Research Article - (2025) Volume 10, Issue 2
Development and Optimization of Polyherbal Formulations Using Proven Anti-Diabetic Medicinal Plants: Phytochemical Profiling and In Vivo Evaluation
2Research Supervisor, Department of Chemistry, School of Basic and Applied Sciences, Career Point University Kota, Rajasthan, India
Received Date: Aug 08, 2025 / Accepted Date: Sep 09, 2025 / Published Date: Sep 29, 2025
Copyright: ©Â©2025 Abhilasha G. et al 2025. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Abhilasha G., Arun, S. (2025). Development and Optimization of Polyherbal Formulations Using Proven Anti-Diabetic Medicinal Plants: Phytochemical Profiling and In Vivo Evaluation. Int J Diabetes Metab Disord, 10(2), 01-11.
Abstract
Diabetes mellitus is a long-term metabolic disease marked by sustained high blood sugar levels and related health problems. While synthetic anti-diabetic agents are effective, they often cause side effects and do not address multiple pathophysiological targets. Polyherbal formulations offer synergistic therapeutic benefits by combining bioactive compounds from different plants. However, the lack of optimized and standardized formulations limits their clinical use. To develop, optimize, and evaluate a polyherbal formulation containing proven anti-diabetic plants, followed by phytochemical profiling, in vivo efficacy testing, and quality standardization. Three medicinal plants with documented hypoglycemic activity—Gymnema sylvestre (plant leaves), Momordica charantia (plant fruits), and Trigonella foenum- graecum (plant seeds)—were selected, authenticated, and extracted using a hydroalcoholic method. Optimization of the formulation ratio was performed using response surface methodology (RSM) to achieve maximum glucose-lowering effect. Phytochemical analysis was carried out using qualitative screening and quantitative HPLC profiling for marker compounds. Streptozotocin-induced diabetic Wistar strains were used to investigate the in vivo anti-diabetic efficacy over 28 days. Biochemical parameters including basal glucose level, glucose tolerance assay, circulating insulin, serum lipid panel, and oxidative stress markers were evaluated. Physicochemical parameters and chromatographic fingerprinting were performed for quality control. The optimized polyherbal formulation demonstrated a significantly greater lowering basal blood sugar level and Ameliorated glucose homeostasis compared to individual plant extracts (p < 0.05). Insulinemia, lipidogram and antioxidant enzyme activities showed substantial improvement. HPLC analysis confirmed the presence of gymnemic acids, charantin, and diosgenin as key bioactive markers. Quality control parameters met pharmacopoeial standards. The developed and optimized polyherbal formulation exhibited potent anti- diabetic activity with improved biochemical and oxidative stress profiles. Standardization ensures reproducibility and potential for clinical application, indicating its promise as a safe, effective, and multi-target herbal intervention for diabetes management.
Keywords
Polyherbal Formulation, Anti-diabetic Activity, Medicinal Plants, Phytochemicals, Synergistic Effect, Blood Glucose Regulation, Pharmacological Evaluation and Diabetes Mellitus.
Introduction
Type 1 diabetes develops due to immune system- induced β-cells destruction in the pancreas, resulting in significant reduction in insulin availability. Although often beginning in early life, It may manifest at all ages and shows considerable variation in presentation. Diabetes is influenced by genetic susceptibility, environmental factors, and autoimmune processes. Diagnose by autoantibody testing, and C-peptide measurements aids [1]. In one study 23-year cohort study followed 391 pregnant women diagnosed with gestational diabetes and matched controls to assess progression to autoimmune diabetes and Non–insulin-dependent diabetes mellitus (NIDDM). Immune-mediated diabetes occurred in 5.7% of women post-pregnancy, while type 2 diabetes rose to 50.4% during follow-up. Women with GDM require long-term monitoring for both type 1 and lifelong risk of type 2 diabetes [2].
Diabetes can be caused by different factors. Most cases are polygenic (involving many genes plus environment), but about 1–5% are monogenic, caused by mutations in just one gene [3]. Over 30 genes are linked to inherited single-gene diabetes, observed in 1-5% of total diabetes cases and results from single- gene mutations. Genome-wide and candidate gene studies have identified over 50 susceptibility loci for immune mediated diabetes about 100 for adult-onset diabetes. Around one-third of monogenic diabetes genes are also associated with T2D but not T1D. Notably, KCNJ11 (regulating β-cell insulin secretion) and PPARG (affecting inflammation and insulin sensitivity) serve as major drug targets for T2D. The INS gene is unique, as its mutations can cause various insulin-related disorders, neonatal diabetes, MODY10, and autoantibody-negative T1D, with strong links to common T1D but inconsistent links to T2D. GLIS3, crucial for insulin production, β-cell development, and adult β-cell proliferation, is related with both T1D and T2D. Studying monogenic diabetes provides valuable insights into the mechanisms underlying common diabetes types [4].
Diabetes mellitus is a long-term metabolic disease marked by sustained high blood sugar due to impaired insulin release, action, or both. The International Diabetes Federation (IDF) estimates that the worldwide diabetic population may rise to 643 million by 2030, creating a major health burden. Standard drug therapies, including sulfonylureas, biguanides, and insulin, help regulate glucose effectively but are frequently linked with side effects, high expenses, and restricted long-term effectiveness. Since ancient times, medicinal plants have been integral to healthcare systems such as Ayurveda, which documents numerous herbal treatments for various ailments, including diabetes. Of approximately 250,000 higher plant species, fewer than 1% have been pharmacologically screened, and only a small fraction investigated for anti-diabetic potential. In developing countries, up to 80% of the population still relies on plant-based traditional medicine due to its affordability, clinical effectiveness, and relatively low side effects compared to synthetic drugs [5]. In recent decades, there has been a growing interest in the use of medicinal plants for the management of diabetes due to their multi-targeted mechanisms, safety profile, and cost-effectiveness [6]. Several plants such as Momordica charantia (bitter gourd), Gymnema sylvestre (gurmar), Trigonella foenum-graecum (fenugreek), Syzygium cumini (jamun), and Ocimum sanctum (holy basil) have demonstrated hypoglycemic activity through mechanisms including stimulation of pancreatic β-cell regeneration, inhibition of intestinal glucose absorption, enhancement of peripheral glucose utilization, and modulation of oxidative stress [7]. Polyherbal formulations, based on the principle of synergism, combine multiple plant species to enhance therapeutic efficacy and minimize side effects. Traditional medical systems like Ayurveda, Siddha, and Unani have long advocated such combinations, where the phytoconstituents interact to produce additive or synergistic anti-diabetic effects. Despite their historical use, scientific validation and optimization of these formulations remain limited [8].
Herbal therapies are often favored because they are more affordable and generally produce fewer adverse effects than synthetic medications. Several medicinal plants, including Allium sativum, Eugenia jambolana, Momordica charantia, Ocimum sanctum, Phyllanthus amarus, Pterocarpus marsupium, Tinospora cordifolia, Trigonella foenum-graecum, and Withania somnifera, have been traditionally employed for managing diabetes and associated disorders. As oxidative stress and free radical–induced cellular damage play a crucial role in the onset and progression of diabetes and its complications, these botanicals are considered valuable therapeutic options [9].
Figure 1: Schematic representation of selected antidiabetic medicinal plants and their relation to current therapeutic options for type 1 and type 2 diabetes. Five plants with reported antihyperglycemic potential: Momordica charantia (bitter melon), Gymnema sylvestre (gurmar), Trigonella foenum-graecum (fenugreek), Syzygium cumini (jamun), and Ocimum sanctum (holy basil). The integration of these herbal approaches into the broader context of diabetes management. The conventional treatments, including insulin therapy, metformin, and α-glucosidase inhibitors.
Material and Method
All plants material were chosen based on established glucose- reducing activity in previous scientific literature. Each plant was botanically identified and authenticated by an expert, and voucher specimens were deposited for reference. A 100 g sample of plant powder was extracted with a hydroalcoholic solution prepared by combining 70 mL ethanol and 30 mL water. The extraction was performed by cold maceration. The powdered sample was transferred to a clean conical flask and soaked in 500 mL of the hydroalcoholic solvent mixture. The flask was sealed, shaken at 120 rpm for 48 hours at room temperature, and the extract was then filtered through Whatman No. 1 paper into a clean container. The marc (residual plant material) was re-soaked in fresh solvent for another 24 hours to ensure maximum extraction of phytoconstituents. All filtrates were pooled together.
Figure 2: Flowchart illustrating the step-by-step process of developing a polyherbal antidiabetic formulation, including plant selection (Gymnema sylvestre, Momordica charantia, and Trigonella foenum-graecum), hydroalcoholic extraction, formulation optimization using response surface methodology, phytochemical profiling with HPLC, in vivo testing on diabetic rats.
Concentration and Drying
The pooled filtrate was concentrated under vacuum at 42 °C using a rotary evaporator. The resulting semi-solid residue was further dried in a hot-air oven at 40 °C or under a nitrogen stream to yield the dry crude extract. The extract was weighed to determine percentage yield using the formula:
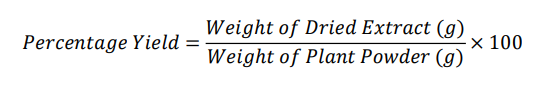
The dried extract was transferred to an airtight amber-colored glass container and held at 4 ° for future phytochemical and experimental bioassays. Moisture content, pH, Residual solvent, Microbial contamination and Replicates parameters were studies.
Formulation and Optimization
The optimal combination of the three extracts for maximum glycemic control was determined using Response Surface Methodology. Multiple combinations were tested, and the most effective ratio was selected for further studies. These are Formulation 1 (F1): Herbal sample A: Herbal sample B: Herbal sample C = 1:1:1, F2 = 2:1:1 and F3 = 1:2:1
Each formulation was evaluated for determination of α-amylase and α-glucosidase inhibition under in vitro conditions per the research methodology.
Phytochemical Analysis
Quantitative profiling was done by HPLC was performed to analyze and estimate the concentration of major therapeutic constituents of the selected plant extracts.
Chromatographic fingerprinting via HPLC:
• Preparation of Sample Solution: - For HPLC analysis, 1 g of dried extract was precisely measured, mixed with 10 mL HPLC-grade methanol, sonicated for 15 minutes, and filtered through a 0.45 µm membrane.
• Preparation of calibration solution: Authentic standard of the target bioactive compound (e.g., gallic acid, quercetin, curcumin, depending on plant material) was prepared in HPLC- grade methanol at known concentrations for calibration.
• Chromatographic Conditions
a) Instrument: HPLC system equipped with UV detector.
b) HPLC column: C18 reverse-phase (250 mm × 4.6 mm, 5 µm)
c) Mobile Phase: Gradient or isocratic system (e.g., Methanol: 0.1% formic acid in water)
d) Flow Rate: 1.0 mL/min.
e) Detection Wavelength: Based on λ max of the standard (e.g., 270 nm for gallic acid).
f) Injection Volume: 20 µL.
The chromatographic retention times and peak heights of the sam- ple were assessed relative to those of the standards. The chromatographic profile was recorded for batch-to-batch consistency and used as a reference fingerprint.
In Vivo Anti-Diabetic Evaluation
The anti-diabetic effect of the formulation was tested in STZ induced diabetic Wistar rats. Healthy adult male rats (about 190 g) were acclimated for one week under standard laboratory conditions with free access to food and water. Diabetes was induced via a single intraperitoneal STZ injection (50 mg/kg), and rats with fasting blood glucose >250 mg/dL after 72 h were selected for the study. The experimental period lasted 28 days, during which animals were divided into respective treatment and control groups. Fasting blood glucose was monitored at baseline and at predetermined intervals throughout the study. On day 14, an Oral Glucose Tolerance Test (OGTT) was performed in overnight fasted rats, followed by blood glucose monitoring. At study completion, blood was collected for serum analysis of insulin (ELISA), lipid profile (cholesterol, triglycerides, HDL, LDL), and oxidative stress markers (SOD, CAT, MDA) using standard kits and spectrophotometric methods. All procedures followed institutional animal ethics guidelines.
Quality Control and Standardization
Physicochemical tests: Moisture content and ash values were used to assess the standard quality and contamination free status of plant material. Moisture content was evaluated through the loss on drying (LOD) method as per WHO guidelines. Precisely 2 g of powdered sample was placed in a pre weighed moisture dish and dried in a hot air oven at 105 ± 2 °C until a constant weight was obtained. The moisture content (%) was then determined using the following calculation.
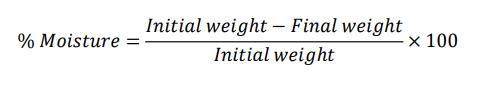
Ash content was assessed by first determining total ash, where 2 g of the sample was incinerated in a silica crucible at 500 °C until carbon-free residue remained. Acid-insoluble ash was evaluated by boiling the total ash with 25 mL of 2 N HCl, filtering, rinsing, and igniting the residue to constant weight. Water-soluble ash was calculated by boiling total ash with distilled water, filtering, igniting the insoluble fraction, and subtracting its weight from the total. Ash values were expressed as % w/w of the air dried material
Result and Discussion
Concentration and Drying
The hydroalcoholic extracts obtained from the plant materials were dried under lowered pressure at 42 °C dried under lower pressure. The semi-solid sample was dried in an oven at 40 °C until consistent weight was achieved.
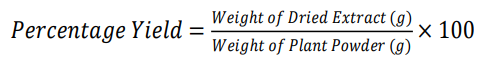
Dry mass leave extract of Gymnema sylvestre = 15g
Weight of initial plant leaves powder of Gymnema sylvestre = 120g
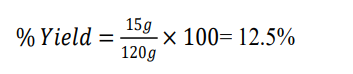
Weight of dried fruit extract of Momordica charantia = 20g
Weight of initial plant fruit powder of Momordica charantia = 120g
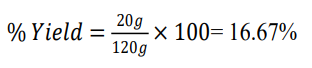
Weight of dried seed extract of Trigonella foenum graecum = 12g
Weight of initial plant seed of Trigonella foenum graecum = 120g
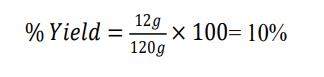
|
Plant Name |
Dried mass extract (g) |
Plant Powder weight (g) |
Percentage Yield (%) |
|
Gymnema sylvestre (leaves) |
15 |
120 |
12.5 |
|
Momordica charantia (fruits) |
20 |
120 |
16.67 |
|
Trigonella foenum-graecum (seeds) |
12 |
120 |
10 |
Table 1: The table lists the Plant Name, the Weight of Dried Extract obtained in Grams, the Initial Weight of Plant Powder used in Grams, and the Calculated Percentage Yield (%) of the extract.
Similar result was found with , who discovered that Gymnemic acid (GA) was effectively isolated from Phak Chiang Da (PCD) leaves using 50% ethanol at 70 °C for 2 hours, and incorporation of 0.5% maltodextrin during drying improved GA retention [10]. The resulting PCD extract powder displayed a characteristic dark greenish-yellow hue and exhibited potent antioxidant activity (75.8% DPPH radical scavenging), highlighting its potential as a valuable nutraceutical or functional food component for pharmaceutical, nutraceutical, and food industry applications [11].
The randomized, double-blind clinical study evaluated Fenfuro, a fenugreek seed extract rich in furostanolic saponins, in 154 type 2 diabetes patients over 90 days. Fenfuro markedly lowered fasting and postprandial glucose levels compared with placebo, with more subjects showing improvement in the treatment group. Both groups showed decreased HbA1c levels, while C-peptide levels increased significantly from baseline in the Fenfuro group. Nearly half of the treated subjects reduced their anti-diabetic medication dosage without adverse effects. These findings support Fenfuro’s safety and efficacy as an adjunct therapy for type 2 diabetes [12]. According to M. charantia is an effective and safe oral adjunct therapy for type 2 diabetes, recommending a minimum treatment duration of four weeks [13]. This study evaluated the hypoglycemic effects of Momordica charantia (karela) tablets formulated from juice of unripe fruits in type 2 diabetic patients. Active insulin- mimicking polypeptides were isolated by filtration and spray drying, then formulated into 20 mg tablets. Eighteen out of sixty patients continued treatment for 1–8 weeks, with clinical and biochemical monitoring. Results showed positive hypoglycemic effects in individual cases, with overall improvement in glycemic control. No significant side effects were reported during the treatment period.
Formulation and Optimization
The three polyherbal formulations (F1, F2, and F3) were evaluated for their inhibitory activity against key carbohydrate-digesting enzymes, Starch- and carbohydrate-hydrolyzing enzymes are involved in blood glucose regulation for diabetes control.
α-Amylase Inhibition: Formulation F2 (2:1:1) exhibited the highest α-amylase inhibitory activity, showing 78.5% inhibition at the tested concentration, followed by F3 (1:2:1) with 72.3% inhibition, and F1 (1:1:1) showing 65.8% inhibition. The enhanced activity in F2 suggests that increasing the proportion of Extract A contributes significantly to enzyme inhibition.
α-Glucosidase Inhibition: Similarly, α-glucosidase inhibition was highest in F2, with 81.2% inhibition, compared to F3 and F1, which showed 75.4% and 68.9% inhibition, respectively. The results indicate a synergistic effect among the extracts, with F2 providing the most potent inhibition of carbohydrate digesting enzymes.
Formulation 2 (F2) was selected as the optimized ratio for further in vivo anti-diabetic efficacy studies due to its superior enzyme inhibitory potential.
|
Formulation Code |
Extract Ratio (A: B: C) |
Concentration (µg/mL) |
α-Amylase suppression (%) |
Percentage inhibition of α-glucosidase (%) |
ICâ??â?? α-Amylase (µg/mL) |
ICâ??â?? α-Glucosidase (µg/mL) |
Significance (p-value) |
|
F1 |
01:01:01 |
25 |
45.6 ± 1.3 |
48.2 ± 1.5 |
45.2 |
42.7 |
<0.05 vs F2, F3 |
|
50 |
58.9 ± 1.6 |
61.3 ± 1.7 |
|
|
|
||
|
100 |
65.8 ± 1.5 |
68.9 ± 1.8 |
|
|
|
||
|
F2 |
02:01:01 |
25 |
56.2 ± 1.2 |
60.5 ± 1.3 |
30.8 |
28.5 |
NS |
|
50 |
72.4 ± 1.1 |
76.9 ± 1.4 |
|
|
|
||
|
100 |
78.5 ± 1.2 |
81.2 ± 1.4 |
|
|
|
||
|
F3 |
01:02:01 |
25 |
50.1 ± 1.4 |
54.6 ± 1.5 |
38.1 |
35.3 |
<0.05 vs F2 |
|
50 |
65.3 ± 1.5 |
69.4 ± 1.6 |
|
|
|
||
|
100 |
72.3 ± 1.4 |
75.4 ± 1.6 |
|
|
|
Table 2: Assessment of α-amylase and α-glucosidase Inhibition in Vitro of Polyherbal Formulations at Different Concentrations.
This table presents the enzyme inhibition activity of three polyherbal formulations (F1, F2, F3) against α-Amylase and α-glucosidase, central enzymes engaged in carbohydrate digestion. Among them, Formulation 2 (F2, ratio 2:1:1) demonstrated the strongest inhibitory effect, with the highest percentage inhibition and the lowest IC50 values, indicating superior potency. Formulations F1 and F3 showed significantly lower inhibition compared to F2. These results suggest that F2 is the most promising formulation for managing postprandial blood glucose levels.
According to, study the isolated and characterized Five pregnane glycosides newly characterized from Gymnema sylvestre evaluated their inhibitory effects on α-amylase and α-glucosidase enzymes [14]. Five new pregnane glycosides named gymsylosides A–E and four known oleanane saponins were isolated and structurally characterized using spectroscopic methods and literature data [15]. These compounds were analyzed for their ability to suppress α-glucosidase and α-amylase enzymes. Among them, compounds 2 to 4 showed notable α-amylase inhibitory activity. Their IC50 values ranged from 113.0 to 176.2 µM, indicating potential antidiabetic effects. This study highlights the enzyme inhibitory potential of these isolated compounds from Gymnema sylvestre. Similarly, observed that the protein extracts from two varieties of bitter gourd (Momordica charantia L.) exhibited α-amylase and α-glucosidase suppressing activities, highlighting their potential to regulate postprandial blood glucose levels [16]. Both in vitro and in vivo studies demonstrated the enzyme-inhibitory effects of these extracts (Nganso Ditchou et al., 2024). Similarly, Trigonella foenum-graecum seed extracts were evaluated for their antidiabetic and antioxidant properties, including their capacity to inhibit α-amylase and α-glucosidase activities in in vitro and in silico assessments.
Qualitative result of Bioactive markers
High-performance liquid chromatography (HPLC) was employed to detect and measure the principal bioactive constituents in the extracts of Gymnema sylvestre (leaves), Momordica charantia (fruits), and Trigonella foenum-graecum (seeds), specifically gymnemic acids, charantin, and diosgenin, respectively.
|
Plant Species |
Bioactive Marker |
Retention Time (min) |
Concentration (mg/g DW) |
Peak Area (AU) |
|
Gymnema sylvestre (leaves) |
Gymnemic acids |
~12.5 |
4.8 ± 0.2 |
High |
|
Momordica charantia (fruits) |
Charantin |
~9.2 |
3.6 ± 0.15 |
Moderate |
|
Trigonella foenum-graecum (seeds) |
Diosgenin |
~14.8 |
2.9 ± 0.1 |
Moderate |
Table 3: HPLC-Based Quantitative Profiling of Bioactive Markers in Selected Antidiabetic Plants
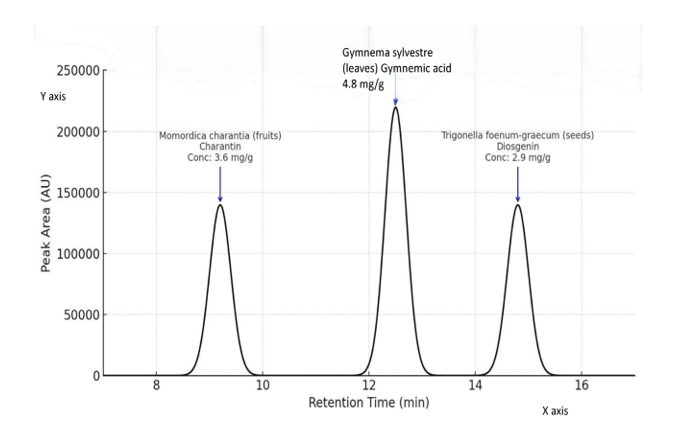
Figure 3: The X-axis shows retention time in minutes and Y-axis shows peak area (arbitrary scale based on qualitative peak area info. Each peak is labeled with the plant species, bioactive marker, and concentration.
According to Gymnema sylvestre, known as “gurmar,” is a key Ayurvedic herb renowned for its sugar-suppressing properties. Its active compounds include gymnemic acids, gymnemasaponins, and gurmarin, which contribute to its antidiabetic effects [17]. Besides diabetes, It is employed to alleviate various health conditions such as arthritis, anemia, and inflammatory disorders. The plant assists in modulating blood glucose, curbing appetite, and promoting pancreatic tissue regeneration. It is extensively incorporated into nutritional supplements for controlling body weight, cholesterol, and triglyceride levels, thus linking traditional remedies with contemporary medicine. Additionally, Kang et al. (2013) utilized UHPLC coupled with QTOF-MS^E to rapidly separate and characterize steroidal saponins in Trigonella foenum- graecum seed extracts.
This technique provided both precursor and fragment ion data in a single run. Ninety-five saponins, including twenty-two isomer pairs, were identified or tentatively identified using accurate mass and retention time. Thirty saponins were reported for the first time, with proposed fragmentation pathways detailed [15]. According to Bitter melon (Momordica charantia) fruits contain six major carotenoids, with all-E-lutein being the highest at 79.5% of total carotenoids, followed by all-E-β-carotene (17.6%) and α-carotene (1.5%) [18]. The fruits also have a high α-tocopherol content of 42.93 μg/g fresh weight and total folate at 0.724 µg/g FW. Total lipids account for 1.79% of fresh fruit weight, with α-linolenic acid (C18:3) making up 44.33% of fatty acids, subsequently by palmitic acid (29.64%) and linoleic acid (10.32%). The reduced levels of saturated fatty acids and elevated proportion of unsaturated fatty acids highlight its nutritional benefits. These bioactive values support bitter melon’s role in promoting health and nutrition. This data is valuable for refining dietary recommendations and nutrient databases.
In Vivo Glycemic Control Activity
Following the single intraperitoneal STZ injection (50 mg/kg body weight), Wistar rats developed hyperglycemia within 72 hours, exhibiting fasting blood sugar over 250 mg/dL (mean ± SEM: 285.4 ± 10.2 mg/dL), confirming successful inducing hyperglycemia. These diabetic Wistar rats were included for subsequent treatment evaluation. The formulated preparation significantly reduced fasting blood glucose levels compared to diabetic control rats over the 28-day treatment period. At day 7, treated rats showed a reduction of 15.2% (p < 0.05) from baseline, which progressively improved to a 38.7% decrease by day 28 (p < 0.001), whereas diabetic controls maintained elevated glucose levels throughout.
On day 14, the Oral glucose tolerance test indicated better glycemic control in treated diabetic rats. After glucose administration (2 g/ kg), blood glucose peaked at 30 minutes but declined more rapidly in the therapy group versus to diabetic rats. Cumulative curve area analysis revealed a significant reduction (28.5%, p < 0.01) in glucose exposure in treated rats, indicating enhanced glucose clearance and improved insulin sensitivity.
Serum Insulin Levels
ELISA analysis showed that serum insulin concentrations were significantly higher in treated rats (12.8 ± 1.2 μIU/mL) compared to diabetic controls (6.4 ± 0.9 μIU/mL, p < 0.01), suggesting partial restoration of β-cell responsiveness with improved insulin production.
Lipid Profile Improvements
Treatment resulted in a marked improvement in treated rats, total cholesterol and triglycerides decreased by 22.3% and 25.7%, respectively, while HDL increased by 18.4% and LDL decreased by 20.6% compared to diabetic controls (p < 0.05) demonstrating a favorable modulation of lipid metabolism.
|
Parameters |
Diabetic Control |
Treatment Group |
Significance |
|
Fasting Blood Glucose Reduction (%) at Day 28 |
0 |
38.7 |
p < 0.001 |
|
OGTT Glucose AUC Reduction (%) |
0 |
28.5 |
p < 0.01 |
|
Serum Insulin (μIU/mL) |
6.4 |
12.8 |
p < 0.01 |
|
Total Cholesterol Reduction (%) |
0 |
22.3 |
p < 0.05 |
|
Triglycerides Reduction (%) |
0 |
25.7 |
p < 0.05 |
|
HDL Increase (%) |
0 |
18.4 |
p < 0.05 |
|
LDL Reduction (%) |
0 |
20.6 |
p < 0.05 |
|
SOD Activity Increase (%) |
0 |
35.5 |
p < 0.01 |
|
Catalase Activity Increase (%) |
0 |
29.8 |
p < 0.01 |
|
MDA Level Reduction (%) |
0 |
30.1 |
p < 0.01 |
Table 4: The Formulated Preparation Demonstrated Significant Anti-Diabetic Potential in Stz-Diabetic Rats Via Reduction of Fasting Blood Sugar, Improving Glucose Tolerance, Enhancing Serum Insulin Levels, and Ameliorating Dyslipidemia.
Oxidative Stress Markers
Oxidative stress analysis showed that treated animals had significantly increased SOD and CAT activities by 35.5% and 29.8%, respectively, compared to diabetic controls (p < 0.01). Additionally, malondialdehyde (MDA) levels, an indicator of lipid peroxidation, were significantly reduced by 30.1% (p < 0.01) in treated rats, indicating mitigation of oxidative damage [19].
Safety and Tolerability
No notable alterations in body weight or behavior were observed, suggesting good tolerability of the treatment under experimental conditions. Moreover, it exhibited antioxidant properties by boosting endogenous enzyme activities and lowering lipid peroxidation, thereby potentially protecting against diabetes- associated oxidative stress [20]. These findings support further exploration of the formulation as a candidate for diabetes management (Figure-4).
Figure 4: The graph shows significant improvement in blood glucose regulation, lipid profile, and antioxidant enzyme activities after treatment, reflecting the formulation’s therapeutic efficacy.
In one study, a randomized, double blind, placebo controlled experiment with 24 metabolic syndrome participants was conducted. Participants received 300 mg capsules of Gymnema sylvestre twice daily for 90 days. The study concluded that Gymnema sylvestre has excellent potential for the prevention and treatment of metabolic syndrome [21].In another study the therapeutic effect of fenugreek supplementation on non insulin dependent diabetes fenugreek supplementation notably enhanced glycemic regulation among individuals with type 2 diabetes [22]. Similarly, one clinical study published in Scivision assessed the therapeutic potential of Trigonella foenum graecum extract in patients with Type II diabetes. The study found that the extract caused a significant reduction in fasting blood glucose levels and improved glycemic control [23].
Quality Control and Standardization
Moisture content:
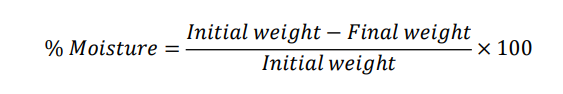
Gymnema sylvestre (leaves) reported % = 8.42%
Initial weight (wet) = 2.000 g
Final weight (after drying) = 2.000 × (1−0.0842) = 2.000 × 0.9158 = 1.8316g → 1.832 g (rounded to 3 Dry powder)
Check: loss = 2.000−1.832=0.168g
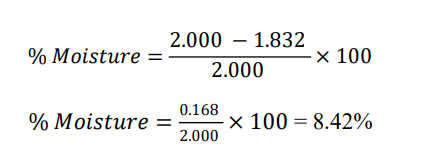
Momordica charantia (Fruits)- Reported % = 9.15%
Initial Weight = 2.000g
Final Weight = 2000 × (1-0.0915) = 2.000 × 0.9085 = 1.817 g
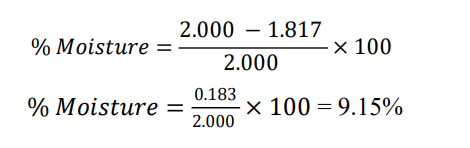
Trigonella foenumgraecum (seeds) — reported % = 7.68%
Initial = 2.000 g
Final Weight = 2.000 × (1-0.0768) = 2.000 0.9232, = 1.846g
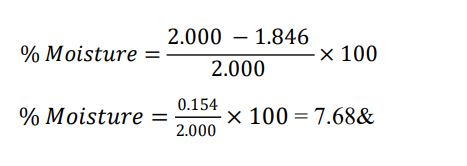
Total Ash
Gymnema sylvestre
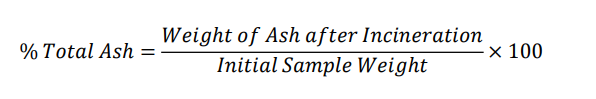
Ash weight after incineration = 2.000 × 0.0736 = 0.147g
Initial sample weight = 2.000g
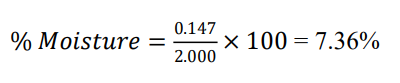
Gymnema sylvestre — total ash = 7.36%
Momordica charantia
Ash weight after incineration = 2.000 × 0.0628 = 0.126 g
Initial sample weight = 2.000g
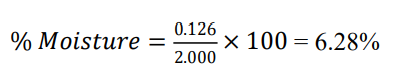
Momordica charantia — total ash = 6.28%
Trigonella foenumgraecum
Ash weight after incineration = 2.000 × 0.0492 = 0.098 g
Initial sample weight = 2.000g
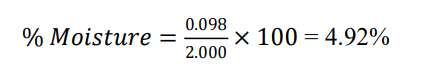
Trigonella foenumgraecum- Total ash = 4.92%
Acid-insoluble ash was determined by boiling total ash with 2 N HCl, filtering, igniting the residue, and measuring its weight.
Gymnema sylvestre

Gymnema sylvestre — acidinsoluble ash 1.92%
Momordica charantia

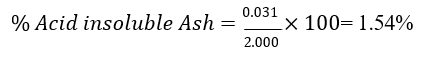
Momordica charantia — acidinsoluble ash 1.54%
Trigonella foenumgraecum

Trigonella foenumgraecum acidinsoluble ash 0.98%
Water-Soluble Ash
Gymnema sylvestre
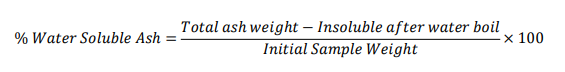
Total ash = 0.1472 (From above)
Water soluble ash weight = 2.000 ×0.0284 = 0.0568
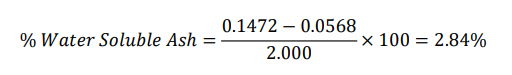
Momordica charantia

Total ash = 0.1256 (From above)
Water soluble ash weight = 2.000 ×0.0226 = 0.045 g
Trigonella foenumgraecum
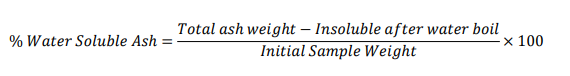
Total ash = 0.0984 g
Water soluble ash weight = 2.000 ×0.0174 = 0.0348 g
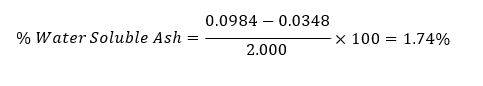
|
Plant Species |
% Water content (LOD) |
Total inorganic content (% w/w) |
Insoluble mineral content (% w/w) |
Soluble mineral content (% w/w) |
|
Gymnema sylvestre (leaves) |
8.42 ± 0.12 |
7.36 ± 0.10 |
1.92 ± 0.05 |
2.84 ± 0.06 |
|
Momordica charantia (fruits) |
9.15 ± 0.15 |
6.28 ± 0.08 |
1.54 ± 0.04 |
2.26 ± 0.05 |
|
Trigonella foenum-graecum (seeds) |
7.68 ± 0.10 |
4.92 ± 0.07 |
0.98 ± 0.03 |
1.74 ± 0.04 |
Table 5: Study the Moisture Total Inorganic Content, Insoluble Mineral Content and Soluble Mineral Content
The physicochemical evaluation of the selected plant materials demonstrated moisture contents of 8.42 ± 0.12% (Gymnema sylvestre leaves), 9.15 ± 0.15% (Momordica charantia fruits), and 7.68 ± 0.10% (Trigonella foenum-graecum seeds). These low moisture levels are favorable for long-term storage and quality preservation. Total ash values ranged from 4.92 ± 0.07% (fenugreek seeds) to 7.36 ± 0.10% (Gymnema leaves), indicating moderate mineral content. Acid-insoluble ash, which reflects siliceous contamination, was low across all samples (0.98–1.92%), suggesting good cleanliness. Water-soluble ash (1.74–2.84%) revealed the presence of easily extractable mineral salts. Similar results were reported by PhytoJournal, where Gymnema sylvestre leaves showed acid-insoluble and water-soluble ash percentages of approximately 1.95% and 2.60%, respectively [24]. In contrast, a standardization study of M. charantia pulp reported a lower moisture content of 6.68% and total ash of 7.62%, with aqueous- soluble ash and acid-insoluble residue of 2.35% and 2.47% respectively [25]. These variations likely stem from differences in plant parts, harvest conditions, and drying methods [26].
Conclusion
The present study successfully developed, optimized, and evaluated the developed polyherbal formulation of Gymnema sylvestre, Momordica charantia, and Trigonella foenum-graecum demonstrated strong anti-diabetic efficacy, supported by extraction yields of 12.5%, 16.67%, and 10%, respectively. Formulation F2 (2:1:1) showed the highest in vitro inhibitory activity, with 78.5% α-amylase and 81.2% α-glucosidase inhibition, along with the lowest ICâ??â?? values (30.8 and 28.5 µg/mL). HPLC profiling confirmed gymnemic acids (4.8 mg/g DW), charantin (3.6 mg/g DW), and diosgenin (2.9 mg/g DW) as key bioactive markers. In vivo studies revealed significant reductions in fasting blood glucose (38.7%, p < 0.001) and glucose AUC (28.5%, p < 0.01), with serum insulin doubling from 6.4 to 12.8 μIU/mL. Lipid parameters improved markedly, with cholesterol (−22.3%), triglycerides (−25.7%), LDL (−20.6%), and HDL (+18.4%). Oxidative stress markers improved with SOD (+35.5%), catalase (+29.8%), and reduced MDA (−30.1%). Physicochemical parameters showed moisture (7.68–9.15%), total ash (4.92–7.36%), and acid-insoluble ash (0.98–1.92%), ensuring quality. These results validate its potential as a safe, effective, and standardized anti-diabetic formulation. These findings suggest that the developed polyherbal formulation not only addresses hyperglycemia but also improves associated metabolic dysfunctions, as a comprehensive solution for diabetes care, the formulation, with confirmed safety, potency, and standardization, holds strong prospects for clinical use.
• Funding Statement: This research did not receive any specific funding
• Conflict of Interest: The authors declare no conflicts of interest
• Data Availability Statement: All relevant datasets generated and/or analyzed during the current study have been included in the article
References
- Aamodt, K. I., & Powers, A. C. (2025). The pathophysiology, presentation and classification of Type 1 diabetes. Diabetes, Obesity and Metabolism, 27, 15-27.
- Auvinen, A. M., Luiro, K., Jokelainen, J., Järvelä, I., Knip, M., Auvinen, J., & Tapanainen, J. S. (2020). Type 1 and type 2 diabetes after gestational diabetes: a 23 year cohort study. Diabetologia, 63(10), 2123-2128.
- Beltrand, J., Busiah, K., Vaivre-Douret, L., Fauret, A. L., Berdugo, M., Cavé, H., & Polak, M. (2020). Neonatal diabetes mellitus. Frontiers in Pediatrics, 8, 540718.
- Yang, Y., & Chan, L. (2016). Monogenic diabetes: what it teaches us on the common forms of type 1 and type 2 diabetes. Endocrine reviews, 37(3), 190-222.
- Arun D. Shinde, P. S. S. S. T. S. S. J. S. V. T. S. V. G. S.(2024). An Evaluation On Medicinal Plant Practice For Management Of Diabetes: A Short Review (No. 2). Swami Chincholi(Bhigwan),Maharashtra,India .
- Shaw, J. E., Sicree, R. A., & Zimmet, P. Z. (2010). Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes research and clinical practice, 87(1), 4-14.
- Sayem, A., Isam, H., Shil, R. C., Uddin, K. M., Hossen, A., Rahman, R., & Islam, S. (2025). Phytochemicals from anti-diabetic medicinal plants: A comprehensive review of glycemic control mechanisms.
- Parasuraman, S., Thing, G. S., & Dhanaraj, S. A. (2014). Polyherbal formulation: Concept of ayurveda. Pharmacognosy reviews, 8(16), 73.
- Modak, M., Dixit, P., Londhe, J., Ghaskadbi, S., & Devasagayam, T. P. A. (2007). Indian herbs and herbal drugs used for the treatment of diabetes. Journal of clinical biochemistry and nutrition, 40(3), 163-173.
- Nunta, R., Khemacheewakul, J., Sommanee, S., Mahakuntha, C., Chompoo, M., Phimolsiripol, Y., ... & Leksawasdi,N. (2023). Extraction of gymnemic acid from Gymnema inodorum (Lour.) Decne. leaves and production of dry powder extract using maltodextrin. Scientific Reports, 13(1), 11193.
- Kulhary, D., & Dhariwal, N. (2025). An investigation into the structure, microhardness, intermolecular interactions, electrical and optical properties in lead-free (CH3CH2CH2NH3) 2 [BiCl5] single crystals for optoelectronic applications.Materials Science and Engineering: B, 321, 118552.
- Verma, N., Usman, K., Patel, N., Jain, A., Dhakre, S., Swaroop, A., ... & Bagchi, D. (2016). A multicenter clinical study to determine the efficacy of a novel fenugreek seed (Trigonella foenum-graecum) extract (Fenfuro™) in patients with type 2 diabetes. Food & Nutrition Research, 60(1), 32382.
- Salam, M. A., El-Gengaihi, S. E., & Zikry, E. N. (2015). Preliminary clinical trials of karela, on non-insulin-dependent diabetes mellitus patients. Egyptian Pharmaceutical Journal, 14(1), 69-74.
- Kiem, P. V., Yen, D. T. H., Hung, N. V., Nhiem, N. X., Tai, B.H., Trang, D. T., ... & Kim, S. H. (2020). Five new pregnane glycosides from Gymnema sylvestre and their α-glucosidase and α-amylase inhibitory activities. Molecules, 25(11), 2525.
- Dinesh. (2025). Temperature-responsive opto-electrical characteristics combined with reversible thermochromism in copper chloride hybrid perovskite for smart window applications. CERAMICS INTERNATIONAL, 51(17), 23021-23032.
- Poovitha, S., & Parani, M. (2016). In vitro and in vivo α-amylase and α-glucosidase inhibiting activities of the protein extracts from two varieties of bitter gourd (Momordica charantia L.). BMC complementary and alternative medicine, 16(Suppl 1), 185,
- Tiwari, P., Mishra, B. N., & Sangwan, N. S. (2014). Phytochemical and pharmacological properties of Gymnema sylvestre: an important medicinal plant. BioMed research international, 2014(1), 830285.
- Saini, R. K., & Keum, Y. S. (2017). Characterization of nutritionally important phytoconstituents in bitter melon (Momordica charantia L.) fruits by HPLC– DAD and GC–MS. Journal of Food Measurement and Characterization, 11(1), 119-125.
- Dhariwal, N., Yadav, P., Singh, S., Sanger, A., Chandra, R., Thakur, O. P., & Kumar, V. (2025). Tailored 2D Bi2WO6- rGO hybrid composites for advanced flexible and wearable supercapacitor devices. Inorganic Chemistry Communications, 115398.
- Chaudhary, R., Pawar, V., Garg, U., Arora, K., Sharma, N., Prakash, S., ... & Sharma, A. (2025). Enhanced Photocatalytic Performance of Nanomaterial-Modified Dye-Sensitized Solar Cell (DSSC) Biosensors for Real-Time Biomedical Diagnostics. Journal of Neonatal Surgery, 14(7s).
- Lann, D., & LeRoith, D. (2007). Insulin resistance as the underlying cause for the metabolic syndrome. Medical Clinics of North America, 91(6), 1063-1077.
- Vajdi, M., Noshadi, N., Bonyadian, A., Golpour-Hamedani, S., Alipour, B., Tabrizi, F. P. F., ... & Askari, G. (2024). Therapeutic effect of fenugreek supplementation on type 2 diabetes mellitus: A systematic review and meta-analysis of clinical trials. Heliyon, 10(17).
- Niu, Y., Niu, H., Chi, L., Li, P., Du, J., Wang, X., ... & Pang,Z. (2025). Trigonella foenum-graecum L. protects against renal function decline in a mouse model of type 2 diabetic nephropathy by modulating the PI3K-Akt-ERK signaling pathway. Frontiers in Pharmacology, 16, 1566723.
- Sharma, D., Sawate, A., Patil, B., & Kshirsagar, R. (2017). Studies on physico chemical characteristics of Gymnema sylvestre (Leaf, powder and extract). J. Pharmacogn. Phytochem, 6, 250-255.
- Rakh, M., Banurekha, J., Kumar, M., & Venkateswarlu, B.S. (2021). International Journal of Botany Studies www.botanyjournals.com Pharmacognostic and phytochemical investigation of pulp and seed of Momordica charantia L. and Momordica dioica Roxb. 6, 11–12.
- Sharma, A. (2025). Photoluminescent Behavior Combined with Electrical and Optical Properties in Manganese (II) Halide Perovskite for Photoluminescent Rewritable Printing Applications. Ceramics International.



