Research Article - (2025) Volume 4, Issue 1
Conjecture of the Marquee on Cube Surface of the Virtual Atomic Orbital and its Implementation of the Chemical Reactions System
Received Date: Jan 15, 2025 / Accepted Date: Feb 10, 2025 / Published Date: Feb 13, 2025
Copyright: ©©2025 Jer-Fong Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Chen., J. F. (2025). Conjecture of the Marquee on Cube Surface of the Virtual Atomic Orbital and its Implementation of the Chemical Reactions System. J Math Techniques Comput Math, 4(1), 01-06.
Abstract
Atomic orbitals have always been a popular research topic. The mainstream theory uses wave functions to predict the position of electrons in atoms. However, this paper proposes a new hypothesis that uses a large cube composed of small cubes, along with a method similar to a running light and projector on the surface of the cube to explain the phenomenon. In addition, this paper explains how to use this hypothesis to distinguish whether a given chemical formula within a specified range will undergo a chemical reaction.
Keywords
Atomic Orbital, Cube Surface, Virtual System
Ccs Concepts
• Human-Centered Computing • Visualization • Visualization Design and Evaluation Methods
• Computing Methodologies • Modeling and Simulation • Model Development and Analysis • Model Verification and Validation
Introduction
As early as 1926, Austrian physicist Erwin Schrödinger proposed the wave function of atomic orbitals. The wave function explains the wave behavior of electrons in atoms. Although the electron waves cannot be directly observed, we can infer the probability of finding an electron at a particular location within the atom. Consequently, scientists often use the concept of an electron cloud to represent the regions within an atom where electrons are likely to be found.
In 1964, due to the wave function's inability to handle the simulation of multiple electrons in iron atoms or within molecules, Pierre Hohenberg and Walter Kohn proposed Density-Functional Theory (DFT). DFT assumes that electrons do not interact with each other. The 'Hohenberg-Kohn theorems' state that for a given electron system in its ground state, all its properties are determined by the electron density. In 1965, Kohn, together with Lu Jeu Sham, published the Kohn–Sham equations, which simplified the wave function based on DFT. This simplification enabled subsequent scientists to understand and predict the most stable arrangements of all electrons in large atoms or molecules.
This paper hypothesizes that electrons on the surface of atoms are presented in a manner similar to a running light. It represents all possible electron orbitals of the 118 elements using 3D graphics on a cube. This hypothesized orbital is then applied to the Kohn– Sham equations to predict whether a chemical reaction can occur. However, whether the chemical reaction requires energy input or releases energy is beyond the scope of this paper.
Conjecture of the Marquee on Cube Surface of the Atomic Orbital
Through the paper 'Discovery of the Characteristics of the Cubic Othello Chessboard and its Implementation of Visualization Expert System', it is known that the cubic Othello chessboard uses three different attack lines [1]. By referring to the illustrations in that paper and placing them in Figure 1, the first type is a blue line passing through every face, the second type is purple and orange lines that turn along the edges, and the third type is red and yellow lines that turn around the vertices. Additionally, this paper uses a fourth type of line, as shown by the green line in Figure 2.
Figure 1: Attack Lines of the Cubic Othello Chessboard
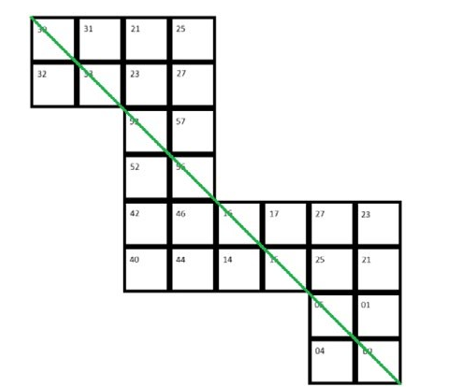
Figure 2: From the Figure, it Can be Seen that the Green Line Rotates Between the Face and its Opposite Face
The first type of line can be classified as the s orbital of an atom. Assuming that the electrons can be divided into three lines running (Zeeman effect), in an environment with only one cube, the s orbital can accommodate at most one electron and its mirror image electron. In a non-overlapping situation, this results in a perfect cubic coverage. This type of cube can be analogous to the element Helium (He), as shown in Table 1.
The second type of line can be classified as the p orbital of an atom. Similarly, by dividing each electron into three lines running, in an environment where a large cube is composed of eight small cubes with two small cubes per edge, the p orbital can accommodate at most four electrons and their four mirror image electrons without overlapping, resulting in a perfect cubic coverage. This type of cube can be analogous to the element Neon (Ne), as shown in Table 2.
The third type of line can be classified as the d orbital of an atom. Likewise, by dividing each electron into three lines running, in an environment where a large cube is composed of twenty-seven small cubes with three small cubes per edge, the d orbital can accommodate at most nine electrons and their nine mirror image electrons without overlapping, resulting in a nearly perfect cubic coverage. This type of cube can be analogous to the third layer of the electron orbitals of the element Copper (Cu).
The fourth type of line can be classified as the f orbital of an atom. Similarly, by dividing each electron into three lines running, in an environment where a cube is composed of 64 small cubes, the f orbital can accommodate at most sixteen electrons and their sixteen mirror image electrons without overlapping, resulting in a nearly perfect cubic coverage. This type of cube can be analogous to the fourth layer of the electron orbitals of the element Ytterbium (Yb).
Finally, following this rule, all 118 elements can be constructed, and according to DFT, it is possible to predict whether a chemical reaction will occur between each element [2].
|
|
1st electron |
Mirror 1st electron |
|
above side of the cube |
1, 2, 6 |
4, 3, 5 |
|
below side of the cube |
4, 5, 3 |
1, 6, 2 |
|
left side of the cube |
6, 1, 2 |
5, 4, 3 |
|
right side of the cube |
3, 4, 5 |
2, 1, 6 |
|
front side of the cube |
2, 6, 1 |
3, 5, 4 |
|
behind side of the cube |
5, 3, 4 |
6, 2, 1 |
Table 1: The Numbers From Small to Large Represent the Sequence of the Running Light, Returning to 1 After Reaching 6. From the Table, it Can be Seen that there is No Instance Where more than one-third of an Electron Overlaps on Any Given Face.
|
|
1st e |
2nd e |
3rd e |
4th e |
|
Mir 1st e |
Mir 2nd e |
Mir 3rd e |
Mir 4th e |
|
|
above-left of the above large cube (No. 30) |
1, 0, 8 |
|
4, 0, 0 |
5, 0, 0 |
|
|
6, 3, 0 |
0, 7, 0 |
2, 0, 0 |
|
|
above-right of the above large cube (No. 31) |
2, 0, 0 |
0, 7, 0 |
|
6, 0, 3 |
|
0, 5, 0 |
0, 4, 0 |
0, 8, 1 |
|
|
|
below-left of the above large cube (No. 32) |
0, 0, 7 |
2, 0, 0 |
3, 0, 6 |
|
|
0, 0, 4 |
5, 0, 0 |
|
1, 8, 0 |
|
|
below-right of the above large cube (No. 33) |
|
1, 8, 0 |
0, 0, 5 |
0, 0, 4 |
|
0, 6, 3 |
|
0, 0, 2 |
0, 7, 0 |
|
|
above-left of the below large cube (No. 16) |
0, 0, 4 |
5, 0, 0 |
8, 0, 1 |
|
|
0, 0, 7 |
2, 0, 0 |
|
6, 3, 0 |
|
|
above-right of the below large cube (No. 17) |
|
6, 3, 0 |
0, 0, 2 |
0, 0, 7 |
|
0, 1, 8 |
|
0, 0, 5 |
0, 4, 0 |
|
|
below-left of the below large cube (No. 14) |
6, 0, 3 |
|
7, 0, 0 |
2, 0, 0 |
|
|
1, 8, 0 |
0, 4, 0 |
5, 0, 0 |
|
|
below-right of the below large cube (No. 15) |
5, 0, 0 |
0, 4, 0 |
|
1, 0, 8 |
|
0, 2, 0 |
0, 7, 0 |
0, 3, 6 |
|
|
|
above-left of the left large cube (No. 40) |
8, 1, 0 |
0, 0, 5 |
|
4, 0, 0 |
|
|
0, 2, 0 |
3, 6, 0 |
0, 0, 7 |
|
|
above-right of the left cube (No. 42) |
0, 2, 0 |
3, 0, 6 |
0, 0, 7 |
|
|
0, 0, 5 |
|
4, 0, 0 |
0, 1, 8 |
|
|
below-left of the left large cube (No. 44) |
7, 0, 0 |
|
0, 2, 0 |
3, 6, 0 |
|
4, 0, 0 |
0, 1, 8 |
0, 5, 0 |
|
|
|
below-right of the left large cube (No. 46) |
|
4, 0, 0 |
0, 1, 8 |
0, 5, 0 |
|
3, 0, 6 |
0, 0, 7 |
|
0, 2, 0 |
|
|
above-left of the right large cube (No. 23) |
0, 5, 0 |
8, 0, 1 |
0, 0, 4 |
|
|
0, 0, 2 |
|
7, 0, 0 |
0, 6, 3 |
|
|
above-right of the right large cube (No. 21) |
3, 6, 0 |
0, 0, 2 |
|
7, 0, 0 |
|
|
0, 5, 0 |
8, 1, 0 |
0, 0, 4 |
|
|
below-left of the right large cube (No. 27) |
|
7, 0, 0 |
0, 6, 3 |
0, 2, 0 |
|
8, 0, 1 |
0, 0, 4 |
|
0, 5, 0 |
|
|
below-right of the right large cube (No. 25) |
4, 0, 0 |
|
0, 5, 0 |
8, 1, 0 |
|
7, 0, 0 |
0, 6, 3 |
0, 2, 0 |
|
|
|
above-left of the front large cube (No. 52) |
0, 3, 6 |
0, 0, 7 |
2, 0, 0 |
|
|
|
4, 0, 0 |
5, 0, 0 |
8, 0, 1 |
|
|
above-right of the front large cube (No. 53) |
0, 4, 0 |
0, 1, 8 |
|
0, 0, 5 |
|
|
0, 7, 0 |
|
6, 0, 3 |
0, 0, 2 |
|
below-left of the front large cube (No. 56) |
0, 0, 5 |
|
1, 8, 0 |
0, 4, 0 |
|
2, 0, 0 |
3, 0, 6 |
|
7, 0, 0 |
|
|
below-right of the front large cube (No. 57) |
|
0, 2, 0 |
0, 7, 0 |
0, 3, 6 |
|
1, 8, 0 |
0, 0, 5 |
0, 0, 4 |
|
|
|
above-left of the behind large cube (No. 01) |
0, 7, 0 |
0, 6, 3 |
|
0, 0, 2 |
|
0, 4, 0 |
|
1, 0, 8 |
0, 0, 5 |
|
|
above-right of the behind large cube (No. 00) |
0, 8, 1 |
0, 0, 4 |
5, 0, 0 |
|
|
|
7, 0, 0 |
2, 0, 0 |
3, 0, 6 |
|
|
below-left of the behind large cube (No. 05) |
|
0, 5, 0 |
0, 4, 0 |
0, 8, 1 |
|
6, 3, 0 |
0, 0, 2 |
0, 0, 7 |
|
|
|
below-right of the behind large cube (No. 04) |
0, 0, 2 |
|
6, 3, 0 |
0, 7, 0 |
|
5, 0, 0 |
8, 0, 1 |
|
4, 0, 0 |
Table 2: Together with Figure 2, the Following Table Can Be Obtained. The Numbers From Small to Large Represent the Sequence of the Running Light, Returning To 1 After Reaching 8. Number 0 Indicates that One-Third of an Electron does Not Pass Through that Position
Method
Projection
Chemical reactions between atoms depend not only on the electrons on the surface but also on the internal structure of the atoms. Therefore, if we assume that the internal electrons of an atom can be projected onto the surface of the atom like light and decay according to the inverse-square law, we can determine the maximum charge that each atom's surface can bear. If the surface charge of an atom is less than the maximum charge of its orbital, the atom exhibits an attractive force—the smaller the surface charge, the greater the attractive force. To facilitate the calculation of electron projection, we choose an edge length of 2 cubes as the first orbital layer and an edge length of 4 cubes as the second orbital layer that encloses the first layer. Similarly, each successive layer increases by 2 cubes in edge length. Thus, by the seventh orbital layer, the edge length will be 14 cubes, with each inner layer enclosed by the next outer layer [3].
Prediction
As long as the average charge caused by the distance between atoms does not reach the maximum charge, the molecule can connect with other atoms. Therefore, it is possible to predict whether the structure of the molecule can exist.
Implementation
The Marquee on Cube Surface of the Atomic Orbital and the Prediction System for Chemical Reactions implementation URL is: https://www.cpapeijer.com/home/autolearn3. It is written in JavaScript and uses the public packages p5.js[3].
Results
For example, taking the element Og with an atomic number of 118, the number of electrons from the innermost to the outermost layers are 2, 8, 18, 32, 32, 18, and 8. Following the previously mentioned method, Figures 3 to 9 can be obtained.
Figure 3: Og First Layer from the Inside to the Outside has Two Electrons on The Surface of the First Layer.
Figure 4: Og Second Layer From the Inside to the Outside has Eight Electrons on the Surface of the Second Layer.

Figure 5: Og Third Layer from the Inside to the Outside has 18 Electrons on The Surface of the Third Layer.

Figure 6: Og Fourth Layer from the Inside to the Outside has 32 Electrons on The Surface of the Fourth Layer

Figure 7: Og Fifth Layer from the Inside to the Outside, there are 32 Electrons on the Surface of the Fifth Layer
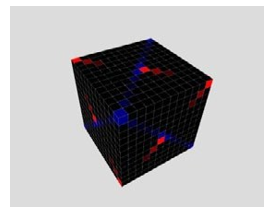
Figure 8: Og Sixth Layer from the Inside to the Outside, there are 18 Electrons on the Surface of the Sixth Layer

Figure 9: Og Seventh Layer from the Inside to the Outside, there are 8 Electrons on the Surface of the Seventh Layer
In addition, by applying the known chemical formulas into the system according to the above method, Table 3 can be obtained.
|
Basic Chemical Formula |
System Guess |
Is it true? |
|
H + H → H2 |
OK |
Yes |
|
H + H + H → H3 |
OK |
Yes |
|
4H → H4 |
Error! |
Yes |
|
2He → He2 |
Error! |
Yes |
|
Li + H → LiH |
OK |
Yes |
|
Li + He → LiHe |
Error! |
Yes |
|
Be + 2H → BeH2 |
OK |
Yes |
|
Be + He → BeHe |
Error! |
Yes |
|
B + 3H → BH3 |
OK |
Yes |
|
C + 2H + 2H → CH4 |
OK |
Yes |
|
N + H3 → NH3 |
OK |
Yes |
|
O + 2H → H2O |
OK |
Yes |
|
F + H → HF |
OK |
Yes |
|
Ne + H → HNe |
Error! |
Yes |
Table 3: Chemical formulas are Diverse and Varied; this Table Lists only a Portion of the Reactions (Atomic Numbers 1 to 10).
Conclusion and Discussion
This paper proves that simplifying DFT using the cubic running light projection is feasible. Additionally, this paper discovers a remarkable number, as shown in Table 4, indicating that there is significant potential for future development of the cubic running light projection.
|
The average projected charge from the 1st surface to the 7th surface from the inside out |
-0.00510204081632653/3 e |
|
The average projected charge from the 2nd surface to the 7th surface from the inside out |
-0.02040816326530612/3 e |
|
The average projected charge from the 3rd surface to the 7th surface from the inside out |
-0.04591836734693878/3 e |
|
The average projected charge from the 4th surface to the 7th surface from the inside out |
-0.08163265306122448/3 e |
|
The average projected charge from the 5th surface to the 7th surface from the inside out |
-0.08163265306122448/3 e |
|
The average projected charge from the 6th surface to the 7th surface from the inside out |
-0.04591836734693878/3 e |
|
The average charge without other layer projections on the 7th surface |
-0.02040816326530612/3 e |
|
The average charge after summing each layer projections and the 7th surface |
-0.30102040816326530/3 e |
Table 4: For the Element Og with an Atomic Number of 118, Using a Cube to Simulate its Surface, the Average Surface Charge is Found to be an Interesting Number: 0.30102040816326530
References
- Chen, J. F., & Hsu, F. R. (2024, May). Discovery of the Characteristics of the Cubic Othello Chessboard and its Implementation of Visualization Expert System. In Proceedings of the 2024 International Conference on Information Technology, Data Science, and Optimization (pp. 1-5).
- Kohn, W., & Sham, L. J. (1965). Self-consistent equations including exchange and correlation effects. Physical review, 140(4A), A1133.
- McCarthy, L., Reas, C., & Fry, B. (2015). Getting started with P5. js: Making interactive graphics in JavaScript and processing. Maker Media, Inc.



