Research Article - (2025) Volume 3, Issue 5
Correlation between Urinary β2-MG Level in Renal Tubular Injury and Non-Free Posture
Received Date: Sep 10, 2025 / Accepted Date: Oct 20, 2025 / Published Date: Oct 31, 2025
Copyright: ©2025 Dao Zheng. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Zheng, D. (2025). Correlation between Urinary ?2-MG Level in Renal Tubular Injury and Non-Free Posture. Int Internal Med J, 3(5), 01-08.
Abstract
Objective: To observe and analyze the correlation between urinary β2 -MG levels and non-free position during renal tubular injury. Explored the possible mechanism of renal tubular injury caused by ipsilateral renal pelvis hydrostatic pressure.
Method: A prospective study was conducted on 85 randomly selected adults to explore the etiology of kidney stones. 75 patients in non-free position and 10 healthy volunteers in free position were randomly selected and grouped according to different sleeping positions. We collected morning urine separately. The levels of urinary β2 -MG were measured using radioimmunoassay. Among them, 24 patients in non-free position were randomly selected for a second urine test. The urine test method was the same, and the interval between the two urine tests was about one month. The urine test results of the same person twice before and after form a paired sample.
A database was established using SPSS 26.0 software and statistical analysis was conducted. Non parametric tests are used for continuous numerical variables that do not follow a normal distribution, while metric data with skewed distributions are represented by M (P25, P75). Mann Whitney U rank sum tests are used to compare differences between groups. The difference is statistically significant with P<0.05 (bilateral).
Result: In the non-free position group, 69 male subjects were randomly selected, with a median urinary β2 -MGlevel of 0.19 (0.16-0.33) MG. Four female participants were randomly selected, with a median urinary β2 -MGlevel of 0.13 (0.02-0.55) Mg. We conducted an independent sample Z-test on two sets of data, P>0.05. In the non-free position group, the median value of urinary β2 -MG levels in 75 randomly selected patients was 0.19 (0.16-0.33) Mg. The median value of urinary β2 -MG levels in 10 randomly selected patients in the free position group was (0.19 (0.16-0.22) Mg. We conducted an independent sample Z-test on two sets of data, P<0.05. There are 75 research subjects in the observation group, of which 51 did not undergo a second urine test; 24 cases underwent two urine tests. This article conducted a paired sample Z-test on the results of two urine tests. The results showed that among them, the median value of the second urineβ2 -MG level was higher than the first urine test result in 12 patients who continued to adhere to non-free position for about a month, and the difference was statistically significant [0.28 (0.09-0.44) mg/L vs. 1.105 (0.45-1.41) mg/L, Z=-3.059, P=0.002];And in the other 12 patients, after about a month of transitioning to a free position, the median value of urine β2 -MG levels in the second urine test was lower than that in the first urine test, and the difference was statistically significant [0.36 (0.25-0.65) mg/L vs. 0.19 (0.15-0.20) mg/L, Z=-3.059, P=0.002].
Conclusion: The study found that non free position is closely related to the median value of urinary β2-MG levels. Tip: Non free position is closely related to impaired renal tubular reabsorption function. The renal tubular injury and changes in urine composition caused by non-free position may be related to the formation of kidney stones. Free position helps prevent kidney stones.
Keywords
Kidney stones; Non free position; Hydrostatic pressure of ipsilateral renal pelvis urine; Urinary β2-MG levels; Renal tubular injury
Introduction
Kidney stones are a common and frequently occurring disease, with a high recurrence rate after treatment such as stone dissolution or fragmentation. A study has found that renal tubular injury is associated with the occurrence of stones, but the cause of the injury is unknown. At present, research directions mainly focus on renal tubular oxidative damage, calcium channels, and local inflammatory reactions [1]. The synthesis rate and release amount ofβ2-MG from the cell membrane in normal human urine are quite constant.β2-MG can be freely filtered from the glomerulus, absorbed 99.9% in the proximal renal tubules, and decomposed and destroyed in the renal tubular epithelial cells; Therefore, under normal circumstances, the excretion of β2-MG is very trace. When the renal tubular reabsorption function is damaged, the urinary β2-MG content increases significantly. As long as the renal tubular reabsorption rate decreases by 1%, the urinary β2- MG excretion can increase by about 30 times, and the degree of increase is closely related to the degree of renal tubular injury [1]. Our previous research has shown that the onset of kidney stones is related to the patient's non free position. The so-called non free position specifically refers to positions that can cause:![]() short-term or sustained urinary retention in the ipsilateral renal pelvis,
short-term or sustained urinary retention in the ipsilateral renal pelvis, ![]() short-term or sustained inability to urinate in an upright position,
short-term or sustained inability to urinate in an upright position,![]() positions that can cause the coexistence of
positions that can cause the coexistence of![]() and
and ![]() ¸. From the perspective of anatomical features, the most unfavorable positions for urine drainage are upright, unilateral supine, supine, and a combination of unilateral and supine positions in the kidneys, which can lead to urinary stasis in the ipsilateral renal pelvis; The bladder is in a non-upright position; Therefore, it is collectively referred to as non-free position. Other sleeping positions are free positions.
¸. From the perspective of anatomical features, the most unfavorable positions for urine drainage are upright, unilateral supine, supine, and a combination of unilateral and supine positions in the kidneys, which can lead to urinary stasis in the ipsilateral renal pelvis; The bladder is in a non-upright position; Therefore, it is collectively referred to as non-free position. Other sleeping positions are free positions.
According to research, the non-free position causes kidney tissue damage and the formation of kidney stones through the sustained and limited increase in urinary hydrostatic pressure [2]. To confirm this hypothesis, the author selected urinary β2-MG levels, which can sensitively display changes in renal tubular function, as the observation index during their work in the Rehabilitation Department of Tangshan Sanatorium for Workers in Jiangsu Province (from September 2016 to June 2022). We conducted a prospective observational study on the relationship between non free position (sleeping position) and urinary β2-MG levels in a randomly selected population. The research results are reported as follows.
Data and Methods
Clinical Observation Object
Selection criteria: All randomly selected observation subjects have normal liver and kidney function, normal abdominal ultrasound, and no history of other urinary system diseases. All randomly selected observation subjects have not used aminoglycoside antibiotics or drugs that affect kidney function within the month prior to urine testing.
Exclusion criteria: Medical history includes other kidney diseases, inflammation, congenital malformations, or diseases accompanied by connective tissue disease, tumors, etc., except for urolithiasis.
Observation group: All patients randomly selected were from: from May 2017 to June 2022, among the hospitalized rehabilitation treatment patients and randomly selected outpatient physical examination patients in our hospital's rehabilitation department, 75 patients were randomly selected to have a non-free sleeping position. Among the randomly selected hospitalized rehabilitation patients: those who have been hospitalized for rehabilitation treatment for more than 1.5 months after bone and joint injury surgery (21 patients were able to get out of bed with or without crutches when they came to the hospital); Seven patients with stage 1 silicosis and six patients with mild carbon disulfide poisoning have both been hospitalized and received cumulative rehabilitation treatment for over two years. Among the outpatient physical examination patients, 41 were randomly selected who had a history of urolithiasis, had undergone lithotripsy treatment, and had been cured for more than a month after B-ultrasound follow- up. Among them, there are 71 males and 4 females. Age group from 23 to 68 years old. The body weight is 49-72 Kg.
Control group: Among the healthy volunteers who came to our hospital during the same period and had normal physical examination results, normal abdominal ultrasound, and no history of other urinary system diseases, 10 individuals were randomly selected to sleep in a free position, including 6 males and 4 females. Age group from 27 to 59 years old. The body weight is 51-71 Kg.
The members of the observation group and the control group are comparable in terms of gender, age, biochemical indicators, etc.
Method
The observation method of this study complies with the requirements of the Helsinki Declaration, and all research subjects provided informed consent. For each observation group and control group member, clearly inform them of the purpose and method of this examination: usually, patients with renal tubular injury have reduced renal tubular reabsorption function and urinary β2-MG levels exceeding normal. Our goal is to examine whether the patient has renal tubular injury by examining the level of urinary β2-MG After obtaining the patient's consent, collect the first urine sample and immediately send it for testing. After obtaining the results of the urine β2-MG test, carefully inform the research subjects of the following three points: ![]() Urinary β -MG is one of the components of kidney stones. An increase in urinary β2-MG components is beneficial for the formation of kidney stones.
Urinary β -MG is one of the components of kidney stones. An increase in urinary β2-MG components is beneficial for the formation of kidney stones. ![]() ¸.The normal value of normal adult urine β -MG test is less than 0.3mg/L. When the urine test result exceeds normal, it indicates that the patient's renal tubular reabsorption function has decreased, and there may be renal tubular injury in the body. It is necessary to correct it in a timely manner and clarify the specific correction methods.
¸.The normal value of normal adult urine β -MG test is less than 0.3mg/L. When the urine test result exceeds normal, it indicates that the patient's renal tubular reabsorption function has decreased, and there may be renal tubular injury in the body. It is necessary to correct it in a timely manner and clarify the specific correction methods.![]() Our research shows that 84.4% of patients who are accustomed to unilateral or/and supine positions have stones located in the ipsilateral kidney or ureter, while 15.6% have stones on the contralateral side. There is no significant difference in the distribution of stones on the left and right sides in supine positions, while 7% of patients are in free and prone positions. The X2 test showed significant differences between them [3]. Tip: Regularly changing between sleeping and free lying positions is of great significance in reducing renal tubular injury and preventing the occurrence of kidney stones. The purpose of our experiment is to reduce renal tubular injury and prevent the occurrence of kidney stones by changing the sleeping position. After the observation group patients expressed understanding, it is explicitly required that all observation group patients must replace their non free position when sleeping with a free position, without changing their original sleep schedule, diet, lifestyle, work, and treatment patterns. That is: When sleeping, first lie on the left (or right) side, then switch to the right (or left) side, then switch to the left (or right) side, and then switch to the right (or left) side, alternating left and right, at least every 2 hours, every night. Set an alarm on the patient's mobile phone to wake up at a scheduled time, or ask colleagues or family members in the same dormitory to supervise and consciously adhere to it. There were 75 study subjects in the observation group, of which 51 underwent one urine test and 24 underwent two urine tests. Among them, 12 patients followed the doctor's advice and changed their non free position to a free position about a month after the first urine sample was collected. They then collected urine samples using the same method for the second time and immediately sent them for testing. However, after repeatedly emphasizing the importance of changing the sleeping position, the other 12 patients were still unable to do so. About a month after the first urine sample was collected, a second urine sample was collected using the same method and immediately sent for testing. Combine the results of two urine tests before and after the same patient into a paired sample.
Our research shows that 84.4% of patients who are accustomed to unilateral or/and supine positions have stones located in the ipsilateral kidney or ureter, while 15.6% have stones on the contralateral side. There is no significant difference in the distribution of stones on the left and right sides in supine positions, while 7% of patients are in free and prone positions. The X2 test showed significant differences between them [3]. Tip: Regularly changing between sleeping and free lying positions is of great significance in reducing renal tubular injury and preventing the occurrence of kidney stones. The purpose of our experiment is to reduce renal tubular injury and prevent the occurrence of kidney stones by changing the sleeping position. After the observation group patients expressed understanding, it is explicitly required that all observation group patients must replace their non free position when sleeping with a free position, without changing their original sleep schedule, diet, lifestyle, work, and treatment patterns. That is: When sleeping, first lie on the left (or right) side, then switch to the right (or left) side, then switch to the left (or right) side, and then switch to the right (or left) side, alternating left and right, at least every 2 hours, every night. Set an alarm on the patient's mobile phone to wake up at a scheduled time, or ask colleagues or family members in the same dormitory to supervise and consciously adhere to it. There were 75 study subjects in the observation group, of which 51 underwent one urine test and 24 underwent two urine tests. Among them, 12 patients followed the doctor's advice and changed their non free position to a free position about a month after the first urine sample was collected. They then collected urine samples using the same method for the second time and immediately sent them for testing. However, after repeatedly emphasizing the importance of changing the sleeping position, the other 12 patients were still unable to do so. About a month after the first urine sample was collected, a second urine sample was collected using the same method and immediately sent for testing. Combine the results of two urine tests before and after the same patient into a paired sample.
Collection and processing of specimens: Urine specimens should be collected on an empty stomach in the morning according to the instructions, and the author should directly send them to the Laboratory Department of the First Affiliated Hospital of Nanjing Medical University for examination. Sample detection method![]() Instrument and reagent kit: Beckman Coulter IMAGE 800 specific protein analyzer from the United States; Reagent kit: Beckman Coulter matching reagents and standards from the United States Specimen testing: According to the instructions of the reagent kit, use radioimmunoassay to measure urinary β2-MG The normal value of normal adult urine β2-MG test is less than 0.3mg/L. The control group was synchronized.
Instrument and reagent kit: Beckman Coulter IMAGE 800 specific protein analyzer from the United States; Reagent kit: Beckman Coulter matching reagents and standards from the United States Specimen testing: According to the instructions of the reagent kit, use radioimmunoassay to measure urinary β2-MG The normal value of normal adult urine β2-MG test is less than 0.3mg/L. The control group was synchronized.
Results
In the randomly selected non free position group, there were 69 males with a median urinary β2-MG level of 0.19 (0.16-0.33) Mg. The median value of urinary β2-MG levels in the female group was 0.13 (0.02-0.55) Mg. We conducted an independent sample Z-test on two sets of data, P>0.05. This indicates that there is no significant statistical difference in the median value of urinary β2- MG levels between male and female group the number of samples compared in this group. See Table 1.
|
|
N |
urinary β2-mg, M (p25, p75) |
Z P |
|
Male |
69 |
0.19(0.16ï½?0.33) |
0.68 0.497 |
|
Female |
4 |
0.13(0.02ï½?0.55) |
|
Table 1: Inter Group Comparison of Urinary β2-MG Levels Between Male and Female groups in the Non-Free Position Group (unit: mg/L)
The median value of urinary β2-MG levels in a randomly selected non free position group of 75 cases was 0.19 (0.16-0.33) Mg. The median value of urinary β2-MG levels in a randomly selected free position group of 10 cases was 0.09 (0.06-0.22) Mg. We conducted an independent sample Z-test on two sets of data, P<?0.05. Indicating that non free position is closely related to elevated levels of urinary β2-MG . Reminder: Non free position is closely related to damage to renal tubular epithelial cells. See Table 2.
|
|
N urinaryβ2-MG, M (P25, P75) |
Z |
|
P |
|
Non free position group |
75 0.19 (0.16-0.33) |
|
-2.193 |
0.028 |
|
Free position group |
10 0.09 (0.06-0.22) |
|
|
|
Table 2: Comparison of urinaryβ2 MG Levels Between Non Free Position Group and Free Position Group (unit: mg/L)
In the randomly selected non free position group, 12 patients underwent a second urine test after continuing to adhere to the non- free position for about a month after the first urine test. The median value of urine β2-MG level in the first urine test was 0.28 (0.09- 0.44) Mg. The median value of urine β2-MG level in the second urine test is 1.105 (0.45-1.41) Mg. The median value of urinary β2-MG levels in the second urine test was found to be higher than that in the first test. Paired sample Z-test was conducted on two sets of data from the same person's urine tests before and after, with P<0.05. This suggests that the persistent presence of non- free position is closely related to the continued increase in urinary β2-MG levels. Indicating that the persistent presence of non- free position is closely related to the worsening of renal tubular epithelial cell damage. See Table 3.
|
|
N |
urinaryβ2-MG, M (P25, P75) |
Z |
P |
|
|
Non free position |
group |
12 |
0.28 (0.09-0.44) |
-3.059 |
0.002 |
|
continue non-free |
|||||
|
Position group |
12 |
1.105 (0.45-1.41) |
|||
Table 3: Comparison of Urinary β2-MGlevels between Non Free Position Group and Continued Non Free Position Group (unit: mg/L)
Table 3: Comparison of Urinary β2-MGlevels between Non Free Position Group and Continued Non Free Position Group (unit: mg/L)urine test was found to have decreased compared to the first time. Paired sample Z-test was conducted on two sets of data from the same person's urine tests before and after, P<0.05. Tip: Free position is closely related to decreased levels of urinary β2-MG. Indicating that the free position is closely related to preventing further damage to renal tubular epithelial cells. See Table 4.
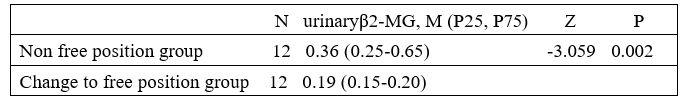
Table 4: Comparison of Urinary β2MGlevels between the Non-Free Position Group and the Free Position Group (unit: mg/L((unit: mg/L)
Discussion
The results of this study indicate that non free position is closely related to impaired renal tubular reabsorption function. The continued existence of non-free position is closely related to the worsening of renal tubular epithelial cell damage. The free position is closely related to preventing further damage to renal tubular epithelial cells. The damage mechanism may be: Increased abdominal pressure leads to reduced renal blood flow, decreased glomerular filtration rate, and tubular damage in the ipsilateral kidney.
Continuous lateral positioning can lead to an increase in intra- abdominal pressure at the ipsilateral kidney. According to research, the intra-abdominal pressure of patients was monitored using intracapsular pressure measurement technology when they were in a supine position and when the head of the bed was raised by 15 ° and 30 °, respectively, for 4 consecutive hours. The Zero position relationship point was set at the intersection of the midaxillary line and the iliac spine. The monitoring results showed that there were significant differences in the average abdominal pressure at different bedside angles (P<0.0001); Compared with the supine position, when the head of the bed is raised by 15 °, the abdominal pressure increases by 1.5mmHg (1.3-1.7mmHg, 1mmHg=0.133KPa), and when it is raised by 30 °, the abdominal pressure increases by 3.7mmHg (3.4-4.0mmHg). Researchers believe that raising the head of the bed can lead to a sharp increase in abdominal pressure. As is well known, the internal pressure of the same liquid increases with depth. Solids only generate pressure on their supporting surface, and the direction is always perpendicular to the supporting surface. The author speculates that the mechanism by which elevated bed heads lead to increased abdominal pressure may be that the vertical distance of all chest and abdominal contents relative to the Zero position relationship point increases, which is the true cause of increased intra-abdominal pressure. Therefore, when in a continuous lateral position, an increase in the vertical distance between the visceral organs of the contralateral abdominal cavity and the ipsilateral kidney can also lead to an increase in intra-abdominal pressure at the ipsilateral kidney.
Increased intra-abdominal pressure can lead to reduced renal cortical blood flow, decreased glomerular filtration rate, and tubular damage. The degree of damage is positively correlated with pneumoperitoneum pressure and time. According to research, a sharp increase in pneumoperitoneum pressure has adverse effects on renal function. Harman reported that when abdominal pressure increases from 0 to 20mmHg, renal blood flow decreases by 23.0%, glomerular filtration rate decreases by 21.0%, and renal vascular resistance increases by 555.0% [4]. Chiu found through animal experiments that under normal abdominal pressure, the blood flow in the renal cortex and medulla was (50.1 ± 17) ml/ min • 100g and (8.8 ± 3.3) ml/min • 100g, respectively. When the pneumoperitoneum pressure reached 15mmHg, the blood flow in the renal cortex decreased from (50.1 ± 17) ml/min • 100g to (20 ± 5) ml/min • 100g, with an average decrease of 60.0% [4]. According to research, N-acetyl -β- D- glucosaminidase (NAG) is a sensitive indicator for evaluating renal function in urine excretion, and its increase often suggests potential tubular damage. Koifusalo reported that urinary NAG increased by 153.0% in laparoscopic surgery patients compared to before pneumoperitoneum, which persisted until 3 hours after the release of pneumoperitoneum, confirming the potential renal dysfunction associated with pneumoperitoneum [4]. Ji Wu observed the damage to the ultrastructure of rabbit kidneys caused by different levels of pneumoperitoneum pressure and duration, and found that the degree of damage was positively correlated with pneumoperitoneum pressure and duration [4]. Tip: Non free position (continuous lateral position) may lead to reduced ipsilateral renal blood flow, decreased glomerular filtration rate, and tubular damage due to increased intra-abdominal pressure.
Persistent and limited elevation of renal pelvis urine hydrostatic pressure leads to reduced blood supply to ipsilateral renal tubules and other tissues The phenomenon of positional renal retention is indeed present. The normal rate at which the kidneys secrete urine is 1 ml/min. Wang et al. used the exploration method of taking a supine coronal longitudinal section or a prone abdominal elevation longitudinal section for all subjects, measured the maximum separation value of the renal system, and performed urine routine examination [5]. Ultrasound image analysis was performed on 355 patients with renal sinus separation.
The results showed that in group B (a total of 105 cases, all without urinary system diseases), the maximum liquid dark area width of renal sinus separation was 0-6 mm in 84 cases and 7-10 mm in 18 cases, 3 cases of 11-14 mm. After the examination is completed, instruct the patient to urinate. After 10 minutes of urination, 96 cases were reduced to 4-6 mm and 9 cases were reduced to 7-9 mm. There were no cases of renal sinus separation exceeding 10 mm after urination. This result indicates that: (1) Due to the inclusion of measurement results from prone position testing in the pre urination test data, even if all subjects were to switch to supine position testing, there may be more than but not less than 3 cases of renal sinus separation>10 mm; (2) The renal sinus separation data of 105 B-ultrasound subjects in Group B (whether measured 10 minutes after urination or compared before and after urination) indicate that in normal individuals with a duration of more than 10 minutes in a unilateral supine or/and supine position, there is positional renal retention in the ipsilateral kidney.
Non-Free position individuals with ipsilateral renal pelvis urinary stasis can cause sustained and limited elevation of ipsilateral renal pelvis hydrostatic pressure. Wang et al. studied the effect of double-J tube on renal pelvis pressure [6]. The baseline value of renal pelvis pressure was recorded using a urodynamic analyzer as (33.07 ± 7.04) cm H2O=(2.43 ± 0.52) mmHg, which the author referred to as the baseline renal pelvis hydrostatic pressure, denoted as A. The normal adult urine density is about 1.020. This article takes the depth of the left middle kidney calyx in males as an example (the same applies below) for analysis. According to research, the depth of the left middle renal calyx in males is (3.93 ± 1.01) cm [7]. The urine static pressure formed by it is 1.020 × (3.93 ± 1.01)=(4.01 ± 1.03) cm H2O=(0.29 ± 0.08) mm Hg, abbreviated as the deep urine static pressure of the renal pelvis by the author, denoted as B. In non-free position individuals, lying on the side leads to an increase in intra-abdominal pressure on the same side. The hydrostatic pressure of urine at the opening of the renal papilla in the ipsilateral kidney increases accordingly. The increased portion of urine hydrostatic pressure is represented by C. It at least includes: the basal renal pelvis hydrostatic pressure A of the contralateral kidney vertically stacked due to lateral position, and the deep renal pelvis hydrostatic pressure B of the ipsilateral kidney, that is: C≥A+B.
Persistent and limited elevation of ipsilateral renal vesicle urine hydrostatic pressure leads to a decrease in ipsilateral renal glomerular filtration rate. According to research [9], the effective filtration pressure of the glomerulus can be represented by the following equation:
Effective glomerular filtration pressure=glomerular capillary blood pressure - (plasma colloid osmotic + Renal capsule pressure). Among them, the glomerular capillary blood pressure is 45 mm Hg, the plasma colloid osmotic pressure at the end of the afferent arteriole is 20 mm Hg, the plasma colloid osmotic pressure near the end of the efferent arteriole is 35 mm Hg, and the renal capsule pressure is 10 mm Hg. The effective filtration pressure at the scoring end is 45- (20+10) =15 (mm Hg), The effective filtration pressure at the exit of the ball is 45- (35+10) =0 (mm Hg). Therefore, for non-free position individuals, in the above equation, the actual glomerular effective filtration pressure of the ipsilateral kidney should be subtracted by the newly increased hydrostatic pressure of the ipsilateral renal capsule (i.e. subtracting C ≥ A+B). Now calculated as C=A+B, the result is: At the scoring end, the actual effective glomerular filtration pressure decreases sequentially to: Actual glomerular effective filtration pressure=15- [(2.43 ± 0.52) +(0.29 ± 0.08)] (mmHg); At the exit end, the actual glomerular effective filtration pressure decreases sequentially to: Actual glomerular effective filtration pressure=0- [(2.43 ± 0.52) +(0.29 ± 0.08)] (mmHg).
It can be seen that the lateral position leads to a significant decrease in the actual glomerular effective filtration pressure of the ipsilateral kidney. According to research, the filtration effect of glomerular capillaries can only exist from the end of the afferent arteriole to the point where filtration equilibrium occurs. The closer the equilibrium point of filtration is to the end of the afferent arteriole, the shorter the capillaries that can filter and form ultrafiltrate, the smaller the total effective filtration area, and the lower the glomerular filtration rate [8]. For non-free position individuals, the actual glomerular effective filtration pressure at the glomerular capillary at the entrance is lower than normal due to the increase in intra renal pressure on the ipsilateral renal capsule, and the glomerular effective filtration volume is reduced compared to normal; The glomerular capillaries located at a considerable distance from the exit end of the glomerulus, due to the increase in pressure within the ipsilateral renal capsule, also increase in length of the capillaries that cannot generate ultrafiltration fluid, resulting in a decrease in total glomerular filtration. The oxygen consumption of renal metabolism mainly depends on the activity of proximal tubular transport. Experiments have shown that the renal tubules are the most obvious site of renal ischemic injury. Due to the high solute transport activity of renal tubular epithelial cells (RETCs), which require energy support from mitochondria, RETCs are highly sensitive to ischemia and hypoxia [9]. According to research, the blood supply to the renal tubules comes from the glomerulus, so compared to the glomerulus, the renal tubules are more susceptible to ischemic injury [10]. Therefore, when the total filtration of the ipsilateral renal glomerulus is lower than normal, it will directly lead to a decrease in the blood supply of the ipsilateral renal glomerulus and its associated renal tubules, causing damage to the renal tubular epithelial cells due to ischemia and hypoxia.
It can be seen that: (1) It is generally believed that the pressure limit that causes renal parenchymal reflux is 40 cmH2O [11].When the hydrostatic pressure of urine in the ipsilateral renal pelvis is ≥ 40cm H2O, this pressure can force the opening of the renal papilla opening, causing an increase in the pressure inside the ipsilateral renal capsule, resulting in a lower than normal total glomerular filtration in the ipsilateral kidney, and directly leading to a decrease in the blood supply to the corresponding renal tubules. This may be the true cause of reduced tubular reabsorption function and renal parenchymal atrophy in the ipsilateral kidney. The elevated and continuously increasing levels of urinary β2-MG in non-free position patients in this group may be due to this. (2) As the position changes, the effect of position on the effective filtration pressure of the contralateral renal glomerulus also changes in reverse. If the left lateral position is changed to the right lateral position, the urine static pressure in the left renal capsule is lower than normal, and the total effective filtration of the left renal glomerulus increases accordingly. The blood supply to the left renal glomerulus and renal tubules also increases, and their reabsorption function is restored or recovered. In this experiment, after switching from a non free position to a free position, the urinary β2-MG levels of patients were able to decrease and return to normal, which may be due to this.
(3) When the depth of the ipsilateral renal pelvis is small, the basal renal pelvis urine hydrostatic pressure and the urine hydrostatic pressure formed by the depth of the renal pelvis are less than the threshold for renal reflux (urine hydrostatic pressure<40 cm H2O ), and the renal pelvis nipple opening is not opened due to hydrostatic pressure compression, and the pressure inside the ipsilateral renal small sac is not increased, and the blood flow of the renal tubules is not forced to decrease, then the renal tubular epithelial cells are not injured due to ischemia, and therefore the reabsorption function will not change due to changes in position. Among the 75 patients who participated in the experiment in a non-free position, 52 patients had urinary β2-MG levels less than 0.3mg/L (of which 42 patients had urinary β2-MG levels less than 0.2mg/L), and were not affected by the non free position. The reason may be that the urine static pressure of the ipsilateral kidney is lower than the threshold for renal reflux (urine static pressure < 40cmH2O).
The sustained and limited increase in hydrostatic pressure in the ipsilateral renal small sac urine may be closely related to the renal pathological changes in patients with kidney stones. According to research, the pathological changes in the kidneys of patients with kidney stones are mainly renal compression atrophy caused by hydronephrosis and secondary chronic pyelonephritis [12]. According to research, a sustained and limited increase in urinary static water pressure within the renal small cystic cavity and collecting system may lead to functional impairment of glomeruli, renal tubules, and cellular tissue ischemia, atrophy, and apoptosis. Periglomerular fibrosis of renal capsule is a special structure that exists in the short term and is a pathological manifestation of glomerular injury (the glomeruli disappear from ischemia, atrophy to apoptosis) [2].The author believes that there are 8 pathological characteristics in the formation process of renal glomerular fibrosis:  According to pathological research, renal glomerular fibrosis is not an infectious lesion [13,14]
According to pathological research, renal glomerular fibrosis is not an infectious lesion [13,14]  Traditionally, it is believed that kidney stones and glomerular lesions are nonimmune injuries [13].
Traditionally, it is believed that kidney stones and glomerular lesions are nonimmune injuries [13].  According to "no cases of thickening of the renal renal interstitium, renal pelvis mucosa, or renal blood vessels, or vasculitis" it suggests that it is not caused by extraglomerular vascular factors [14].
According to "no cases of thickening of the renal renal interstitium, renal pelvis mucosa, or renal blood vessels, or vasculitis" it suggests that it is not caused by extraglomerular vascular factors [14]. ![]() According to "the proliferation of fibrous tissue outside the glomerular sac surrounds the renal glomerulus in concentric circles, and the structure of the glomerular sac wall is intact; there is varying degrees of lymphocyte infiltration outside the glomerular sac" rather than fibrous tissue proliferation or inflammation on one side of the glomerular sac wall, it suggests that it cannot be caused by local pathogenic factors on either side of the renal glomerulus lumen [14].
According to "the proliferation of fibrous tissue outside the glomerular sac surrounds the renal glomerulus in concentric circles, and the structure of the glomerular sac wall is intact; there is varying degrees of lymphocyte infiltration outside the glomerular sac" rather than fibrous tissue proliferation or inflammation on one side of the glomerular sac wall, it suggests that it cannot be caused by local pathogenic factors on either side of the renal glomerulus lumen [14]. ![]() According to "It is difficult to observe histologically the dilation of renal glomeruli caused by increased pressure in the glomerular sac due to obstruction of renal pelvis stones" At the same time, "the structure of the glomerular capsule wall is intact", indicating that renal small capsule wall lesions are not caused by rapid increase in hydraulic pressure in the renal pelvis (the latter can lead to tubular rupture and local scar formation, such as scar areas in reflux nephropathy) Research has shown that the formation process of periglomerular fibrosis in renal small cysts is a slowly progressing pathological process that requires time (approximately 6 weeks after obstruction) [14,15].
According to "It is difficult to observe histologically the dilation of renal glomeruli caused by increased pressure in the glomerular sac due to obstruction of renal pelvis stones" At the same time, "the structure of the glomerular capsule wall is intact", indicating that renal small capsule wall lesions are not caused by rapid increase in hydraulic pressure in the renal pelvis (the latter can lead to tubular rupture and local scar formation, such as scar areas in reflux nephropathy) Research has shown that the formation process of periglomerular fibrosis in renal small cysts is a slowly progressing pathological process that requires time (approximately 6 weeks after obstruction) [14,15].  ¸ Because the remaining glomeruli are constantly producing urine, in order to maintain the hydraulic pressure in the renal pelvis from continuously increasing or rapidly increasing, it is necessary that the urinary tract is not obstructed or completely obstructed, and urine in the renal pelvis can still flow out of the urinary tract, in order to maintain a relative dynamic balance between the inflow and outflow of urine in the renal pelvis
¸ Because the remaining glomeruli are constantly producing urine, in order to maintain the hydraulic pressure in the renal pelvis from continuously increasing or rapidly increasing, it is necessary that the urinary tract is not obstructed or completely obstructed, and urine in the renal pelvis can still flow out of the urinary tract, in order to maintain a relative dynamic balance between the inflow and outflow of urine in the renal pelvis![]() . The author analyzed the characteristics of scar and non-scar lesions in reflux nephropathy, and believed that in order to maintain the renal tubular wall from breaking due to excessive hydraulic pressure (if the renal tubules break due to compression and urine enters the interstitium, it can lead to local scar formation), the increase in renal small cystic space and urinary static water pressure in the renal tubules where kidney stones can occur can only be limited [2]. The pressure range after its increase is approximately: When reflux nephropathy occurs, it can cause rupture of renal tubules in renal small sacs and increase the hydrostatic pressure of urine in renal tubules > Urinary static pressure in the glomeruli and tubules where kidney stones can occur > Normal glomerular and tubular urine static pressure. Just like the placental villi are immersed in the placental sinusoids, the anatomical and physiological relationship between the glomerular sac and the glomerular capillary plexus can be seen as the simultaneous "immersion" of glomerular capillaries and mesangial tissue in the original urine of the renal small sac. According to:
. The author analyzed the characteristics of scar and non-scar lesions in reflux nephropathy, and believed that in order to maintain the renal tubular wall from breaking due to excessive hydraulic pressure (if the renal tubules break due to compression and urine enters the interstitium, it can lead to local scar formation), the increase in renal small cystic space and urinary static water pressure in the renal tubules where kidney stones can occur can only be limited [2]. The pressure range after its increase is approximately: When reflux nephropathy occurs, it can cause rupture of renal tubules in renal small sacs and increase the hydrostatic pressure of urine in renal tubules > Urinary static pressure in the glomeruli and tubules where kidney stones can occur > Normal glomerular and tubular urine static pressure. Just like the placental villi are immersed in the placental sinusoids, the anatomical and physiological relationship between the glomerular sac and the glomerular capillary plexus can be seen as the simultaneous "immersion" of glomerular capillaries and mesangial tissue in the original urine of the renal small sac. According to:  the anatomical and physiological characteristics of the kidney;
the anatomical and physiological characteristics of the kidney; ¸ According to the renal pathological characteristics of patients with kidney stones, including ischemic atrophic lesions in the glomerular capsule wall, glomerular capillaries, and glomerular mesangial area;
¸ According to the renal pathological characteristics of patients with kidney stones, including ischemic atrophic lesions in the glomerular capsule wall, glomerular capillaries, and glomerular mesangial area;  According to the 8 pathological characteristics that exist during the formation of perirenal fibrosis in renal glomeruli;
According to the 8 pathological characteristics that exist during the formation of perirenal fibrosis in renal glomeruli; ![]() According to Pascal's law (the pressure applied to any part of a sealed liquid must be transmitted from the liquid in all directions according to its original size), the author believes that only the sustained and limited increase in urine liquid pressure inside the renal capsule (which provides both compressive and tensile stress) can lead to ischemic atrophy damage to both the glomerular capsule wall and the tissues inside the glomerulus. In other words, renal tissue ischemic atrophic lesions (including perirenal fibrosis of renal glomeruli) in patients with kidney stones may be closely related to long-term limited elevation of urinary static water pressure in the glomeruli and tubules. That is to say, the degree of tissue damage to the glomerulus and renal tubules is related to both the magnitude of hydraulic pressure in the renal pelvis and the duration of elevated hydraulic pressure in the renal pelvis (i.e., the degree and extent of tissue damage "intensify over time"). I believe that among all known (physical, chemical, biological) factors that cause injury, almost only sustained and limited elevation of urine hydraulic pressure within the renal capsule has this potential for injury. The hydrostatic pressure of renal pelvis urine generated in non-free position can precisely meet this requirement. Moreover, it has three characteristics:
According to Pascal's law (the pressure applied to any part of a sealed liquid must be transmitted from the liquid in all directions according to its original size), the author believes that only the sustained and limited increase in urine liquid pressure inside the renal capsule (which provides both compressive and tensile stress) can lead to ischemic atrophy damage to both the glomerular capsule wall and the tissues inside the glomerulus. In other words, renal tissue ischemic atrophic lesions (including perirenal fibrosis of renal glomeruli) in patients with kidney stones may be closely related to long-term limited elevation of urinary static water pressure in the glomeruli and tubules. That is to say, the degree of tissue damage to the glomerulus and renal tubules is related to both the magnitude of hydraulic pressure in the renal pelvis and the duration of elevated hydraulic pressure in the renal pelvis (i.e., the degree and extent of tissue damage "intensify over time"). I believe that among all known (physical, chemical, biological) factors that cause injury, almost only sustained and limited elevation of urine hydraulic pressure within the renal capsule has this potential for injury. The hydrostatic pressure of renal pelvis urine generated in non-free position can precisely meet this requirement. Moreover, it has three characteristics:  Limited elevation: basal renal pelvis urine static pressure A + elevated portion of urine static pressure C.
Limited elevation: basal renal pelvis urine static pressure A + elevated portion of urine static pressure C.  ¸ Persistent existence: It exists when it is in a non-free position.
¸ Persistent existence: It exists when it is in a non-free position.  Repetitive occurrence: It changes with daily changes in posture. Based on this, it is speculated that when renal pelvis hydrostatic pressure causes tissue ischemia injury, this repetitive characteristic will lead to ischemia-reperfusion injury of the damaged tissue.
Repetitive occurrence: It changes with daily changes in posture. Based on this, it is speculated that when renal pelvis hydrostatic pressure causes tissue ischemia injury, this repetitive characteristic will lead to ischemia-reperfusion injury of the damaged tissue.
The Non-Free Position is Closely Related to the Formation of Kidney Stones, While the Free Position can Prevent Kidney Stones
The results of this experiment suggest that non free position is closely related to urinary β2-MG levels, which depend on both intra-abdominal pressure and renal calyx depth, as well as the duration of non-free position. When the ipsilateral renal pelvis urine hydrostatic pressure A+C ≥ 40 cmH2O, it has two characteristics:  the ipsilateral renal pelvis urine hydrostatic pressure continues to increase and is limited. When the pressure is greater than the glomerular capillary filtration pressure, it leads to a decrease in blood supply to the renal unit tissue in that part;
the ipsilateral renal pelvis urine hydrostatic pressure continues to increase and is limited. When the pressure is greater than the glomerular capillary filtration pressure, it leads to a decrease in blood supply to the renal unit tissue in that part;  as long as the non-free position exists, the ipsilateral renal pelvis urine static pressure persists simultaneously. From this, the author associates pressure therapy for burn scars in clinical practice. According to research when using pressure therapy for burn scars, it is necessary to control the pressure level [16]. Generally, the appropriate pressure range is 3.30-5.32Kpa (25-40mmHg). If the pressure is too low (<3.3 KPa), edema is difficult to reduce, and the therapeutic effect is difficult to achieve. If the pressure is too high (>5.32 KPa), it is easy to cause local tissue ischemia and hypoxia, leading to local tissue necrosis. According to research , the most suitable duration for wearing elastic sleeves is 7-12 months [17]. Because the complete set of pathological changes caused by the compression of burn scars requires prolonged external pressure to complete. According to research, the principle of scar compression therapy is to reduce the number of blood vessels, narrow or even close the lumen, cause endothelial cell degeneration and degeneration, worsen vascular wall damage, and reduce the blood supply to scar tissue [17]. The above research results suggest that:
as long as the non-free position exists, the ipsilateral renal pelvis urine static pressure persists simultaneously. From this, the author associates pressure therapy for burn scars in clinical practice. According to research when using pressure therapy for burn scars, it is necessary to control the pressure level [16]. Generally, the appropriate pressure range is 3.30-5.32Kpa (25-40mmHg). If the pressure is too low (<3.3 KPa), edema is difficult to reduce, and the therapeutic effect is difficult to achieve. If the pressure is too high (>5.32 KPa), it is easy to cause local tissue ischemia and hypoxia, leading to local tissue necrosis. According to research , the most suitable duration for wearing elastic sleeves is 7-12 months [17]. Because the complete set of pathological changes caused by the compression of burn scars requires prolonged external pressure to complete. According to research, the principle of scar compression therapy is to reduce the number of blood vessels, narrow or even close the lumen, cause endothelial cell degeneration and degeneration, worsen vascular wall damage, and reduce the blood supply to scar tissue [17]. The above research results suggest that:  The pressure applied in pressure therapy should be greater than the capillary pressure of the scar tissue under pressure, and the magnitude of the pressure must be appropriate to achieve ischemia, hypoxia, degeneration, and atrophy of endothelial cells; Too much or too little pressure is not appropriate;
The pressure applied in pressure therapy should be greater than the capillary pressure of the scar tissue under pressure, and the magnitude of the pressure must be appropriate to achieve ischemia, hypoxia, degeneration, and atrophy of endothelial cells; Too much or too little pressure is not appropriate;  The pressure applied in stress therapy must be sustained for a long period of time (7- 12 months or more). If we consider the process of using pressure therapy to inhibit the proliferation of burn scar tissue as a "clinical trial" on the joint damage of renal tubular epithelial cells caused by increased intra-abdominal pressure in the ipsilateral kidney and increased hydrostatic pressure in the ipsilateral renal pelvis, and compare it with the theory and practice of pressure therapy for burn scars, since applying sustained and moderate pressure to the limbs of burn patients can effectively reduce their blood supply to the injury site and achieve the goal of inhibiting scar tissue proliferation. The author believes that under the dual effects of sustained and limited increase in intra-abdominal pressure at the ipsilateral kidney and renal pelvis hydrostatic pressure at the ipsilateral kidney, all kidney tissues directly affected may also experience ischemia, hypoxia, functional decline, structural changes, and even cell apoptosis due to reduced blood supply. This helps us understand the causal relationship between non free position and kidney stone formation:
The pressure applied in stress therapy must be sustained for a long period of time (7- 12 months or more). If we consider the process of using pressure therapy to inhibit the proliferation of burn scar tissue as a "clinical trial" on the joint damage of renal tubular epithelial cells caused by increased intra-abdominal pressure in the ipsilateral kidney and increased hydrostatic pressure in the ipsilateral renal pelvis, and compare it with the theory and practice of pressure therapy for burn scars, since applying sustained and moderate pressure to the limbs of burn patients can effectively reduce their blood supply to the injury site and achieve the goal of inhibiting scar tissue proliferation. The author believes that under the dual effects of sustained and limited increase in intra-abdominal pressure at the ipsilateral kidney and renal pelvis hydrostatic pressure at the ipsilateral kidney, all kidney tissues directly affected may also experience ischemia, hypoxia, functional decline, structural changes, and even cell apoptosis due to reduced blood supply. This helps us understand the causal relationship between non free position and kidney stone formation:  After the reduction of renal tubular function, both organic and inorganic components in the original urine, including urinary β2-MG, will change accordingly depending on the changes in renal tubular reabsorption function. Those that continue to remain in the original urine will form terminal urine, manifested as an increase in the concentration of these substances. The urine β2-MG detected in this experiment is only one of the many components in its final urine. Jinjiang Tao et al. studied the relationship between the composition of renal pelvis urine and the duration of hydronephrosis in patients with unilateral upper urinary tract obstruction induced hydronephrosis (32 cases), and found that the levels of endothelin, creatinine, microalbumin β2 -MG the concentrations, pH values, and conductivity of sodium ions, magnesium ions, potassium ions, and chloride ions are higher than those of bladder urine [18]. The difference between the two was statistically significant (P<0.05). The author believes that the organic and inorganic components mentioned in the article by Jinjiang Tao et al. all come from original urine. The direct consequence is that certain components of the final urine become the material basis for the formation of kidney stones [18].
After the reduction of renal tubular function, both organic and inorganic components in the original urine, including urinary β2-MG, will change accordingly depending on the changes in renal tubular reabsorption function. Those that continue to remain in the original urine will form terminal urine, manifested as an increase in the concentration of these substances. The urine β2-MG detected in this experiment is only one of the many components in its final urine. Jinjiang Tao et al. studied the relationship between the composition of renal pelvis urine and the duration of hydronephrosis in patients with unilateral upper urinary tract obstruction induced hydronephrosis (32 cases), and found that the levels of endothelin, creatinine, microalbumin β2 -MG the concentrations, pH values, and conductivity of sodium ions, magnesium ions, potassium ions, and chloride ions are higher than those of bladder urine [18]. The difference between the two was statistically significant (P<0.05). The author believes that the organic and inorganic components mentioned in the article by Jinjiang Tao et al. all come from original urine. The direct consequence is that certain components of the final urine become the material basis for the formation of kidney stones [18].  When the renal tubular epithelial cells affected by intra-abdominal pressure in the ipsilateral kidney and hydrostatic pressure in the ipsilateral renal pelvis are structurally damaged due to ischemia and hypoxia, it will facilitate the formation of kidney stones. According to research in various disease conditions, such as acute ischemic injury, acute tubular necrosis, etc., the expression of hyaluronic acid (HA) is elevated and appears in places that have never been seen before, such as the luminal surface of renal tubules [19]. Research has shown that HA has an affinity for calcium oxalate crystals, especially after injury, the surface of renal tubular epithelial cells is more prone to adhere to calcium oxalate crystals, leading to stone formation
When the renal tubular epithelial cells affected by intra-abdominal pressure in the ipsilateral kidney and hydrostatic pressure in the ipsilateral renal pelvis are structurally damaged due to ischemia and hypoxia, it will facilitate the formation of kidney stones. According to research in various disease conditions, such as acute ischemic injury, acute tubular necrosis, etc., the expression of hyaluronic acid (HA) is elevated and appears in places that have never been seen before, such as the luminal surface of renal tubules [19]. Research has shown that HA has an affinity for calcium oxalate crystals, especially after injury, the surface of renal tubular epithelial cells is more prone to adhere to calcium oxalate crystals, leading to stone formation After structural damage and atrophy of renal tissues affected by intra-abdominal pressure in the ipsilateral kidney and hydrostatic pressure of urine in the ipsilateral renal pelvis due to ischemia and hypoxia, the space occupied by the original compressed renal tissue is replaced by stagnant urine. Over time, the space for stagnant urine to replace it continues to expand, becoming clinically visible hydronephrosis. on the other hand, after switching from a non-free position to a free position, the sustained and limited increase in urinary static water pressure in the renal tubules and collecting system no longer exists. As a result, a series of kidney tissue injuries caused by the non-free position stop, or the reabsorption function of the renal tubules is restored, and urinary β2-MG levels will also decrease or return to normal. Our observation found that in the non-free position group, the first urine test results of 12 randomly selected patients showed that the urine β2-MG levels of 8 members were higher than the normal value (>0.3mg/L.), and the urine β2-MG levels of 2 members were higher than 0.2mg/L. After they changed to a free position for one month, a urine test was conducted again, and the urine β2-MG levels of 10 members decreased to below 0.2mg/L, with one member's result being 0.21mg/L and another member's result being 0.228mg/L. Pairing the two sets of data for sample Z-test and comparing them between groups, there was a significant difference (P<0.05). Tip: After removing non free position factors, urine β2-MG levels mostly return to normal levels as there are no new renal units that continue to be damaged, or the reabsorption function of damaged renal tubules is restored in a short period of time. In other words, the free position may prevent further damage to renal tubular epithelial cells. In this sense, kidney stones can occur due to non-free position and will no longer occur due to free position.
After structural damage and atrophy of renal tissues affected by intra-abdominal pressure in the ipsilateral kidney and hydrostatic pressure of urine in the ipsilateral renal pelvis due to ischemia and hypoxia, the space occupied by the original compressed renal tissue is replaced by stagnant urine. Over time, the space for stagnant urine to replace it continues to expand, becoming clinically visible hydronephrosis. on the other hand, after switching from a non-free position to a free position, the sustained and limited increase in urinary static water pressure in the renal tubules and collecting system no longer exists. As a result, a series of kidney tissue injuries caused by the non-free position stop, or the reabsorption function of the renal tubules is restored, and urinary β2-MG levels will also decrease or return to normal. Our observation found that in the non-free position group, the first urine test results of 12 randomly selected patients showed that the urine β2-MG levels of 8 members were higher than the normal value (>0.3mg/L.), and the urine β2-MG levels of 2 members were higher than 0.2mg/L. After they changed to a free position for one month, a urine test was conducted again, and the urine β2-MG levels of 10 members decreased to below 0.2mg/L, with one member's result being 0.21mg/L and another member's result being 0.228mg/L. Pairing the two sets of data for sample Z-test and comparing them between groups, there was a significant difference (P<0.05). Tip: After removing non free position factors, urine β2-MG levels mostly return to normal levels as there are no new renal units that continue to be damaged, or the reabsorption function of damaged renal tubules is restored in a short period of time. In other words, the free position may prevent further damage to renal tubular epithelial cells. In this sense, kidney stones can occur due to non-free position and will no longer occur due to free position.
The author believes that if we promote in depth among the population, change the non free position to a free position, or allow kidney stone patients to adhere to the free position after dissolution, lithotripsy, or stone removal treatment, it will be beneficial for the prevention and treatment of kidney stones.
References
- Sheng Bin-Wu, He Dalin, Chen Xing-fa, etc. (2010). The significance and mechanism of elevated urinary NAG, γ - GT, and β2-MG levels in patients with kidney stones [J]. ShaanxiMedical Journal, 39ï¼?1ï¼?ï¼?70.
- Zheng Dao. (2013). Non free position can cause kidney tissue damage and kidney stone formation through the use of urinary hydrostatic pressure [J]. Chinese Medical Innovation, , 10 (20): 164, 159.
- Zheng Dao. (1990). The relationship between non-free position and urinary drainage and urinary stone distribution [J]. Jiangsu Medical Journal, 16 (24): 484.
- Yang Ming-Xing. (2009). The effect of human body position on abdominal pressure [J]. CT Care Med. 37 (7): 2187-2190
- Li Xing-bin. (2008). The impact of artificial pneumoperitoneum on the urinary system [J]. Western Medicine, 20 (3): 651-652
- Wang Jin, Li Hai-chao, Li Rong-Jun, etc. (2010). The effect and clinical significance of double-J tube on renal pelvis pressure [J]. Journal of Shanxi Medical University, 41 (7): 648.
- Zheng Dao, Lu Bi-qing, Li Yu-Nian, et al. (2013). X-ray measurement of healthy renal calyx depth in 237 normal individuals and 138 patients with urolithiasis [J]. Chinese Journal of Medical Innovation, 10 (1): 60.
- Wang Ting-Huai. Physiology [M]. (2018). 9th edition Beijing: People's Health Commission: two hundred and thirty-three.
- Zou Wan-Zhong. Renal biopsy pathology [M]. 2nd edition Beijing: Peking University Medical Press, 2006:44.
- Peng Dandan, Cheng Liping. (2013). Research progress on renal tubular epithelial cell injury [J]. Journal of Clinical Pediatrics, 31 (6): 584.
- Pan Jian-gang, Yan Jiajun, Tang Ai-Juan, etc. (2008). The relationship between changes in renal pelvis pressure during minimally invasive percutaneous nephrolithotomy and the incidence of early postoperative complications [J]. Journal of Clinical Urology, 23 (11): 817.
- Pathology Teaching and Research Office of Wuhan. (1983). Medical College Surgical Pathology [M]. Wuhan: Hubei People's Publishing House : 579.
- Guo Lin-lang, Guo Ying. (1996). Study on changes of extracellular matrix in renal calculi and glomerular lesions [J]. Journal of Clinical Urology, 11 (3): 175.
- Ding Xiaoyu, Zhang Renli, Yang Xuemei. (1995). Renal pelvis stones - renal histopathological histological study [J]. Journal of Xuzhou Medical College, 15 (1): 22.
- Ye Da-fu, Zhang Jie, Yao Yi. (2010). Morphological changes of renal tissue in rats with acute and chronic obstructive hydrocephalus [J]. Journal of Wuhan University (Medical Edition), 31 (2): 168,166.
- Wang Guanfa, Long Bin. (2013). Clinical Observation of Pressure Therapy for Burn Scar Hyperplasia [J]. Journal of Yibin University, 35 (9): 27
- Hu Yong-Cai, Li Jinhong, Ou Caisheng. (2000). Evaluation of the therapeutic effect of elastic compression therapy on burnscars [J]. Modern rehabilitation, 4ï¼?1ï¼?ï¼?33.
- Jinjiang Tao, Deng Guo-Xiang. (2009). The relationshipbetween the degree and duration of hydronephrosis and the composition of renal pelvis urine [J]. Journal of Shanxi University of Traditional Chinese Medicine, 10 (2): 54.
- Xin Wen-hu, celebrity, Yue Zhong-Jin. (2013).Research progress on the mechanism of renal tubular epithelial cell injury and calcium oxalate kidney stone formation [J]. International Journal of Urology, 33 (4): 540.
- Wang Kaijie, Wang Rui-qing. (2000). Ultrasound image analysis of 355 patients with renal sinus separation [J]. Shandong Medicine, 40 (21): 22.
- Tang Siyuan. Physiology [M]. (2017). 4th edition Beijing: People's Health Commission,: two hundred and forty-six.




