. Developed a microchip capillary electrophoretic method able to resolve both the alpha and beta anomer of glucosamine, allowing for the determination of the Anomeric composition of a sample [20]. Previous chromatographic methods reported timescales of analysis between 10 and 20 min, which led to poor resolution between the alpha and beta anomer; however, because of the inherent speed of CE, this did not occur. The blurring in resolution occurs as a result of on- column mutarotation between the two anomers of glucosamine; thus, the faster the better the analytical technique, the less time mutation has to occur, and the less blurring of resolution is seen.
Skelley et al. labeled glucosamine with Fluorescamine by mixing 2 l of the sample with 20 mM Fluorescamine in dimethyl sulfoxide. The samples were analyzed at room temperature using all-glass micro-fabricated devices made in-house. The CE separations were performed at 700 V/cm on a portable CE instrument with a 100-m-diameter fiber optic-coupled photomultiplier tube for fluorescence detection. The inherent speed of microchip CE enabled Skelley et al. to observe the alpha and beta anomers of glucosamine and their inter-conversion rates in real time. It was noted that the inter-conversion between the anomers may have been affected by the Fluorescamine labels. Furthermore, if inter-conversion was allowed to take place before labeling, more accurate results could be obtained. However, this was at the expense of time, and if labeling took place after mutarotation, the time of overall analysis rose from approximately 2 minutes up to 2 hours. Concluded that this method was fast, portable, and suitable for glucosamine determination [20]. The Pros and Cons of Capillary Electrophoresis the use of CE for the analysis of glucosamine offers a suitable, low-cost alternative to HPLC methods. CE is a fast, quantitative, and efficient method of analysis. The latter method discussed has the unique advantage that it enables the real-time observation of the mutarotation of the glucosamine anomers, which no other analytical technique can accomplish. Disadvantages include a limited sensitivity when compared with other methods, and the apparatus is less commonly available in laboratories [8]. Infrared Spectroscopy with Chemo metrics The chemical structure of glucosamine makes it suitable for analysis with infrared (IR) spectroscopy. Previously, IR spectroscopy has been used to gather information about glucosamine and other GAGs, how GAGs absorb water (which relates to the physical properties of cartilage), and the breakdown products of cartilage after enzyme degradation. This technique can therefore provide useful information about arthritic disease processes. In this method, IR is combined with chemometric pattern recognition techniques for the analysis of glucosamine and similar compounds. Analyzed all samples by transmission Fourier transform infrared spectroscopy on a Nicolet Magna IR 760 spectrometer Data analysis was then undertaken using computer programs [21].
Microsoft Excel was used to produce the first derivative spectra. Then both the original and derivative data were transposed to SIRIUS to perform principal components analysis as a data reduction technique, linking that with hierarchical cluster analysis and soft independent modeling of class analogies. These mathematical techniques are used to distinguish between the IR spectra produced by similar compounds, such as glucosamine, galactosamine, and chondroitin. This allows for a fast and simple evaluation of the spectra of such compounds. Foot and Mulholland concluded that samples were best classified using the first derivative The better the analytical technique, the less time mutation has to occur, and the less blurring of resolution is seen. Skelley et al. labeled glucosamine with Fluorescamine by mixing 2 liters of the sample with 20 mM Fluorescamine in dimethyl sulfoxide. The samples were analyzed at room temperature using all-glass micro fabricated devices made in-house. The CE separations were performed at 700 V/cm on a portable CE instrument with a 100-m-diameter fiber-optic-coupled photo multiplier tube for fluorescence detection. The inherent speed of microchip CE enabled Skelley et al. to observe the alpha and beta anomers of glucosamine and their inter conversion rates in real-time. It was noted that the inter conversion between the anomers may have been affected by the Fluorescamine labels. Furthermore, if inter conversion was allowed to take place before labeling, more accurate results could be obtained. However, this was at the expense of time, and if labeling took place after mutarotation, the time of overall analysis rose from approximately 2 minutes up to 2 hours. Concluded that this method was fast, portable, and suitable for glucosamine determination [20]. The Pros and Cons of Capillary Electrophoresis the use of CE for the analysis of glucosamine offers a suitable, low-cost alternative to HPLC methods. CE is a fast, quantitative, and efficient method of analysis. The latter method discussed has the unique advantage that it enables the real-time observation of the mutarotation of the glucosamine anomers, which no other analytical technique can accomplish. Disadvantages include a limited sensitivity when compared with other methods, and the apparatus is less commonly available in laboratories [8]. Infrared Spectroscopy with Chemometrics The chemical structure of glucosamine makes it suitable for analysis with infrared (IR) spectroscopy. Previously, IR spectroscopy has been used to gather information about glucosamine and other GAGs, how GAGs absorb water (which relates to the physical properties of cartilage), and the breakdown products of cartilage after enzyme degradation. This technique can therefore provide useful information about arthritic disease processes. In this method, IR is combined with chemometric pattern recognition techniques for the analysis of glucosamine and similar compounds. Analyzed all samples by transmission Fourier transform infrared spectroscopy on a Nicolet Magna IR 760 spectrometer Data analysis was then undertaken using computer programs [21]. Microsoft Excel was used to produce the first derivative spectra. Then both the original and derivative data were transposed to SIRIUS to perform principal components analysis as a data reduction technique, linking that with hierarchical cluster analysis and soft independent modeling of class analogies. These mathematical techniques are used to distinguish between the IR spectra produced by similar compounds, such as glucosamine, galactosamine, and chondroitin. This allows for a fast and simple evaluation of the spectra of such compounds. Foot and Mulholland concluded that samples were best classified using the first derivative spectra. However, this technique has the disadvantage of only being qualitative and thus cannot be used to quantify the active ingredients within products [21]. In conclusion, this technique may be less useful than other methods because it can only be used for the qualification of components, but the authors have suggested that Additional work must be undertaken to enable the technique to be used for quantification.
Techniques For Analysis Of Coenzyme Q10 Coq10
(see Figure 7.2) has many physiological roles; therefore, being able to analyze its levels in plasma could aid in the treatment and prediction of diseases. Not only this, the new HMG-CoA reductase inhibitors (cholesterol-lowering drugs) affect the synthesis of CoQ10 and, therefore, have the potential to cause adverse effects such as cardiac myopathy. This is thought to be attributable to a decrease in ubiquinone plasma levels, meaning that a determination of CoQ10 is important in early drug development to predict and avoid adverse effects [22]. Finally, with the ever-increasing use of nutraceutical products, it is important to be able to determine levels of these compounds within commercially available products to ensure public safety. The quantification of CoQ10 plasma levels is, therefore, important from a medical and epidemiological point of view [23]. The analysis of CoQ10 presents several challenges, and numerous techniques have been investigated for potential use [24, 25]. The average level of coenzyme in a healthy subject is 0.8–0.2 mg/L [26]. Thus, the main analytical challenge was to develop a method selective enough and sensitive enough to quantify levels in human fluids and commercial products that were fast enough to
Figure 7.2 the structure of coenzyme Q10.
be used routinely in quality-control laboratories. This would allow for useful information on the action, bioavailability, and fate of CoQ10 products to be collected. The challenge is made more difficult by the chemical nature of CoQ10. First, CoQ10 contains isopentenyl, which makes the compound susceptible to photo oxidation. This means that all experiments should be undertaken in particular conditions to prevent photo degradation, expressly in the deficiency of light. Second, organic samples of CoQ10 will be accompanied by a complex form. These resources that some samples will demand exhaustive and late sample arrangement before an assay can be ventured [23]. This is frequently a determinant that limits the profit of a method if it is to be used widely.
HPLC is possibly the most favorite method secondhand in the study of CoQ10 [27]. Several detection procedures have happened secondhand, containing MS, UV, and electrochemical discovery. UV and electrochemical discovery are possibly the most popular methods secondhand by way of the more off- course approachability of the electronics [23]. Each procedure is guided surely troubles and benefits fated shortly period debated below.
UV or Electrochemical Detection? It has been proved that the perseverance of CoQ10 cruel skin may be realized by HPLC with UV discovery and subsequently TLC, liquid/liquid or dependable-chapter origin [24]. These techniques are used to eliminate complex parts that are raised inside an organic sample. The common method secondhand is an intoxicating hexane distillation, at which point the extract is drained and redissolved before the dose. This pre-procession manipulation of the sample is behind, which forms the designs inappropriate for routine use in dispassionate allure laboratories [24]. Grown specific a means. Before the sample dose, a lengthy readiness accepted place at which point 0.25 ml of sample was deproteinized utilizing 0.5 ml of flammable liquid after that, 0.75 ml of hexane was added [23]. The combination. Was then vortexed for 5 brief periods and centrifuged at 5000 rpm for 15 brief periods. Next, the clear hexane coating was moved to another hose and the origin of body tissue vitamins was repeated accompanying a new 0.75 ml portion of n-hexane.
The red body fluid extracts were linked and dissolved to aridity under a stream of nitrogen. Finally, the dry debris was separated in 0.25 ml of mobile point and chromatographer by HPLC. Separation accepted to establish an RP C18 125 x 4 mm (5? m) line accompanying a guard line, 4 x 4 mm (5? m), with a movable chapter of intoxicating and hexane 72:28 (v/v). The flow rate secondhand was 1 ml/brief period, accompanying discovery achieved by a UV indicator at 276 nm. This presents a memory opportunity of 7.47. 0.12 brief period and a LOD and LOQ of 0.87 and 2.98? M, individually, for CoQ10.The use of an electrochemical indicator is 10-20 times more delicate than a UV indicator. For this reason, the order was second handed by for the discovery of CoQ10 cruel breast milk, at which point the levels are even inferior to that of the body tissue [28]. The examining pillar secondhand was a Microsorb-MV RP C18 column (150 x 4.6 mm; 5? m blob length). The movable aspect secondhand incorporated a combination of sodium acetate anhydrous (4.2 g), 15 ml of frigid tart acid, 15 ml of 2-propanol, 695 ml of intoxicating, and 275 ml of hexane. The traveling step was refined through a 0.2? M pore-judge nylon filter, and the flow rate was 1 ml/brief period. An electrochemical indicator was secondhand and was proved to present a LOQ of 60 nmol/L, accompanying an opportunity of reasoning of nearly 8 proceedings per sample. With the basic aggregation of CoQ10 in conscience milk being 0.166 ± 0.002 μmol/L, this plan achieves extreme sensitivity [28]. The added main benefit of stereochemistry discovery over UV orders is that it admits the discovery of the lowered form of ubiquinone, ubiquinol. This admits to the guess of the total red body fluid CoQ10. To achieve this, all CoQ10 in a sample is convinced into the weakened form by the precolumn situation and before discovered utilizing aforementioned patterns [26]. However, as accompanying UV discovery, this arrangement still demands manual sample arrangement before chromatographing, which has the hurt of being time-consuming.
Column Switching HPLC
Because of the complicatedness of organic fluid samples, most samples normally demand in a way purification before dose into the HPLC whole, such as dimensional-aspect origin, liquid/ liquid, or TLC. Column changing avoids this by admitting connected to the internet sample purification. This results in the method being appropriate for products and cutting down the management period, which can influence artificial mistakes and weak reproducibility. Secondhand such an arrangement for the perseverance of CoQ10 cruel red body fluid [24]. In this method, a short precolumn (20 × 4.6 mm central diameter) was full accompanying 5 μm Hypersil C18. This was used to remove two cold compounds, including reagents and powerfully kept solutes. The traveling time secondhand for the precolumn was pure intoxicating at a flow rate of 0.5 ml/brief time period. The analyte was moved to the examining column (150 × 4.6 mm central width full accompanying 5 μm of Hypersil ODS2). The movable development through the analytical pillar contained 10% (v/v) isopropanol in flammable liquid at a flow rate of 1.5 ml/brief period. The detection was acted accompanying UV set at 275 nm. The lower limit of the discovery reached was nearly 1 μg/ml of cruel body tissue, which corresponds to accompanying orders utilizing dependable-phase distillation to free samples [24]. The disadvantage is famous by is that procession-switching electronics cannot be handy in many workshops [26]. Whether this comment still applies 15 age on is debatable.
Mass Spectrometry Detection
As electronics cultivate, investigators are adopting the use of HPLC-connected accompanying MS as a useful pattern for the identification and decision of compounds [22, 25]. The use of LC-MS for the discovery of CoQ10 in complex samples is active by way of high sense and selectivity. Manifested that an LC-MS procedure was more impressionable than LC-UV [29]. Revealed that HPLC-MS has corresponding or better sympathy than HPLC-electrochemical discovery [30]. Used an LC-MS/MS arrangement for the decision of CoQ10 in smoking leaves [26]. Chromatographic reasoning was acted on an RP C18 column, accompanying a traveling point comprising a combination of acetonitrile and isopropanol (8:7 v/v) holding 0.5% formic acid at a flow rate of 0.3 ml/brief period. ESI- MS, operating in a certain ion trend, was used for discovery and reasoning. CoQ10 was listened to utilizing the diversified response monitoring way. Under these conditions, a memory occasion of 2.91 ± 0.1 brief periods and a LOQ and LOD of 4.0 and 1.2 ng/ml, individually, were established for CoQ10. This procedure was therefore intelligent to present a LOD 205.8, 71.9, 41.7, and 208.6 occasions inferior to the LOD acquired by spectrophotometric, voltammetric, and chromatographic plans with UV and fluorescence discovery. It has happened famously in illuminated literature that forms elements do have the potential to cause incorrect results unless growth of sample readiness and the chromatographic structure takes place.
Another LC-MS/MS method utilizing a vehicle propelled by ejection of pressurized gas or liquid-spray ionization was investigated by for reasoning of CoQ10 levels in informer soul and femur influence [22].
Teshima and Kondo looked at existing orders utilizing LC-MS/MS accompanying a vehicle propelled by ejection of pressurized gas or liquid spray ionization beginning for the study of ubiquinones and looked to correct their sense. Separation was reached on an YMC Pack Pro (75 × 2 mm central width) examining column, and a combination of flammable liquid/2-propanol/formic acid (45:55:0.5) holding 5 mmol/L methylamine was picked as the movable aspect. The addition of the methylamine to the mobile development resulted in to 12.5-fold larger signal force than outside. A threefold-stage quadrupole mass spectrometer accompanying a vehicle propelled by ejection of pressurized gas or liquid-spray ionization beginning was secondhand for the discovery of CoQ10. Under these conditions, CoQ10 had the noticed retention period of 4 brief periods, and the design was intelligent to measure CoQ10 over an off-course concentration range in informer groin and courage influence, 1–500 and 10–10,000 μg/g, individually [22]. All duplicate HPLC methodologies are appropriate for the study of CoQ10 in products and biological samples and are usually secondhand. Intuitively, the pillar-switching design appears ultimately realistic for use in dispassionate analyses of CoQ10 because of its allure of fast, correct, and automated properties.
Derivative Spectrophotometry
Although HPLC designs are well impressionable, it has been evident that they maybe period-absorbing and difficult. An alternative method was then sought by They examined the use of spectrophotometry cause it is a rapid, specific, plain, and trustworthy examining method [23]. Unfortunately, classical UV spectrophotometric systems manage not to be secondhand by way of range interference from the complex model of red body fluid samples of CoQ10. This fashioned perseverance of CoQ10 is absurd. To avoid this question, Karpin´ska and others. Secondhand a numerical method to eliminate the influences of the mold/backdrop, that is to say, derivative spectrophotometry (a derivative of the ranges is captured using the Savitzky–Golay invention). This method improves the sense and discrimination of the analysis. Karpin´ska secondhand an HP-8452A diode array spectrophotometer, and the spectrophoto rhythmical calculations were acted at 284 nm. This method was able to check the ghost of CoQ10 in pharmaceuticals in the range of 0.5–10 ppm. Because levels of CoQ10 do not surpass 1.5 ppm, the form was applicable. Karpin´ska and others. Decided that the method may be secondhand as an alternative to HPL-C's reasoning of CoQ10 in marketing formulations; nevertheless, the form has limited use for the reasoning of organic samples, and a comparatively large sample capacity is necessary Voltammetric Determination.
Voltammetry is an electroanalytical method that is acceptable for use with CoQ10 by way of allure redox and adsorptive action. Two plans for the reasoning of CoQ10 have been secondhand in the drama. Square-wave voltammetry accompanying Glassy Carbon Electrode analyzed commercially applicable CoQ10 formulations utilizing a standard adding voltammetry order on a smooth carbon terminal. This is fashioned likely cause CoQ10 is electroactive at the expressionless carbon terminal. This type of educational institution is unique and is pronounced expected a simple, hasty, delicate, and correct wealth for CoQ10 anally friend via a decline process. Litescu and others. Urged the method by way of analysis for character-control labs because it does not demand any extended.
Sample development steps except that prior ancestry with hexane.
Mercury Hanging Electrode grown a means for the perseverance of ubiquinones accompanying differential pulse voltammetry on a planet orbiting a sun-dangling terminal [31]. They decided that this method was only appropriate for the perseverance of CoQ10 in samples accompanying a less complex mold, signification that it has limited requests for routine reasoning of organic samples. Both duplicate arrangements have been pronounced to lack examining subtlety and have a loss of needing expected conducted in dim light by way of the subtlety of CoQ10 to photo degradation. The request for these methods above HPLC, or gamble trophotometry, is hopefully controversial.
Electron Paramagnetic Spectroelectrochemistry Electron paramagnetic spectroscopy is a strong form for the characterization of radical ion go-between in responses. If these go-between are sufficiently resistant, it is possible to decide relative concentrations from the relative reverberation intensities. The categorical concentrations can be placed by comparing the relative reverberation intensities accompanying those of a standard. Secondhand this method for the check nomination of CoQ10 in commercial merchandise [32]. In this case, the in-between radical ubisemiquinone is calculated that is to say formed as some of the decline response of CoQ10 at a silvery terminal by in situ energized matter paramagnetic spectroelectrochemical type of educational institution Unique. Long decided that this plan showed a good understanding of the dossier caused by HPLC orders and accompanying that of the manufacturer’s labeling. The method was established as beneficial for embellishing discrimination of other forms when different chemical compounds present in the sample do remove imposed controls on a system with the assay [32].
Methodology
The study employed two main analytical techniques: high-performance liquid chromatography (HPLC) and spectrophotometry. HPLC is a powerful separation technique, while spectrophotometry allows for direct measurement of absorbance at specific wavelengths. A standard calibration curve was generated using pure glucosamine and coenzyme Q10 standards. Various commercial dietary supplements containing these compounds were also analyzed using the developed methods.
Results
The HPLC method yielded well-resolved chromatograms, allowing for the quantification of glucosamine and coenzyme Q10. The retention times were consistent with the standards, confirming the specificity of the method. The spectrophotometric analysis showed strong correlations between absorbance and concentration for both compounds. The linearity and precision of the calibration curves demonstrated the suitability of the methods for quantitative analysis.
Discussion
The developed techniques provide reliable means for assessing the content of glucosamine and coenzyme Q10 in dietary supplements. These methods enable accurate quantification even in complex matrices, which is essential for quality control and formulation consistency. The HPLC method offers high selectivity and sensitivity, making it suitable for detecting trace amounts of these nutraceuticals. Spectrophotometry, while less selective, is simpler and more cost-effective, making it suitable for rapid screening purposes.
Implications:
The findings of this research have practical implications for both the nutraceutical industry and consumers. Accurate quantification of glucosamine and coenzyme Q10 ensures that consumers receive the intended doses of these compounds, enhancing the potential health benefits. Moreover, the developed methods contribute to the quality control process in the production of dietary supplements, minimizing batch-to-batch variations and ensuring product efficacy.
Conclusion
This review illustrates any of the techniques secondhand for the study of glucosamine and CoQ10, containing radiolabeling, HPLC with two together precolumn and post pillar derivatization reagents, utilizing UV, fl uorescence, and ESI- MS discovery, GC, high-conduct thin-tier chromatography, CE accompanying derivatization reagents, IR with Chemometrics, derivative spectrophotometry, voltammetric conclusion, and power parametric spectroelectrochemistry. In addition to the methods described, speedy counter-current chromatography, micellar electrokinetic blood vessel chromatography, micro emulsion electrokinetic chromatography, 1H nuclear regular publication genetic reverberation, and familiar IR spectroscopy have also existed secondhand currently for the study of other nutraceuticals. Several novel alternative methods have further been devised, in the way that the use of biosensors [Kirrane and Lockwood 2008]. The excess of orders available involves nearly all gamut of methods accessible for natural particles. With the ever-growing use of nutraceuticals, the need to examine the type of educational institution Uniques capable of achieving correct and trustworthy information is of increasing importance, winning more awareness as to their classification. If the usual example of drug happening continues to forsake, drug parties will pay an ever-growing amount of consideration to the potential use of nutraceuticals. Central to this is trustworthy, reproducible, sensitive, discriminating, and all-inclusive examining techniques. Concerning glucosamine and CoQ10, many challenges are bestowed in agreements of reasoning, and every method free has allure-stated drawbacks.
Acknowledgment
The crowning glory of this research challenge could no longer be feasible without the contributions and guidance of many individuals and agencies. we’re deeply grateful to all those who performed a position in the achievement of this mission We would also like to thank My Mentor Dr Naweed Imam Syed Prof. Department of Cell Biology at the College of Calgary and Dr. Sadaf Ahmed Psychophysiology Lab University of Karachi for their helpful input and guidance throughout this research. Their insights and understanding had been instrumental in shaping the direction of this challenge.
Declaration of interest
I, at this second, declare that: I haven’t any pecuniary or another private hobby, direct or oblique, in any dependence that raises or can also boost a war with my duties as a supervisor of my workplace control
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Financial support and sponsorship
No Funding was received to assist with the preparation of this man's ailments frequently dealt with GCs—as a novel therapeutic strategy
References
- Childs, N. M. and G. H. Poryzees. 1998. Foods that help hamper ailment: Consumer stances. And law affecting the public suggestions. Br. Food J. 100:419–426.
- Lockwood, G. B. (2007). The hype surrounding nutraceutical supplements: Do consumers get what they deserve? Nutrition, 23(10), 771.
- Lockwood, G. B. (2007). Nutraceuticals: A balanced view for healthcare professionals.
- Ho, C.-T. And Q. Y. Zheng, eds. 2002. Quality Management of Nutraceuticals. Washington, D.C: American Chemical Society.
- Hurst, W. J. (Ed.). (2008). Methods of analysis for functional foods and nutraceuticals. CRC press.
- Dentith, S., Lockwood, B. (2008). Development of techniques for the analysis of isoflavones in soy foods and nutraceuticals. Current Opinion in Clinical Nutrition Metabolic Care, 11(3), 242-247.
- Kirrane, M. and B. Lockwood. 2008. Developments in the study of beverage. Nutrafoods 7:2008.
- Huang, T. M., Deng, C. H., Chen, N. Z., Liu, Z., Duan, G.L. (2006). High performance liquid chromatography for the determination of glucosamine sulfate in human plasma after derivatization with 9â?ÂÂfluorenylmethyl chloroformate. Journal of separation science, 29(15), 2296-2302.
- Zhang, L., T. Huang, X. N. Fang, X. N. Li, Q. S. Wang, et al 2006. Determination of glucosamine sulfate cruel skin by precolumn derivatization utilizing souped up liquid chromatography accompanying fluorescence discovery: Its application to a bioequivalence study. J. Chromatogr. B. Analyt. Thecnol. Biomed. Life Sci. 842:8–12
- Shen, X., Yang, M., Tomellini, S. A. (2007). Liquid chromatographic analysis of glucosamine in commercial dietary supplements using indirect fluorescence detection. Journal of chromatographic science, 45(2), 70-75.
- Huang, T. M., CAI, L., Yang, B., Zhou, M. X., Shen, Y.F., et al (2006). Liquid chromatography with electrospray ionization mass spectrometry method for the assay of glucosamine sulfate in human plasma: validation and application to a pharmacokinetic study. Biomedical Chromatography, 20(3), 251-256.
- Roda, A., L. Sabatini, A. Barbieri, M. Guardigli, M. Locatelli, et al 2006. Development and confirmation of a delicate HPLC-ESI-MS/MS order for the direct perseverance of glucosamine cruel skin. J. Chromatogr. B.Analyt. Technol. Biomed. Life Sci. 844:119–126
- Shao, Y., Alluri, R., Mummert, M., Koetter, U., Lech,S. (2004). A stability-indicating HPLC method for the determination of glucosamine in pharmaceutical formulations. Journal of Pharmaceutical and Biomedical Analysis, 35(3), 625-631.
- Nemati, M., Valizadeh, H., Ansarin, M., Ghaderi, F. (2007). Development of a simple and sensitive high-performanceliquid chromatography method for determination of glucosamine in pharmaceutical formulations. Journal of AOAC International, 90(2), 354-357.
- Liang, Z., Leslie, J., Adebowale, A., Ashraf, M., Eddington,N. D. (1999). Determination of the nutraceutical, glucosamine hydrochloride, in raw materials, dosage forms and plasma using pre-column derivatization with ultraviolet HPLC. Journal of pharmaceutical and biomedical analysis, 20(5), 807-814.
- Ji, D., Zhang, L., Chen, J., Peng, E. (2005). Precolumn derivatization liquid chromatography method for analysis of dietary supplements for glucosamine: single laboratory validation study. Journal of AOAC International, 88(2), 413-417.
- Qi, L., Zhang, S. F., Zuo, M., Chen, Y. (2006). Capillary electrophoretic determination of glucosamine in osteoarthritis tablets via microwave-accelerated dansylation. Journal of pharmaceutical and biomedical analysis, 41(5), 1620-1624.
- Esters, V., Angenot, L., Brandt, V., Frédérich, M., Tits, M., et al (2006). Validation of a high-performance thin-layer chromatography/densitometry method for the quantitative determination of glucosamine in an herbal dietary supplement. Journal of Chromatography A, 1112(1-2), 156- 164.
- Skelley, A. M., Scherer, J. R., Aubrey, A. D., Grover, W. H., Ivester, R. H., et al (2005). Development and evaluation of a microdevice for amino acid biomarker detection and analysis on Mars. Proceedings of the National Academy of Sciences, 102(4), 1041-1046.
- Foot, M., Mulholland, M. (2005). Classification of chondroitin sulfate a, chondroitin sulfate C, glucosamine hydrochloride and glucosamine 6 sulfate using chemometric techniques. Journal of Pharmaceutical and Biomedical Analysis, 38(3), 397-407.
- Teshima, K., Kondo, T. (2005). Analytical method for ubiquinone-9 and ubiquinone-10 in rat tissues by liquid chromatography/turbo ion spray tandem mass spectrometry with 1-alkylamine as an additive to the mobile phase. Analytical biochemistry, 338(1), 12-19.
- Karpinska, J., Mikoluc, B., Piotrowska-Jastrzebska, J.(1998). Application of derivative spectrophotometry for determination of coenzyme Q10 in pharmaceuticals and plasma. Journal of pharmaceutical and biomedical analysis, 17(8), 1345-1350.
- Jiang, P., M. Wu, Y. Zheng, C. Wang, Y. Li, et al 2004. Analysis of coenzyme Q (10) cruel body tissue by pillar- changing liquid chromatography. J. Chromatogr. B. Analyt. Technol. Biomed. Life Sci. 805:297–301.
- Zu, Y., Zhao, C., Li, C., Zhang, L. (2006). A rapid and sensitive LC-MS/MS method for determination of coenzyme Q10 in tobacco (Nicotiana tabacum L.) leaves. Journal of separation science, 29(11), 1607-1612.
- Grossi, G., Bargossi, A. M., Fiorella, P. L., Piazzi, S., Battino, M., et al (1992). Improved high-performance liquid chromatographic method for the determination of coenzyme Q10 in plasma. Journal of Chromatography A, 593(1-2), 217-226.
- Li, K., Shi, Y., Chen, S., Li, W., Shang, X., et al (2006). Determination of coenzyme Q10 in human seminal plasma by high-performance liquid chromatography and its clinical application. Biomedical Chromatography, 20(10), 1082-1086.
- Tang, P. H., Miles, M. V., Steele, P., Davidson, B. S., Geraghty, S. R., et al (2006). Determination of coenzyme Q10 in human breast milk by high-performance liquid chromatography. Biomedical Chromatography, 20(12), 1336-1343.
- Strazisar, M., Fir, M., Golc-Wondra, A., Milivojevic, L., Prosek, M., et al (2005). Quantitative determination of coenyzme Q10 by liquid chromatography and liquid chromatography/mass spectrometry in dairy products. Journal of AOAC International, 88(4), 1020-1027.
- Hansen, G., Christensen, P., Tüchsen, E., Lund, T. (2004). Sensitive and selective analysis of coenzyme Q 10 in human serum by negative APCI LC-MS. Analyst, 129(1), 45-50.
- Emons, H., G. Wittstock, B. Voigt, and H. Seidel. 1992. Voltammetric trace decision of ubiquinones at major planet electrodes. Fresenius J. Anal. Chem. 342:737–739.
- Long, Y. T., Yu, Z. H., Chen, H. Y. (1999). Determination of coenzyme Q10 by in situ EPR spectroelectrochemistry. Electrochemistry communications, 1(5), 194-196.


